Abstract
The MedTech Funding Mandate (MTFM) policy was introduced in 2021 by the National Health Service (NHS) England, to facilitate the adoption of innovative, cost-saving medical technologies approved by the National Institute for Health and Care Excellence (NICE) within the NHS. This policy aligns with the objectives of the Getting It Right First Time (GIRFT) initiative, which aims to improve clinical effectiveness by reducing unwarranted variations in healthcare practices. In April 2022, the MTFM policy included four technologies for the treatment of bladder outlet obstruction (BOO). These technologies offer clinical effectiveness, resource efficiency, and affordability, as recommended by NICE. Despite the availability of NICE-approved minimally invasive surgical treatments (MISTs) for BOO, transurethral resection of the prostate (TURP) remains the commonest surgical treatment for BOO, although there is debate as to whether this remains the gold-standard of care. The inclusion of the four BOO technologies in the MTFM policy aims to address the existing inequalities in patient access to NICE-approved treatments and support the adoption of these effective alternatives to monopolar TURP. Key stakeholders, including GIRFT, Urology Area Networks (UANs), and the 15 Academic Science Networks (AHSNs), play a crucial role in implementing the MTFM policy. The policy has significant implications for clinical practice, promoting pathway transformation, resource and cost savings, and a comprehensive range of treatment options for patients with BOO.
Introduction
The management of bladder outlet obstruction (BOO) secondary to benign prostatic hyperplasia (BPH) poses significant challenges for the National Health Service (NHS) in the United Kingdom. With an ageing population and a rising prevalence of BPH, the demand for surgical interventions to alleviate BOO symptoms has increased, putting immense pressure on NHS surgical waiting times. This has prompted the need for innovative approaches to improve clinical effectiveness, enhance patient care, and optimise resource utilisation.
According to recent statistics, BPH affects a substantial number of men in the United Kingdom. It is estimated that approximately 40% of men over the age of 50 years and 70% of men over the age of 70 years experience lower urinary tract symptoms associated with BPH. 1 These symptoms can significantly impact the quality of life and require medical or surgical intervention. In the year 2021–2022, there were approximately 32,000 surgical procedures performed for BPH-related conditions, including transurethral resection of the prostate (TURP), laser prostatectomy, and other minimally invasive techniques. 2 However, despite this significant number of surgeries, there is still a substantial backlog of patients awaiting treatment, leading to prolonged waiting times and increased patient dissatisfaction.
Similarly, the costs associated with surgical BPH treatment pose a considerable financial burden on the NHS. Apart from the direct surgical costs, there are additional expenses related to managing post-operative complications and treating cases of surgical failure. It has been estimated that the average cost of managing a TURP-related complication ranges from £1500 to £4000 per patient, depending on the severity and required interventions.3,4 Furthermore, managing cases of surgical failure, where initial treatment does not provide satisfactory relief, adds to the overall cost burden.
Recognising the economic impact and the need for efficient, cost-effective patient pathways for BPH management, the implementation of innovative approaches such as novel minimally invasive techniques and the development of Urology Area Networks (UANs) have gained attention. These initiatives aim to optimise resource utilisation and improve patient outcomes while reducing the financial burden on the NHS. However, the development of UANs also presents potential challenges and drawbacks. One of the primary concerns is the need for effective coordination and communication among different stakeholders within the networks. Establishing robust governance structures and clear pathways for decision-making and resource allocation will be crucial to overcoming these challenges. In addition, UANs must navigate existing organisational structures and hierarchies, ensuring that all urology departments have equal representation and opportunities to contribute to network activities. There are many examples of good practices across urology departments which can be used to support the improvement and development of urology services. 5
In recent years, the national programme, ‘Getting It Right First Time’ (GIRFT), has gained prominence within the NHS. GIRFT aims to reduce unwarranted variations in health service and practice, enhance patient outcomes, and maximise resultant resource and cost savings. By addressing inefficiencies and standardising care pathways, GIRFT seeks to ensure that patients receive the most appropriate treatment in a timely manner. Aligning with the goals of GIRFT, the MedTech Funding Mandate (MTFM) 6 was introduced in 2021 by NHS England to promote the adoption of innovative, cost-saving, National Institute for Health and Care Excellence (NICE)-approved medical technologies across the NHS.
While clinicians and NHS providers acknowledge the value of adopting innovative technologies for the treatment of BPH and ensuring equal access to treatment options, funding barriers have limited the widespread implementation of these advancements. The MTFM policy specifically mandates patient access to included technologies through a dedicated funding mechanism. Funding for the technologies included in the policy comes from existing local allocations and commissioners are expected to reimburse providers on a ‘pass through’ or cost and volume approach. 7 The policy encompasses four technologies for the treatment of BOO, recommended by NICE as clinically effective and capable of reducing resource utilisation and surgical waiting times. UANs can play a vital role in the implementation of the MTFM policy, ensuring the effective adoption of the included BOO technologies and reducing variations in care across regions.
The aim of this article is to discuss the implementation of the GIRFT initiative, the MTFM policy, and the proposed development of UANs for the management of BPH in the United Kingdom.
Implementation of MTFM and inclusion of BOO technologies
In April 2022, the MTFM policy included four technologies for the treatment of BOO, which have been recommended as clinically effective and cost saving by NICE.8–11 These technologies offer advantages such as reduced resource utilisation, shorter theatre time, and shorter hospital stays compared with offering TURP as the only surgical modality. By promoting the adoption of these technologies, more patients can be treated promptly, reducing waiting times and improving overall patient care. As well as mandating commissioners to fund the MTFM technologies when clinically appropriate, the policy aims to direct the NHS on which technologies are effective and cost saving and support their adoption at scale.
The four BOO technologies included in the MTFM policy are UroLift, GreenLight XPS, Rezum, and the PLASMA system. These technologies met specific criteria for inclusion, which are as follows:
Effective: As demonstrated through positive NICE medical technology guidance.
Cost-saving within 3 years of implementation: As demonstrated by NICE modelling and a NICE resource impact assessment.
Affordable to the NHS: The NICE budget impact analysis total costs should not exceed £20 million in any of the first 3 years.
Challenges in surgical management of BOO
Despite the availability of NICE-approved minimally invasive surgical treatments (MISTs) for BOO, TURP, remains the predominant surgical intervention for this condition. Hospital Episode Statistics (HES) data from England in 2021–2022 revealed that TURP accounted for over 70% of surgical procedures for BOO. 2 TURP is traditionally associated with an average length of stay of 2 days and 60–90 minutes of theatre time plus a general/regional anaesthetic. This indicates the existing inequalities in patient access to NICE-approved MISTs. We conclude that TURP, particularly monopolar TURP, is associated with longer hospital stays and greater resource utilisation compared with MISTs and bipolar TURP, which are more often performed as day-case procedures or in an outpatient setting.
TURP has been recommended by GIRFT and The British Association of Day Surgery (BADS) as a suitable elective procedure for the development of a day surgery pathway. However, it is notable that fewer than 7% of patients are treated as a day case. 12 This compares with the MISTS or, in some units bipolar TURP, which can typically be performed as a day-case procedure and even provide the opportunity to treat patients under a local anaesthetic9,10 or move the setting to outpatients. 10 This leads to the consideration of whether TURP alone continues to be the appropriate treatment for a large proportion of men suffering from BOO, given the significant resource and financial implications and the availability of effective minimally invasive alternatives when used in the correct patient group.
The inclusion of the four BOO technologies in the MTFM policy presents an opportunity to improve patient experience, increase theatre capacity, and reduce inpatient bed utilisation by offering access to a comprehensive portfolio of effective NICE-approved technologies (UroLift System, Rezum, PLASMA System, Greenlight XPS). One of the barriers to the large-scale adoption of MISTs has been the higher cost of consumables associated with these procedures. The MTFM policy aims to address this with the aim of removing funding barriers and facilitating the widespread adoption of these technologies.
Role of stakeholders in MTFM implementation
To successfully implement the MTFM policy, collaboration among national and regional stakeholders is essential. GIRFT, alongside BADS, the British Association of Urological Surgeons (BAUS), and the British Association of Urological Nurses (BAUN), 13 has published a practical guide for improving the management of bladder outlet obstruction (Figure 1). The guide emphasises the need for a comprehensive range of treatment options within UANs as a critical component of high-quality care. The 15 Academic Science Networks (AHSNs) also play a pivotal role in facilitating the adoption of BOO technologies at the local level.
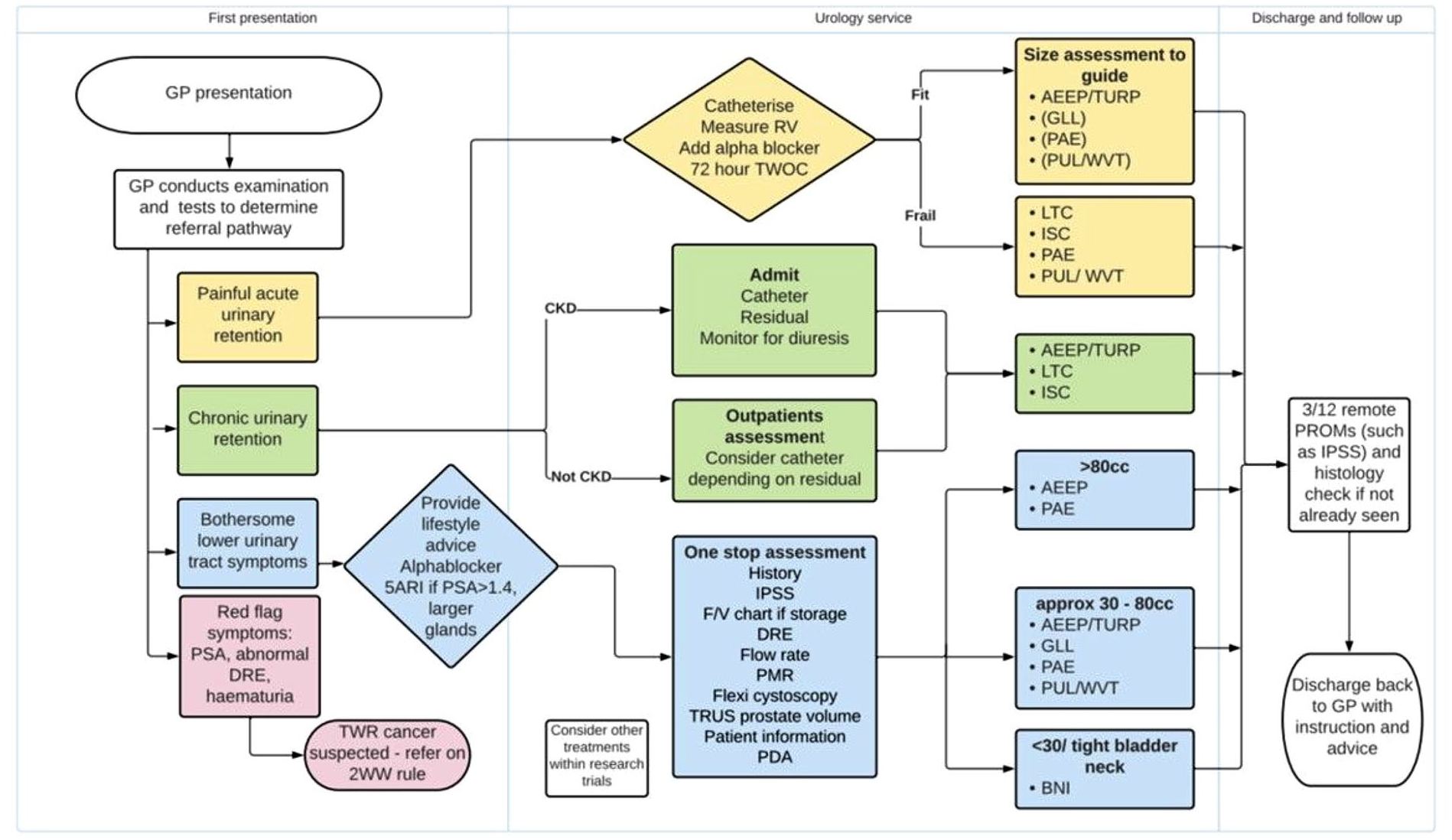
The bladder outlet obstruction pathway. This putative clinical pathway proposes the rapid and focussed assessment of patients with BPH LUTS to assist treatment selection based on specific clinical and patient factors. The inclusion of MISTs alongside TURP or anatomical endoscopic enucleation procedures (AEEP), demonstrates the potential array of therapies that collective services should offer as part of a UAN.
The combined initiatives of GIRFT, the UANs, and the MTFM policy support a coordinated regional approach to ensure patients have access to a full range of NICE-approved treatment options for BOO. The MTFM policy helps to enable the implementation of GIRFT recommendations to transform care pathways; moving from main theatres to day surgery units, urology treatment centres, community hospitals, and outpatient clinics. The establishment of one-stop lower urinary tract symptoms clinics further streamlines patient care by enabling a comprehensive assessment and discussion of treatment options in a single appointment.
Furthermore, the implementation of the MTFM policy can lead to significant resource and cost savings. NICE resource impact modelling estimates that treating 100 patients with minimally invasive surgical treatments such as UroLift or Rezum can save 87 hours of theatre time, 300 bed days, and approximately £214,000 in costs associated with this resource utility. 14 The authors accept that the limitations of the article do not allow us to address the impact of emerging technologies that may be added to the list of approved MISTs or indeed may supplant current technologies in the future. Likewise, the brief of the article does not allow a discussion to include the advantages and disadvantages of prostate enucleation surgeries, such as HoLEP, which may be associated with improved longer-term clinical outcomes but at the risk of higher setup costs for individual centres and longer training curves for surgeons. These latter aspects speak of the potential benefits of creating well-structured UANs that can develop one or two high-volume BPH centres offering more complex inpatient or day-case BPH surgeries. This is likely to positively impact unnecessary procurement costs for the NHS across multiple sites and thereby affording other centres to focus on delivering MISTs in the outpatient setting under the MTFM policy. In this way, we believe that the NHS would no longer have to steal from Peter to pay Paul, which will allow clearer financial structuring for commissioning groups and fairer access to treatments for patients based on individual needs and improving patient choice.
Conclusion
The MTFM policy, implemented in conjunction with the GIRFT initiatives and other key stakeholders, supports the adoption of NICE-approved BOO technologies and has the potential to address existing inequalities in patient access. The policy promotes pathway transformation, resource efficiency, and cost savings. By leveraging the available support systems and collaborating with stakeholders, NHS Trusts can overcome barriers to implementation and ensure equitable access to all included BPH technologies.
Footnotes
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.K. and O.K. are paid consultants of Teleflex. No conflicts are declared by J.M.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Guarantor
O.K. takes full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Contributorship
O.K. and L.K. devised and constructed the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability statement
Not applicable.
