Abstract
Objectives:
Chest computed tomography (CT) is recommended by the European Association of Urology guidelines as part of the evaluation of patients presenting with renal tumours. There are no direct recommendations for the use of computed tomography (CT) pelvis, but this is often included routinely in the assessment for metastatic disease. The incidence of metastatic disease in cT1a renal cell carcinoma (RCC) is low. Predictive algorithms may be able to guide the selective use of chest CT in the pre-operative evaluation of patients. We sought primarily to assess the clinical utility of these algorithms in predicting lung and pelvic metastases in a national cohort of purely cT1a tumours.
Patients and Methods:
Patients with sporadic, unilateral cT1a renal tumours diagnosed between January 2012 and December 2017 were identified from a prospectively collected national database. Patient clinico-pathological parameters and treatment type were recorded. Details on clinical presentation and bloods at diagnosis were taken from local electronic records retrospectively. Differences between those with and without metastatic disease were assessed.
Results:
Of the total 696 patients, 7 (1.0%) patients had metastatic disease exclusively outside the abdomen. Indeterminate lung lesions were present in 114 (16.6%) patients, with 2 (1.8%) progressing to presumed metastatic disease. Patients with metastatic disease were more likely to be anaemic than those without metastatic disease (85.7% and 25.7%, respectively, p = 0.020).
Conclusion:
Metastatic disease at presentation with cT1a renal cancer was uncommon in our national multi-centre series, with limited impact on clinical management. Patients with cT1a without intra-abdominal metastatic disease could safely avoid CT chest or pelvic CT at diagnosis, with benefits in terms of radiation exposure and resource utilisation.
Level of evidence:
3.
Introduction
One-third of patients with renal cell carcinoma (RCC) present with metastatic disease. 1 Consequently, the European Association of Urology (EAU) recommends staging with cross-sectional imaging of the chest and abdomen to detect metastases. 2 Acknowledging the risk of additional ionising radiation and increased resources required from full staging computed tomography (CT), Larcher et al. 3 and Voss et al. 4 have presented risk stratification tools for predicting lung metastases in patients with RCC and stratifying the need for staging CT chest at diagnosis. Both groups recommend that CT chest can be omitted safely for patients with cT1a tumours without evidence of nodal or visceral metastases on CT abdomen and pelvis provided there are no systemic symptoms or raised platelet/haemoglobin ratio. The potential for risk stratification and avoiding a pre-operative CT chest in selected patients is acknowledged in the most recent EAU RCC guidelines. 2 There are currently no guidelines on the need for CT pelvis for staging of suspected RCC patients, although this is routine practice in the United Kingdom.
We aim to use a prospectively collected national database of patients diagnosed with RCC to determine whether (1) the risk stratification tools proposed by Larcher et al. 3 and Voss et al. 4 hold true for predicting lung metastases in a large cohort of purely cT1a renal tumours; (2) the rate of indeterminate pulmonary nodules (IPNs) in this cohort and their clinical significance; and (3) whether CT pelvis has any clinical utility in patients with cT1a RCC.
Patients and methods
Study population
Data from patients presenting with suspected RCC across the Scottish Renal Cancer Consortium (ScotRCC) are collected prospectively on a regional level and key performance indicators reported nationally. 5 Data include basic demographics, clinical and pathological TNM staging, treatment decision and outcomes. Data were available from 7 of the 10 hospitals across Scotland who manage patients with RCC.
Patients with sporadic, unilateral cT1a RCC diagnosed between January 2012 and December 2017 were identified from this prospectively collected database. All cases were reviewed at local multi-disciplinary team meetings (MDM) with specialist uro-radiology assessment. A cT1a tumour was defined as a solid or cystic enhancing mass of the renal parenchyma less than 4 cm in maximal tumour diameter without any measurable vascular invasion on CT. An IPN was defined as a well-demarcated round opacification <1 cm of unknown nature following review at the MDM. Baseline patient demographics, clinical and pathological TNM stage, maximum tumour diameter in centimetres and treatment type were recorded. Details on clinical presentation (incidental, haematuria, loin pain or systemic symptoms) and bloods at diagnosis were taken from local electronic records retrospectively. Blood results recorded included haemoglobin (Hb; g/dL), neutrophil (Neu; ×−109/L), lymphocyte (Lymph; ×109/L), platelet counts (Plt; ×109/L) and C-reactive protein (CRP; mg/L). Absolute values, as well as whether they were above the upper limit of normal (ULN) or below the lower limit of normal (LLN), corrected for gender when appropriate, were also recorded based on local hospital reference ranges. The results of staging and follow-up imaging were reviewed, including the presence of lung metastases, IPNs and alternative pathologies.
Data were collected by individuals within each participating centre after local Caldicott Guardian approval. This project was assessed and deemed to be a clinical audit not requiring formal ethics board review. Patient data were anonymised before collation centrally by the first and senior authors prior to analysis.
Analysis
Differences in baseline patient characteristics between those with and without metastatic disease were compared using a t-test for continuous data and a chi-square test for categorical data. With all tests, a two-sided p-value of <0.05 was considered statistically significant. All statistical analyses were performed using the software Statistical Package for the Social Sciences (SPSS) for Windows version 25.0 (IBM Corp, Armonk, NY, USA).
Results
A total of 707 patients were diagnosed with cT1a RCC between January 2012 and December 2017. All patients underwent CT abdomen and pelvis. CT chest was omitted from staging in 51 (7.2%) patients at the time of diagnosis, but these patients were managed with presumed localised disease. None of these patients had metastatic disease on first follow-up imaging and were included in the study. Metastatic disease was present in 16 (2.26%) patients at diagnosis. A total of 10 patients had intra-abdominal metastases either alone (n = 7), with associate intra-pelvic metastases (n = 2) or with intra-thoracic metastases (n = 1). One further patient who presented with neurological symptoms had further investigation which led to a diagnosis of metastatic RCC with intra-cranial metastases. These 11 patients were excluded from further analysis. Five (0.7%) of the remaining 696 patients had isolated metastatic disease exclusively outside the abdomen.
Of the 691 patients with localised disease, 114 (16.5%) patients had IPNs on CT chest. This finding did not affect initial management of any patient. Two (1.8%) of these patients were subsequently diagnosed with lung or bronchial carcinoma and 2 (1.8%) patients had progression of their lung nodules and were presumed to have metastatic RCC, without biopsy confirmation. The remaining 110 (96.5%) patients were stable on surveillance with a median follow-up of 41.2 months (range = 1–212 months).
The median age of patients was 68 (range = 23–92) years and 376 (54%) were male. The clinical characteristics of all patients are detailed in Table 1. Most patients presented incidentally (78.4%). Pathological confirmation of RCC was made in 493 (70.8%) patients, while 203 (29.2%) patients underwent surveillance or watchful waiting without biopsy because of patient comorbidities (Table 2). Overall, active treatment was undertaken in 463 patients (66.5%; Table 2). Of the seven patients included with metastatic disease, four patients had lung metastases at diagnosis (one biopsy confirmed mRCC and three presumed mRCC without biopsy), two patients initially had IPNs with subsequent growth leading to a diagnosis of mRCC and one patient had bone metastases in the L1 vertebrae and sacrum detected on CT pelvis. Only one of the seven patients with a diagnosis of mRCC underwent biopsy for diagnostic confirmation. This patient had a lung biopsy confirming mRCC and was commenced on systemic anti-cancer therapy (SACT) and did not undergo any surgical intervention. The patient with progressive IPN after laparoscopic radical nephrectomy also commenced SACT following an extended period of surveillance. All other patients were managed symptomatically, in view of their comorbidities, as they were not considered fit for surgical or oncological intervention at diagnosis. Give this, only one (0.14%) patient had a change to management given the identification of mRCC.
Clinical characteristics of patients presenting with cT1a renal cancer.
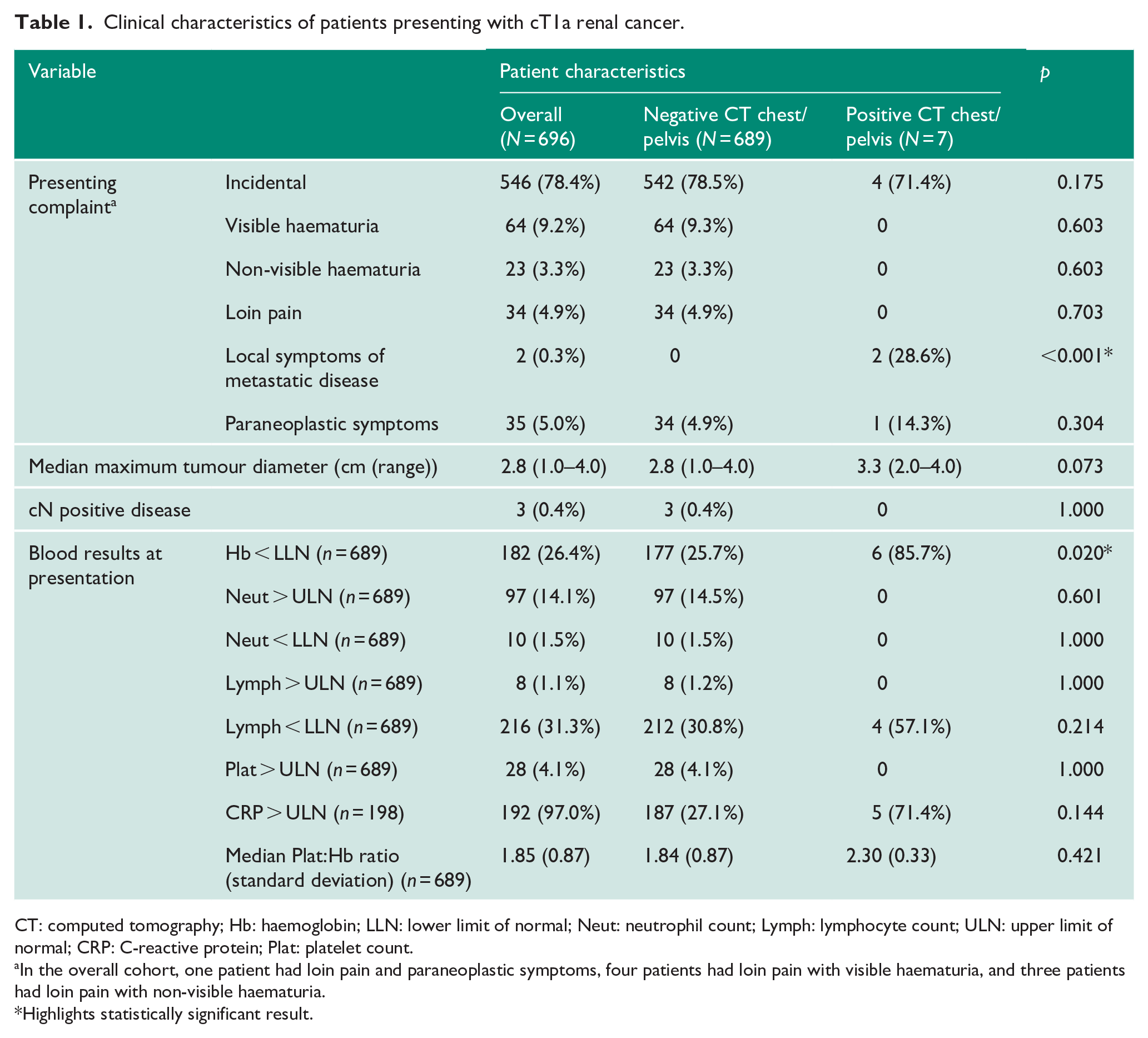
CT: computed tomography; Hb: haemoglobin; LLN: lower limit of normal; Neut: neutrophil count; Lymph: lymphocyte count; ULN: upper limit of normal; CRP: C-reactive protein; Plat: platelet count.
In the overall cohort, one patient had loin pain and paraneoplastic symptoms, four patients had loin pain with visible haematuria, and three patients had loin pain with non-visible haematuria.
Highlights statistically significant result.
Initial management and pathological diagnosis of patients presenting with cT1a renal tumours across Scotland.
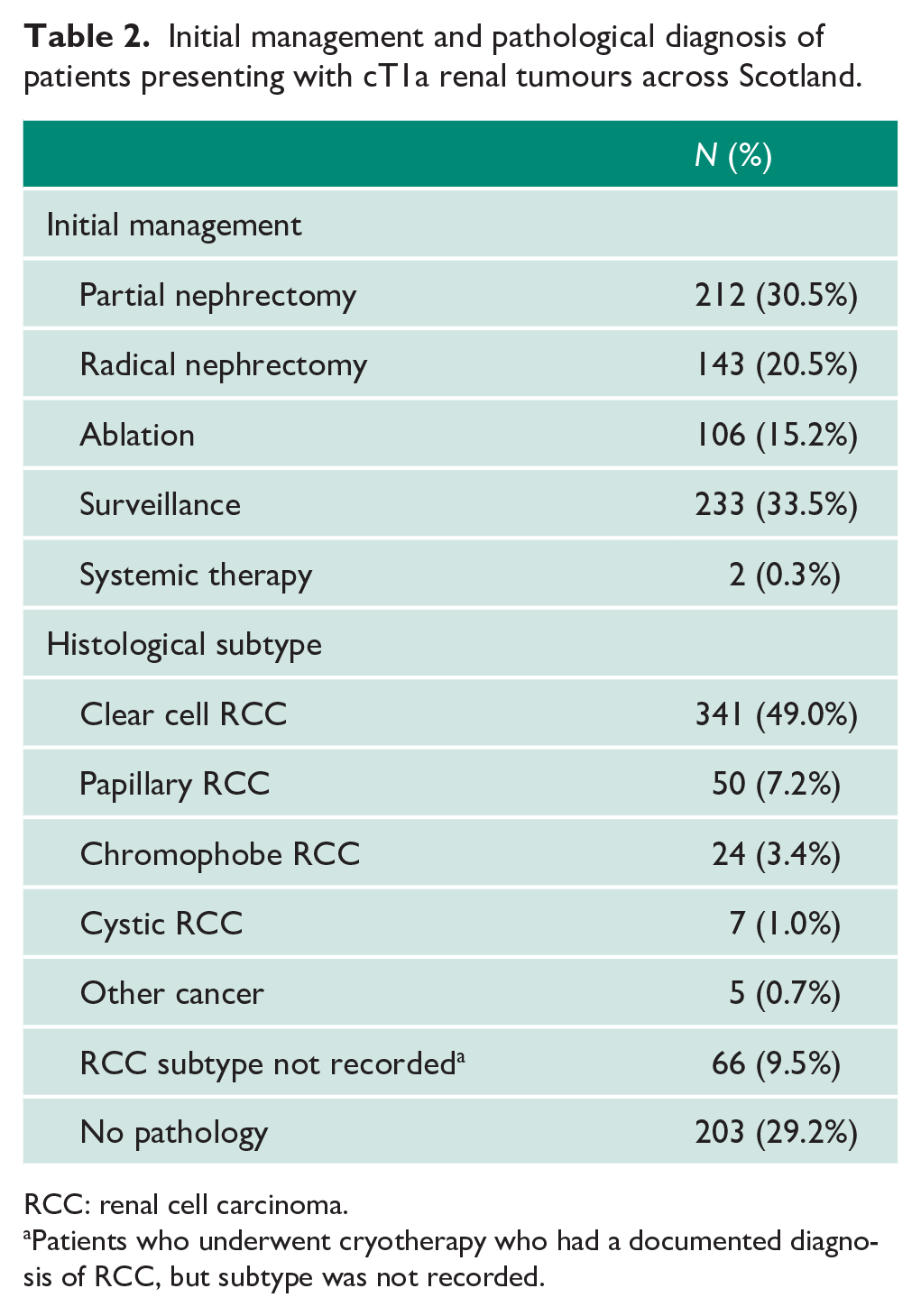
RCC: renal cell carcinoma.
Patients who underwent cryotherapy who had a documented diagnosis of RCC, but subtype was not recorded.
Four of these patients presented incidentally, while two patients presented with local respiratory symptoms leading to a diagnosis of lung metastases and the subsequent identification of a renal primary. One patient with initially IPNs and later diagnosed with metastatic disease had systemic paraneoplastic symptoms, which settled after initial laparoscopic radical nephrectomy. Interestingly, this patient was staged as cT1a pre-operatively, although final pathology after nephrectomy was The International Society of Urological Pathology (ISUP) grade 2, stage pT3a clear cell RCC with small segmental vein tumour thrombus.
Those patients with metastatic disease were more likely to be anaemic than those without metastatic disease (80% and 25.8% Hb < LLN, respectively, p = 0.019). Those with metastatic disease were no more likely to have abnormal neutrophil, lymphocyte count, platelet count or CRP (Table 1). There was no significant difference in the Plt:Hb ratio between metastatic and non-metastatic RCC patients (2.30 and 1.84, respectively, p = 0.495). Due to the overall small number of events, many of the variables were underpowered to determine whether they were predictive of metastatic disease on CT chest or CT pelvis, and therefore, no regression analysis was performed. Interestingly, two patients did have symptomatic lung metastases which led to the initial diagnosis, but incomplete recording of respiratory symptoms from all patients limited the analysis.
Given the difference in rate of anaemia between those with localised and metastatic disease, we assessed the predictive power of this marker for deciding upon whether to perform full staging cross-sectional imaging. Using anaemia as a marker for full staging CT in patients with cT1a renal cancer without intra-abdominal metastases, 514 (73.9%) patients with a normal Hb would have avoided additional CTs, missing 1 (0.2%) diagnosis of metastatic disease. Staging CTs would be performed for 182 (26.1%) patients, detecting 85.7% cases in this cohort with an overall positive predictive value of 3.3%.
Discussion
Our study of a national multi-centre series of patients with cT1a renal tumours confirms that metastatic disease is uncommon in this group and the rate of pulmonary or pelvic metastases in the absence of intra-abdominal metastases is particularly uncommon (0.86% and 0.14%, respectively). Given the uncommon rate of metastatic disease, staging CT chest or pelvis could be omitted in this group of patients, with a very low impact on patient management. Anaemia may be a useful marker to select patients to undergo further staging CT, but this will still subject a high number of patients to unnecessary imaging and a larger study may be required to validate this potential biomarker.
We present a rate of metastases in cT1a renal tumours without the presence of intra-abdominal metastases similar to that reported by both Larcher et al. 3 and Voss et al. 4 (8/859 (0.9%) and 4/385 (1.0%)), respectively. Larcher et al. 3 recommend a threshold for further staging when the risk of metastatic disease is > 1%, a threshold which is not met in our present study, or the cT1a renal tumour subgroups in the Larcher or Voss studies. Both Larcher and Voss use a larger cohort of all-stage localised renal cancer patients to determine other factors beyond stage, which are predictive of the presence of metastases. Unsurprisingly, in this all-stage analysis, clinical node status, systemic symptoms, lower Hb and higher platelet count, as well as a surrogate measure of both with the Plt: Hb ratio, reported to be associated with advanced disease in other studies, predict the presence of metastases.6–8 However, when analysis is confined to small renal masses alone, with the low overall rate of metastatic disease, node status, paraneoplastic symptoms, elevated platelet count and thus Plt:Hb ratio were not significantly different in patients with metastatic disease, compared with the whole cohort, in our series. Voss et al. 4 define the possible difficulties interpreting cN stage on CT as well as the lack of validation of the Plt:Hb ratio and the resultant limitations associated with use of these parameters in a nomogram. Similarly, the breadth of systemic symptoms and paraneoplastic syndromes attributable to metastatic RCC may have been difficult to catalogue in the presented retrospective studies and may be easily overlooked in the clinic, potentially limiting the reproducibility of this risk stratifier. 9 We would caution our analysis of the predictive capability of these factors in our cohort, given the small number of events overall. A larger study or meta-analysis may be of benefit. The presence of local or systemic symptoms is difficult to interpret in our study because of potential missing data and small number of events, although clearly if there is clinical suspicion of metastatic disease, then this should be considered when deciding on the need for further imaging. We did find the rate of anaemia to be different between those with and without metastatic disease. The presence of anaemia may be useful for sub-selection of patients requiring full-staging CT, increasing the diagnostic yield to 3.3% in our study with the risk of missing one case (0.14%) of metastatic disease. Given the small number of patients with metastases overall, the use of anaemia as a stratifier requires further validation. Nonetheless, this could allow a more cautious approach to changing staging protocols for these patients, with clinical rational given that we would not necessarily expect a small localised RCC to cause anaemia and the finding of anaemia may result in additional investigations, including further CT scans.
The identification of metastatic disease may not influence initial management decisions. In our cohort, the majority of patients were managed symptomatically because of comorbidities, while one patient proceeded to surgery as planned and only one patient had a change from surgery to systemic therapy. Clearly, ensuring our patients are fully informed of their diagnosis and the significantly different outcomes associated with management for localised and metastatic RCC is key.10–12 For patients who are fit for surgical intervention for their small renal mass, sub-stratification with assessment of Hb would allow an easy and reproducible means of identifying patients at a higher risk of metastatic disease. The identification of anaemia may also be important in the context of mRCC. Patients with anaemia, who are identified to have metastatic disease, would have at least intermediate risk mRCC using the International Metastatic RCC Database Consortium (IMDC) risk classification and could arguably have a change in their treatment plan from (cytoreductive) nephrectomy to systemic therapy.13–15
This rationalisation of CT imaging has several potential benefits. A CT chest typical effective radiation dose is between 6 and 8 mSv compared with 10 mSv for a CT abdomen and pelvis, although significant variation exists in reported doses in the literature.16,17 A CT chest is associated with a 1 in 2500 additional lifetime risk of a fatal cancer. 17 Clearly, the additional cancer risk of a single CT chest/pelvis in a patient in their 60s could be debated but the principles of keeping ionising radiation dosing as low as reasonably achievable (ALARA) while ensuring that medical benefits are as high as reasonably achievable (AHARA) are central to the practice of radiologists across the globe. The significance of reducing radiation by omitting a single scan could be argued, particularly given that patients are likely to continue post-operative CT surveillance. Perhaps more significantly, is the cost and resource associated with additional cross-sectional imaging. Inclusion of the chest and pelvis requires more time within the scanner as well as reporting time, and some patients may be recalled for a separate CT chest depending on the mode of detection of their initial tumour. These are important considerations in a resource stretched national health service. Reducing the use of CT chest and CT pelvis in patients with cT1a renal tumours may therefore have additional economic benefits, while liberating additional radiological capacity.
In addition to the detection of metastatic disease, CT chest also often identifies non-specific pulmonary nodules of uncertain significance in patients with kidney cancer. This can result in additional follow-up imaging and may lead to significant anxiety for patients. IPNs are classified as being rounded opacities of uncertain aetiology which are less than 3 cm in maximal diameter.18,19 Mano et al. 20 reported an incidence of IPN in 51% of study patients presenting with localised renal cell cancer. The presence of IPN in RCC patients can present a diagnostic dilemma, representing a range of potential entities from benign lesions to metastatic RCC or primary lung cancer. However, the prognostic significance of IPN in the context of a diagnosis of apparently localised RCC remains unclear. Mano et al. 20 noted a size cut-off of 1 cm for IPN, above which the risk of progression to pulmonary and indeed distant metastases was significantly increased. Xu et al. 21 noted that the presence of IPN has a detrimental effect on disease-specific survival but failed to demonstrate a correlation with size. Abidi et al. 22 reported a similar finding upon developing a predictive nomogram, with no size cut-off but rather an increase in risk of developing lung metastases with increasing IPN size and number.
Our series confirms that indeterminate lung lesions are also common in our population (16.6%). Patients can, however, be reassured that in this group, significant pathology was relatively low (4/116, 3.5%) with two cases of primary lung cancer identified on resection and two cases presumed to be metastatic RCC, although no pathology was obtained. The potential alternative pathology of these pulmonary progressive nodules or presumed lung metastases more generally is highlighted further by Voss et al. 4 who demonstrated that in the four patients with renal tumours ⩽4 cm and positive CT chests (1.0%), three were resected and all were confirmed to be primary lung carcinoma and not mRCC. Further work is required to stratify and determine best follow-up for patients with indeterminate lung lesions, although importantly, these often do not affect initial treatment decisions. Given the low risk overall of isolated lung metastases in cT1a RCC, consideration should be given to biopsy growing or presumed pulmonary metastatic disease in this patient group, particularly to exclude primary lung cancer which may have alternative treatment options, such as those with epidermal growth factor receptor (EGFR) mutations. 23
Limitations
As with all retrospective analyses, there are several limitations with this analysis. One key consideration is that only 70.8% of patients included had pathological confirmation of underlying RCC. Based on other data, we presume up to 20% of those without biopsy (5.7% total from the cohort) may have benign disease, which is a relatively small proportion of the cohort. 24 We believe this reflects real-world data in which a decision to perform further staging would be based on initial radiological assessment of the kidney given the low rates of diagnostic renal biopsy generally. 25 Unfortunately, there are missing data within the cohort. This includes only a small number of patients without a full blood count but a larger sub-group without a CRP assessment, which could significantly alter the analysis for this marker. While local symptoms of metastatic disease were found in two of the patients with a positive CT chest, thorough documentation of chest symptoms, such as cough, haemoptysis or shortness of breath, in other patients without metastases were not documented. This prevents any valid conclusion on the utility of local chest symptoms as a screening tool to select patients for CT chest. Overall, there are a low number of events, and this limited the power to identify all markers predictive of metastatic disease. Further larger-scale studies or meta-analysis of current studies may prove useful. Finally, while metastatic disease was detected in five patients pre-operatively and subsequently in two patients with initially described indeterminate lung nodules, only one of these patients had a lung biopsy to confirm metastatic disease. Indeed, as discussed, the two patients with indeterminate lung lesions who had local treatment had biopsy confirmed adenocarcinoma of the lung. Consideration to biopsy should be given when presumed metastatic disease is identified in the presence of cT1a RCC.
Conclusion
In our multi-centre series of cT1a tumours, the rate of isolated pulmonary or pelvic metastases, in the absence of abdominal metastases, is low and staging CT chest and pelvis can be safely omitted in this group at diagnosis. The presence of anaemia may have clinical utility in risk stratifying patients who are at higher risk of metastases and further work to investigate this and other predictors would be useful. Reducing the need for additional CT imaging in this group of patients may have benefits in terms of both reducing exposure to ionising radiation as well as resource utilisation.
Footnotes
Acknowledgements
The authors thank the South East Scotland, West of Scotland and North of Scotland Cancer Network teams for their assistance in identifying appropriate patients. For the purpose of open access, the author has applied a Creative Commons Attribution (CC BY) licence to any Author Accepted Manuscript version arising from this submission.
Authors’ note
Matthew Trail, Daniel W Good and Jennifer B Ayers are also affiliated to Department of Urology, Western General Hospital, NHS Lothian, UK.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: J.B. is supported by an SCREDS Clinical Lectureship, University of Edinburgh. A.L. is supported by a Chief Scientist’s Office of Scotland (CSO) NRS Career Researcher Fellowship.
Ethical approval
As an audit of standard of care, the study did not require specific ethical approval as per NHS Health Research Authority guidance. Caldicott Guardian approval was sought prior to local data collection.
Informed consent
Written informed consent was obtained from a legally authorised representative (Caldicott Guardian) for anonymised patient information to be published in this article.
Guarantor
A.L.
Contributorship
A.L. contributed to conceptualisation. A.L. contributed to methodology. E.A.A.M., S.N., E.M., A.M., M.T., D.G., J.A., F.R. and J.B. contributed to data collection or management. E.A.A.M. and A.L. contributed to data analysis. E.A.A.M. and A.L. contributed to writing – original draft. E.A.A.M., S.N., E.M., A.M., M.T., D.G., J.A., F.R., J.B., J.H., K.Q., S.L., S.P., S.A.M., A.M., K.J., J.F.D., B.G.T., A.C., G.A., M.Z.A., G.L., G.O. and A.L. contributed to writing – review and editing. J.H., K.Q., S.L., S.P., S.A.M., A.M., K.J., J.F.D., B.G.T., A.C., G.A., M.Z.A., G.L., G.O. and A.L. contributed to supervision.
