Abstract
Objective:
To determine if periodic triamcinolone injections into Hunner’s lesion in patients with bladder pain syndrome (BPS) reduces the need for opioids and other pain treatments.
Methods:
This is a retrospective analysis of 28 patients receiving endoscopic injections of 0.5–1.0 cc of triamcinolone acetate into Hunner’s lesions between 2010 and 2018. Wilcoxon signed-rank test was used to compare pain regimens before and after injections.
Results:
Median age at first triamcinolone injection was 63 (IQR 54–73). Median number of injections/patient was 3 (IQR 2–5.5), at a mean of 8-month intervals (2–80). The median number of pain treatments prior to triamcinolone was 4 (0–13), and 25% of patients were using opioids. With one or more injections, 92.9% had improvement in pain symptoms. There was a significant decrease in number of pain treatments following triamcinolone injections (4.1 vs 0.8, p = 0.006). Fifty-seven percent managed with triamcinolone injections alone, with no other pain treatments. Of the seven patients using opioids, four discontinued opioids altogether.
Conclusions:
Repeat triamcinolone injections into Hunner’s lesions are associated with a significant reduction in the number of pain treatments used for BPS, with an associated decrease in opioid use.
Level of evidence:
4
Introduction
Bladder pain syndrome (BPS), previously referred to as interstitial cystitis, is found in 3–6% of American women and places a significant economic burden on the health care system. 1 The mean annual per-person healthcare cost of BPS is greater than that for diabetes mellitus, depression, hypertension, and asthma, and is 2.0–2.4 times higher than age matched controls.2,3
There are guidelines for the management of patients with BPS. These include conservative therapies, medical therapies, minimally invasive surgical procedures, radical surgery and phenotype-directed multimodal therapy.4,5 Despite these guidelines, satisfaction/quality-of-life (QOL) assessments remain low and patients’ perceptions of treatment success are not in keeping with the efficacy reported from clinical trial data.6–8 Furthermore, many patients resort to opioid use for BPS related pain, which has been associated with significant long-term side effects and further burden to the health care system.9,10
Based on cystoscopic findings, 10–20% of patients with BPS have Hunner’s lesions.11–13 Treatment options include fulguration or resection, with an alternative treatment of triamcinolone injections into the lesions. 14 Triamcinolone is a synthetic glucocorticoid that has been shown to improve the appearance of hypertrophic scars through inhibition of fibroblast growth and promotion of collagen degradation.15,16
Several clinical studies have evaluated triamcinolone injections into Hunner’s lesions in case series and have reported improvement in voiding and patient satisfaction scores.14,17–19 However, studies have not evaluated the effect of repeated triamcinolone injections over a prolonged period of time on mitigating the use of pain treatments such as opioids for BPS pain. The primary objective of this study was to assess whether the recurrent use of triamcinolone injections for Hunner’s lesions in BPS leads to a consistent reduction in the use of pain treatments.
Methods
Research ethics board approval was obtained at Sunnybrook Health Sciences Centre prior to initiation of this retrospective case series (REB# SUN-2565). Data were collected retrospectively from both electronic and paper patient charts. We identified a total of 28 patients with BPS and Hunner’s lesions who underwent triamcinolone injections between 2010 and 2018 by two surgeons at a single institution. BPS was diagnosed based on a combination of patient history, cystoscopy and urodynamic findings, and Hunner’s lesions were diagnosed via cystoscopy. Data collection included patient characteristics, pre- and post-BPS pain treatments, triamcinolone treatment number, follow-up time, injection complications and patient reported reduction in bladder pain. Pain treatments were defined as oral, physical therapy, minimally invasive surgery, intravesical therapy, suppository, or topical, and are listed in Supplemental Tables 1 and 2. Treatment schedule was determined on a case-by-case basis, depending on individual response to triamcinolone. Repeat triamcinolone injection was offered as early as 3 months from initial injection if urinary symptoms recurred, or if they did not have an initial response. Patients receiving greater than five treatments were analysed separately to assess change in frequency of injections over time in those receiving a greater number of injections.
Surgical technique
All patients gave informed consent for treatment. The procedure was done under local anaesthesia in the lithotomy position with a rigid cystoscope for women and supine position with a flexible cystoscope for men, or under general anaesthesia in the lithotomy position with a rigid cystoscope for both men and women. Local anaesthesia consisted of instillation of 50–100 cc of 2% lidocaine solution for 20 minutes. The bladder was emptied and 0.5–1.0 cc of triamcinolone acetate (40 mg/mL diluted to 4 mg/mL with normal saline; max. 10 mL) was injected into Hunner’s lesions with an endoscopic needle under cystoscopic vision. Hydrodistention and fulguration were not done.
Data analysis
Descriptive statistics were used to describe the patient cohort and treatment schedule. The primary outcome was defined as the difference in number of pain treatments used before and after repeat triamcinolone injection use. This was assessed using the Wilcoxon signed-rank test. Secondary outcomes included patient-reported decrease in bladder pain (recorded in patient chart), and complications following injections. All data analysis was generated using SAS Enterprise Guide 6.1 (SAS Institute Inc). A p-value of <0.05 was considered significant.
Results
Demographics
A total of 28 patients received triamcinolone injections into Hunner’s lesions. 82.1% were female, 42.9% had undergone prior pelvic surgery, and 14.3% had an additional diagnosis of a pain syndrome. The median age at first triamcinolone injection was 63 (IQR 54–73) (Table 1). The mean follow-up time from first treatment was 38.6 months (range 7–99 months).
Patient characteristics.
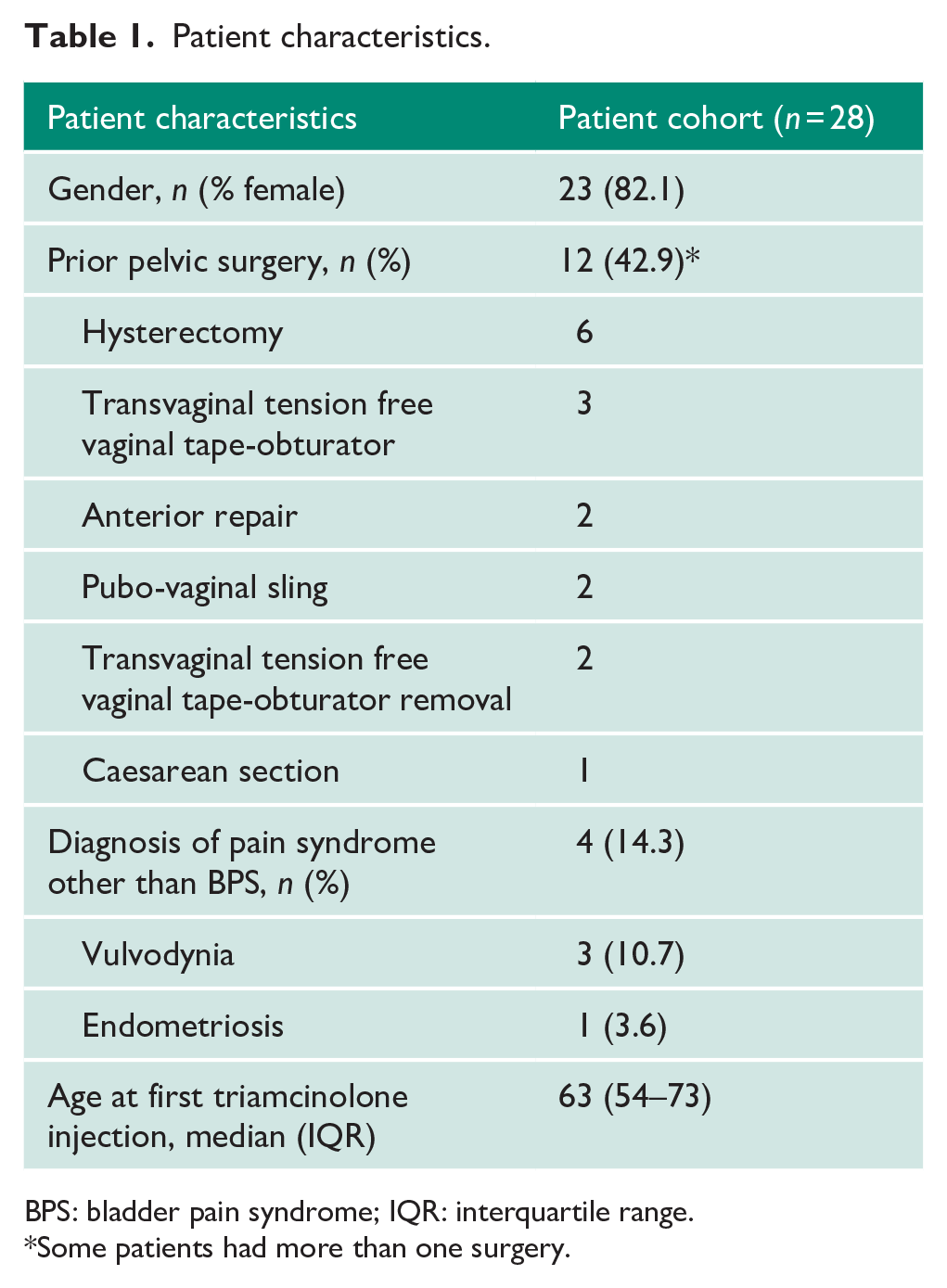
BPS: bladder pain syndrome; IQR: interquartile range.
Some patients had more than one surgery.
Triamcinolone treatment schedule
The mean number of triamcinolone treatments at the time of study end date was 4.3 per patient (range 1–16), with a median of three treatments (IQR 2–5.5). At the time of study end date the number of triamcinolone treatments ranged from 1 to 16, with seven patients having one treatment, seven patients having two treatments, three patients having three treatments, three patients having four treatments, three patients having five treatments, four patients having six treatments, one patient having eight treatments and two patients having 16 treatments. For patients who had more than one treatment, the average time between injections was 8 months, with a range of 2–80 months. For patients who had greater than five triamcinolone treatments (7/28), there was no observed increase in frequency of procedures due to earlier symptom recurrence. In these patients, the average time between treatments was 5 months, with a range of 2–21 months.
Effect of triamcinolone treatment on pain treatments
A total of 22 patients were on pain medications for BPS prior to triamcinolone treatment. Patients had tried a mean number of 4.1 different pain treatments (range 0–13) prior to triamcinolone injections. These treatments are listed in Supplemental Tables 1 and 2; 10 different oral medication categories were used, for a total of 23 unique medications. Nineteen non-oral medication treatments were used. The most-used oral treatment was pentosan polysulfate sodium (PPS), with 16 patients (57.1%) either currently using the medication or having tried it in the past. 32.1% and 35.7% of patients tried intravesical instillations and lesion fulguration, respectively. Seven out of 28 (25%) patients had used opioids for BPS management.
After one or more triamcinolone treatments 16 patients were not using any pain medications (57.1%), while 12 patients continued one or more pain treatments (42.9%). Seventeen patients had decreased overall pain medication use, seven had no change (six of which had not been on any pain medication prior to triamcinolone treatments) and three patients had an increase in pain medication use (Figure 1). Of the 16 patients who had tried PPS, only two patients were using PPS after starting triamcinolone treatments. Of the seven patients using opioids for pain management, four discontinued the use of opioids altogether, and one transitioned from daily use to as needed use. In comparing the mean number of pain treatments before and after triamcinolone injection, there was a significant difference on Wilcoxon signed-rank test (mean 4.1 vs 0.8; p = 0.006).
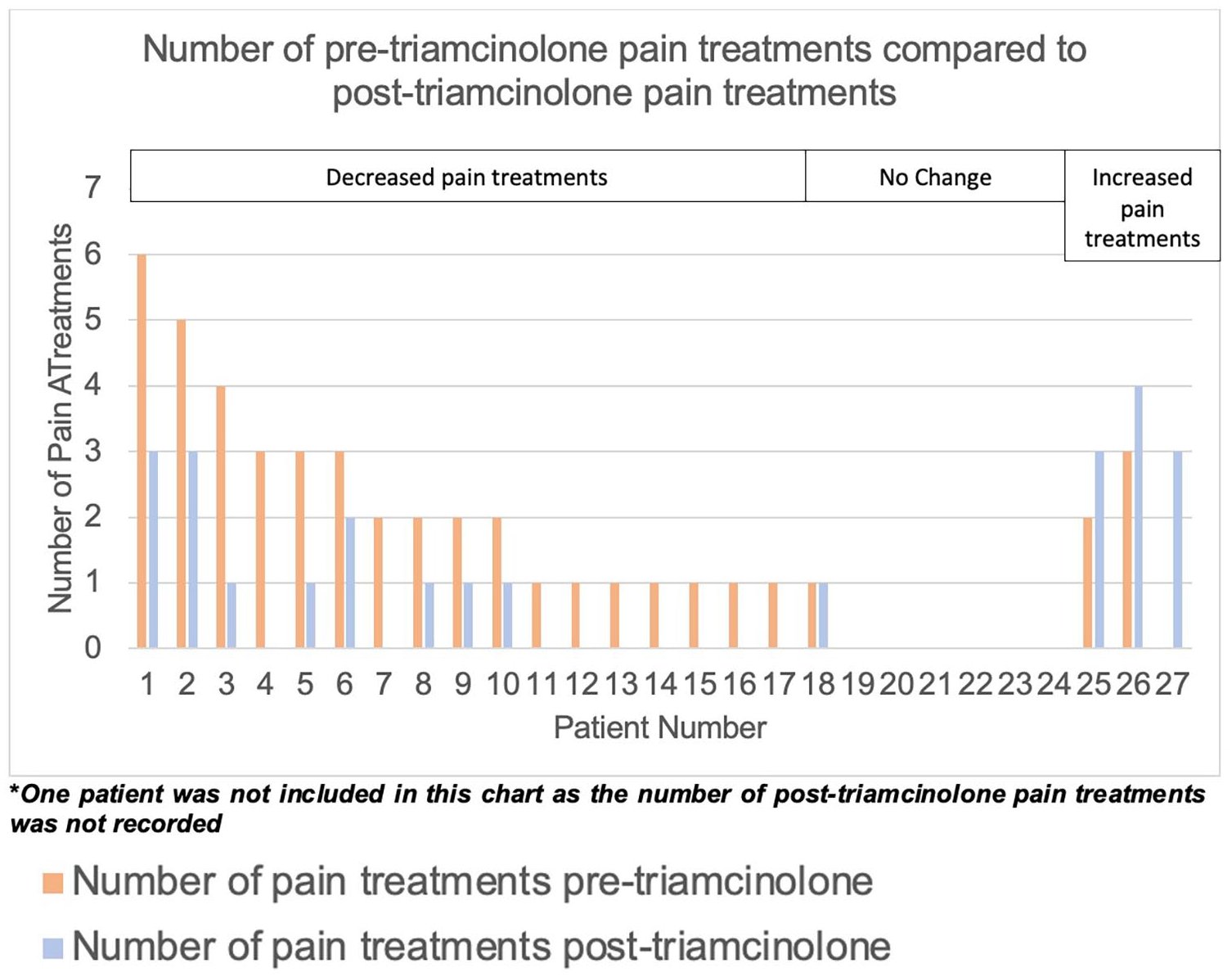
Number of pre-triamcinolone pain treatments compared to post-triamcinolone pain treatments.
Efficacy
92.9% of the patients reported improvement in pain symptoms after triamcinolone treatment, based on patient history in follow-up clinic. 7.1% had complete resolution of Hunner’s lesions on follow-up cystoscopy (after six treatments in one patient and two treatments in the other patient). Patients who had more than five triamcinolone treatments continued to have subjective pain improvement. Of those who had just one triamcinolone treatment (7/28), one stopped due to a diagnosis of bladder cancer while six have had a good response not requiring another triamcinolone treatment to date. Of the two patients who did not have improvement in pain after triamcinolone treatment, one went on to have cystectomy and ileal conduit creation after six injections, and one is pending a re-trial of triamcinolone injections.
Safety
Three patients presented to the emergency room with gross hematuria, of which two required an overnight hospital admission for continuous bladder irrigation. One of these three patients was on coumadin. No systemic effects were noted with the use of triamcinolone.
Discussion
BPS is a difficult-to-manage chronic pain syndrome often requiring multiple treatments. Triamcinolone injections are treatment option for the 10–20% with Hunner’s lesions. Patients describe improved voiding and pain outcomes with this treatment.9,10,16 In this study, 92.9% of the patients had improved subjective pain, and in those who had at least five triamcinolone treatments, the subjective improvement of pain has continued to date. Furthermore, we demonstrate that patients who have triamcinolone injections require fewer number of other pain treatments afterwards (mean 4.1 vs 0.8; p = 0.006). Sixteen of the 28 (57%) patients required no pain treatments after triamcinolone injections.
Twenty-five percent of the patients in this cohort tried opioids for their BPS pain. Similarly, Zillioux et al. 10 found that 28% of patients with BPS received opioids (in a cohort of 6884 patients with BPS from the Virginia All Payers Claims Database). Zillioux et al. 9 also found that there is a significant subset of patients who receive 10 or more opioid prescriptions, suggesting that some BPS patients remain on opioids long-term with possible long-term side effects and burden on the health care system. In our study, fewer than half of those initially on opioids remained on them after triamcinolone injections. Thus, triamcinolone injections into Hunner’s lesions appear to have significant clinical benefit.
The most used oral medication in this study was PPS, at 57.1%. Pigmentary maculopathy has been reported in long-term use of PPS, with visual symptoms manifesting as difficulty reading, slow adjustment to low or reduced light, and blurred vision. Health Canada issued a safety alert in October 2019 with regard to this issue, and the Food and Drug Administration (FDA) followed in June 2020.20,21 This highlights the need for further effective treatment options for patients with BPS.
BPS treatments specific to the treatment of Hunner’s lesions include transurethral fulguration with electrocautery or laser, or transurethral resection (TUR) of the lesion. Like triamcinolone injections, these procedures often need to be repeated for durability of effect. A randomised-controlled trial comparing TUR to fulguration of Hunner’s lesions in 126 patients showed good efficacy, with no difference between the two groups, and a recurrence free time of 11.5–12 months. 22 However, there is concern that repeat treatment with TUR/fulguration results in bladder wall scarring and contracture, leading to decreased bladder capacity. A recently published study of 118 BPS patients with Hunner’s lesions compared the frequency of electrocautery of lesions to maximum bladder capacity (MBC). They found MBC decreased by 50cc per treatment session. 23 We have not observed any bladder wall scarring with repeat triamcinolone injections, and scarring has not been reported in the literature.14,17,19 Histological studies of repeat injections with botulinum toxin A, a procedure similar to repeat injection of triamcinolone, have not demonstrated evidence of bladder fibrosis after four repeat injections.24,25
The appropriate regimen for triamcinolone treatment remains to be determined. In other studies, repeat injections were required with a median time between injections ranging from 224 to 313.5 days.14,17 In our series, patients who had more than five triamcinolone treatments did not see a decrease in the time in between injections and remained at a median of 168 days. This treatment option appears to be similar to botulinum toxin A injections for overactive bladder (OAB), where the expectation of treatment is for symptom management, not cure. An algorithm for repeat use of triamcinolone injections has been included (Figure 2).
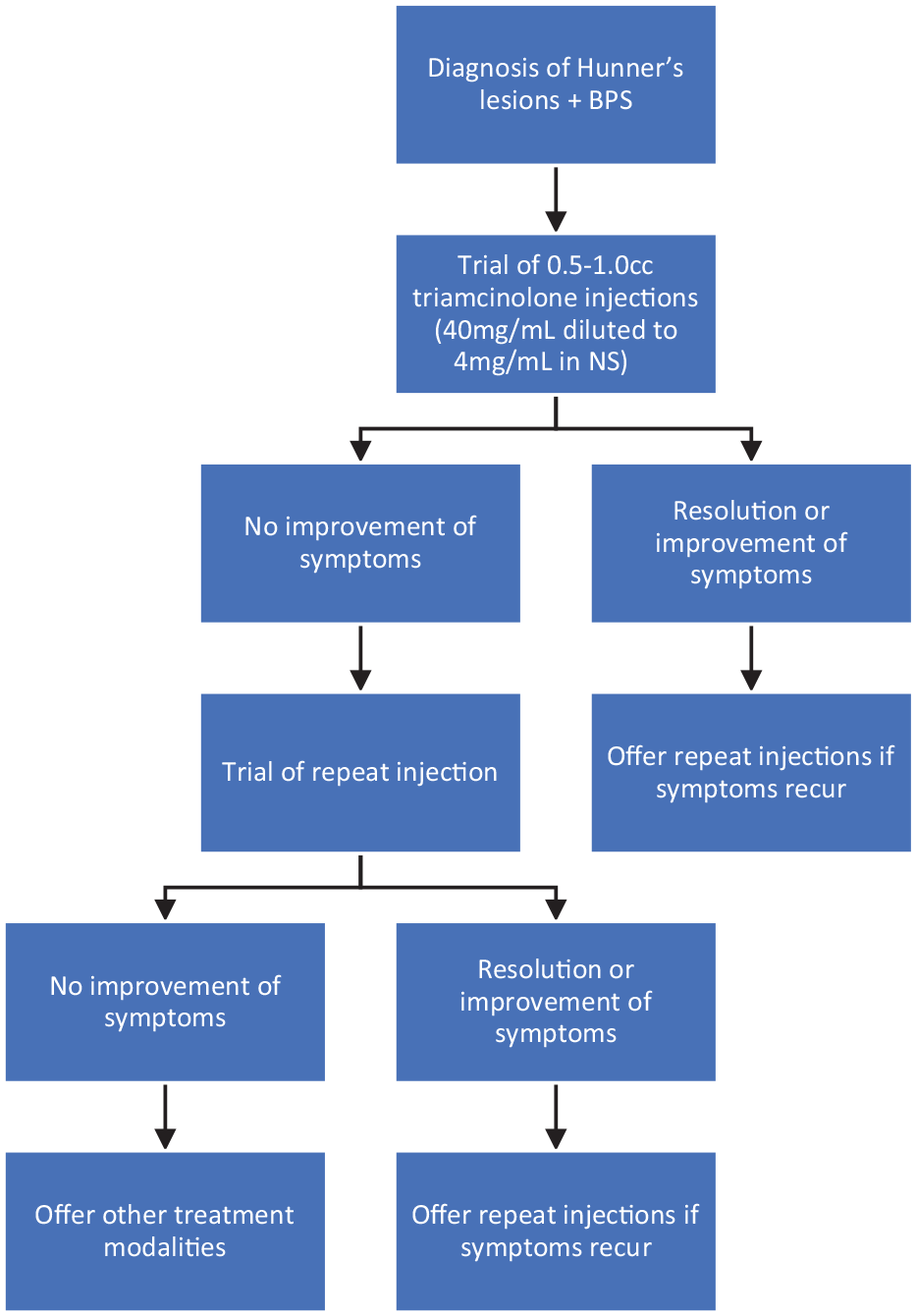
Algorithm for repeat triamcinolone injections.
Limitations to our study include the relatively small sample size. It is a retrospective case series without a control group. Furthermore, triamcinolone injections were delivered by two surgeons at a single institution. Validated pain scales were not used, as this was a retrospective review. Future large-scale studies should focus on determining the appropriate regimen for triamcinolone injections as well as overall safety and durability of this treatment option.
Repeated triamcinolone injections into Hunner’s lesions are associated with a significant and consistent reduction in the number of pain treatments used in patients with BPS, with an associated decrease in opioid use. Patients may experience a durable response in symptom improvement with repeat injections, with minimal safety concerns.
Conclusion
Triamcinolone injections into Hunner’s lesions are associated with a significant reduction in the number of pain treatments used in patients with BPS, with an associated decrease in opioid use. Patients may experience a durable response in symptom improvement with repeat injections, with minimal safety concerns.
Supplemental Material
sj-docx-1-uro-10.1177_20514158221122521 – Supplemental material for Recurrent triamcinolone injections for the treatment of Hunner’s lesions in bladder pain syndrome
Supplemental material, sj-docx-1-uro-10.1177_20514158221122521 for Recurrent triamcinolone injections for the treatment of Hunner’s lesions in bladder pain syndrome by Sarah Neu, Jennifer A Locke, Karla Rebullar, Lesley Carr and Sender Herschorn in Journal of Clinical Urology
Footnotes
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The protocol for this research project has been approved by a suitably constituted Ethics Committee of the institution and it conforms to the provisions of the Declaration of Helsinki (Sunnybrook Health Sciences Centre, REB# SUN-2565).
Informed consent
Not required in accordance with the below mentioned REB approval.
Guarantor
S.N.
Contributorship
S.N. and S.H. researched literature and conceived this study. S.N. developed the protocol, gained ethical approval, collected the data, and completed data analysis. S.N., J.L. and K.R. completed the manuscript. All authors reviewed and edited the manuscript and approved the final version.
Disclosures
Dr S.H. reports grants from Astellas Pharma Canada, during the conduct of the study; and grants from CIHR, grants from Allergan, grants and other from AMI, other from Boston Scientific, and other from Pfizer, outside the submitted work.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
