Abstract
The paper discusses the CF-industries ammonia plant in Donaldsonville, Louisiana. The plant is framed as an exemplary site from which the Anthropocene can be observed and understood. In doing so, a proposal for a “chemical cultural theory” is set out, to allow us to understand such molecular planetary technologies and interpret their (geo)historical significance. As one of the largest fertilizer plants in the world in terms of its output, and one of the largest chemical plants along the “Petrochemical Corridor,” a cluster of chemical industries situated between Baton Rouge and New Orleans, Donaldsonville typifies the relations between the nitrogen and hydrocarbon industries. Catalysis is here used both as a chemical concept and as a metaphor central to the proposed chemical cultural theory. As key to the Haber-Bosch process and refinery technologies in general, investigating the role of catalysis allows us to connect the history of the Petrochemical Corridor to that of German industrialism. This relation reveals how, from the late 19th century through to the World Wars, an ambivalent industrial co-operation between the US and Germany not only transformed local and planetary environments, it also contributed to the Anthropocene condition.
Keywords
“When a chemist wishes to speed up a reaction, he has generally speaking, three ways to turn: he can increase the temperature; he can increase the pressure or concentration of the reaction materials; most useful of all, he can try to find a substance which will act as a ‘middleman’ to bring the reacting substances into the most intimate contact and thus facilitate their union or interaction. The middleman is called a catalyst.”
Prologue: A visit to the ammonia plant in Donaldsonville, Louisiana
In November 2019, participants of the “Anthropocene River Campus: The Human Delta” visited Donaldsonville. This small Louisianian town lies at Mississippi River mile 175, halfway between Baton Rouge and New Orleans, and is home to one of largest chemical factories in the United States. Here atmospheric nitrogen (N) and hydrogen (H) derived from natural gas are synthesized into ammonia (NH3) with the help of an activated iron catalyst called “ammonia iron,” using the Haber-Bosch process. The site, situated on the banks of the Mississippi River, consists of six ammonia plants, five urea plants, and four nitric acid plants. Owned by the 1946-founded parent company CF Industries, Donaldsonville’s plant is considered one of the largest ammonia producing complexes in the US and the third largest in the world, processing 1.55 megatons of nitrogen per year (Smil, 2001: 125, see Figure 1).
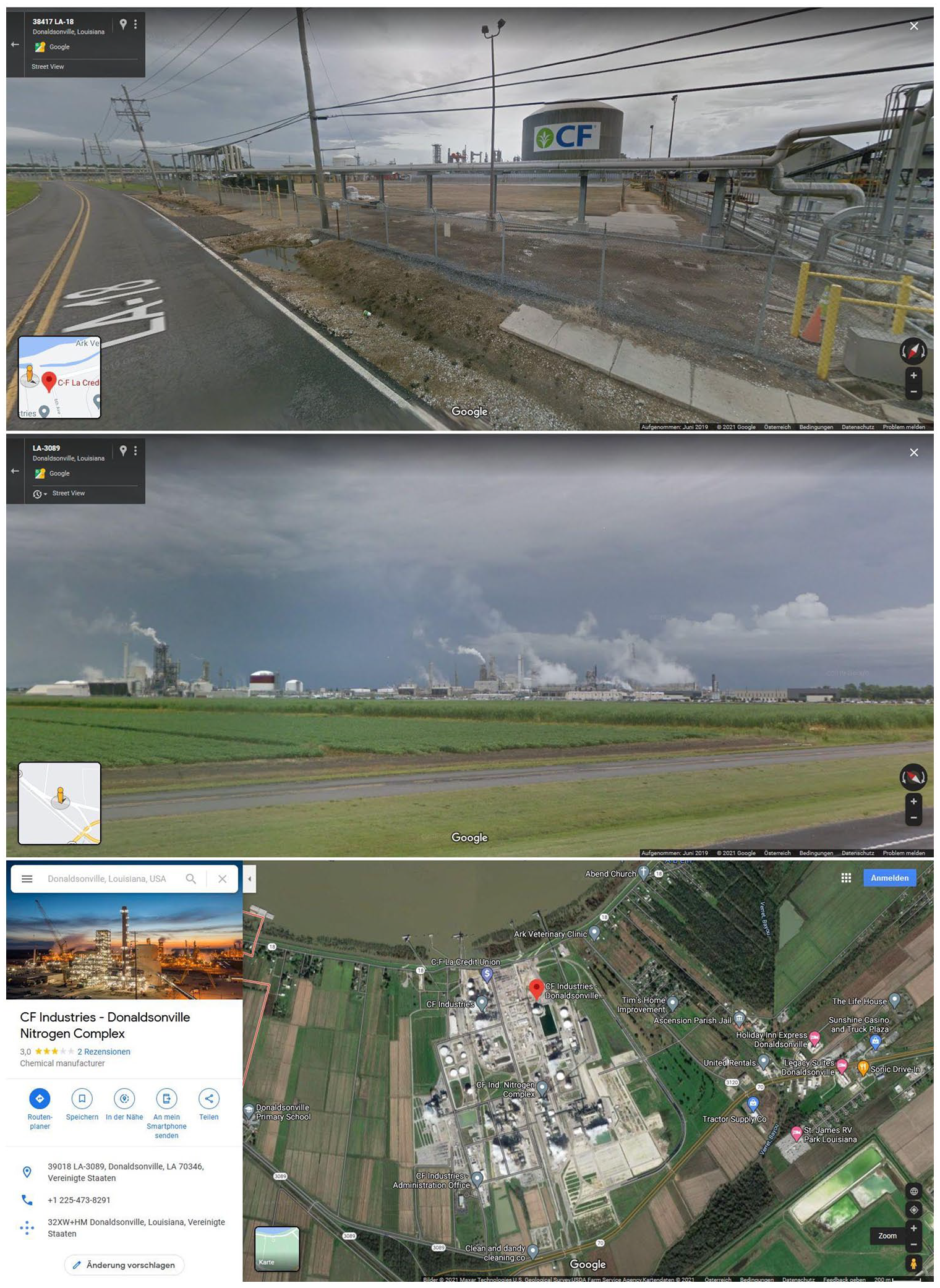
“A Google street view of CF Industries reveals almost nothing about these institutions” (after Bertolt Brecht).
Our excursion to the Petrochemical Corridor was both impressive and vacuous. The plant loomed large from the bus, but the chemical technology and its planetary impact remained invisible due to the strictures of corporate secrecy. This was despite a number of concerted attempts from Campus organizers to arrange a tour of the facility through correspondence with CF Industries, Director of Corporate Communications. As German playwright Brecht (1988) famously noted of such facilities, “a photograph of the Krupp works or AEG reveals almost nothing about these institutions” (p. 469). Even an in-person visit to such a site would have probably failed to offer further insights into what it is that plants like Donaldsonville actually do.
This essay addresses the CF Industries’ nitrogen plant in Donaldsonville as an exemplary site from which to discern the Anthropocene. This industrial facility is cast as planetary reactor, a place in which chemical reactions take place combining molecules from the lithosphere and the atmosphere. The resulting “chemifertilizers” are then shipped up- and downriver, to be applied to the soils of various biospheres across Earth’s surface. In order to understand the plant and the extended geography of the petrochemical corridor, both the technical principles of these sites and the various layers of history of technology, science, and politics which lie behind this technology need to be revealed.
Towards a chemical cultural theory of the Anthropocene
Ammonia synthesis is one of the most important industrial inventions of the 20th century. The abundance of food and agricultural products that the resulting artificial fertilizers have created have allowed the planet’s population to grow far beyond the given limits of an organic economy. As geographer Smil (2001) puts it, since the point of this discovery, we have been “enriching the Earth.” It is this demographic growth, in turn, which lies behind those phenomena that constitute the “Great Acceleration” of various throughputs of energy and resources within the Earth system, which since the mid-20th century, have become characteristic of the Anthropocene (Steffen et al., 2015). Well-known drivers—including urbanization, land use change, biodiversity loss, and economic growth, to name just a few—have been dramatically accelerated by the production of synthetic fertilizers. But agriculture is only one part of this product’s violent impacts. Ammonia synthesis has also been crucial for ammunition production since the beginning of World War I (Friedrich et al., 2017). Moreover, as a technological platform, the development of high-pressure reactors and artificial catalysts for ammonia synthesis helped further open up the productive scope of the petrochemical industries. Products like refined fuels, plastics, and pharmaceuticals affect all aspects of modern human life and become artifacts of politics, economy, health, transportation, mobility, and consumerism, enfolding within planetary systems such as the biosphere, soils, the hydrosphere, and the atmosphere (Williams et al., 2015). In the 1950s, ammonia synthesis also truly became a part of the petrochemical complex because it began to make use of hydrogen derived from natural gas. Such petro-chemical technology evolved into a crucial agent of planetary change and a necessary subject for Anthropocene scholarship.
Chemical technologies have planetary effects. Or put differently, technical interventions on the level of chemical processes affect planetary and geohistorical processes. With the rise of the chemical industries, matter has itself become a technical product. As they are neither more or less naturally given materials like plants, wood, stone, or natural fiber, nor products of those chemical technologies that emerged largely via trial-and-error such as steel or ceramics, these technologies are rather products of an explicitly molecularly informed industry. In the 20th century, molecular chemical technologies became central to historical processes of change: These “science-fashioned molecules for top performance,” as a Standard Oil film advertisement put it in 1946, became historical actors (Fairbanks (Jerry) Inc., 1946).
Both the fact that the chemical industry redefined the boundaries between technology and nature, and that its rise is marked by both broad and intimate connections to the political history of the 20th century qualify this topic as exemplary for developing an understanding of Anthropocene history and theory. In fact, this shift in the technological realm touches upon a central interest of cultural theory, here understood as the focus on the versatile borderlines and zones of encounter that occur between culture and nature, be it in bodies, societies, geographies, or materials. In the Anthropocene, an age in which the boundaries between natural and technological processes have become blurred, reflection upon these boundaries is of great importance.
Materials provide explicit zones of encounter between nature and technology. Despite the long tradition of correlating historical epochs with materials since Greek poet Hesiod (around 750 and 650 BCE), who identified past “Golden,” “Silver,” and “Iron” Ages, and the long tradition of materialism in philosophical thinking (Lange, 1950), materials have drawn comparatively little scholarly interest in cultural theory. Since the 19th century, “historical materialism,” and thus the political economy of “materials” became crucial for the socialist and communist movements and thus for the political history of modernity (Bloch, 1985). Despite this prominent political role and subsequent so-called “material turns” in various epistemological schools, the attention has often touched more upon things, or objects, rather than on base materials themselves (Ingold, 2012). Studies of substances are booming, but philosophically informed approaches such as Jens Soentgen’s programmatic books, on carbon dioxide, nitrogen, and others (Ertl and Soentgen, 2015; Soentgen, 2019; Soentgen and Reller, 2009), and moves to address the agency of material things, such as Bennett’s (2010) neo-vitalist notion of “vibrant matter” remain exceptions.
This essay contributes to this agenda, and proposes the need for a “chemical cultural theory.” In doing so, it is a similar intervention as that of geographer Barry’s (2017) programmatic manifesto for a “chemical geography.” Barry’s research agenda proposes to study the concrete distribution and dissipation of chemical substances in landscapes and cultures to detect their enfoldment with mechanisms of power. “Chemical cultural theory” enters this same domain but with a different programmatic intent. Its seeks to observe the specific technicalities of modern materiality and the historical settings that enabled this.
For cultural theorists and historians, this agenda is intended to give insight into a material and industrial sphere, an arena that deserves more than just specialists’ attention. And for Anthropocene scholarship, it adds a necessary perspective on the epoch as an age of industrially produced artificial molecules. Chemical substances and their production sites appear as switching points in the dynamics of the age. They are agents of technology and history and they are products of technology and history. Bottom-up and top-down dynamics are both relevant, and furthermore unfold on the background of a convoluted history of the understanding of the entanglement between microworld and macroworld, from Greek atomism, via early modern speculations about atoms and molecules, the later opposition to atomism even by leading pioneers of modern chemistry such as Wilhelm Ostwald, to the broad acceptance of microscopic theories of matter in the early twentieth century. Still, chemical ontology cannot be reduced to reductionist assumptions about a microworld of atoms and molecules. It is not only the case that developments in the understanding of such a microworld enabled novel macro-historic processes, but that such processes enabled in turn chemical ontology the microsphere to be understood in new ways (Klein and Lefèvre, 2007). Chemical industry has been the historical result of an interplay of all layers of history: politics, the sciences, geopolitics, and economics. At the same time, these layers are affected by the molecular products of this industry. History both enters molecules and emanates from them.
As an academic endeavor, “chemical cultural theory” is both based on and expands materialist approaches in cultural theory, epistemology, and media theory. In particular, German media theory such as the work of Friedrich Kittler, Hans-Ulrich Gumbrecht, and others have established a strong focus on materiality and on the history of technology since the 1980s (Berz, 2001; Gumbrecht and Pfeiffer, 1994; Kittler, 1990, 1997, 1999, 2014; Siegert, 1999; Vismann, 2000). These scholars, it has been argued, have a similarity to approaches in epistemology, such as that of Rheinberger (1997, 2010), particularly with regard to the need to investigate the material basis of culture and science for an understanding of specifically modern thought. Only by addressing the historical, material, and media-technical conditions of science can a more comprehensive philosophical interpretation of modern and present subjectivity be pursued. One of the basic terms for these approaches is Foucault’s (2002) concept of a “historical a priori” (Chapter III, p. 5) which qualifies all knowledge as fundamentally historical. Media theorists followed and extended this figure of thought by arguing that the “media a priori,” the media-technological conditions of history, also need to be investigated to interpret their respective epistemological settings (Engell and Vogl, 2001: 6). These approaches allow a programmatic shift from the study of the materiality of technical media to the study of the mediality (the mediating and defining role) of materials and the technical materiality of history. This leads from a focus on “modernity” as a distinct epoch in human history, to that of the “Anthropocene” as a special epoch in Earth history. The epistemological interest of “chemical cultural theory” therefore aligns with the general goals of cultural theory: to investigate human culture as a historically evolved, complex, and often contradictory totality. Such study is carried out via the analysis of material artifacts and abstract concepts, from infrastructures and institutions to belief systems, from the real to the imaginary, with all their mechanisms of effect.
Chemical cultural theory doesn’t follow the money, but the molecules. It recognizes that chemical technology is crucial for the complex “matter and energy” which, just like “information”—the classical subject of media theory—is fundamental to all abstract aspects of human activity. A typewritten note by Nietzsche (1986) from 1882 serves as a powerful slogan for media theory, “You are right—Our writing utensils work on our thoughts” (p. 172). It can also be stated that our fuels also work on our thoughts. Investigating the “chemical a priori” as an extension of both the historical and the “media a priori” (Engell and Vogl, 2001: 6) would combine geographical and historical but also philosophical approaches, and synthesize insights into infrastructures and landscapes (Steininger and Klose, 2020) with a yet to be written critique of “fossil reason” (Steininger and Klose, 2018).
Catalysis as a key principle of Anthropocene materiality
Catalysis marks a crucial point for reflecting the technicality typical of Anthropocene materiality, from the cascades of molecular to planetary accelerations, and for the types of history that have played a role in establishing their respective industries (Steininger, 2008, 2013, 2014, 2018). Following catalysis through industrial and historical processes offers a model case for what chemical cultural history and theory can contribute to Anthropocene scholarship. Moreover, focusing on catalysis can address pars pro toto the principles of industrial chemistry which have proved so central to such dramatic shifts in terrestrial composition.
Catalysts are substances which accelerate and control chemical reactions. The principle is ubiquitous in biochemistry, and was central to the beginning of life on Earth and the geohistorical shift from an inorganic to organic geochemistry. Only with the help of metal catalysts was the complex chemistry of cells and their respective metabolisms able to emerge some 3.5 billion years ago (Belmonte and Mansy, 2016). Complex processes in cells are only possible because biocatalysts steer their reactions. However, the history of catalysis as a science did not begin until the 19th century. In 1835 Swedish chemist Berzelius (1835) noticed a common feature in biochemistry and some inorganic reactions. In these reactions there was no numerical relation between the reaction partners and other agents such as platinum, or an organic ferment which contributed to the reaction. Unlike more normal chemical reactions where the ratio of elements involved can, following the laws of stoichiometry, be easily written down with small integers, the agency of catalysts falls below scales of measurement. Platinum, for example, even if present only in tiny amounts, is able to ignite oxyhydrogen gas, as the German chemist—and friend of Goethe—Johann Wolfgang Döbereiner first showed in 1823 in Weimar. Berzelius (1835: 245) named this type of chemical reaction with an artificial Swedish-Greek word, formed according to the word “analysis”: “Katalys.”
Following Berzelius has been a struggle for a precise understanding of catalysis which continues to this day. It was only at the end of the nineteenth century that chemical reactions became accessible to thermodynamic calculations and catalysis increasingly turned into an industrial tool (Baracca, et al., 1979). Ostwald, professor for physical chemistry in Leipzig, used the principles of thermodynamics to measure energetic equilibria and reaction kinetics and in 1894 redefined the catalyst accordingly: “Catalysis is the acceleration of a slow chemical process through the presence of a foreign substance” that is not part of the product of the reaction (1894: 706). This definition foreshadows the key modernist and Anthropocene concept of acceleration in chemical science and industry. What had first been seen as a somewhat inexplicable chemical miracle already around 1900 became the subject of systematic experimentation and industrial probity. A whole school of new catalysis research was established around Ostwald. Even today, catalysis research, as it has branched into fields such as surface and enzyme chemistry, remains one of the most dynamic areas of research and application in chemistry (Schlögl, 2017). Less than two decades after his definition, the laudatory 1909 speech recognizing Ostwald’s Nobel prize noted that “key sections of industry such as sulfuric acid manufacture, the basis of practically the whole chemical industry, and the manufacture of indigo [. . .] [had become] based on the action of catalysts” (Nobel Foundation, 1966: 148). Although, Ostwald himself would not have admitted it because of his skeptical stance towards the existence of a microworld, molecular mobilization had begun to take command of the material world.
Ammonia synthesis
Since the 19th century, the German chemical industry had been marked by its successes in creating substitute substances. As a “belated nation,” whose comparatively late formation in 1871 had afforded comparatively few colonial possessions, Germany made use of innovative chemical substitutions, particularly involving coal, to create resources rather than seizing them from others. Wissenschaft bricht Monopole: Der Forscherkampf um neue Rohstoffe und neuen Lebensraum (Science breaks monopolies: The explorers’ battle for new raw materials and living-space) was a telling book title which describes this national attitude of aggressive autarchy (Zischka, 1936). In fact, the self-image of a nation without resources but with a strong chemical industry is still present today (Soentgen, 2019: 131–134). The most paradigmatic colonial resource, sugar, was replaced, from the 19th century onward, by the industrial combination of domestic beet plants and fossil energy. Products in which coal was both used as a material and energy resource were Germany’s industrial substitutes for the dye indigo, later rubber, and even crude oil. These substitutes transformed global markets and material flows. Around 1910, artificial ammonia and saltpeter mark a crucial point where the catalytic manipulation of chemical processes began to interfere with the processes of global history. The development of ammonia synthesis began as a specifically German science and industry. In Leipzig in the 1890s, Ostwald, who held a chair of one of the first institutes for physical chemistry in the world, identified the idea of the catalyst as an accelerator. If it was the German chemists Fritz Haber and Walter Nernst who first argued about the complex thermodynamics of ammonia synthesis reactions, from the beginning, ammonia synthesis was recognizably an act of global history making and a topic of planetary concern (Renn et al., 2017, Johnson, 2021).
Transforming atmospheric nitrogen into NH3 is a planetary project, not least because this molecular technology itself emerged from geostrategic questions of planetary proportions. Chemists such as William Crookes in England and Ostwald had made prominent warnings about possible supply bottlenecks of saltpeter, an agricultural and military necessity for the industrial North, both for its great demand and as it came from far and strategically difficult to access, specifically Chile in the Southern Hemisphere (Crookes, 1899; Ostwald, 1904). While Crookes’s warning had focused upon the fate of wheat-eating Europeans and North Americans, Ostwald’s discussed the threat that Chilean saltpeter might be blocked by the British Navy as a means to restrict the growing armory of the German military.
Chemical operations solved these geopolitical concerns. In just a few years, an industry emerged that could be described as both chemical-molecular and strategic-planetary. In order to fix atmospheric nitrogen to ammonia molecules, innovations were being developed which went far beyond the nitrate chemistry of ammonia and which ultimately formed the basis for a catalytic chemistry of hydrocarbons. The chemical elements nitrogen and hydrogen were combined in a high-pressure apparatus which could operate at temperatures and pressures that were not previously thought possible. Nitrogen could be relatively easily produced by the “Linde” process (named after German inventor Carl von Linde), in which air was cooled and the liquified gases could be separated again by their respective boiling points. A procedure to produce high amounts of hydrogen was found by the “Wasserstoffkontaktverfahren,” a catalytic process that used water gas and coke-oven gas to produce pure hydrogen (Smil, 2001: 97). A crucial element was the development of artificial catalyst masses. At the laboratory scale, in 1909 German chemist Fritz Haber had already managed to synthesize ammonia using a catalyst from the light bulb industry, the filament material osmium. But there were only 40 kilograms of osmium available on the entire world market. Following tens of thousands of experiments, Alwin Mittasch (1953), head of the catalysis laboratories at chemical company Badische Anilin und Soda Fabrik (BASF), developed a multi-component catalyst made of iron oxide, with additives from alumina (Al2O3) and potash (K2O). This was the innovation that would play a role in changing the mechanisms of the planet, and it was based on insights into the deep mechanisms of chemical engineering and on second and third order chemical reactions which involved not only one catalytic substance, but a system of several chemical elements in the catalyst’s mass. BASF had experience with this type of chemical technology. In the 1890s the company developed the so called “contact process” to produce sulfuric acid with the help of platinum catalysts. This could only work as a result of understanding and taming so-called “catalyst poisons,” substances such as arsenic that weaken catalytic activity. Profiting from this research experience, second order substances like catalyst poisons and catalyst promotors opened a new agenda.
Mittasch’s multi-component catalyst marked a further shift from natural to artificial materiality on several levels. Of course, historically such shifts have occurred since Neolithic times, from ceramic via metallurgic technologies to the production of beer and wine by fermentation. But in the context of chemical industry, the shift took on a new dimension. To replace the naturally occurring substance saltpeter, the naturally occurring chemical element osmium needed to be substituted by an artificial mixed-material. Only with this second-order artificial material did the first order technicality of fertilizers and food abundance come within reach. Only via this specific manipulation on the microsphere was the manipulation of the macrosphere possible. In retrospect, this innovation marks one of the most important steps into the Great Acceleration and into the Anthropocene. Even still, the science is not yet closed: the exact mechanism by which this mixed material catalyst works in ammonia synthesis remains under investigation. The 2007 Nobel prize for chemistry, awarded to Ertl (2008), recognized this ongoing work.
By 1914 the chemical innovation of synthetic ammonia was already making history: ammonia, with just one additional catalytic step (the “Ostwald-Process”), meant saltpeter could be produced artificially in the German armament factories of Oppau and Ludwigshafen. The abundance of explosive material they generated meant the First World War could last 4 years, with all its bloody consequences (Szöllösi-Janze, 2000; Friedrich et al., 2017). That which had been synthesized under high-pressures and high-temperatures was then discharged on Europe’s battlefields with similar force. Without ammonia synthesis, the war could have been over for Germany after a few months due to a lack of adequate saltpeter stockpiles. Haber himself was promoted to work as a middleman between the chemical industry and the military to help establish the necessary industrial facilities in record time, such as the Leuna plant (Szöllösi-Janze, 2000).
The militaristic impacts of the First World War were one of the first planetary effects of this chemical technology. No less important was the longer-term transformation of global agriculture by synthetic fertilizers. Capacities that had been developed during the war for ammunition manufacturing soon made Weimar Germany a net exporter and major player in the artificial fertilizer market (Szöllösi-Janze, 2000). And as a result of the Versailles treaty, Germany’s Haber-Bosch patents became a global scientific good, helping to globalize this scientific breakthrough. Ammonia factories were licensed in France, the United Kingdom, and the US. Retrospectively, German wartime production of 96,000 tons of fixed nitrogen in 1918 seems small compared to the 16,250,000 tons produced in the US in 1997. Global production—also accounted for by the mass of fixed nitrogen produced per year, a quantity which increased from 150,000 tons in 1920 to 85,000,000 in 2000 (Smil, 2001: 242–245; Friedrich, 2017).
A complex technological ensemble had been established: it combined feedstock methods such as catalytical hydrogen production with high-pressure technologies and multi-component catalysts. Such complexes opened up industrial science to further opportunities, first for the German chemical industry, and then, from 1920 onward, for global petrochemistry. In interpreting this industrial and scientific evolution, the move from ammonia industry to petrochemistry, and the respective political and societal effects, historian of technology Hughes (1969) coined the concept of the “technological momentum” to describe the dynamism of the determinate forces that had been unleashed.
The German petrochemical coast
As early as in 1913, the high-pressure technologies and catalysts were used at BASF in Ludwigshafen to experimentally synthesize various hydrocarbons from carbon monoxide (CO) and hydrogen (H) (Farbenindustrie, 1927). Synthetic hydrocarbons appeared as the logical next step in the evolution of industrial chemistry. In the early 1920s, the first industrial product of the procedure was methanol (CH3OH), which was widely used as an industrial solvent. The technological apparatuses required to create synthetic hydrocarbons very much resembled the infrastructures of Haber-Bosch, but the role of the catalyst had changed. Catalysts evolved from a device to accelerate the reaction rate to a tool to steer reactions, and to decide what particular hydrocarbon molecules would be synthesized from one and the same set of feedstocks. Like building blocks, hydrogen and carbon atoms could now be arranged in desired combinations.
A whole new branch of industrial chemistry was about to be established: petrochemistry. Fuels, lubricants, plastics, fibers, and solvents were all “science-fashioned” hydrocarbons; these materials would become important chemical actors in the Great Acceleration (Steininger and Klose, 2020). Petrochemistry forms a key subject for a chemical cultural theory of the Anthropocene. With these hydrocarbons, not only a new set of raw materials became the subject of industrial catalysis, but a new aspect of the dynamics of catalytic processes became historically crucial for science and technology. With that step, the chemical industry became a factor within a whole new set of political and geostrategic issues. The interdependencies of chemistry and politics require interpretation in both directions. Chemistry enables political and economic power, but it can only do so because of the geostrategic setting and power structures that have afforded the means to establish such chemical tools. The complex of industrial petrochemistry, with all its impact on the dynamics of the Anthropocene, is built from the dynamics of all these interdependent scales.
The following chapter of this history gives insight into the establishment of petrochemistry by presenting the complex and occasional abysmal cooperation between German chemical industry and the US oil industry from the 1920s until the 1940s. Nestled in the Louisianan petrochemical corridor, interdependencies between the German and US chemical industries appear numerous, so much so that North American historians of chemistry sometimes have to counter the wrong impression that US industrial chemistry was predominantly a German import (Haynes, 1945: xiii). In the region south of Baton Rouge, the chemical industry remains situated at places named after German towns such as Geismar and is clearly built on a longer history of German regional influence. This area had been called the “German Coast” since the 18th century, as a number of Germans had settled in the area and even ran plantations in sparsely populated areas, granting them German names like Haydel or Augsburg. Compared to the history of French settlement in the region, the German influence upon the area is comparatively understudied (Blume, 1990; Le Conte and Conrad, 1967).
In 1920, the German chemical industry and US oil business knew relatively little about each other. Their first close encounter was not even triggered by oil. In 1923 the head of BASF, Carl Bosch, travelled to the United States (Holdermann, 1953: 223–225). Bosch was already world-famous as a chemist and mechanical engineer. In the US it was not oil but a lead compound which had caught his attention: Tetra-Ethyl-Lead (TEL). Shortly before that in 1921, Charles Kettering, a bustling and legendary inventor in many fields of automobilism who was at that time head of General Motors Research Corporation, demonstrated, together with his collaborators Thomas Midgley and Thomas A. Boyd, that tiny quantities of this additive could decisively improve the combustion of motor fuels (Boyd, 1957). It was found that just “one part of lead-tetra-ethyl in 1500 parts of petrol” could reduce the inefficiencies of engine knocking, and could reputedly helping to conserve gasoline (Egerton, 1928). Carl Bosch—himself an inventor—immediately recognized that such molecules had enormous potential as a kind of second-order fuel. He immediately started a BASF program to test a range of similar fuel additives, such as iron carbonyl, and a rig for testing engine fuels was developed.
Tetraethyl lead was an industrial development that would trigger interest in developing liquid gasoline from domestic coal, a potential geostrategic gain for a country such as Germany without its own oil resources. Alongside his fellow chemist Friedrich Bergius, Carl Bosch would win the Nobel Prize for Chemistry in 1931 for the invention, of chemical high-pressure methods, which were crucial for the establishment of global modern petrochemistry. The branches of nitrogen chemistry from Haber-Bosch, its extension into coal-based hydrocarbon chemistry, and oil chemistry were about to merge. In documenting the industrial history of I.G. Farben and the extension of this technique into the domain of petrochemistry, the historian Hughes (1969) provided further evidence for the concept of “technological momentum.”
In 1926, Frank Howard, North American research director of Standard Oil of New Jersey, was invited to Ludwigshafen. He was thrilled. “I was plunged into a world of research and development on a gigantic scale such as I had never seen” (Howard, 1947: 13). He was shown BASF’s attempts to produce liquid hydrocarbons from solid coal under high-pressure and with the help of catalysts. This procedure seemed able to not only liquify coal but also convert inferior crudes into the more desired fuels of gasoline and kerosene. He stated: “It seemed clear that the German hydrogenation processes, and the new horizons they opened, were tremendously significant—perhaps more significant than any technical factor ever introduced into the oil industry up to this time” (Howard, 1947: 17).
The frontiers of the industrial macrosphere were about to be transformed by a very significant new step in the catalytic manipulation of the molecular sphere. Compared to ammonia synthesis, the requirements in coal chemistry were even more complex. Sulfur, which is inextricably present in both coal and crude, acts as a strong catalyst poison. But the technology of multi-material artificial catalysts offered a solution. Discovered in 1924 by BASF chemists led by Matthias Pier, a multi-material iron sulphide catalyst (Fe2S3) was found to be resistant to sulfur poisoning, and has been described as “one of the most important breakthroughs in the study of catalysis and especially in high-pressure coal hydrogenation” (Stranges, 2000: 174). This was the first of a long list of innovative multi-compound catalysts, where the poison—sulfur—was integrated as sulfide into the catalyst, so that sulfur from the raw material would no longer harm the industrial process (Stranges, 2000: 174–195). The implications of this trick for a theory of Anthropocene technical materiality are compelling: high performance gasoline from coal and inferior grades of crude oil became possible because of an anthropogenic catalyst. In fact, these catalysts can even be described as dialectical chemical tools: a functioning industrial catalyst results from a dialectical integration of opposing chemical actors into a synthetic tool. Like a double agent, the raw material’s catalyst poison is re-applied and is used as a tool that works for, rather than against, industrial goals.
These encounters between humans, ancient fossilized organic material, and chemical substances had significant historical consequences. At an institutional level, these relations would go on to encourage close cooperation between I.G. Farben (the company which had absorbed BASF in 1925) and Standard Oil. An agreement committing both companies to continued collaboration was signed in 1929. This intervention from the spheres of big business was crucial for the implementation and upscaling of experimental findings within the industrial process. In fact, it was this cooperation, and the American capital which was invested in it, which made the German project of a coal-based liquid fuel industrially feasible. Without this collaboration, it is believed that the high costs of such a complex endeavor would have killed the project had it been carried out by I.G. Farben alone (Abelshauser, 2002: 241).
The joint laboratories of I.G. Farben and Standard Oil were built in Baton Rouge (Abelshauser, 2002: 240; Stranges, 2000: 188–190). The city became something like a hub between the German and US chemical industries which led to close personal connections (Morris, 1985). In 1931 a hydrogen plant licensed by BASF was built in Baton Rouge (Smil, 2001: 113). In 1938 eight companies (Standard Oil of New Jersey, Standard Oil of Indiana, Texas Co., Shell, Anglo-Iranian, M. W. Kellogg, UOP, and I. G. Farben) founded the Catalytic Research Associations (CRA) in order to develop a new way of cracking crude oil (Fletcher, 2008). Through the CRA not only did the long-standing catalysis expertise of I.G. Farben become shared knowledge, but also the principle of a fluidized bed, a technology first developed by BASF in 1921 in which a solid catalyst is pumped through the reactor in the form of a fine powder to catalytically crack crude oil (Winnacker and Küchler, 1971: 228). As a result, in 1942 the first fluid bed reactor was built in Baton Rouge (Peterson, 2000: 41), which is now recognized as a National Chemical Landmark. With large facilities to produce aviation fuels, tetraethyl lead, and synthetic rubber—another product of German-American cooperation (Morris, 1985)—the city became a sprawling center for catalytic chemistry and the chemical industries that fueled the war.
The historical and political ambivalences of this collaboration were clear. Standard Oil was one prominent partner of I.G. Farben; the other was the Nationalsozialistische Deutsche Arbeiterpartei (NSDAP, National Socialist German Workers’ Party), who had embraced the military potential of coal gasoline. With the so-called “Benzinvertrag” or gasoline treaty (Onderka, 1997: 8), from 1933 onward the German NS government imposed a political price on I.G. Farben’s coal gasoline, and the company became a close industrial ally of this fascist leadership, irrespective of Carl Bosch’s personal opposition to Nazism (Hayes, 2000). It was not until as late as 1940 that international cooperative bodies such as the Catalytic Research Associates (CRA) and the Joint Study Company (Jasco), crucial to developing the fluid-bed technologies central to the region’s innovative petrochemistry, stopped cooperating with I.G. Farben: a company which from 1941 onward had even built plants at the Auschwitz concentration camp (Hayes, 1987, 2000; Wagner, 2000). The Italian chemist and author Primo Levi wrote about his experience as a prisoner at the laboratory (Levi, 2008).
The disturbing circumstances of this cooperation arise from bizarre details and historical twists. North American money had enabled a German chemical company to produce coal gasoline and to wage a war against its donor; this conflict was eventually won, in part as a result of US aviation gasoline, which itself was produced by help of a decade long and close cooperation with its German industrial partner. Even the Mississippi estuary became a place of conflict. The fuel tanker Virginia was sunk less than 8 kilometers from the Gulf coast on May 12, 1942 by a German submarine, U-507 (List of Shipwrecks in May 1942, 2020).
Another bizarre detail of the political material entanglement is that Frank Howard, as a collaborator who connected the two industrial powers, lost his position as research director of Standard Oil of New Jersey in 1945. Allegedly this was due to his overseeing a large shipment of tetraethyl lead (TEL) to Germany, which he was said to have arranged in 1939 in the capacity of his additional function as director of Ethyl, the leading producer of TEL (Morris, 1985: 34). The substance, potentially a vital input into the German war machine, marked the beginning of one important industrial chapter of the region’s history, and closed a biographical one.
For the perspective of Anthropocene scholarship and chemical cultural theory, this historical narrative shows how different and temporarily even opposing agents were brought together to establish petrochemistry as a planetary technology. German coal chemistry and the US-oil business were in the first place global competitors on the fuel market. As shown, they became close collaborators and then wartime enemies, both using technology they had developed together (Howard, 1947, Stranges, 2000). As is well known, German and North American societies became allies and scientific collaborators once more after the war—at least with regard to West Germany. When in 1945 the United States Strategic Bombing Survey (USSBS, 1976) carried out research on the effects of aerial warfare on the chemical industries and compiled the report “Oil Division” about the fuel sector of German industry, US chemists and reporters spoke of old German colleagues as “friends” (Morris, 1985: 28). The further evolution of petrochemistry shows how opposing agents contributed to the establishment of a whole new technology, helping establish the post war reign of “hydrocarbon-man” (Yergin, 2008: Chapter 27) and the era of the Great Acceleration.
Fertilizer plants in the petrochemical corridor
The molecular technology of catalysis was a necessary condition for the planetary dynamics of the Great Acceleration. At the same time, it took concrete plants in concrete landscapes in concrete societal circumstances to produce these artificial molecules; The Petrochemical Corridor between Baton Rouge and New Orleans is such a concrete landscape. With about a 150 chemical plants, the area appears as an industrial super-organism. It processes every layer of the geography and of the history of the region. Placed at the banks of one of the biggest waterway systems in the world, it makes use of the fluvial geography of the Mississippi river as a transport system, as a provider of a thermal sink for cooling and water for the generation of steam, and, from the very beginning, as a tremendous industrial sewer system. The surrounding industries use all types of underground raw materials from the region, such as oil and gas but also sulfur from large salt domes, that once fully exploited serve as vast tanks for the storage of hydrocarbons. These materials, forming aspects of the environmental geography of the region, are then put into the process landscape of interconnected chemical plants. It is in such an interdependent system, rather than in single units, that the processing of chemical molecules is possible. Pipeline networks for gas and oil, and also for many other pumpable substances such as hydrogen, nitrogen, oxygen, ethylene, and numerous other hydrocarbons and refined products, alongside rail and shipping routes, connect the plants as the vital organs of this industrial super-organism. This organismic idea is echoed in the USSBS (1976) report, which described the German chemical industry as “a vast interlinking complex, so that attacking them was like fighting an octopus” (p. 1). The core units of its physiology are the chemical reactors, where fossil or other substances are transformed, largely if not always, with the help of chemical catalysts.
What was once “nature” when it abounded in the atmosphere (nitrogen), or in reservoirs of rocky matter (carbon, hydrogen, sulfur), at the reactor becomes a product of technology. A further systemic repositioning of these artificial molecules is, in most cases, achieved by processes of dissipation, or their proliferation and interaction with a wide variety of other material spheres. In the case of fertilizers artificial molecules are spread predominantly within the biosphere to make organisms grow in the planned way, but in the Anthropocene biosphere, the effects exceed the planner’s intentions (Williams et al., 2015).
With 16 “world scale” plants, the petrochemical corridor remains one of the major producers of fertilizers in the world (Peterson, 2000: 133). The region shows how artificial molecules have been concretely produced in the Anthropocene. Louisiana’s ammonia synthesis industries were mainly established around the 1960s. Plants were built at Luling (1954), Geismar (1962), Hahnville (1964), Taft, (1965), New Orleans (1966) Faustina, (1967), Plaquemine (1971), and in various steps in Donaldsonville (1966, 1968, 1975) (Peterson, 2000: 136). Given the early history of Haber-Bosch technology, which began in 1909, these developments came quite late. They also seem late compared to the first hydrocarbon refinery developments, which began in 1909 at Baton Rouge, and in 1916 at Norco, part of St. Charles Parish, Louisiana. But the plants built from the 1950s onwards were signs of a global boom in demand for ammonia fertilizers, with large plants also built in the Soviet Union, China, and Indonesia (Smil, 2001: 124). Managed by less chemical than engineering companies, specifically M. W. Kellogg, the plants deployed newly developed process technologies. The chemistry and the reaction process of high-pressure synthesis remained the same, but so called “single-train plants” used a central centrifugal compression for all high-pressure purposes and could produce higher outputs at lower energy consumption and costs (Smil, 2001: 123–127).
On the material level this boom marked an important shift. Whereas the early Haber-Bosch plants had emerged from flows of coal energy and worked with hydrogen from so called “water gas” (C + H2O → CO + H2) and thus from water, this generation of plants uses hydrogen from natural gas. Since the 1920s, BASF chemists had experimented with reformation of methane with a nickel catalyst, and in 1931 I.G. Farben licensed a plant to use this natural gas procedure. This plant was built at the Baton Rouge-based Standard Oil refinery of Baton-Rouge (Smil, 2001: 113). When large natural gas deposits were discovered in Louisiana in the 1960s, the region became the perfect place to produce fertilizers. Natural gas from the region could be transformed into a product which could then be easily shipped to some of Earth’s most abundant agricultural land, just upriver in the Midwest. This kind of fertilizer production became closely connected to the petrochemical industry thanks to the historical evolution of catalysis industries at Baton Rouge, with all their intercontinental and cross-industry connections. Fertilizer plants became part of the material flows of petrochemicals and natural gas derived hydrogen, occupying a new role in the Anthropocene’s bio- and techno-spheres.
Natural physical resources, such as waterways and hydrocarbon deposits, play an important role in the location of the chemical industry in Lower Louisiana. But they are not the only reason for the geography of this industrial agglomeration. Societal, and in fact, highly problematic factors play a role in how companies produce here. In this case it is not strong structures such as the mighty river or thick sediments of oil and gas but, on the contrary, weak and systemically weakened regulatory structures that benefit the industry. As historians Colten (2012) and Allen (2006) have shown, weak pollution regulations and tax-exemption programs have helped foster the region’s world-changing industrial expansion, particularly between 1940 and 1970. Contamination by huge damage events like explosions or leakages, or by the legal disposal of wastes into the river, created a landscape known amongst activists, citizens, and historians as “Cancer Alley” given the region’s effects on human health (Misrach and Berry, 2001). The region appears as a “sacrifice zone” (Colten, 2012: 92, Markowitz and Rosner, 2002: 248), in which industrial units produce with little respect for the needs of the surrounding communities. Modern chemical industries and refineries are automatized, and measured against their material output and economic value, companies create few jobs and have little need to invest in local infrastructures and amenities. It is an important and brutal part of this narrative that the geographical possibility of the industrial “sacrifice zone” is intimately connected to the local (and yet global) history of the sugar plantation regime and its enslaved workforce (Bullard, 2015: 85). The communities suffering most from the close vicinity to petrochemical plants remain largely African American, living in spaces inhabited by descendants of the enslaved who stayed after the Civil War (Allen, 2006: 112f). The vicious circle of the plants’ contamination and then tax avoidance in the very communities who enabled industrial production is clearly a continuation of racist policy. As is the case of the Dow Chemical Company project in Plaquemine Parish from 1956 onward, the location of large petrochemical plants in the formerly rural area between Baton Rouge and Norco was part of a strategy to avoid strong union opposition (which Dow already faced in Texas) and to profit from a disenfranchised African American population. The Plaquemine’s protest grew and in 1963 the town saw demonstrations and police violence (Markowitz and Rosner, 2002: 240). This unrest was partly fueled by the demand of the Civil Rights movement, but given the local impacts of intensive chemical industry, it was clearly also connected to what later would be rightly termed an environmental justice movement.
From the 1970s onwards, industrial expansion became increasingly connected to the history of petrochemical industry in the region and, in a way, to the corporate origins of the German Haber-Bosch technology. German and Japanese chemical companies transferred the production of increasingly more obviously polluting product lines to the Lower Mississippi region. If, in the 1930s, Baton Rouge had been established as a place of cutting-edge German-American industrial chemical research, the petrochemical corridor would become an extended and unloved workbench for the worst practices of international companies. Such a geographical race to the regulatory bottom is a problematic practice well established with regard to the Global South, but one which becomes even more apparent when applied to the US heartland and cradle of petrochemical development.
The infamous label “Cancer Alley” is itself the effect of this industrial setting. The term was first used on BASF workers’ billboards during a bitter labor dispute at the biggest BASF plant in the US, at Geismar, part of Ascension Parish, Louisiana, between 1984 and 1989. What started as a dispute over wages and health care provisions led to one of the longest lockouts in US history. The workers’ struggle engaged with questions of pollution and health, but also aspects of German history. The German Green Party were contacted by the Oil, Chemical and Atomic Workers Union (OCAW), and even BASF’s former relations with German National Socialism were raised in the ensuing controversy (Markowitz and Rosner, 2002: 247).
All these socio-historical circumstances are part of the costs and the production processes in the Petrochemical Corridor and are therefore intimately built into the artificial molecules synthesized there. The layering of environmental and historical burdens in the area is part of a distinct geo-industrial complex. As with catalysis, where unstable, precarious structures are best suited to forming active intermediates to drive the chemical reaction, in Louisiana weak social structures catalyze the output of the industry.
Even the most correctly-run and clean Haber-Bosch plant which has greatly expanded food production still leads to the overfertilization of land and biodiversity loss (Ertl and Soentgen, 2015). As the Midwest drains into the Lower Mississippi, the most intensely farmed agrarian district on the planet sheds excess nitrates. What had been shipped upriver as fertilizer is then dispersed downriver by the hydrological cycle. The resulting algal blooms in the Gulf of Mexico create hypoxic “dead zones” in the marine environment. Hydrocarbon molecules, perhaps once lifted from reservoir formations at the Gulf or at the Delta, which have made the area sink and caused land loss, are now causing further marine ecological problems. Paradoxically, it was the establishment of the Louisiana Offshore Oil Port (LOOP), a platform for conveying domestic and imported petroleum offshore situated about 30 kilometers south of Fourchon, which first contributed with measurements to the detection and investigation of such dead zones (Gorman, 2013: 151).
Epilogue: Catalytic history
The chemical process of catalysis, which allows reactions to be accelerated and controlled through the addition of minute amounts of specific substances, has become indispensable at almost all levels of the industrial practices described here. A close analysis of all levels of the actual chemical catalytic action of substances has emerged as an important means for interpreting the geo-historical situation of this industry.
One of the historically earliest and at the same time most far-reaching attempts to gain a general term from this form of chemistry came from the physician Robert Mayer, known for his formulation of the law of energy conservation. Shortly before industrial catalysis began, in 1876, he wrote a text of only seven dense pages, with the opening passage: Many natural processes only take place if they are initiated by means of an impulse, and this circumstance is what the science of the day calls “unloosening.” One of the readiest examples is certainly oxyhydrogen gas (knallgas). As is well known, a mixture of oxygen and hydrogen gas in the ratio in which they yield water enters by itself no chemical combination until the latter is initiated by means of heat or an electric spark or by platinum black. (Mayer, 1893: 440)
The ignition of oxygen and hydrogen via the use of “platinum black” which Mayer (1948a: 553) discussed was exactly one of the experiments Berzelius used to coin the term catalysis; some even consider “unloosing” as a translation of the term catalysis.
Mayer also speculated about nerve impulses, drugs, firearm triggers, and explosions, where a spark has such a violent effect. All these examples could be classified as the sudden discharge of an energetic potential and placed under the concept of unloosening (Auslösung). Mayer’s principle of triggering has aroused great interest, particular in German media theory (Schäfer and Vogl, 2004). Achieving great causal effects with little effort, an action exerting great influence is a core motif of every technology, but especially of technical media: from the alphabet, to letter press, to electronics, they are materially small but achieve highly specific impulses that launch cultural processes in different directions. Another powerful reference for media theory is that Friedrich Nietzsche was an enthusiastic reader of this Mayer essay. Alwin Mittasch (1948b, 1952) head of the catalysis laboratories of BASF from 1909 to 1933, was aware of Nietzsche’s interest, and investigated his interest in this description of catalysis in a number of studies. The absence of a translation into English has unfortunately slowed down further dissemination of Mayer’s “Ueber Auslösung” essay.
The catalytic aspects of Mayer’s speculations—apart from Mittasch’s also largely forgotten work—have been widely neglected. But considering catalysts as powerful chemical agents, analogous to the understanding of the role of media by media theorists, is not far-fetched. Since Ostwald’s time, chemists have discussed how catalysts form “intermediates” to promote a chemical process. Catalysts not only work in a temporal sense as an “intermediary.” They appear in the form of catalytic solid surfaces (Schlögl, 2017), as highly specialized spaces where highly specialized processes can occur, and as topological zones of in-betweenness. The environment of a reaction is described in chemistry as the medium of a reaction (e.g. “in an aqueous medium”), similar to what is done in biology, where the “milieu” (from the Latin medius locus) describes the environment of a species as media (Berz, 2010).
We live in a world and in landscapes that have been transformed by chemical media in a sense much broader than Mayer could have imagined. The study of the Anthropocene and the work of cultural theory need to catch up with what has, for many decades, been clear: industrial super-organisms like the Petrochemical Corridor have world-changing capacities. Studying the science and technology of catalysis offers far-reaching insights into the systems of interconnected substances, process landscapes, and historical actors that have created the contemporary geohistorical condition. From Nazi Germany’s attempts at autarky to Louisiana-based industries’ willingness to engage in the environmental racism of sacrifice zones, from global fertilizer markets to chemical engineering, from surface chemistry to dead zones in our oceans, it takes a holistic approach to interpret holistic phenomena.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work on this article was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy – EXC 2008/1 (UniSysCat) – 390540038.
