Abstract
Background:
Conflicting information is available regarding the stability of glucose concentrations in frozen plasma samples. Clinical trials could benefit from such long-term storage because it would allow usage of a central laboratory with higher-quality laboratory analyzers in contrast to mobile analyzers in a decentralized setting.
Methods:
In this study, venous blood samples were collected in lithium-heparin gel tubes. Plasma was separated immediately after blood was drawn, and from each of the 21 plasma samples, 6 aliquots were prepared for measurement at 6 time points: immediately and after 2, 4, 6, 8, and 12 weeks. Between sampling and measurement, aliquots were stored at less than −20°C. Transport on dry ice was simulated by placing aliquots in a −80°C freezer for 5 days between weeks 8 and 12. Measurements were performed on a hexokinase-based laboratory analyzer.
Average relative differences and corresponding 99% confidence intervals (CIs) were calculated between the stored aliquots’ and the immediately measured aliquots’ glucose concentrations. Glucose concentrations were deemed stable as long as average relative differences were ≤±2.5%.
Results:
Over the whole 12-weeks duration, the largest average relative difference was −1.82% (99% CI: –2.25% to −1.39%). Shorter storage durations tended to lead to less bias.
Conclusion:
In this study, the stability of glucose concentrations in frozen plasma samples obtained with lithium-heparin gel tubes could be shown for up to 12 weeks. Future studies should be performed to assess whether this is independent of the glucose analyzer and the type of sampling tube used.
Introduction
In clinical trials with glucose concentration measurements, like accuracy or performance studies for blood glucose (BG) monitoring systems or continuous glucose monitoring (CGM) systems, the ability to perform reference measurements is paramount. Larger studies may require a multicenter design, leading to the question of whether performing decentralized high-quality measurements, that is, using separate devices at different sites, is possible. The alternative, that is, performing measurements at a central laboratory, would require suitable means for storage and transport of blood samples.
In a decentralized setting, devices have to be selected carefully. It is well established that different brands of laboratory analyzers may yield different results; up to 8% difference was reported in the past. 1 However, even the same type of device used at different sites may yield systematically different results, Bailey et al, for example, found >4% difference. 2 A decentralized setting benefits from using small benchtop devices, because, in contrast to large laboratory analyzers, they are mobile. However, those benchtop analyzers tend to provide lower-quality measurements than larger laboratory analyzers.3-5
Literature indicates that glucose concentrations in native whole blood samples are not sufficiently stable.6,7 After sampling, glucose concentration is lowered by approximately 5%-7% per hour due to glycolysis in blood cells. 7 Additives like sodium fluoride citrate can be used to effectively inhibit glycolysis, 6 but long-term stability over more than 72 hours is not well established.8,9 In addition, care has to be taken during blood sampling and handling of samples. Alternatively, whole blood samples may be centrifuged to separate cells from plasma. However, literature is inconsistent regarding the stability of glucose concentrations in plasma samples.10-12
To provide further insight and for potential validation of a storage procedure, this study aimed to assess the stability of glucose concentrations in separated blood plasma frozen at less than −20°C.
Materials and Methods
This study was approved by the local ethics committee (Ethikkommission bei der Landesärztekammer Baden-Württemberg, Stuttgart, Germany) and exempted from approval by the competent authority (Bundesinstitut für Arzneimittel und Medizinprodukte, Bonn, Germany). The study was performed in consideration of national laws and regulations as well as the World Medical Association’s Declaration of Helsinki in its latest revision.
Study Design
A total of 21 subjects were enrolled in this study after providing informed consent. Subjects had type 1 diabetes (
To simulate transport on dry ice between a study site and the central laboratory in a clinical study, aliquots designated to be measured after 12 weeks were transferred to a −80°C freezer 10 weeks after sample collection for 5 days, before being returned to the less than −20°C freezer.
On the day of the measurements, the respective aliquots were thawed for approximately 30 minutes before measurements. Because of precipitation in the aliquots, they were recentrifuged, and the liquid supernatant was separated for glucose concentration measurement.
Glucose Concentration Measurements
Glucose concentration measurements were performed in triplicate on a hexokinase (HK)-based laboratory ana-lyzer (Cobas Integra 400 plus; Roche Instrument Center, Rotkreuz, Switzerland). This laboratory analyzer is traceable according to ISO 17511. 13 Internal and external quality control measures were performed according to the Guideline of the German Medical Association on Quality Assurance in Medical Laboratory Examinations (Rili-BÄK). 14 In addition, the internal quality control was assured by performing daily measurements with Standard Reference Material 965b (National Institute of Standards and Technology, Gaithersburg, MD, USA), with acceptance criteria of ±5% for individual results. In these additional measurements, bias was ≤2.16% and imprecision was ≤1.56%. Internal and external quality control measures were well within the respective acceptance criteria. The analyzer also participated in the International Federation of Clinical Chemistry and Laboratory Medicine’s proficiency program for reference laboratories. 15
Data Analysis
Averages of triplicate measurements were calculated for each aliquot and each time point. For each aliquot, the relative difference in glucose concentration was calculated in comparison to the result of the immediate measurement (“0 weeks”). An additional analysis was performed in which duplicate measurements were simulated by calculating averages of only the first 2 values in each triplicate. The aim of this analysis was to assess the potential need for additional measurements in the same sample.
Mean values as well as 2-sided 99% confidence intervals (CI) based on the 21 aliquots’ relative differences were calculated for each of the 6 time points.
An acceptance criterion of ±2.5% bias was defined a priori, with the aim to ensure more reproducible results than those expected when using different analyzers. This criterion was chosen based on experience from expected systematic differences between lab devices. It is lower than the initial bias found by Bailey et al, 2 which exceeded 4.0%, or bias between different brands of laboratory analyzers, which is reported to be up to 8%. 1 Even if laboratory analyzers in a multicenter study adhered to the POCT12-A3 criterion, 16 that is, exhibiting ≤2.2% bias, up to 4.4% systematic difference between analyzers might occur in the worst case.
Results
Initial glucose concentrations ranged from 89 mg/dL to 289 mg/dL, and the mean glucose concentration was 143 mg/dL. Of the 21 samples, 10 were in a range of 80 to <120 mg/dL, 7 within 120 to <200 mg/dL, and 4 within 200 to <300 mg/dL. Table 1 and Figure 1 show the bias of subsequent measurements at 2, 4, 6, 8, and 12 weeks of storage, respectively. Mean bias was found to be within ±1.82% at all times for triplicate as well as duplicate measurements, indicating acceptable bias. The lowest end of a 2-sided 99% CI was found after 12 weeks of storage (–2.25%), still within the bias acceptance criterion. Regarding individual samples, similar stability was observed, and there did not seem to be an influence of initial glucose concentration (Figure 2).
Mean Relative Bias and 99% Confidence Intervals for Glucose Concentrations in Plasma Samples Stored for up to 12 Weeks.
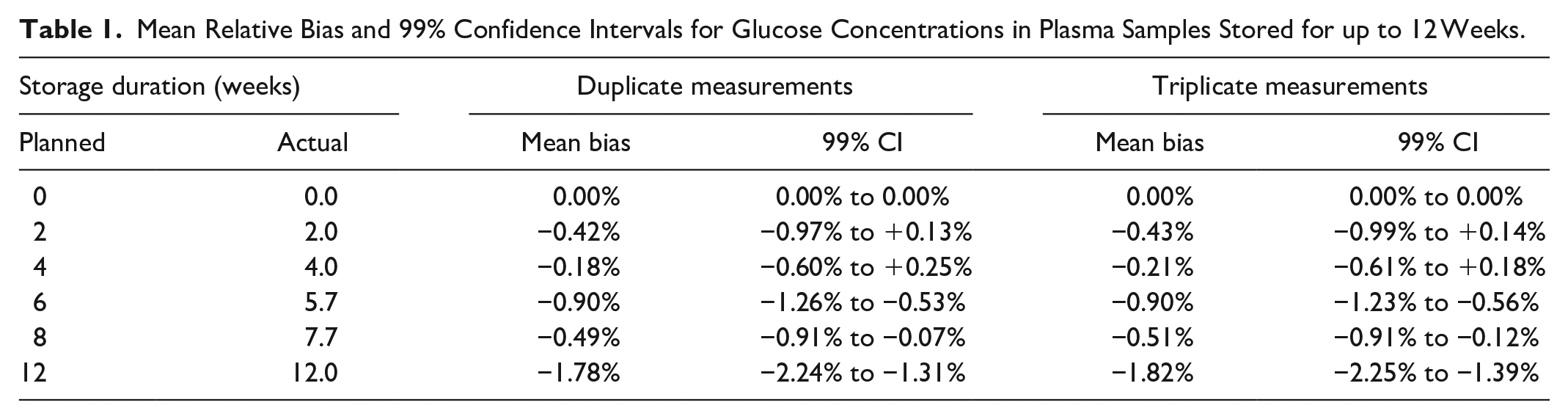
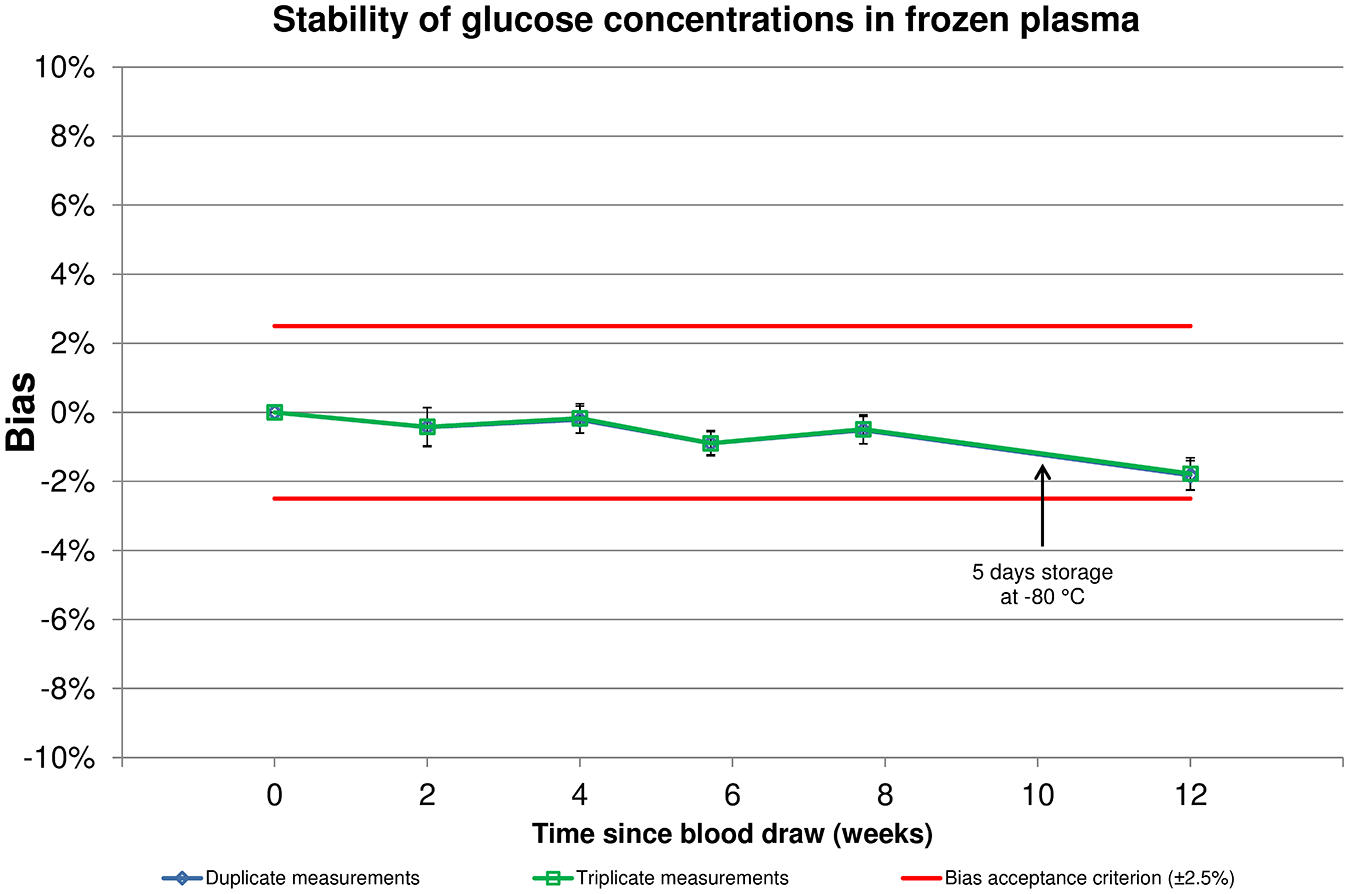
Mean relative bias and 99% confidence intervals for glucose concentrations in plasma samples stored for up to 12 weeks. Predefined bias acceptance criteria are shown in red horizontal lines.
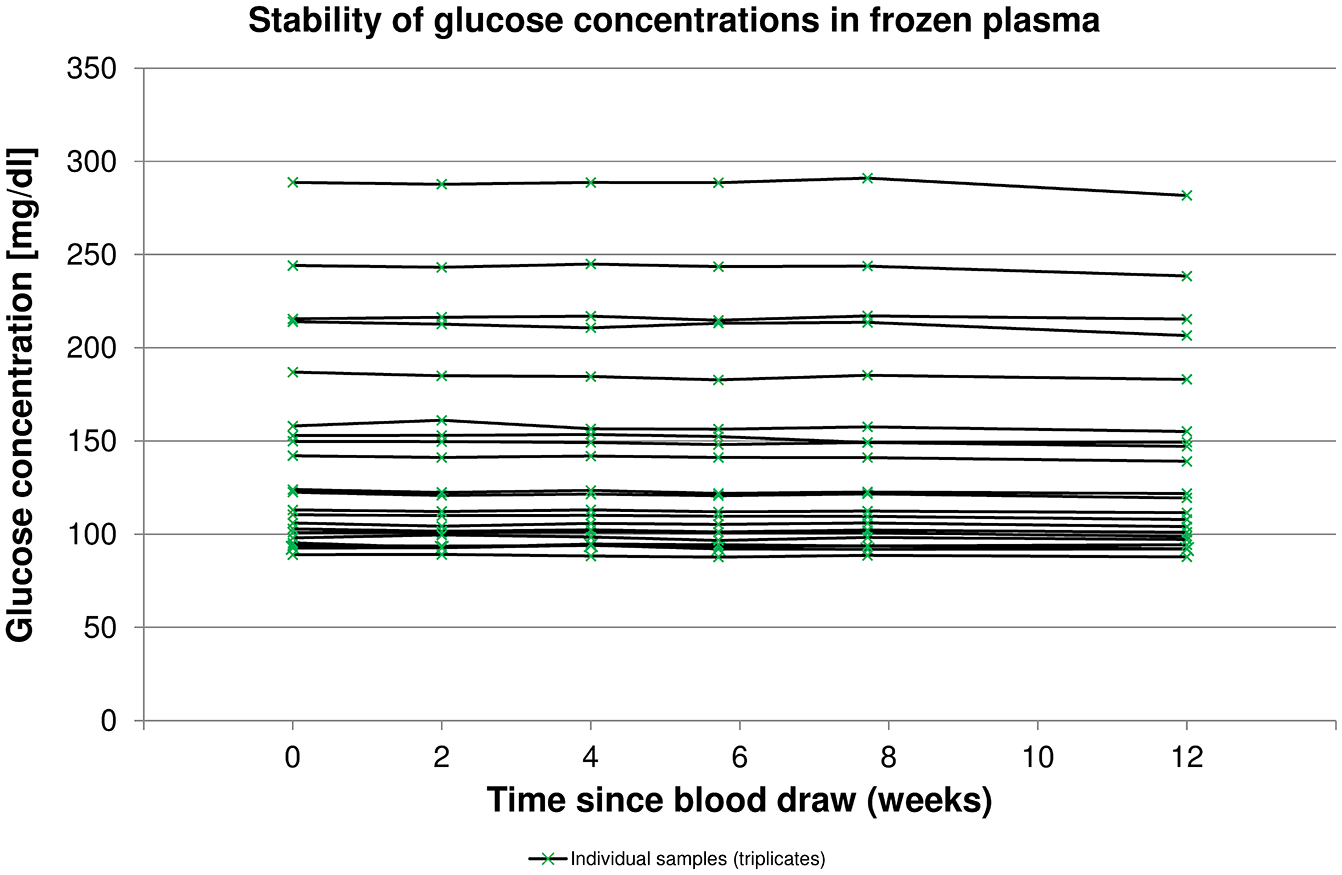
Glucose concentrations in 21 individual plasma samples stored for up to 12 weeks (means of triplicate measurements).
Discussion
This study showed that the storage of plasma samples at less than −20°C did not lead to relevant instability in glucose concentrations. Even after 12 weeks, the mean relative bias was −1.82%, with the lowest 99% CI bounds being −2.25%. Although all results were found within the acceptance criterion of ±2.5%, shorter storage durations tended to lead to less relative bias. No relevant differences were observed between duplicate and triplicate measurements. Although the sample size was not sufficient to compare different glucose ranges, there did not seem to be an influence of initial glucose concentration on stability. Transport on dry ice was simulated in this study by storing the samples in a −80°C freezer for 5 days between weeks 8 and 12. In that time span, the slope in mean bias change was slightly steeper than before. However, it remains unclear whether this is due to the storage at very low temperatures that potentially affected the sample or due to normal sample degradation that might not be linear over that time span or due to other influences.
It has to be acknowledged that sample storage and transport may introduce a systematic difference, albeit small, if, for example, the procedure is used to centralize BG measurements in a clinical trial. However, even using the same type of device at different sites may yield larger systematically different results, for example, Bailey and colleagues reported a >4% difference in bias a few years ago. 2 According to their report about 2 studies, systematic differences could be minimized with substantial effort. Their YSI analyzers were calibrated before each subject sample was tested, and Bailey et al performed additional measurements with serum samples immediately before each subject sample. 2 Both steps increase the amount of time and consumables needed for adequate measurement of patient samples. In clinical trials with CGM systems, where the number of reference measurements in BG samples can be in the several thousand, this approach to minimize bias might not work anymore. Another potential influence factor contributing to systematic differences is caused by changes in the laboratory analyzer, for example, due to performing calibrations or using different calibrators or reagent batches. In a clinical trial, potential systematic differences due to prolonged storage would have to be assessed in contrast to potential systematic differences due to repeated calibration and changes in calibrator and reagent batches.
In this study, lithium-heparinized gel tubes were used to facilitate plasma separation after obtaining venous samples. If, however, immediate centrifugation is a problem in the clinical trial, containers with added glycolysis inhibitors may be better suited.
This study only assessed the stability of glucose concentrations with 1 specific glucose analyzer and 1 specific type of sampling container. Whether these findings are transferable to other models of glucose analyzers and/or other types of sampling containers remains unclear and should be investigated separately.
Conclusion
This study showed that the storage of lithium-heparin plasma samples at less than −20°C did not lead to relevant instability in glucose concentrations. Even after 12 weeks and inclusion of a simulated transport on dry ice, mean relative bias was found within the predefined acceptance criterion of ±2.5%. Further investigations are required to assess whether these findings can be transferred to other glucose analyzers and/or other types of sampling containers.
Footnotes
Acknowledgements
The authors would like to thank Mrs Martina Tesar, Mrs Tuba Alkan, and Mrs Natalie Neuburger (all IfDT) who contributed to the performance of this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is the general manager of the Institut für Diabetes-Technologie Forschungs-und Entwicklungsgesellschaft mbH an der Universität Ulm (IfDT, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF (IfDT) has received speakers’ honoraria or consulting fees from Abbott, Ascensia, Dexcom, i-SENS, LifeScan, Menarini Diagnostics, Metronom Health, Novo Nordisk, PharmaSense, Roche, Sanofi, Sensile, and Ypsomed. SP, BA, and CH are employees of IfDT.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
