Abstract
The striking influence humans are exerting on their environment will likely result in the stabilization of a new climatic equilibrium of the Anthropocene, possibly without historical precedent. Many conceivable outcomes would reshape the planet’s biodiversity. If the Earth-human interaction is to endure in its current state, which still shares characteristics with the Holocene, one necessary development is that humans close the various biogeochemical cycles (C, N, P, K, etc) they have fundamentally altered (i.g. Haber-Bosch). Many of the technologies required to close the chemical cycles, such as the emissions-free production of methanol from industrial exhaust, already exist. Historical examples show, however, that deployment of technology can lag behind innovation resulting in an implementation gap that hinders our ability to mitigate climate change. However, assuming we close this gap, biogeochemical cycles can act as a gage for a “successful” Anthropocene in which mitigation strategies stave off much of what will otherwise become widespread forced adaption to a new, possibly hostile climate. Closed chemical cycles supporting human consumption can be causally linked to human action and precisely marked in time; they will leave an indelible global stratigraphic record, namely in that human influence decreases. Such a development would be a sign that humans had achieved a managed, stable (or at least steady) state within acceptable planetary boundaries of the Earth-human system. This article focuses on closing the carbon cycle over the following decades and proposes, as a measure of progress, the flattening of the Suess effect, a well-known indicator of human impact.
Keywords
Introduction
In the preface to the 1957 book The Next Hundred Years, Lee A. DuBridge, then president of CalTech, wrote, . . .the question of what constitutes a resource is really the question of how intelligently man uses the materials with which the Earth is endowed and of how successfully he discovers new ways to use these materials ( Brown et al., 1957).
Sixty-five years later, his words sound like the best advice we never took. Although we have developed methods for the intelligent use of our resources–ranging from social strategies to highly technical solutions–we have not been proficient at implementing these methods. Much to the contrary, in fact, our detrimental influence on the biosphere is increasing. Our flagrant choices and behavior have endangered the current form of human existence with the energy and climate crises being the most acute examples. I would broaden DuBridge’s statement, then, by adding that the intelligent use of our resources depends on how successfully we discover and implement new ways to use these materials.
Our use of carbon demonstrates our decisions and our predicament (Masson-Delmotte et al., 2021). Carbon, at current concentrations, is detrimental in certain chemical forms. Yet, it is a building block in a large number of molecules essential to our daily lives (and to our physical existence). We have precise chemical control over these molecules and we can skillfully manipulate them. This ability has given us many ways to use carbon intelligently, especially in conjunction with another abundant resource: energy from the sun. Have we discovered a “new way” to use carbon? I argue we have known how to use carbon intelligently since DuBridge penned his advice. But it seems there is too much daylight between knowing how to do something intelligently and actually doing it intelligently.
Our history with carbon certainly starts before DuBridge. Progress toward a modern understanding of the element and its compounds began in the nineteenth century. In 1825, Michael Faraday published his observations on the decomposition of oil by heat, describing one of the substances he found as bi-carburet of hydrogen (Faraday, 1825). He claimed the compound was composed of two parts carbon and one part hydrogen at a time when natural philosophers were beginning to make headway through the labyrinthine field of organic chemistry (Gräbe, 1920; Ihde, 1964; Scholz, 1987). Soon, other substances were identified with the same ratio of carbon-to-hydrogen as found by Faraday but displaying different properties. The concept of the isomer did not yet exist and the results were baffling. Faraday also did not know that hydrogen is a diatomic molecule. His assumed weight for the element was off by a factor of two and meant that his new compound actually consisted of equal proportions of carbon and hydrogen. Throughout the next half century, painstaking studies revealed the actual make-up of many organic molecules (Klein, 2003). Of particular note, Friedrich August Kekulè succeeded in predicting the form of bi-carburet of hydrogen in 1865: it was a ring of six carbon and six hydrogen atoms (Kekulè, 1865). This is the benzene ring, a structure which has played a conspicuous role in humans’ interference with biogeochemical cycles. The composition of this molecule alone, C6H6, shows the carbon and hydrogen cycles are inherently and inextricably linked.
In the middle of the nineteenth century, the complexity and diversity of organic bodies made them not only difficult to classify but also thwarted attempts to explain their behavior. Even the most sophisticated mechanical theories could not reliably predict the outcomes of chemical reactions. A holistic approach was needed, one that could smooth out the details of individual bodies and sum up everything with one overarching concept. By 1865, thermodynamics, based on the concepts of energy and entropy, seemed a likely candidate and had been applied successfully to the description of gases. Over the next four decades, the theory was extended to chemical reactions in solution, giving birth to the hybrid field of physical chemistry (Girnus, 1987; Laidler, 1993; Ostwald, 1927). The products and yields of chemical reactions could finally be predicted.
During the same period, the benzene ring was used to produce aniline, the organic molecule at the heart of the British and German chemical and dye industries. By the turn of the twentieth century, this enterprise had built up a vast infrastructure for the production of synthetic chemicals along with heavy financial clout (Abelhauser et al., 2003; Travis, 1993; Travis and Travis, 2015, 2018). The knowledge from physical chemistry and the means provided by the chemical industry established an entirely new playing field for scientists like Henry Le Chatelier, Wilhelm Ostwald, Fritz Haber, and Walther Nernst to combine the knowledge into new forms and dimensions of chemical manipulation (Johnson, 2022b). One consequential outcome was ammonia synthesis from the elements, a process that transformed the fixed nitrogen industry and provided the foundation for today’s high pressure catalytic industry (Haber, 1971; Szöllösi-Janze, 2000). These technologies have allowed us to solve our limitations on fertilizer production and to refine oil into fuels. They furnish the energetic basis of our modern world.
A common lament, however, is that these successes of science and technology have had unintended consequences and placed us again in the position of having to solve overwhelming, perhaps existential problems. By harnessing the power of chemical manipulation, we have altered the biogeochemical cycles (carbon, nitrogen, phosphorous, potassium, rare earths, etc.) through the biosphere, atmosphere, and lithosphere. As per the nature of the linear economy, the result has been a build-up of substances that become detrimental at certain concentrations. Carbon, in the form of carbon dioxide and methane, strengthens the greenhouse effect; nitrogen run-off causes eutrophication.
While the perturbation of chemical cycles themselves has been discussed as a geologic marker for the Anthropocene (Bernhardt et al., 2017; Carson, 1962; Crutzen, 2002; Fairchild et al., 2021; Rubino et al., 2013; Ruddiman, 2003; Slaveykova, 2019; Waters et al., 2016; Wolff, 2011), another development during a “successful” Anthropocene will be that humans reduce material and energetic waste by driving biogeochemical cycles in closed modus. The word “successful,” though perhaps cynical, denotes that if we do not close the biogeochemical cycles, the Anthropocene, as it exists now with many characteristics of the Holocene, will only be a transitionary period to a new climatic state.
To achieve a closed carbon cycle, we need to defossilize as well as decarbonize. Electrification of manufacturing, mobility, and other processes is important and must be combined with further reductions in carbon use to the extent technical limits allow (decarbonization and carbon direct avoidance) However, we cannot electrify everything and we cannot completely limit material use. Our daily necessities depend on many chemical compounds that we will continue to manufacture and although we cannot undo past industrial developments, we can make future manufacturing practices sustainable. Therefore, we must also implement ways of driving the remaining, carbon-dependent, non-electrifiable processes with net-zero emissions solutions (defossilization). Otherwise we will continue to pollute. Closed chemical cycles, the linchpin of a circular economy, are the final step in forming a truly long-term, sustainable energy system. Their implementation can help us gage the trajectory of (sustainable) consumption as we reduce but also control the accelerating exchange of energy and materials between humans and the environment (Geissdoerfer et al., 2017). Closure of the biogeochemical cycles, in contrast to most anthropogenic activity, will decrease apparent human influence on the biosphere and lithosphere. The change will be captured in the stratigraphic and atmospheric record, for example, as a flattening of the Suess effect first described by Roger Revelle and Hans Suess in the 1950s (Fairchild et al., 2021; Johnson, 2022a; Revelle and Suess, 1957; Suess, 1955).
The Suess effect is caused by the use of fossil fuels and identifiable using the concentrations of carbon isotopes in the atmoshphere, of which there are three: 12C, 13C, and 14C. Only the rarest isotope, 14C, is radioactive, but its proportion in the atmosphere is replenished through cosmic rays, which continually transmute atmospheric nitrogen into new 14C. Thus, the Earth’s pre-industrial atmosphere had an approximately stable mixture of carbon isotopes (Dean et al., 2014; Rubino et al., 2013). Fossil fuels, on the other hand, have a different mixture. The organic matter from which fossil fuels were formed millions to hundreds of millions of years ago is many times older than the 5730 years half life of 14C so that fossil fuels are completely depleted of this isotope. Furthermore, plants (and, thus, animals) are more likely to take up 12C via photosynthesis than 13C, which means organic tissue is enriched in 12C compared to the atmosphere (Farquhar et al., 1989). When fossil fuels are burned, 12C-enriched carbon is rereleased in detectable quantities and decreases the atmospheric 13C/12C
Because the Suess effect is dependent on the burning of fossil fuels, it directly reflects human action. The flattening of the Suess effect as a signal for a “successful” Anthropocene will, therefore, reflect future human behavior and our ability to close the carbon cycle. The effect has been tracked around the world, including since the 1970s in Nuvuk (Point Barrow), Alaska. This stretch along the Arctic coast is a powerful illustration of how anthropogenic activity has consequences in even the most remote parts of the globe (Figure 1) (Keeling et al., 2001; Keeling and Piper, 2005).
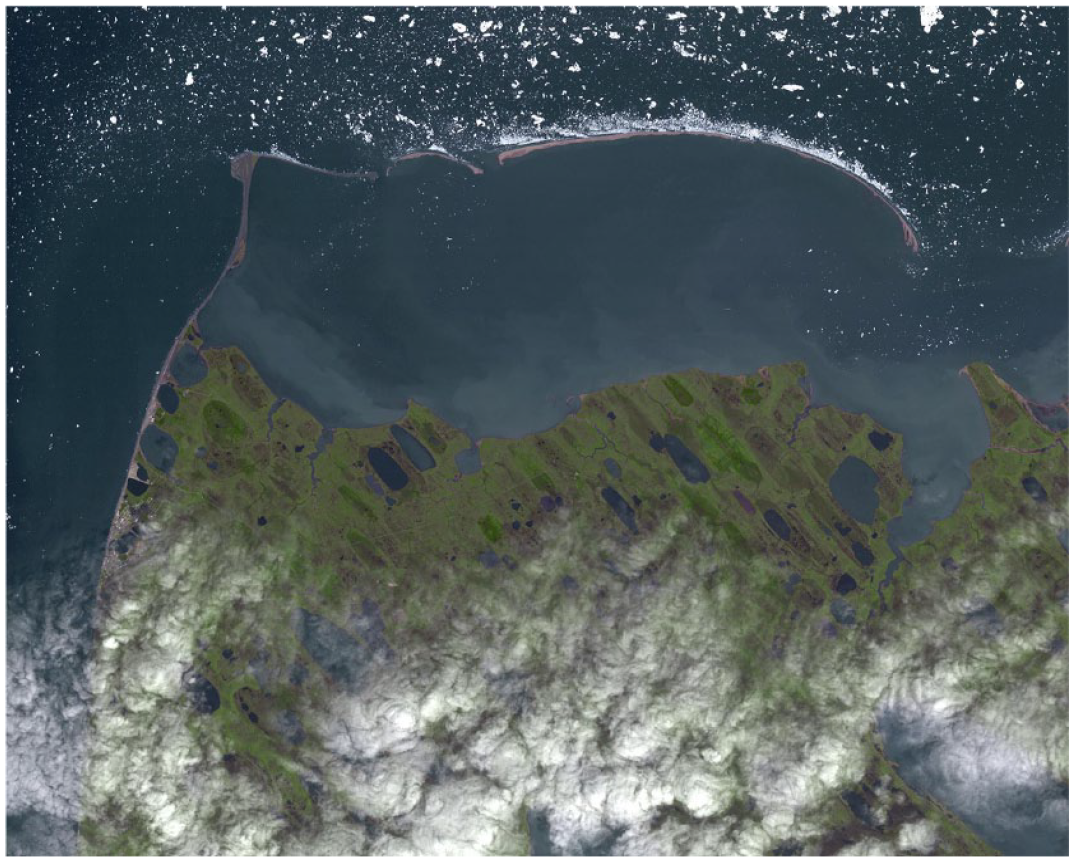
Satellite image of Nuvuk (Point Barrow), Alaska, on the Arctic coast from July 29, 2015. Atmospheric carbon isotope concentrations (Suess effect) have been measured here since the 1970s. Nuvuk is at the upper left land tip bending around Elson Lagoon. The city of Utqiaġvik (formerly Barrow), Alaska is situated down the coast to the southwest (along the left side of the image).
Biogeochemical cycles
Biogeochemical cycles represent complex pathways of chemical change and are driven by no-less complex dynamics. The notion of a “cycle” is actually outdated. Early explanations of the carbon and nitrogen cycles from the first half of the nineteenth century envisioned something like actual step-wise circular behavior (Johnson, 2022b; Jones, 2017; Liebig, 1843; Sprengel, 1839; Youmans, 1856). But modern conceptions describe a set of possible chemical states of an atom (oxidation state), with the distribution of occupied states in a given environment dependent on competition between organisms, natural input and output material fluxes, and, of late, human activity (Rennenberg et al., 2011; Sutton et al., 2011). It is a dynamic equilibrium that can be shifted through external influence.
Before examining the carbon cycle and strategies for closing it, it is important to clarify what is meant by “closed chemical cycles.” In this paper, closing a chemical cycle is with respect to the stability of the biosphere and limiting global warming. How must our material and energy use change so that our consumption is no longer detrimental to life and biodiversity on Earth? Closing chemical cycles means we identify and modify our consumption regime to be sustainable in a special way (“sustainable” and “innocuous” are not synonymous). Even when resource depletion is not an issue, some kind of reuse is required to avoid toxic accumulation. Closure is considered in the following ways:
Material and Chemical Closure. The most straightforward sense of material and chemical closure is that current quantities of elements (stocks) in the atmosphere and biosphere remain constant. This steady state implies that stocks in the lithosphere also remain unchanged. However, closing biogeochemical cycles means considering the amount of an element in circulation along with its chemical state. It is conceivable, for example, that the amount of carbon dioxide in the atmosphere is increased while the amount of methane is decreased to an extent that keeps radiative forcing constant. The overall amount of carbon in the atmosphere would increase, perhaps without negative consequences. A more holistic and realistic sense of material and chemical closure means keeping the chemical and radiative effects of the elements in the biosphere and atmosphere within boundaries that preserve biodiversity and human well-being. This approach is also in line with the managed, stable Anthropocene discussed in this article. It should also be noted that the quantities of some chemical species (carbon dioxide in the atmosphere, nitrogen in coastal regions) are currently too high and net-collection or negative emissions measures are necessary before a steady state can be reached.
Energetic Closure. Chemical cycles are not closed with respect to energy because driving a chemical cycle requires energy input. Energetic closure is a question of waste heat and particles within the system and depends on limiting entropy production (Kümmel, 2011; Kümmel et al., 2018). The biosphere-atmosphere system can endure (and its stability is dependent on) a certain regular energy input. In the absence of human influence, heat is generated by radiation from the sun (low entropy) being converted to infrared heat radiation (high entropy) (Kleidon, 2016, 2023). The process results in a high entropy production and large loss in useful energy. The Earth system is (was) in dynamic equilibrium with this production of entropy from solar influx because it releases (released) entropy into space at the same rate (Rapf and Kranert, 2021). While slow changes in entropy production rates can be compensated for, rapid changes disrupt the balance. The current use of fossil fuels is having this effect by completing the entropy production process that was “deferred” to form fossil chemical bonds in the past. We are overdrawing our entropy budget.
1
Reversing this process requires more energy–but our current energy system only allows for the use of fossil energy carriers, which will increase the rate of entropy production even further. Closing chemical cycles and using renewable energy will return the total entropy production in the Earth system, including our manufacturing and consumption processes, to a rate in equilibrium with the solar influx. For example, capturing sunlight with a solar cell
2
and using the electricity to make heat results in the same entropy production as when natural processes convert the sunlight to heat. But we have exploited the natural entropy production in a way that fulfills our energy needs. Extending the scenario, if the electricity from the solar cell is used to produce hydrogen (electrolysis) and collect carbon to manufacture methanol, we only briefly defer the natural entropy production until we burn the methanol. If ∆
When it comes to human well-being, then, it is perhaps more appropriate to speak of closed bioatmospheric chemical cycles achieved through minimal and decreasing intrusion into the lithosphere. As a first approximation, bioatmospheric chemical cycles can be viewed as individual processes, dependent on an element’s availability, usage and chemical behavior. However, in today’s world of chemically complex, interdependent processes, chemical cycles can only really be managed if they are understood as being interlinked with one another (Barquet et al., 2020; Jarvie et al., 2019; Smith et al., 2020; Sutton et al., 2011; Thomas et al., 2022; Tonini et al., 2019). The connection between the carbon and nitrogen cycles is, for example, illustrated by the Haber-Bosch process. When fertilizer production is included, the phosphorus and potassium cycles come into focus as well. Biogas production, on the other hand, navigates the competition between nitrogen, phosphorus, and cellulose recovery during gas yield optimization (Hoek et al., 2018). This interlinking makes it unlikely we will close just one chemical cycle. Either we will close all of them, or we will close none (Gruber and Galloway, 2008).
Admittedly, closing all bioatmospheric chemical cycles is a formidable task demanding an array of chemical, technical, and policy solutions on land and sea supported by changes in infrastructure and human behavior (Jurgilevich et al., 2016). The phosphorous and nitrogen cycles, as integral parts of industrial agriculture, illustrate the complexity of the challenge and make explicit the difference between sustainability and circularity.
In some ways, the nitrogen cycle is already closed: denitrification of N-species in the soil returns N2 to the atmosphere. Yet, fixed nitrogen washed into coastal regions causes eutrophication. The nitrogen is bound into plant tissue in an open ended process. Complicating the picture still further, both the open and closed parts of the nitrogen cycle have been modified by human intervention through intensive reliance on fossil fuels (Hoek et al., 2018). In contranst, phosphorous consumption remains largely linear. The main source of phosphorous is from phosphate rock mining and attempts at recycling may account for only about one-third of total phosphorous use in the next decade (Elser and Bennett, 2011; Withers, 2019).
One solution for closing the nitrogen and phosphorous cycles is wastewater treatment, in which nitrogen and phosphorous are precipitated out (struvite) or recovered through air-stripping or sludge reuse (Hoek et al., 2018; Withers, 2019). A second, complementary strategy to bridge the “marine-land loop” is through mariculture in coastal regions suffering from eutrophication. Ranging from the growth of wild biomass to non-fed, low-trophic species, these strategies are capable of collecting nitrogen and phosphorous into biomass that can be sold in a future blue-green economy (Barquet et al., 2020; Thomas et al., 2022).
The full solution, whatever it may be, will benefit from differentiating between sustainability and a circular economy (Geissdoerfer et al., 2017). The atmosphere is an inexhaustible and sustainable source of nitrogen. However, the use of fossil fuels in the preparation of fixed nitrogen and the overuse of fertilizer makes continued consumption according to current practices detrimental to human well-being. But we will not run out of nitrogen. Phosphorus, on the other hand, is limited and a recycling regime, or circular phosphorus economy is required to make our use of phosphorus sustainable in any form.
The closed carbon cycle
Carbon is similar to nitrogen in that it is a sustainable resource. We will not run out of carbon, but our flagrant use has become injurious, particularly in the form of the greenhouse gases carbon dioxide and methane. Our ability to close the carbon cycle is closely linked to our methods of energy conversion and energy use. There are several strategies. The most urgent are changes in behavior and the strengthening of communications campaigns to spread the message that a reduction in consumption is needed. These efforts have large regional and cultural fluctuations and must be supported by education, outreach, and self-discipline. However, even together with re- and afforestation (Wich-Konrad et al., 2020), they will not get us to net-zero carbon emissions on their own. The restructuring of manufacturing and consumption processes is also required. Two possibilities are Carbon Capture and Storage, or CCS (possibly sustainable for humans), and Carbon Capture and Use, or CCU (possibly circular) (Abanades et al., 2017; Acatech, 2019; Bui et al., 2018; Deerberg et al., 2018, 2020; EU Commission, 2021; Styring et al., 2011). With current technology, CCS is an option, but storing carbon as carbonate in bedrock or via ocean uptake does not decrease intrusion into the lithosphere and is not a circular strategy. CCS relies on a sink. CCU, though technologically in development, offers the possibility of establishing a net-zero carbon cycle and is a concrete example of the circular economy.
Although there are advantages and criticisms of both CCU and CCS (that CCU is “a monstrous thermodynamic crime,” e.g.), both will likely play a role in our future energy and industrial landscapes. “We need to consider CCS and CCU,” states Peter Styring, “and any other methodology that develops. We are not in a position to exclude any technology as we do not yet have a solution. Indeed, we may need a suite of solutions that depend on industry, environment and geographical location, including resource availability (Wilson et al., 2016, Quotes are on p. 573 and p. 578, respectively).”
CCU, like photosynthesis, has a multi-step production chain with total inefficiency dependent on the steps of the chain. Electricity generated by photovoltaics (PV) and wind can be stored in batteries, fed into the public grid, or the electricity can be consumed immediately or stored as heat or hydrogen. The hydrogen can also be reacted to more complicated fuel molecules or precursor chemicals for industry (Ausfelder and Dura, 2019). For example, hydrogen can be combined with nitrogen in an emissions-free alternative to the Haber-Bosch process or with carbon and oxygen to form any number of organic molecules such as methane, methanol, liquid organic hydrogen carriers (LOHC), polyoxymethylene dimethyl ether (PODE, a diesel alternative) (Schlögl, 2020), or even Michael Faraday’s bi-carburet of hydrogen. The carbon can come from fossil fuels, as it does today, or carbon dioxide can be filtered out of the air via direct air capture (DAC) (Lackner, 2009; Rochelle, 2009; Sanz-Pérez et al., 2016; Zeman, 2007). With the latter option, the carbon dioxide set free when burning carbonaceous fuels is collected and reused in a circular process.
Carbon can also be captured from industrial flue gases, such as from steel manufacturing, and cleaned, adjusted for constitution, and reused as precursors for the chemical industry or in the production of building materials (carbonates). The steel and chemical industries are examples of sectors that cannot be fully decarbonized because their production processes depend on carbon chemistry (Bender et al., 2018; Deerberg et al., 2018, 2020; Styring et al., 2011; BMWi, 2020). However, as with synthetic fuels, the carbon need not come from fossil sources. The carbon can be part of a closed loop. Current efforts focused on a one-time reuse of carbon in the chemical industry can result in up to a 50% reduction in carbon emissions (Abanades et al., 2017). Further reductions can be achieved through carbon direct avoidance measures such as the direct reduction of iron with hydrogen. If the widespread recycling of plastics and chemicals is established, a closed loop is also possible (and needs to be our goal) (Christensen et al., 2019; Dai et al., 2022; Geres et al., 2019; Taylor, 2017). On the other hand, industrial emissions can also be used to manufacture fuels such as methanol, in which case, parts of the carbon cycle can be closed with current technology.
The closed carbon cycle illustrates not only possible sustainable energy and consumption pathways. It also shows how humans’ approach to energy use has changed over time. The establishment of a closed carbon cycle based on CCU depends on the ability (or luxury) to harvest power from inexhaustible sources, such as the sun or wind. Historically, humans have been purposeful in their choice of energy conversion (Billington and Billington, 2006; Smil, 2017). Early processes converted chemical energy into heat from fire or kinetic energy for a canon or steam engine. Later, the generation of electricity in the nineteenth century made possible the transport of mechanical work to regions where coal or the kinetic energy of wind or water was not accessible (Emery, 1896). Generally, it was a linear conversion chain revolving around economic considerations to maximize efficiency.
Into the twentieth century, sources of electricity expanded but remained based on nineteenth century technology (hydropower or steam driving an induction coil). Around mid-century, nuclear power was introduced, but it did not change the basic mechanism of electrical generation. Further changes made up only a small fraction of the energy regime. Wind farms, for example, did away with the conversion step to heat but still depended on induction. All of these processes, harnessing kinetic energy to make electricity for an end-user or burning fuel for heat or motive force, are still based on a linear conversion and consumption chain.
Solar cells, on the other hand, represent a fundamentally different approach to electrical generation. Here, electromagnetic radiation is captured and converted directly into electricity in a single, integrated step (Klenk, 2001; Lewerenz and Jungblut, 1995). Like wind, PV is an emissions-free (at this point, still excluding the manufacturing process), inexhaustible energy source driven by the sun. These two qualities allow us to implement the circular strategy of consumption needed for a “successful” Anthropocene. Similar to some processes that have existed on Earth for millennia, direct reliance on the sun frees us from efficiency as the constant central factor in our choices of energy conversion. Natural processes can be notoriously inefficient when it comes to energy conversion: photosynthesis is about 3% efficient compared with human-made solar-to-hydrogen conversion (10%−20% or 30% under concentrated illumination) or solar-to-fuels (about 13%) (Lewis (2007; Aresta and Dibenedetto, 2020; Biello, 2011; Ganesh, 2011; Jia et al., 2016; Tredici, 2010). 3
The sun provides approximately 1000 W of radiative power per square meter to the Earth’s surface, or about as much energy in 1 hour as humanity uses in a year (Lewis and Nocera, 2006). After harvesting and converting this energy into electricity with PV or wind turbines, we have several choices. We may perform zero conversion steps and use the electricity directly or we may choose a long conversion chain that includes carbonaceous fuels and at the end, still have electricity. We are free to choose the final form of energy suitable for any situation; the inherent inefficiency of the process and practically boundless initial energy source frees us from having to define a maximally efficient, linear conversion pathway.
Here, we can distinguish between process efficiency and system efficiency. While we should try to optimize individual processes as much as possible, the most efficient system does not necessarily presuppose a set of individual components, each of which is in itself maximally efficient. Less efficient processes may lead to system optimization, within realistic technological or regional limits, by allowing the use of other key processes or beneficial choices at different stages (De_Richter et al., 2013; Schlögl, 2017). Barquet et al. (2020), for example, warn of the lock-in effects of relying on one favored technology. “By considering multiple benefits and trade-offs across additional parameters,” they write, “. . .we might be better placed to avoid solving one environmental problem at the expense of another.” As examples, the conversion of natural lands to agriculture has been alleviated at the expense of coastal regions (eutrophication). And deforestation to produce wood as fuel has been solved by polluting the atmosphere and oceans with carbon from fossil fuels. There are also countless examples of solving human-specific (not necessarily environmental) problems at the expense of the environment.
When considering the mix of requirements placed on our future energy system, then, the question about a solution becomes: which configuration produces the most efficient system? Apart from finally regarding waste heat and pollution as expensive consequences to be avoided, the answer will be a shifting optimization, or balance between the available options as bridge-technologies are replaced by maturing, long-term capabilities within the context of changing societal expectations and the occasional disruptive discovery. There is no one superlative fix; it is a dynamic solution, in which a closed carbon cycle will play a role.
The history of the closed carbon cycle
So far, strategies and technology for closing the carbon cycle have been shown to be closely tied to energy production and use. The history of the closed carbon cycle can also be discussed well within this context.
The closed carbon cycle is dependent on two things: a cheap, large source of energy and a way to store that energy in chemical bonds. In the 1950s, both were available for the first in the form of nuclear power plants and water electrolyzers. However, the first attempts at non-fossil chemical synthesis began earlier, around 1900, soon after the discovery of radium and its decay product, radon, and their use in the first radiochemistry experiments (Lind, 1928). Initial investigations were crude: a vile of radium or radon was held in a chemical solution and the
Many chemical processes were observed, but despite some intriguing reactions that fixed nitrogen, reduced CO2, or decomposed water into hydrogen and oxygen, synthetic chemical yields were too small to entertain the notion of scaled-up production. First, larger and more reliable sources of radiation were needed. These appeared in the 1920s when the linear particle accelerator was developed and put into use in Germany and later, in the 1930s, when Ernest Lawrence built the first cyclotron at Berkeley. Another technology-based source of radiation from the period was the betatron. The observation of excess neutrons produced in fission events, however, led researchers back to natural sources of radiation like uranium and thorium when it was realized a nuclear chain reaction was possible. The first reactors were built in the 1940s and 50s and became, along with the radioactive byproduct 60Co, the centerpiece in large-scale plans for non-fossil chemical synthesis (Creager, 2015; Draganić, 1989; Heilbronn and Seidel, 1989; Lanouette and Silard, 2013; Steinberg, 1962). The reactors provided the necessary advances in the generation of radiation and led to the foretelling of a “nuclear-electrochemical age.” Nuclear energy would be the power source of the future (Figure 2) (Gregory et al., 1972).
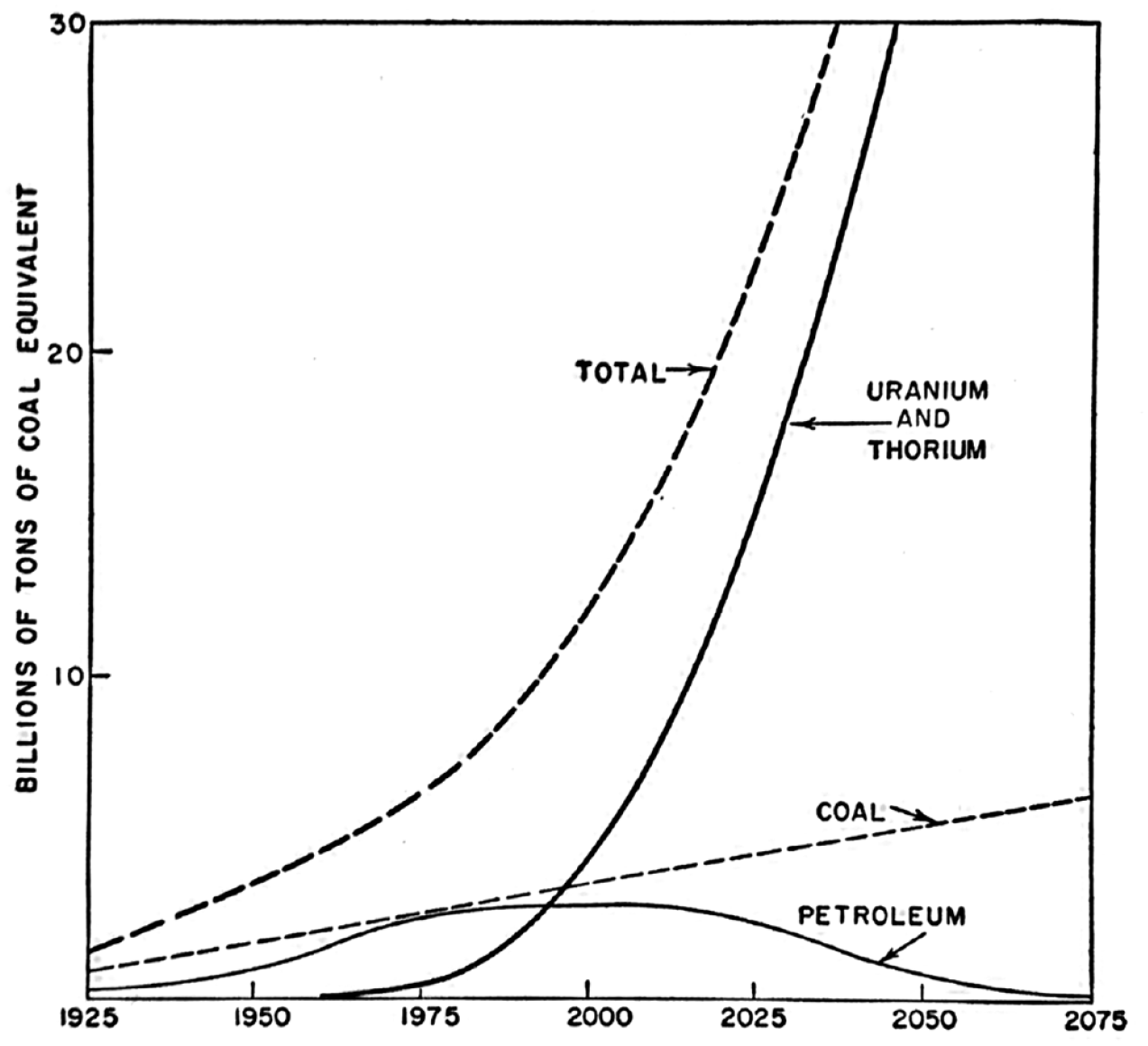
A graph from The Next Hundred Years depicting the increased use of nuclear fuels into the twentieth century (Brown et al., 1957, p. 110). The original caption reads: World Energy Consumption in the Next Century–a Possible Pattern.
A publication from 1961, for example, envisioned a reactor fueled by uranium dust, in which diatomic nitrogen and oxygen circulated with fissioning 235U. The fission fragments were to split the triple-bonded nitrogen molecule and lead to the formation of NO x . It was also thought that such a reactor could produce CO from CO2 (Steinberg et al., 1961). However, economic considerations, decontamination, and product separation posed hurdles that were too severe.
These complex devices using nuclear heat and fission fragments were researched into the 1970s despite the availability of the simpler electrolytic production of hydrogen from nuclear-based electricity. Not all efforts at establishing a fossil-free energy economy had been focused on electrolysis by this time because electrochemistry, too, had had its difficulties. Conceptual progress had been incremental and, at times, stagnant since the 1890s.
There are two ways to release chemical energy: through combustion (the direct release of reaction enthalpy completely as heat) and electrochemically. The burning of fossil fuels in the nineteenth century allowed for unprecedented technological development, but the low efficiency of combustion-to-mechanical work or combustion-to-electricity conversions, dictated by the Carnot limit, was well-known to scientists and engineers. Ways around the limit were sought. One expression of this ambition came from Wilhelm Ostwald. “The way in which the greatest of all technical questions– the procurement of cheap energy– can be solved,” he wrote in 1894, “must be found by electrochemistry (Ostwald, 1894a, 1894b).” Ostwald made clear that all chemical reactions were a consequence of rearranging atomic electrons, but that combustion (burning) only allows partial use of the resulting heat energy. Electrochemical oxidation, on the other hand, was not subject to the Carnot limit and allowed an almost complete conversion of chemical into useful, electrical energy. In contrast to today, much of the effort in electrochemistry at the time was centered on the oxidation of carbon containing compounds in an attempt to make more efficient use of convenient and conventional fossil energy sources.
It was not clear, though, how electrochemical systems (electrolyzers and fuel cells) were to be built in order to take advantage of the higher theoretical efficiencies. Electrochemical reactions were not well-understood and the source of energetic losses could not be pinpointed. When water is decomposed electrochemically into hydrogen and oxygen under standard conditions, the thermodynamically required voltage to “tear the water molecule apart” is 1.229 V. However, at this voltage, no reaction takes place. Because of resistive effects in the electrochemical circuit, an overvoltage is required to drive the decomposition. Similarly, when hydrogen is oxidized (burned) in the reverse electrochemical reaction to form water, the resulting generated voltage is less than the expected 1.229 V. Early on, the phenomenon was referred to as “polarization” and some researchers thought the energy losses were inherent in the electrochemical process. Combustion was, they opined, not an inferior alternative. It took until the 1950s to fully understand the difficulties and sufficiently reduce the overvoltage in working systems (Appleby, 1990; Bockris and Reddy, 1973; Conway, 1989; Tafel, 1905). In particular, an adequate description of charge transfer at the solid-liquid interface was needed. And establishing this description required the production of new knowledge about electrochemistry. The progress in this field reflects the general increase in the understanding of solids, liquids, and gases during the first 50 years of the twentieth century and how matter and different phases of matter interact at an atomic scale. It is a strong example of the scientific process and how breakthroughs require the correct circumstances before they occur (Johnson, 2022b; Riordan and Hoddeson, 1997; Townes, 1999).
It was also in the 1950s that climate change emerged as a pressing global issue after nuclear testing and attempts at weather modification had shown human activity to be global in reach and perhaps detrimental. Scientific explanations of human impact on the climate had changed little since the 1890s when the interplay of CO2, water vapor, and solar radiation had been identified as a possible cause of warming or cooling of the Earth. Over the next half century, however, the actual importance of human activity in the face of climate change caused by mountain building or the Earth’s axial tilt and orbit was called into question (Fleming, 1988, 2010). Also, it was thought that global warming could theoretically stave off a new ice age or, perhaps more realistically, open new lands for agriculture in colder northern regions (Arrhenius, 1896; Callendar, 1938; Crawford, 1996; Hart, 1992; Shulman, 2010). Global warming was not considered a threat.
By the 1950s, the tune had changed and CO2 began again to attract attention. “It has been suspected,” wrote Palmer Putnam in 1953, that the recent world-rise in temperature, accompanied by a rise in sea-level as glaciers melt, has been caused in part by an increasing carbon dioxide content of the atmosphere, for which man is responsible [. . .] If there exists a possibility that the maximum plausible expansion of demands for energy, coupled with our other activities, may inadvertently affect the weather, we should investigate until doubt is removed (Putnam, 1953, p. 170).
The pitfalls of the linear carbon economy were clear. With nuclear energy on the horizon, it was not a large leap of the imagination to see how the marriage of man to combustion, which had been declared wasteful and unjustified by Ostwald over a half century earlier, could come to an end. Numerous ideas were published in the 1950s, 60s and 70s, not as a reaction to the oil crisis of 1973, but as a response to the military and the space race, rapid population increase, economic and energy self-sufficiency, and the pollution or climatic consequences of fossil fuel use. The expectation that fossil-based resources were limited may have become more acute in the mid- and late 1970s after the oil crisis, but the perceived threat of declining fuel reserves was already prominent in the 1950s and 60s (Bockris, 1972; Bockris and Reddy, 1970, 1973; Bolin and Keeling, 1963; Brown et al., 1957; Burger, 1976; Callendar, 1958; Egli and Sherman, 1964; Keeling, 1960; Putnam, 1953; Reed and Lerner, 1973; Revelle and Suess, 1957; Shreve, 1967; Steinberg and Dang, 1977; Weinberg, 1959; Wigg, 1974).
Ideas about circular carbon use, on the other hand, are less apparent in the 1950s and require further historical research. Putnam did discuss recycled carbon in the context of algae cultivation, but it is understandable that early concepts for a closed carbon cycle are rare or vague: in the 1950s, nuclear and wind power were still in their infancy. PV cells jumped from about 0.5% efficiency to between 6 and 10% (Riordan and Hoddeson, 1997)–a dramatic improvement but still not enough to power a closed carbon cycle. Mature concepts had to wait until nuclear power was no longer a military secret (Krige, 2006). After the passage of the 1954 Atomic Energy Act and the 1955 Atoms for Peace Conference in Geneva, information on atomic energy not only became widely available for the first time, but Western governments began actively promoting it. By the end of the decade, scientists had the tools for chemical synthesis based on the newly developed electrochemical cells and electricity from “the recent development of large-scale nuclear sources (Steinberg, 1964).” Early block flow diagrams of electrochemonuclear systems began to appear in the 1960s describing the synthesis of ammonia, hydrazine, methane, methanol, and other chemicals. There were also ideas for manufacturing including an electric furnace to produce metal.
By the 1970s, the closed carbon cycle was being discussed much in the same terms it is today. “The problem of CO2 control in the atmosphere,” wrote Cesare Marchetti in the abstract of his 1977 paper on CO2 ocean storage, “is tackled by proposing a kind of ‘fuel cycle’ for fossil fuels where CO2 is partially or totally collected at certain transformation points and properly disposed of (Marchetti, 1977).”
While Marchetti was describing something more akin to CCS, the technical possibility of stripping CO2 directly from air or point sources had been recognized since the 1930s and has explicitly figured into mature concepts for a closed carbon cycle since at least the 1970s (Figure 3) (Nonhebel, 1936; Rochelle, 2009). 4 “The ability” wrote Meyer Steinberg and Vi-Duong Dang in 1977,
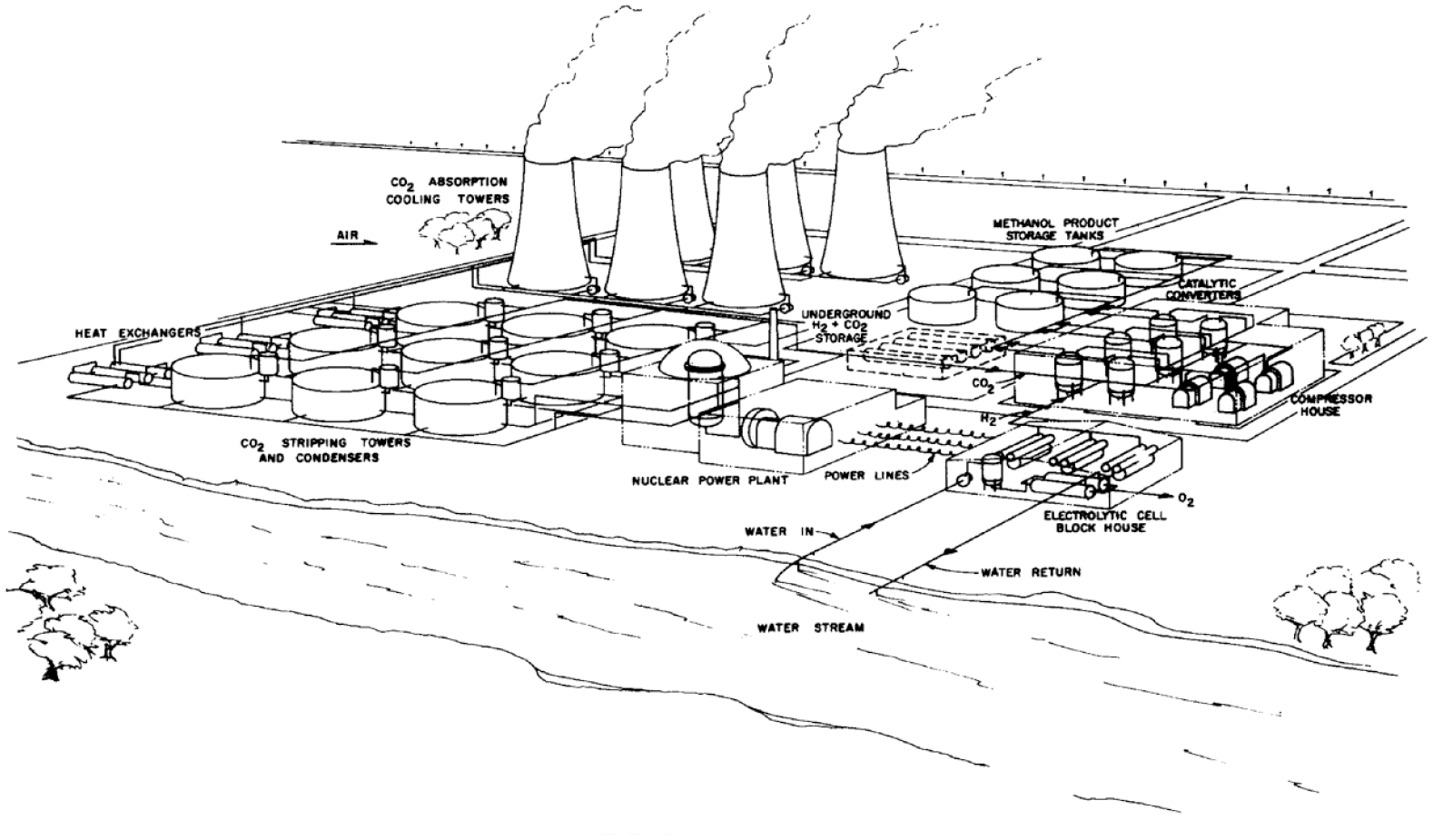
A depiction of a non-fossil methanol production facility from 1977 (Steinberg and Baron, 1977). Analogous to current ideas, the site consists of integrated CO2 collection (stripping), electricity production from nuclear fuel (today with renewables), water electrolysis, and methanol synthesis (See also footnote 2). The original caption reads: Liquid methanol fuel from nuclear power, air and water.
of utilizing nuclear power to form synthetic hydrocarbon fuels from non-fossil fuel sources would provide distinct environmental advantages and add a new dimension to nuclear power. The synthesis of methanol using only CTR [controlled thermonuclear reactor] energy, atmosphere and water comes closest to simulating the solar energy photosynthetic fuel cycle. The carbon fixed from the atmosphere to methanol is returned to the atmosphere by combustion (Steinberg and Dang, 1977).
It is noteable that fusion was the nuclear energy of choice. In the 1970s, many of the scientists promoting a non-fossil fuel economy expected the energy requirements of the future to be provided by nuclear fusion rather than nuclear fission (Thirring, 1958).
Although by this point, much of the conceptual puzzle was in place to establish a non-fossil synthetic fuel infrastructure, economic and political factors were still not favorable. Hydrogen was being produced electrolytically in Canada and other locations with access to cheap hydropower. Otherwise, the price of “conventional” electricity and cheap and widely available natural gas kept steam reformation of methane a more viable option for producing hydrogen. Furthermore, transporting natural gas in a pipeline remained cheaper than hydrogen or electricity and methanol could be easily manufactured from gas, coal or wood (Gregory et al., 1972; Reed and Lerner, 1973). Nevertheless, research toward the nuclear-electrochemical age continued because, as seemed clear at the time, no matter how cheap the natural gas and other fossil resources, supplies would soon dwindle and at that point, hydrogen would be the only alternative.
As it happened, the expectation of a world powered by fusion energy never materialized: an energy positive experiment has only recently been reported (Ball, 2021). Fission, the other option for nuclear power anticipated by the physicists of the 1950s, 60s, and 70s never reached its expected potential either. In the United States, for example, after the initial government push for the acceptance of nuclear power in the 1950s in the wake of the Atomic Energy Act and Atoms for Peace program, skepticism toward atomic energy began to wane; concerns about air pollution from fossil resources and the lack of public willingness to embrace more conservative energy use aided the trend. By the 1970s, over 200 nuclear power plants were online or planned with projections of over 1000 by the end of the century (Del Sesto, 1979; Walker, 2004). However, the 1973 oil crisis, somewhat paradoxically within the context of the United States’ push for energy independence, ended the boom in nuclear energy expansion (Yergin, 1990).The resulting inflation and steep increase in the price of oil severely compromised the energy industry’s ability to invest in the high capital costs of new nuclear power stations. However, many scientists and (according to polls) a majority of the American public supported nuclear power. At the same time, the anti-nuclear movement revived concerns, mainly about radiation exposure, that had first surfaced during the fallout debates of the late 1950s and been quelled by the Limited Test Ban Treaty of 1963. Falling together with the rise of the environmental movement in the early 1970s, the opponents of nuclear energy expressed concern about thermal pollution, radioactive emissions, reactor safety, and the disposal of nuclear waste. It was a well organized and outspoken movement and a bitter and emotional debate arose, lasting into the 1980s. High profile disasters such as Three Mile Island and Chernobyl added to the fervor (Krige, 2006; Walker, 2004).
Succumbing to public pressure, the proliferation of nuclear energy stagnated into the 1990s. Today, it accounts for 10% of global electricity supply, far from supporting a nuclear-electrochemical age (IEA, 2019a). The missing source of cheap, accessible power may now soon become available through solar and wind–if we are able to implement the necessary infrastructure. With these resources, we could close the carbon and other bioatmospheric chemical cycles based on science, technology, and concepts, some of which are more than a century old. As stated at the outset, we have discovered methods of intelligently using our natural resources, but we have not always found ways to implement them. The result is an implementation gap separating our practical technical ability from that needed to reach our emissions goals and combat climate change. It is not a necessary deficit due to a lack of know-how, but one that has arisen through decades of deferred decision making.
In order to place ourselves in the best position for success now, we must be ready to implement coming advances. Beyond the infrastructure changes needed to facilitate a closed carbon cycle economy, essential ongoing research includes improving carbon capture capabilities and catalyst performance along with the optimization of fuel cell and electrolyzer design (Ausfelder and Dura, 2019; Dechema, 2013; Kumar and Himabindu, 2019; Sanz-Pérez et al., 2016; Zeradjanin et al., 2021).We also need effective communications campaigns to inform the public so they can participate in identifying and adopting solutions.
Inspiration for the future
With such lofty goals for our energy future, it may be good to have some inspiration.
Quoting again from The Next Hundred Years, we find a description of the closed carbon cycle from 1957 that sounds remarkably similar to modern ideas: An ingenious system has recently been described for the production of power from algae grown in a closed system containing a high concentration of carbon dioxide. The algae are cultured and then fermented in such a way that methane and hydrogen are produced. These gases are burned in a gas turbine or engine which is used to generate electricity. The carbon dioxide which results from the combustion is returned to the algae culture unit. In this way, under ideal conditions, one would have a closed system which would convert between 1 per cent and 3 per cent of the incident solar energy to electricity (Brown et al., 1957, p. 105).
Surely, this “ingenious system” provided motivation for scientists and engineers of the 1950s and 60s. Looking to some of the more fanciful technological options now on the horizon, perhaps we can also find our own motivation for shaping the future. Options for closing the carbon cycle often reflect the processes driving life on Earth and are, not surprisingly, not only influenced by nature but also derive their names from their natural counterparts. They demonstrate that it is beneficial to look to nature for solutions.
Synthetic trees, for example, can be built and covered with artificial leaves performing artificial photosynthesis (photoelectrolysis) (De_Richter et al., 2013). Early artificial leaves split water photocatalytically to produce hydrogen using only sunlight (Fujishima and Honda, 1972). But they did not chemically reduce carbon. Some later devices were able to use water and light to directly reduce CO2 to hydrocarbons just like a leaf via photosynthesis (although the internal processes are different!). There are also efforts underway today to incorporate photocatalysts into biotechnology to create “bioelectrochemical” systems (Aresta and Dibenedetto, 2020). These technologies are attractive because they rely on charge transfer to split water and reduce carbon in an integrated step without the explicit generation of renewable electricity characteristic of today’s “green” hydrogen (Ganesh, 2014; Lewis, 2016; McKone et al., 2014; Ulmer et al., 2019).
Like a real leaf, the artificial leaves require a steady supply of water and carbon dioxide for continuous hydrocarbon production, while heat or (unlike a leaf) electricity can improve performance. These essentials can be provided by fastening the artificial leaves onto enormous stacks called solar updraft towers. The “synthetic trees” are several 100 m in radius at their base with a chimney that is about 5 m wide and several 100 m tall. A prototype was built in 1982 at Manzanares, Spain (Figure 4) (Schlaich et al., 2005).The solar radiation incident on the base of the tree can generate either electricity from solar panels or can be converted to heat. Because the base is situated above the ground, the heat causes thermal updrafts that suck in air from the perimeter and push it out through the chimney. Thus, the tree produces kinetic energy, which can drive turbines to generate more electricity or move air with 400 ppm CO2 through DAC units attached to the underside of the base. The swing capture and regeneration cycles of the DAC units can be powered with the electricity produced by the solar cells or turbines.

The solar updraft demonstration facility at Manzanares, Spain, built in 1982. Heat from solar radiation causes an updraft from the structure’s perimeter, which exits through the chimney. Image © sbp sonne.
In sum, the tree produces electricity and synthetic hydrocarbons using sunlight and atmospheric CO2. Only water must be supplied as a reducing agent because the trees will likely be situated in remote locations (Bockris, 1973; Goldberg and Lackner, 2015). It is a typical CCU energy conversion chain: synthetic trees capture electromagnet radiation and deliver electricity, heat, or energy stored in chemical bonds. These species of energy can be used where and when we please and may again be converted to other forms of energy. And the tree, or perhaps trees grouped together into an artificial forest, does all this within a closed carbon cycle by taking in CO2 from the atmosphere. Or at least it would if it were economical to build. Perhaps 1 day it will be. Until then, we can let ourselves be inspired by its integrated and responsive design.
Now, beginning with our scientific and technological ideas, we can begin to develop an inclusive vision–or visions–for the future. But the transformation of our energy landscape is dependent on more than just science and technology. More than ever, answers must now come from researchers and policy makers, from industry, and from outreach and communication programs. And from the public. Or, what will surely be more decisive, the answers must come from these groups working together in a candid and constructive way to ensure that new technologies are developed, accepted, and implemented as quickly as possible into a robust and effective future energy system. A successful Anthropocene is more than a question of future energy sources and biogeochemical dynamics in the context of planetary boundaries. It is also a question of resource (re)distribution, a safe and just political and social order, and robust education curriculums (Crutzen, 2002; Gupta et al., 2021; IEA, 2019b; Jones et al., 2017; Rockström et al., 2009, 2021; Schlögl, 2015; Sovacool et al., 2016; Steffen et al., 2015, 2018; Wich-Konrad et al., 2020).
The Anthropocene as a discontinuity
Efficiency, based on economic considerations, was the solution to the fossil-fueled expansion of the last two centuries. Now, however, we are faced with a two-dimensional problem of economic viability and environmental protection. The solution will also be multi-dimensional. The Earth-human interaction represents a complex dynamic, not only in its individual intricacies (tipping points), but, perhaps more urgently, in its feedback mechanisms (tipping cascades). Our task is to find a set of means and strategies for a steady, or preferably stable state of the Earth’s biosphere. Part of the solution will be a controlled equilibrium achieved by closing the carbon cycle. That is, our future prosperity will be within a managed steady state of the Earth-human system (Klose et al., 2021; Rockström et al., 2009, 2021; Slaveykova, 2019; Steffen et al., 2015, 2018; Wolff, 2011).
Beyond the more efficient use of resources, closing the carbon cycle means, in particular, that we find a way to deal with the components of human activity that cannot be electrified or decarbonized, at least not yet. Ships and long-distance aircraft will consume carbon-based fuels in the near term and wide swaths of industrial processes will continue to rely directly on carbon chemistry (Bender et al., 2018; Schlögl, 2017). Estimates of CCU to recycle and reduce carbon use are on the order of gigatons of CO2-eq. per year, or, by 2040, up to about 10% of current annual global emissions (including carbon directly avoided by the restructuring of industrial processes). The actual quantity of carbon that is recycled will depend on the expansion of non-fossil sources of electricity and on science-based policy incentives (Aresta and Dibenedetto, 2020; BMWi, 2020; Dechema, 2013; Geres et al., 2019; Mac Dowell et al., 2017; Song, 2006; Styring et al., 2011; Ueckerdt et al., 2021). One prognosis finds, for example, that exhausting the potential of CCU in the chemical industry alone will require between half and all the total electrical production planned for 2030 (Kätelhön et al., 2019).
Even without taking into account the decarbonization of energy generation, which will make the largest contribution to closing the carbon cycle, the reduction and reuse of carbon via CCU strategies (defossilization) can make the difference between the IPCC’s representative carbon pathways (RCP), at least in the short term (Schwalm et al., 2020). To reinforce Peter Styring’s earlier sentiment: we need to use all the tools available to reduce the implementation gap and close the bioatmospheric chemical cycles so that the climate remains within a habitable trajectory. In the twentieth century, the carbon cycle was not closed due to a lack of a large, cheap power source–without an adequate renewable energy infrastructure in the near future, history will repeat itself. If a circular carbon economy is achieved, newly-released carbon will have neither the isotope fingerprint of fossil carbon, nor of the pre-industrial atmosphere but rather, a gradually stabilizing average of the two. We will be able to follow the change as less carbon from fossil sources is emitted into the atmosphere and the new mixture of carbon isotopes leads to a flattening of the Suess effect. Apparent human impact will decrease.
If, on the other hand, our efforts fail and the bioatmospheric chemical cycles are not closed, we may reach a tipping point, or set of tipping points that knock the Earth into a climate unsuitable for human life. The Anthropocene, as we know it today, may turn out to be the lead-in to a geologic discontinuity that shares little with what stabilizes in the wake of the shift. Make no mistake, the Earth as a rocky planet will endure no matter what we do. If humans are gone, however, it will be a most ironic facet of the new geologic age.
Footnotes
Acknowledgements
The author would also like to thank two anonymous reviewers for engaging in a critical and exemplary peer-review process.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the German Federal Ministry of Education and Research (BMBF) through the project Carbon2Chem-2 L-KK: Koordination und Kommunikation (Förderzeichen: 03EW0003B) as well as the project WissKommEnergiewende. The author would also like to thank those who took the time to give feedback and suggestions: Prof. Andy Cundy (University of Southampton), Prof. Robert Schlögl (Max Planck Institute for Chemical Energy Conversion), Prof. Ursula Klein (Max Planck Institute for the History of Science), Prof. Jürgen Renn (Max Planck Institute for the History of Science), Prof. Jan Zalasiewicz (University of Leicester), Prof. Reiner Kümmel (University of Würzburg), and Dr. Axel Kleidon (Max Planck Institute for Biogeochemistry).
