Abstract
Objective:
Clinicians have shown variable practice in the diagnosis and management of bladder pain syndrome (BPS). This study assessed pain localisation sites, common co-morbidities, investigations and treatment patterns in clinical practice.
Patients and methods:
We performed a retrospective analysis of 412 patients attending our pelvic pain clinic between 2004 and 2016. Frequency counts were used to summarise findings.
Results:
Pain in women (
Conclusion:
This study demonstrates that patient expectation management and education is essential in BPS, with most achieving good pain control with conservative measures and amitriptyline. Those receiving intravesical treatments will most likely require subsequent revisions.
Level of evidence:
Not applicable for this single-centre audit.
Introduction
In 2014, the American Urological Association described bladder pain syndrome (BPS) as ‘the complaint of suprapubic pain related to bladder filling, accompanied by other symptoms such as daytime and night-time frequency, in the absence of proven urinary infections or other obvious pathology’. 1 The International Continence Society previously reserved the diagnosis of interstitial cystitis (IC) for patients ‘with typical cystoscopic and histological features’. 2 A distinctive feature of BPS is pain on bladder filling and relief on voiding associated with urinary urgency, frequency and nocturia. Pain can be difficult to localise. However, the suprapubic area, groin and lower back are typically affected. Some diagnostic overlap exists with overactive bladder syndrome and urethral syndrome. BPS is often misdiagnosed as arising from other causes of chronic pelvic pain (Table 1), 3 with its true prevalence often underestimated. BPS is a severely disabling condition affecting approximately six to seven million women in Europe at a 10:1 female-to-male ratio. 4 More than 55% IC patients reported depression, and >50% were unable to work full time, 5 demonstrating its emotional and financial burden.
Differential diagnosis and investigations in BPS.
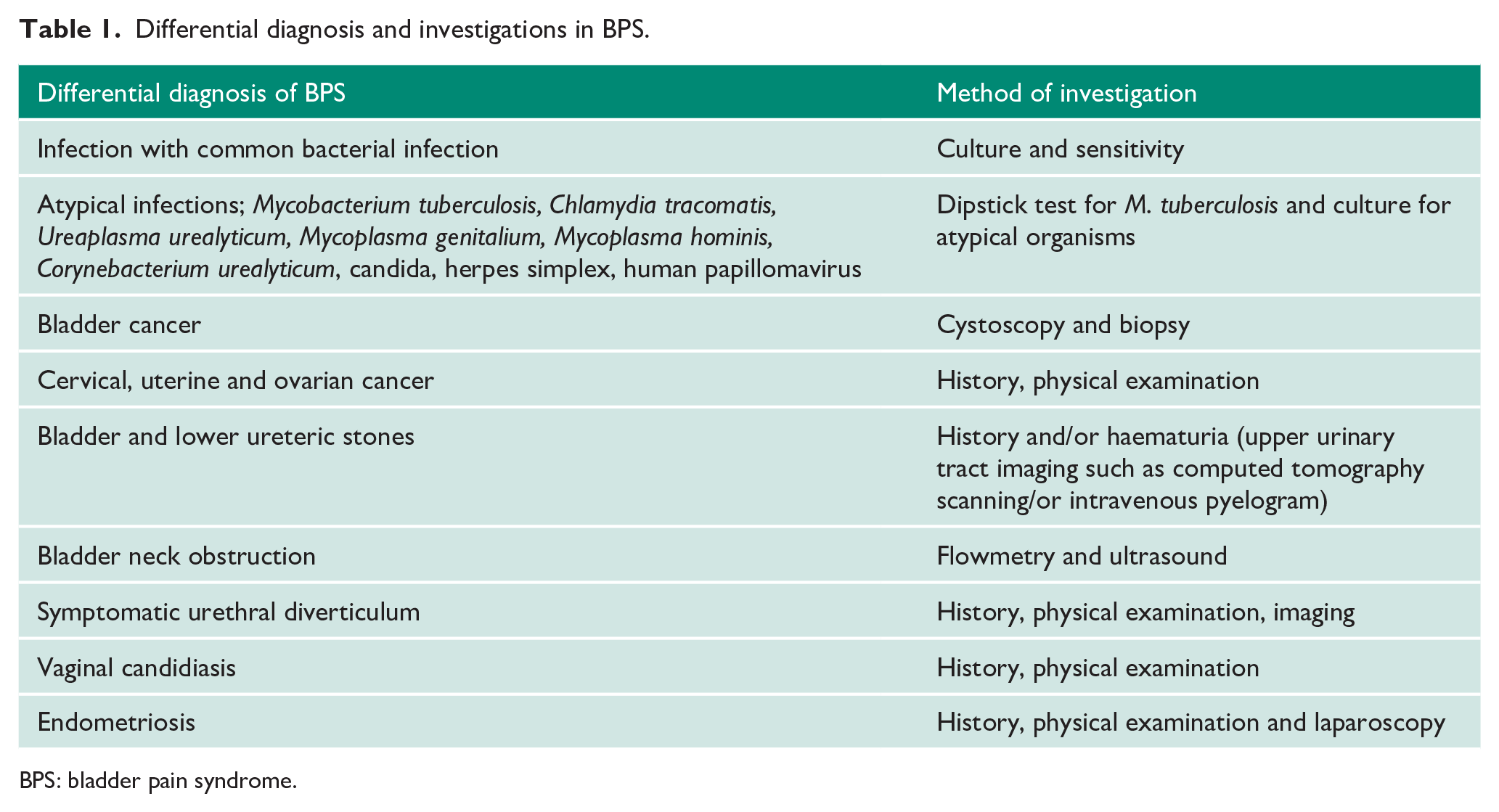
BPS: bladder pain syndrome.
There are several proposed aetiological theories underlying BPS. Cystoscopy and biopsy have demonstrated a defect in the urothelium’s protective glycosaminoglycan layer. 6 This can lead to bladder musculature hyper-permeability, with potassium influx triggering pain and urgency. Biopsies from inflammatory lesions on cystoscopy usually confirm non-specific inflammation. BPS is rarely isolated and has been associated with certain human leucocyte antigen groups and autoimmune and neuropathic pain disorders. 7 With limited data on BPS in the UK and clinicians showing variable practice regarding its diagnosis and management, 8 we aimed to review initial pain localisation, common co-morbidities, investigations and treatment patterns for BPS patients in clinical practice.
Methods
A retrospective single-centre analysis was conducted of patients diagnosed with BPS. This analysis was performed on patients attending our pelvic pain clinic between 2004 and 2016. Patients’ electronic record and clinic letters were reviewed. Data on presenting symptoms, particularly pain localisation sites, as well as overall duration of pain and duration of flare-ups, were collected. Records were reviewed to determine common autoimmune and visceral neuropathic pain co-morbidities, with eczema, psoriasis or allergic conditions of the skin categorised as skin lesions.
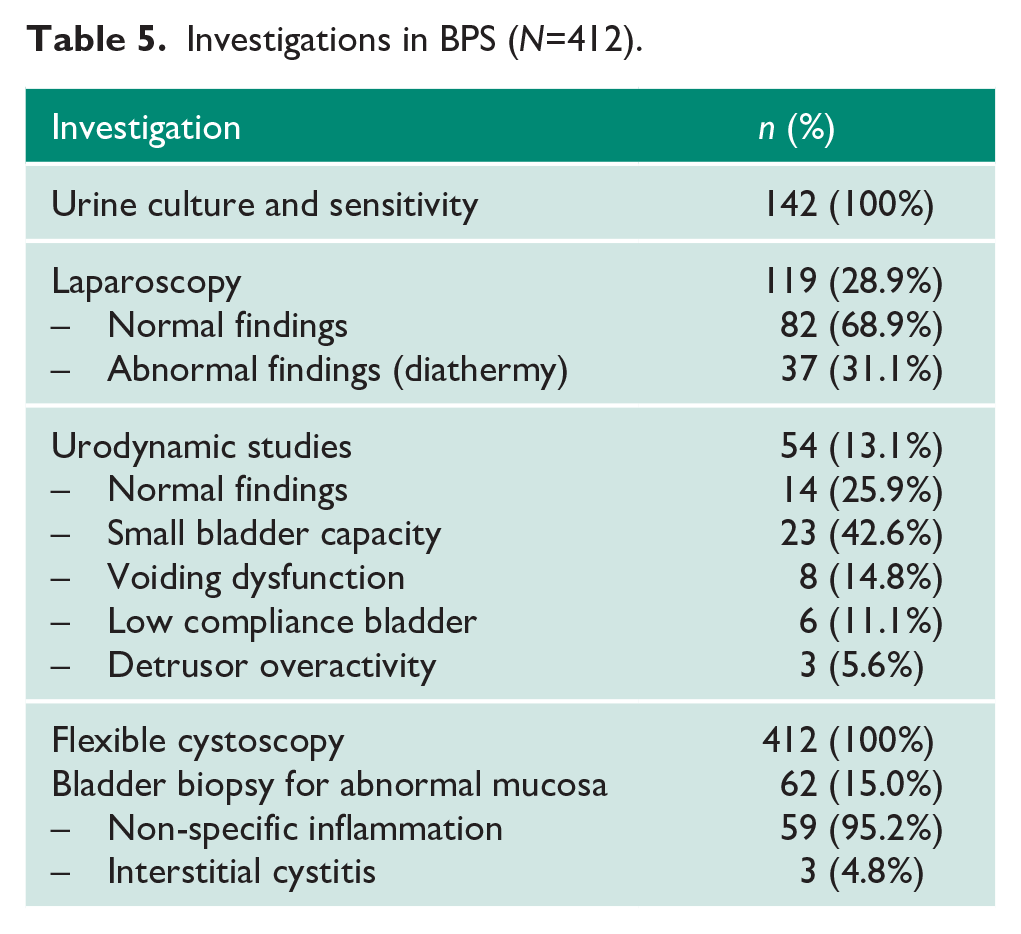
Frequency counts were used to summarise the methods of investigations and findings in our cohort. All patients had urine culture and sensitivity to exclude any possibility of acute chronic bacterial cystitis. Laparoscopy findings by our gynaecology colleagues were categorised as normal or abnormal, with the latter receiving diathermy for scattered endometriosis. Urodynamic studies are not routinely performed in BPS patients. However, the decision to investigate patients with urodynamic studies further was based on the severity of bladder overactivity (frequency >12 times during the day and more than three or four times during the night) refractory to conservative treatment. Urodynamic study findings were classified as normal, small bladder capacity <230 mL, voiding dysfunction in relation to poor flow with high abdominal straining but normal voiding pressure, low compliance bladder and detrusor overactivity. In conjunction with the fact that 230 mL could be considered approximately 50% of normal capacity, we therefore used this urodynamic figure as a marker of a small-capacity bladder. Flexible cystoscopy was performed in all patients; those with abnormal looking mucosa were recalled for bladder biopsy under general anaesthesia. A deep bladder biopsy was taken from any red or ulcerative mucosa, with two additional biopsies from healthy mucosa. Histology findings for abnormal mucosa were classified as non-specific inflammation or IC with detrusor mastocytosis (>28 mast cells/mm2).
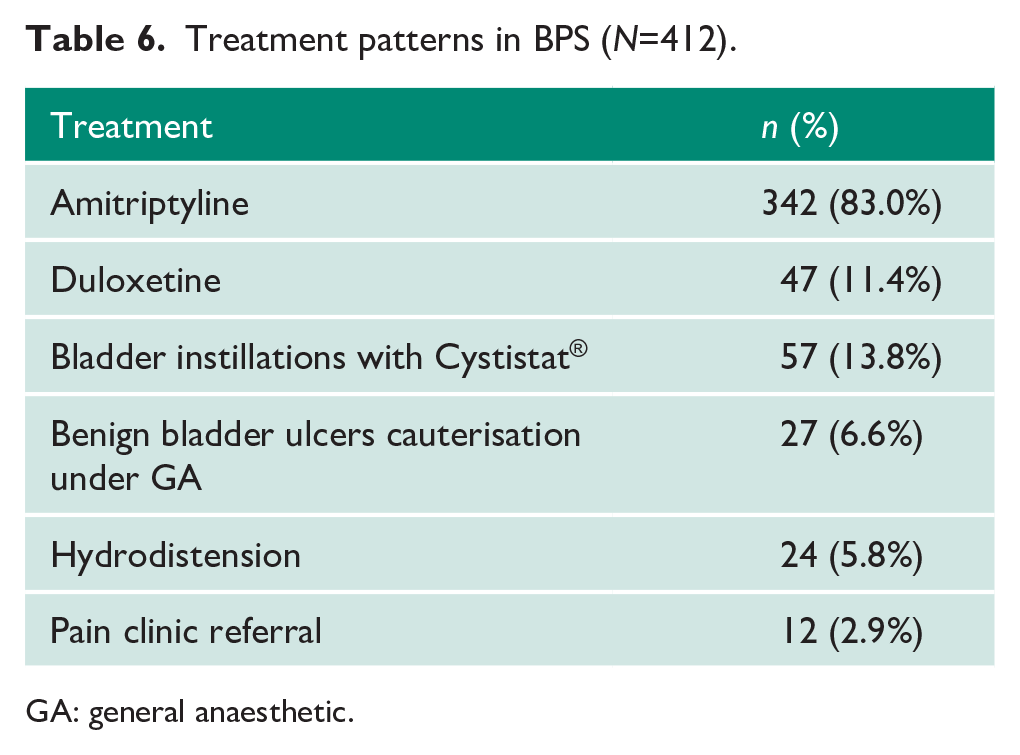
Frequency counts were also used to summarise treatment patterns in our cohort: amitriptyline, duloxetine, pain clinic referrals, bladder instillations with Cystistat® (Teva UK Ltd, Castleford, UK; 40 mg dose in a 50 mL solution), cystoscopy with hydro-distension, benign bladder ulcers cauterisation under general anaesthesia, Botox injections, pentosan polysulfate sodium (PPS; Elmiron®) and surgery. When conservative measures failed, we recommended a starting dose of 10 mg amitriptyline q.h.s., gradually increasing over two to three weeks to the 25 mg maintenance dose. A maximum of 50 mg q.h.s. for three to five days was recommended during flare-ups. Patients were educated that symptoms may improve four to six weeks after commencing pharmacological treatment. For those who failed to respond, we recommended duloxetine at a starting dose of 60 mg, which could be increased to 60 mg b.i.d. if required. Cystistat® consisted of four weekly instillations followed by monthly therapy until symptoms resolved. Patients with two to four benign bladder ulcers on cystoscopy underwent cauterisation under general anaesthesia, with the majority advised to reduce or stop amitriptyline after each cauterisation.
Results
Patient characteristics
A total of 687 patients attended our pelvic pain clinic between 2004 and 2016. Of these, 275 patients were excluded due to insufficient retrospective data, leaving 412 patients for analysis. The mean age was 32 years (range 17–49 years). A total of 388 (94.2%) patients were female, and 24 (5.8%) were male. The mean overall duration of pain was 4.2 years (range 4 months–8 years), with a mean flare-up duration of five weeks (range 1–14 weeks). The mean flare-up for our cohort was three times a year (range 2–6 times). Flare-ups were short in duration, with a mean of three days (range 2–7 days), and patients reported minimal effects on quality of life, work or physical activity.
Pain localisation sites
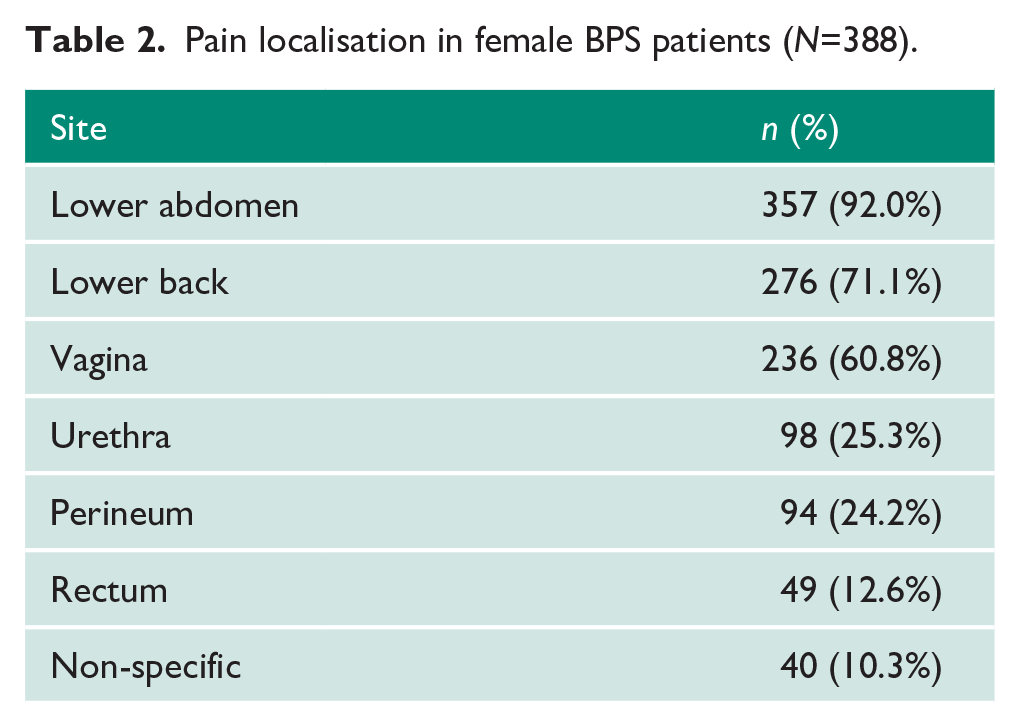
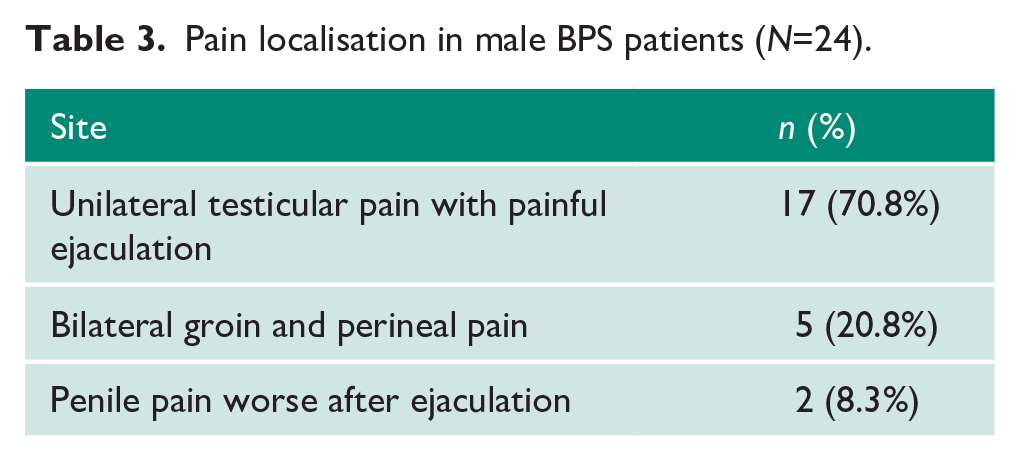
The 388 women reviewed predominately presented with lower abdominal pain (92.0%). However, pain was often localised to secondary sites: lower back (71.1%), vagina (60.8%), urethra (25.3%), perineum (24.2%), rectum (12.6%) and non-specific sites such as the arm, thigh or upper chest (10.3%; Table 2). A total of 17/24 (70.8%) men presented with unilateral testicular pain with painful ejaculation, with 5/24 (20.8%) presenting with bilateral groin and perineal pain and 2/24 (8.3%) presenting with penile pain worse after ejaculation (Table 3).
Pain localisation in female BPS patients (
Pain localisation in male BPS patients (
Co-morbidities
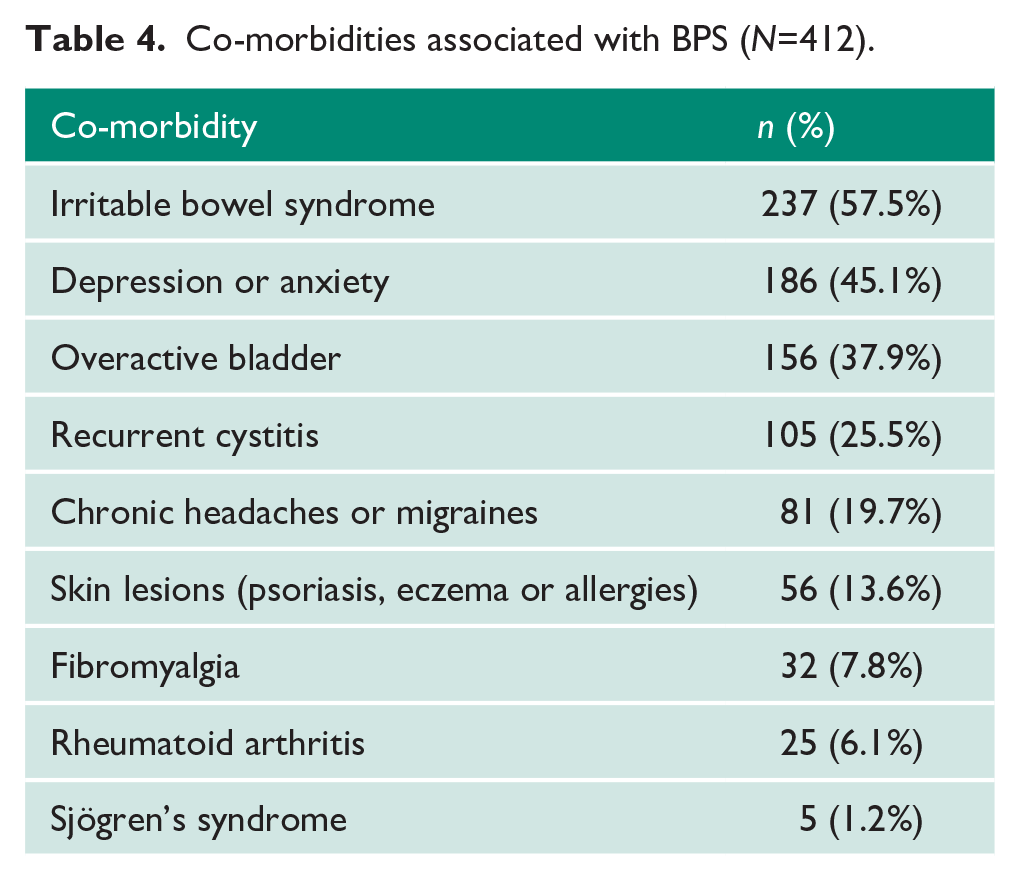
A total of 393/412 (95.4%) patients presented with sexual dysfunction. Of all 412 patients, common visceral neuropathic pain and autoimmune co-morbidities included: irritable bowel syndrome (57.5%), depression or anxiety (45.1%), overactive bladder (37.9%), recurrent cystitis (25.5%), chronic headaches or migraines (19.7%), skin lesions (13.6%), fibromyalgia (7.8%), rheumatoid arthritis (6.1%) and Sjögren’s disease (1.2%; Table 4).
Co-morbidities associated with BPS (
Investigations
On initial assessment, all 412 patients had urine culture and sensitivity performed (Table 5). A total of 119/388 (30.7%) women were assessed by our gynaecology colleagues with laparoscopy, with normal findings in 82/119 (68.9%). Those women with abnormal laparoscopy findings (37/119; 31.1%) reported little symptomatic relief following diathermy for scattered endometriosis. A total of 54/412 (13.1%) patients were investigated with urodynamic studies: 14/54 (25.9%) had normal findings, 23/54 (42.6%) had small bladder capacity, 8/54 (14.8%) had voiding dysfunction, 6/54 (11.1%) had a low compliance bladder and 3/54 (5.6%) had evidence of detrusor overactivity. All 412 patients had flexible cystoscopy performed, with 62/412 (15.0%) recalled for bladder biopsy for abnormal mucosal appearances. Of these, three (4.8%) had histologically confirmed IC on bladder biopsy. In our series, no patients had the potassium sensitivity test due to post-instillation pain.
Investigations in BPS (
Management
Most patients were advised to use alkalising agents, such as sodium bicarbonate sachets two to three times a day, to ease bladder pain during flares, with some also using short-acting narcotic medications, such as paracetamol, codeine and opioids. Patients were also advised to avoid potential triggers such as alcohol, fluids containing citric acid, strong coffee, carbonated drinks, fresh tomatoes and spicy foods. Patients were either treated with single agents or a combination. A total of 342/412 (83.0%) patients and 47/412 (11.4%) patients achieved good pain control on amitriptyline and duloxetine, respectively (Table 6). A total of 57/412 (13.8%) patients had additional bladder instillations with Cystistat®, with 49/57 (86%) on monthly instillations with some improvement for 28 months. Benign bladder ulcers were cauterised in 27/412 (6.6%) patients, alleviating pain for an average of 6.8 months (range 4–16 months). However, 18/27 (66.7%) patients required two to three re-cauterisations. No patients required more than three cauterisations. Temporary symptomatic relief of three to four months was achieved for 24/412 (5.8%) patients undergoing hydro-distension. A total of 12/412 (2.9%) patients failed to respond to these treatment modalities and were therefore referred to local pain clinics for further assessment and treatment. No patients in our cohort were treated with PPS, Botox injections or surgery, which is only recommended in severe refractory forms of BPS.
Treatment patterns in BPS (
GA: general anaesthetic.
Discussion
In our series, the presence of other autoimmune or neuropathic pain disorders may raise the clinical suspicion of BPS, after exclusion of other common causes of chronic pelvic pain. Subsequent investigations, such as laparoscopy, urodynamic studies, cystoscopy and bladder biopsy, mostly yielded normal findings. In primary care, symptom validation and education on conservative measures were critical in the management of BPS. Most patients achieved good pain control on oral amitriptyline. Bladder instillations with Cystistat® have been shown to be effective, with temporary symptomatic relief achieved from hydro-distension and cauterisation.
A systemic review of IC symptoms reported pain (63–92%), urgency (84–98%), frequency (80–92%), nocturia (61–89%), dyspareunia (45–57%) and incontinence (26%). 9 They also found similar pain localisation sites, supporting our data. Prevalence of sexual dysfunction varies across the literature due to differing definitions; we defined this as vulvodynia, dyspareunia or painful ejaculation. Although pain forms part of the definition, one study showed that 41% patients with early IC did not initially experience pain, but rather urgency, frequency and/or nocturia. 10 As many as 41% of BPS patients experienced one episode of haematuria over 18 months. 11 This demonstrates the broad spectrum of symptoms in BPS. A study into the prevalence of 12 concomitant diseases in 2682 IC patients noted a higher frequency of allergies (40.6%), irritable bowel syndrome (25.4%), sensitive skin (22.6%) and fibromyalgia (12.8%) compared to the general population. 12 Similar findings regarding these common visceral neuropathic pain and autoimmune disorders were seen in our cohort.
In a study of 150 patients undergoing laparoscopy for pelvic pain lasting more than six months, 75% were diagnosed with endometriosis, 43% of whom also had BPS.
13
After investigative laparoscopy and subsequent endometriosis treatment, all groups experienced a significant reduction in pelvic pain, regardless of the presence or absence of endometriosis. This contrasts with our cohort, where 30.7% (
On urodynamic studies, patients often demonstrate hypersensitivity on bladder filling, a reduced first sensation to void and reduced bladder capacity, in keeping with our data. Only 13.1% of our patients had urodynamic studies performed, with 25.9% displaying normal findings. Detrusor overactivity, seen in 5.6% of our cohort under urodynamic studies and 14% IC patients in another study, 14 does not exclude BPS. Cystoscopy without hydro-distension is predominately normal in BPS patients, with 85.0% of our cohort demonstrating healthy mucosa on cystoscopy. Interestingly, only 4.8% of our cohort with bladder biopsies had features of IC. Cystoscopic features include post-distension glomerulations, reduced bladder capacity and bleeding, whilst histology shows detrusor mastocytosis, haemorrhage and fibrosis, which have been poorly correlated to symptom severity. 15 The role of cystoscopy and biopsy is limited to excluding other pathologies and can identify Hunner lesions in type 3 BPS.
There is no definitive treatment for BPS, with 183 different therapies recorded. 16 In primary care, acknowledgement and validation of symptoms and setting expectations are critical. Conservative measures include physiotherapy, bladder retraining exercise and stress management. Reducing exposure to aggravating factors can alleviate symptoms, such as stress (61%), sexual intercourse (50%), acidic beverages (54%) and coffee (51%). 17 If this fails, oral amitriptyline is recommended. A systematic review of two randomised controlled trials involving 281 patients showed improvements in lower urinary tract symptoms when treated with increasing titrated doses of amitriptyline between 10 and 100 mg over four months. 18 In our series, 342/412 (83.0%) patients achieved good pain control on our carefully titrated regime.
In keeping with our data, bladder instillations with Cystistat® have been shown to be effective in BPS, with a number needed to treat of 1.31. 19 Benefits of hydro-distension were short-lived in our series, with efficacy ranging from 22% to 67% and reports of bladder rupture.20,21 Most patients with cauterisation of benign bladder ulcers required two to three revisions; a similar cohort of 42 IC patients treated with ablative therapy for Hunner lesions reported symptomatic relief in 88% patients within two to three months, and 45% required additional treatment within 23 months. 22 No patients in our cohort received PPS, with National Institute for Health and Care Excellence guidance stipulating specific indications of use, including secondary care management of patients with moderate to severe pain with glomerulations or Hunner’s lesions refractory to oral treatment and bladder instillations. 23 Its side effect profile of diarrhoea, vomiting, rectal bleeding and alopecia makes it an unfavourable treatment option. Furthermore, there have been reports of no statistical significance between oral PPS and placebo, 24 with some patients developing maculopathy after a median treatment duration of 186 months. 25 A total of 12/412 (2.9%) patients in our cohort failed to respond to these treatment modalities and were subsequently referred to local pain clinics for further assessment and treatment.
Our large retrospective analysis of 412 patients evaluates the initial presenting symptoms, co-morbidities, investigations and treatment patterns of BPS seen in clinical practice over a 12-year period. Early exclusion of common chronic pelvic pain pathologies and implementation of conservative measures and pain relief is essential.
Conclusion
BPS is a chronic, disabling diagnosis of exclusion, predominately presenting in females with lower urinary tract symptoms and pain on bladder filling with relief on voiding. BPS has significant psychological and financial implications. Therefore, a multidisciplinary approach must be adopted to exclude other causes of chronic pelvic pain early. In primary care, patient expectation management and education is essential, with most patients achieving good pain control with conservative measures and oral amitriptyline. Those referred to secondary care receiving intravesical treatment will most likely require subsequent revisions.
Footnotes
Acknowledgements
None.
Conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because it was conducted retrospectively from data obtained for clinical purposes.
Informed consent
Informed consent was not sought for the present study because it was conducted retrospectively.
Guarantor
H.R.H.P.
Contributorship
W.A. and H.R.H.P. reviewed the literature, conceived the study and were responsible for data collection. W.A. and R.P. wrote the first draft of the manuscript. U.S. and H.R.H.P. edited the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.
