Abstract
Summary
Objectives
To establish whether blood samples taken from used peripheral intravenous cannulae are clinically interchangeable with venepuncture.
Design
Systematic review. PubMed, Web of Science and Embase were searched for relevant trials.
Setting
Trials which compared blood samples from used peripheral intravenous cannulae to venepuncture and provided limits of agreement or data which allowed calculation of limits of agreement.
Participants
Seven trials with 746 participants. Blood tests included 13 commonly ordered biochemistry, haematology and blood gas measurements.
Main outcome measures
95% limits of agreement. Data were pooled using inverse variance weighting and compared to a clinically acceptable range estimated by expert opinion from previous trials.
Results
Limits of agreement for blood samples from used peripheral intravenous cannulae were within the clinically acceptable range for sodium, chloride, urea, creatinine and haematology samples. Limits of agreement for potassium were ±0.47 mmol/L which exceeded the clinically acceptable range. Peripheral intravenous cannula samples for blood gas analysis gave limits of agreement which far exceeded the clinically acceptable range.
Conclusions
Blood sampling from used peripheral intravenous cannulae is a reasonable clinical practice for haematology and biochemistry samples. Potassium samples from used peripheral intravenous cannulae can be used in situations where error up to ±0.47 mmol/L is acceptable. Peripheral intravenous cannula samples should not be used for blood gas analysis.
Introduction
Venepuncture carries a certain amount of pain and a small risk of complications. Given many patients have an in-situ peripheral intra-venous cannula, sampling blood from this obviates the need for repeated venepuncture if there are clinically equivalent and reliable results. In general, this is not common practice due to concerns regarding the validity of results. 1 Other considerations include the method used for obtaining samples, prior or concurrent use of the cannula for fluid administration, the aspiration volume needing to be discarded and any need for special equipment.
Whether assay results from peripheral intravenous cannula and venepuncture are clinically equivalent is quantified using limits of agreement, expressed as the range within which 95% of values will lie in comparison to a reference standard measurement. 2 The limits of agreement are then compared with a clinically acceptable range usually defined by consensus.
In order to assess the quantity and quality of studies comparing blood samples from existing PIV with venepuncture, we undertook a systematic review. We focused on studies of sufficient quality to influence clinical practice.
Methods
Search strategy
Relevant keywords and terms were developed through a scoping search in PubMed, eventually expanding to three databases (PubMed, ISI Web of Science and Embase). Two reviewers screened titles and abstracts (FL and DL). The abstracts were categorised into ‘not relevant’ and ‘potentially relevant’ and all ‘potentially relevant’ studies were reviewed in full. References of included studies were also hand searched (Figure 1). The full search strategy for PubMed is shown in Appendix 1.
Flow diagram showing search strategy. Source: Adapted from PRISMA flow diagram.
11

Study selection
Inclusion criteria
Studies that compared human blood samples drawn from peripheral intravenous cannulae and venepuncture. Studies reporting numerical results for at least one of the following tests: sodium, potassium, chloride, urea (or blood urea nitrogen), creatinine, haemoglobin, haematocrit, white cell count, platelets, international normalised ratio, pH, partial pressure oxygen, partial pressure carbon dioxide. Studies using the Bland–Altman method for limits of agreement or providing data which allowed the calculation of limits of agreement.2
Exclusion criteria
Articles not in English. Studies which used newly inserted peripheral intravenous cannula for blood sampling unless intravenous fluids had been infused through the cannula prior to sampling. Studies which took samples while infusions were running through the cannula or did not wait after stopping infusions. Delay of greater than 5 min between samples for comparison. Studies which did not discard at least 2 mL of aspirate prior to blood sampling. Studies which required special equipment for blood sampling from peripheral intravenous cannula (for example double stopcock techniques).
Data collection and extraction
Relevant data were extracted from included papers in duplicate (FL and DL), including publication year, patient population, number of patients, blood tests carried out, discard volume prior to sampling, wait time between stopping infusions and sampling, aspiration method and cannula gauge as well as the assay results. Tests measured in non-SI units were converted to SI units. Blood urea nitrogen results were converted to urea by multiplying by 2.14 and then converting to SI units.
The 95% limits of agreement was the primary outcome of interest. If not reported directly, it was calculated
Limits of agreement values were then pooled using inverse variance weighting
Clinically acceptable limits of agreement
The clinically acceptable errors in blood sampling are not fully established and vary depending on patient situation and the clinicians’ tolerance for error. Four studies specified such ranges established through clinician survey.3–6 We used a mean of these values to define clinically acceptable limits for this review.
Results
Summary of included studies.
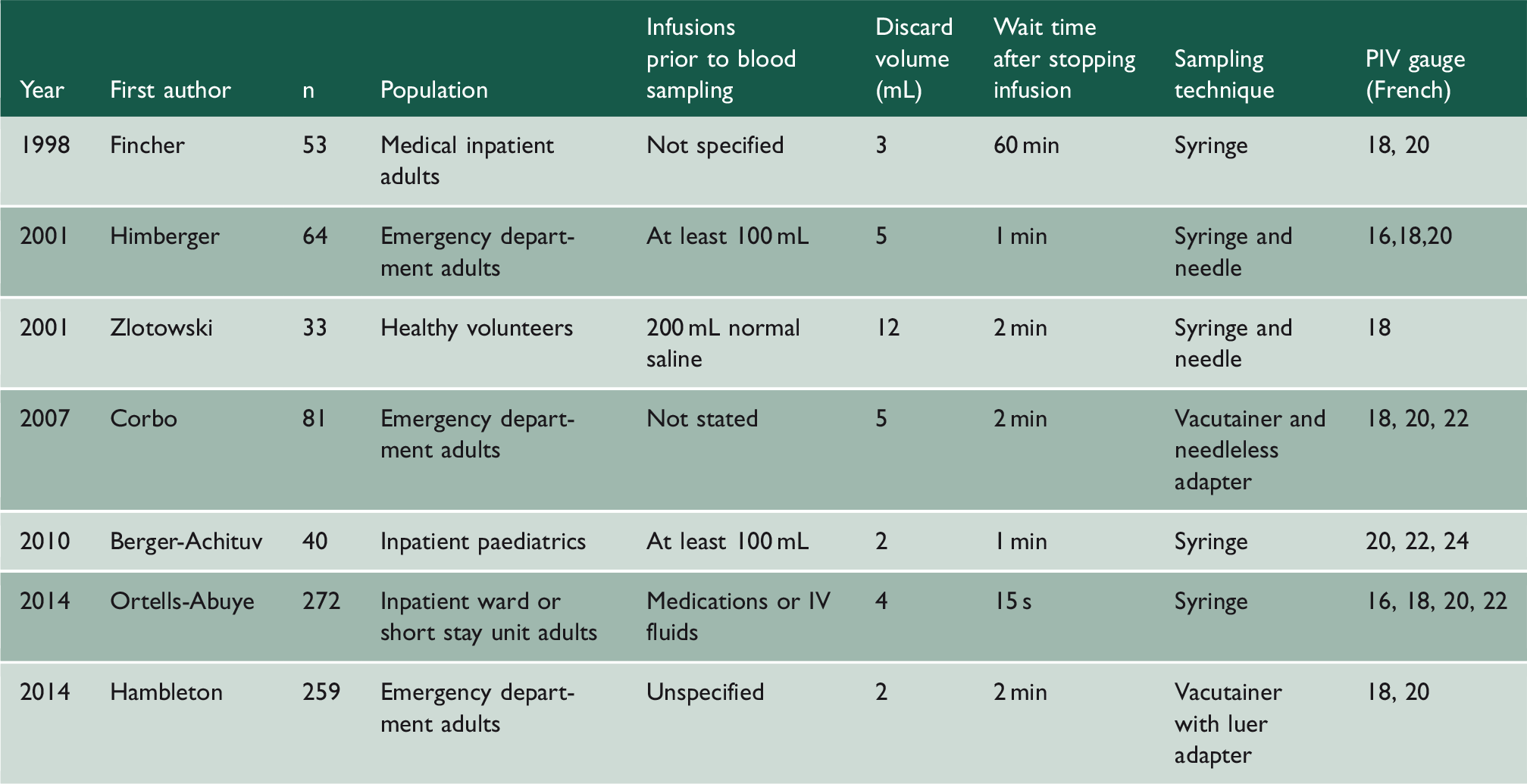
Characteristics of included studies
Studies comparing venepuncture to peripheral intravenous cannula used different methods and protocols (Table 1). The minimum discard volume in the studies was 2 mL. Cannula sizes varied from 16 to 22 French. All cannulae had been used prior to sampling but there was variation in volume and contents of infusion prior to sampling. Sampling devices used were either syringe or vacutainer systems.
Biochemistry
Results for renal function and electrolytes. Showing 95% limits of agreement for included papers, pooled 95% limits of agreement and clinically accepted range for comparison.

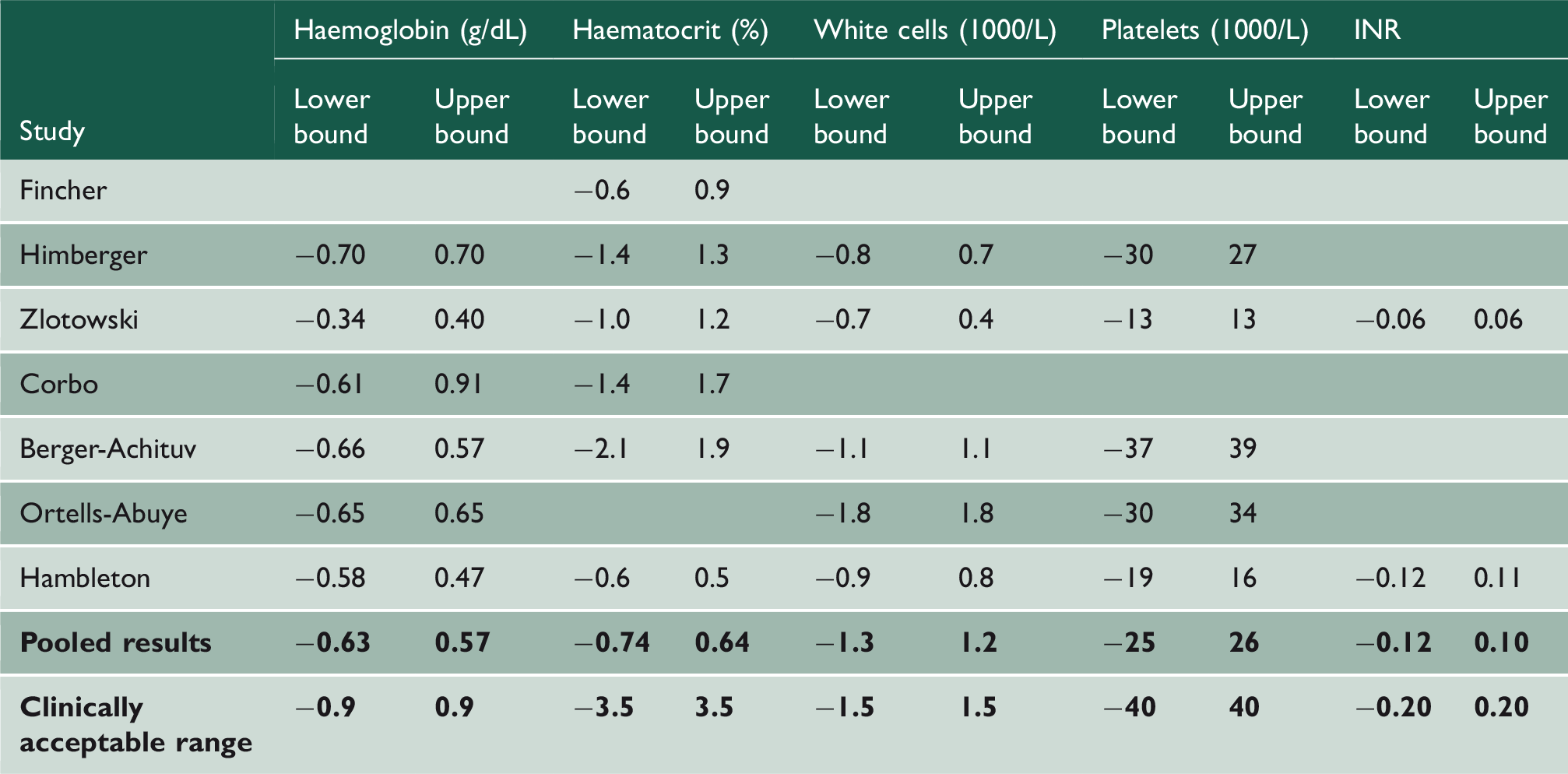
Haematology and international normalised ratio
Results for haematology and international normalised ratio (INR). Showing 95% limits of agreement for included papers, pooled 95% limits of agreement and clinically accepted range for comparison.
Results for blood gas and pH. Showing 95% limits of agreement for included papers, pooled 95% limits of agreement and clinically accepted range for comparison.

Blood gases and pH
The pooled limits of agreement for pH was within clinically acceptable range (Table 4). However, the pooled limits of agreement for pCO2 exceeded the clinically acceptable range (in kPa limits of agreement = −0.9, +1.3; clinically acceptable range = ±0.7) and limits of agreement pO2 dramatically exceeded the clinical acceptable range (in kPa limits of agreement = −4.5, +3.0; clinically acceptable range = ±0.7).
Discussion
Across pooled studies, we showed that assays from used peripheral intravenous cannula were reliable and clinically consistent with fresh venepuncture samples, except in the case of potassium and blood gases. Taken together, our findings suggest that peripheral intravenous cannula sampling could be given greater consideration in clinical practice – at least for the tests described.
The results for potassium levels were not within clinically acceptable agreement limits for some patients. The 95% limits of agreement for potassium of −0.46 to + 0.47 mmol/L shows that, for patients where a tight control of potassium is essential, samples from used peripheral intravenous cannula should be used with caution. For most other patients, a sample from a peripheral intravenous cannula would be sufficient and the level of error ±0.47 mmol/L is unlikely to affect patient outcomes. The cause for the higher level of error is not clear, though haemolysis or haemodilution was excluded as causes in the studies considered here. For blood gas analyses (pO2 and pCO2), the two studies reporting these suggested errors were due to contamination of the samples with atmospheric air post-collection.6,7
There were some limitations to this study. There was heterogeneity in study populations, protocols and equipment. It is not clear whether our findings are generalisable to other sampling techniques, e.g. with narrower gauge cannula.
In terms of haemolysis degrading the sample quality, we show that for most blood tests it does not lead to significant errors. However, if the laboratory or analysers do not check for haemolysed samples it could lead to errors in results. 8
We did not assess some commonly ordered blood tests. Our findings relate to specific assays and may not be generalisable to other haematology and biochemistry investigations.
The clinical impact of these findings will be greatest in those situations in which patients require repeated blood tests where samples from peripheral intravenous cannula would be suitable. For example, if a patient were admitted with symptomatic anaemia and needed serial haemoglobin measurements samples from a cannula could be used. Peripheral intravenous cannula sampling can be an alternative for patients who find venepuncture intensely distressing. There are also patients in which venepuncture is technically difficult and peripheral intravenous cannula samples can provide easier access to blood.
Our findings do not explain why some blood tests are not reliable when taken from a used peripheral intravenous cannula and this could be the subject of further research. Further studies could also be considered to assess other assays which were not included in this paper.
Conclusions
Peripheral intravenous cannula samples are interchangeable with venepuncture for sodium, chloride, urea, creatinine and haematology tests. Peripheral intravenous cannula samples can be used for potassium measurement in situations where error of ±0.47 mmol/L is acceptable. Blood gas analysis for pO2 and pCO2 can show clinically significant differences between peripheral intravenous cannula and venepuncture and so peripheral intravenous cannula samples should not be used. Overall, peripheral intravenous cannula sampling is a reasonable clinical practice for a range of common assays.
Declarations
Competing Interests
None declared.
Funding
DD is supported by a Wellcome Trust Intermediate Clinical Fellowship (WT107467). DL is supported as a UCLH CEO clinical research fellow.
Ethics approval
Not applicable.
Guarantor
FL.
Contributorship
FL and DL both conceived the research. FL led the planning of the research. FL and DL contributed equally to the literature search, review of literature and data analysis. All authors were involved in drafting and have approved the final version. FL is the guarantor and principal investigator, accepting responsibility for the study.
Supplemental Material
sj-pdf-1-shr-10.1177_2054270419894817 - Supplemental material for Blood sampled from existing peripheral IV cannulae yields results equivalent to venepuncture: a systematic review
Supplemental material, sj-pdf-1-shr-10.1177_2054270419894817 for Blood sampled from existing peripheral IV cannulae yields results equivalent to venepuncture: a systematic review by Finnian D Lesser, David A Lanham and Daniel Davis in JRSM Open
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


