Abstract
Granulomatosis with polyangiitis (GPA) is an autoimmune small to medium vessel inflammatory vasculitis that can affect multiple organ systems, with predominantly pulmonary, renal and musculoskeletal manifestations. Rarely, there have been cases involving the genitourinary system, particularly causing urethritis and prostatitis. In some incidences, this may mimic prostate abscess or malignancy. We present a case of a 36-year-old man with refractory urinary retention secondary to prostate abscess due to GPA. This case necessitated transurethral resection of the prostate (TURP) and supra-pubic catheterisation, later complicated by a peri-anal abscess and severe urethritis. A potential recto-urethral fistula was noted on magnetic resonance imaging (MRI) and cystourethroscopy, which was managed conservatively with long-term catheter drainage until resolution. The patient ultimately displayed many severe clinical manifestations of a systemic vasculitis and this case report emphasises the importance of considering autoimmune vasculitis conditions as a differential diagnosis when managing inflammatory genitourinary conditions. This case report highlights the importance of obtaining sufficient tissue to make a timely diagnosis in order to initiate immunosuppressant therapy and disease-modifying anti-rheumatic drugs (DMARDs).
Case report
A fit and well 36-year-old man sought a private virtual consultation in the middle of the 2020 COVID-19 pandemic because of mixed storage and voiding urinary symptoms along with dysuria. He subsequently developed acute retention of urine and was noted to have a small tender prostate on digital rectal examination (DRE). A provisional diagnosis of prostatitis was made and antibiotic therapy was commenced while awaiting magnetic resonance imaging (MRI) of the prostate. He was seen face-to-face in a National Health Service (NHS) outpatient clinic shortly after where his repeat DRE revealed a tender fluctuant prostate, suspicious of abscess formation, which matched the appearance of his MRI. The gland was wholly abnormal, graded LIKERT 5, with abnormal peripheral and transitional zones raising the suspicion of either severe inflammation or malignancy.
Two weeks later, the patient presented with pyrexia and shortness of breath. Chest x-ray and thoracic computed tomography (CT) revealed multiple bilateral cavitating lung lesions. Testing for acid-fast bacilli (AFB) was negative from numerous sputum cultures. Due to his significant symptoms from the indwelling urethral catheter, inability to void spontaneously and his recent MRI, a transurethral resection of the prostate (TURP) was performed.
At cystourethroscopy, florid necrotic tissue was seen in the entire prostatic urethra to the external urethral sphincter at the level of the verumontanum. The abnormal prostatic tissue was excised to gain ample tissue for histological analysis, and in doing so, a large cavity full of amorphous material was de-roofed, which extended cranially and posteriorly under the bladder neck. Post-operatively, the patient successfully voided after catheter removal.
Histology from the TURP was difficult to interpret but was made easier by the presence of ample tissue for analysis. Sections showed inflammatory necrosis with multinucleate giant cells and granulomatous infiltrates, as well as micro-abscesses (Figures 1 and 2). Differentials included tuberculosis (TB), other atypical infection, inflammatory tumour and vasculitis. Slides tested negative for AFB using Ziehl–Neelsen staining, and negative periodic acid-Schiff staining investigating for fungal infection. Elastin Van Gieson (EVG) stains which can highlight a vasculitis process was suggestive but not diagnostic. GPA vasculitis was suspected because of the distribution of the cavitating lung lesions and raised c-ANCA and anti-PR3 levels, as well as his previously reported back and pelvic pain.
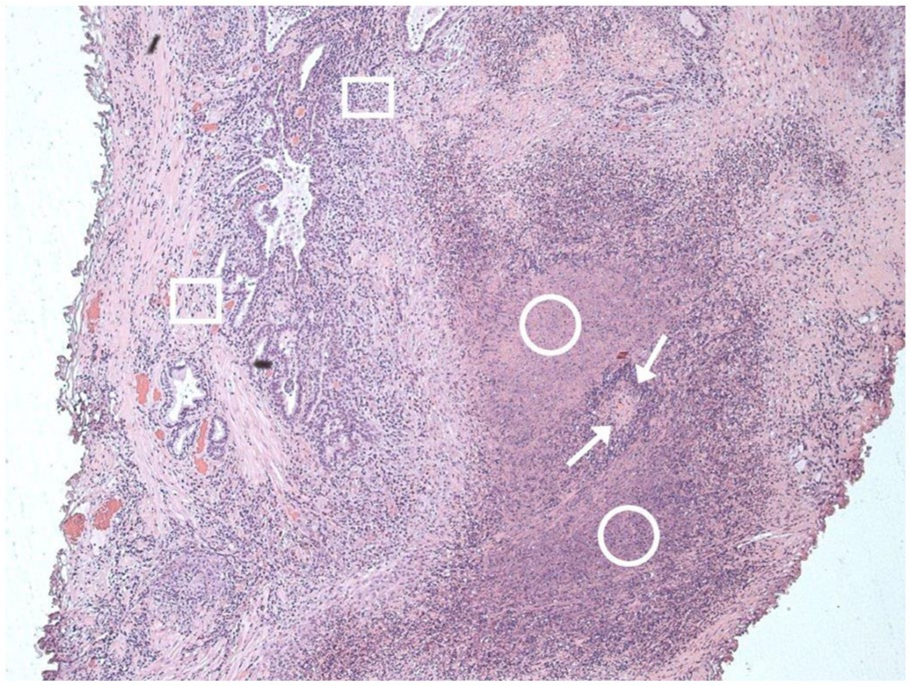
H&E staining, 125× power.
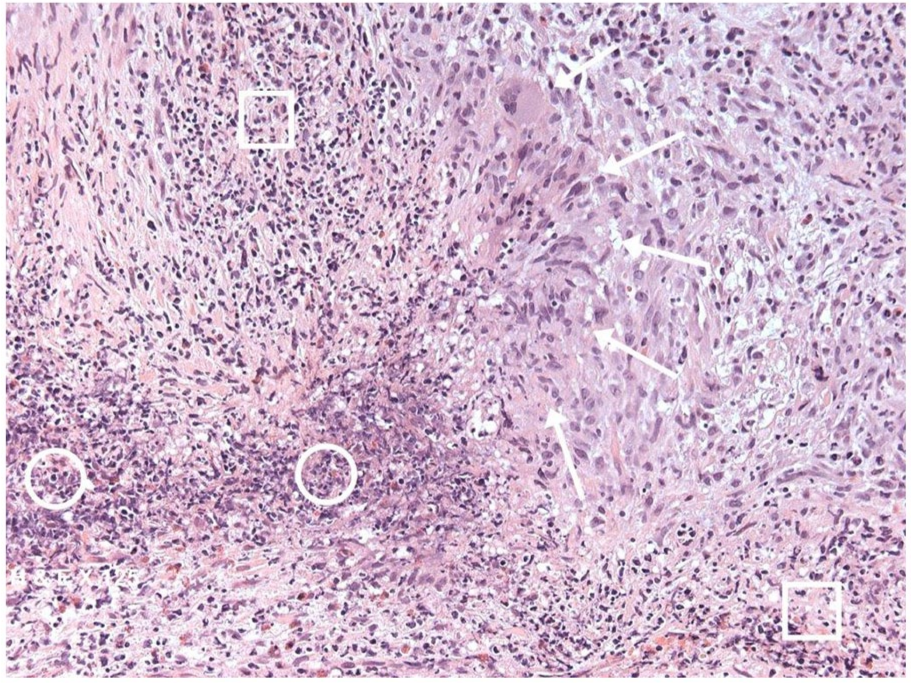
H&E staining, 400× power.
Rheumatology discharged the patient after starting high-dose oral prednisolone (60 mg per day) and a plan was made for regular outpatient review.
Within 3 weeks, Mr X went on to develop peri-anal pain, swelling and rectal discharge. A diagnosis of peri-anal abscess was made by the admitting surgical team; however, due to his recent TURP, a pre-operative MRI was obtained (Figure 3). This identified a large high T2 signal abnormality in the left inter-sphincteric plane beside the anal canal, which extended up to the anorectal junction and appeared connected with the prostatic urethra, suggestive of fistulation. This prompted an incision and drainage of peri-anal abscess along with examination under anaesthetic (EUA), which could not identify any obvious internal or external fistulous tract. The abscess cavity was washed out and packed, and clinically his symptoms resolved with oral antibiotics and district nurse-led packing changes.
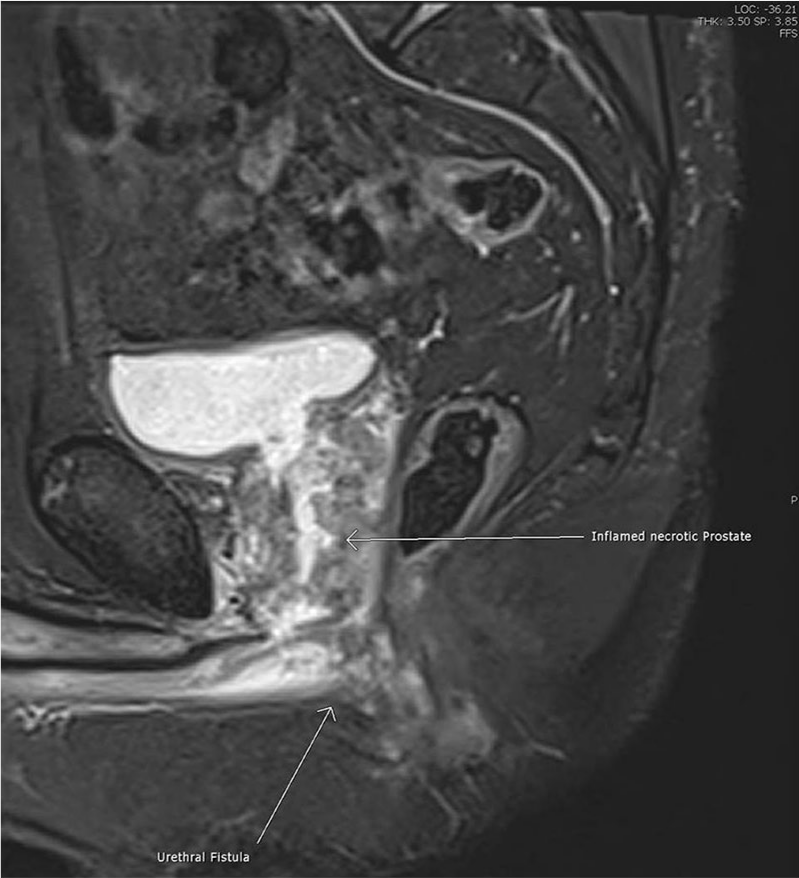
MRI pelvis STIR image T1 weighting – necrotic prostate tissue with fistula tract demonstrated.
One week after discharge, the patient re-presented with severe dysuria due to the passage of pus and debris urethrally and imminent urine retention. Due to the level of discomfort, the patient was taken to theatre for an EUA, cystoscopy and supra-pubic catheter (SPC) insertion. EUA revealed no residual peri-anal abscess and no new perineal swelling. Cystoscopy revealed a penile urethral flap, downstream of the sphincter, at the level of the bulbar urethra which was thought to be the fistulous tract to have communicated with the previous peri-anal abscess. The prostate cavity appeared to have healed satisfactorily. An SPC was inserted and a urethral catheter left in situ. Rheumatology started the patient on mycophenolate mofetil and advised slow tapering of oral prednisolone.
A month later, Mr X re-presented with bleeding and pain around the urethral catheter. Retrograde cystourethrography (RGU) alongside the urethral catheter revealed an irregular anterior penile urethra with contrast ascending to the bladder, with a small, blind-ending sinus tract extending posteriorly that was felt to be a healed fistula (Figure 4). Subsequent MRI of the perineum confirmed the irregular, inflamed anterior urethra and identified the posterior sinus tract from the bulbar urethra towards the peri-anal region. These appearances were postulated to be due to complications of severe urethritis.
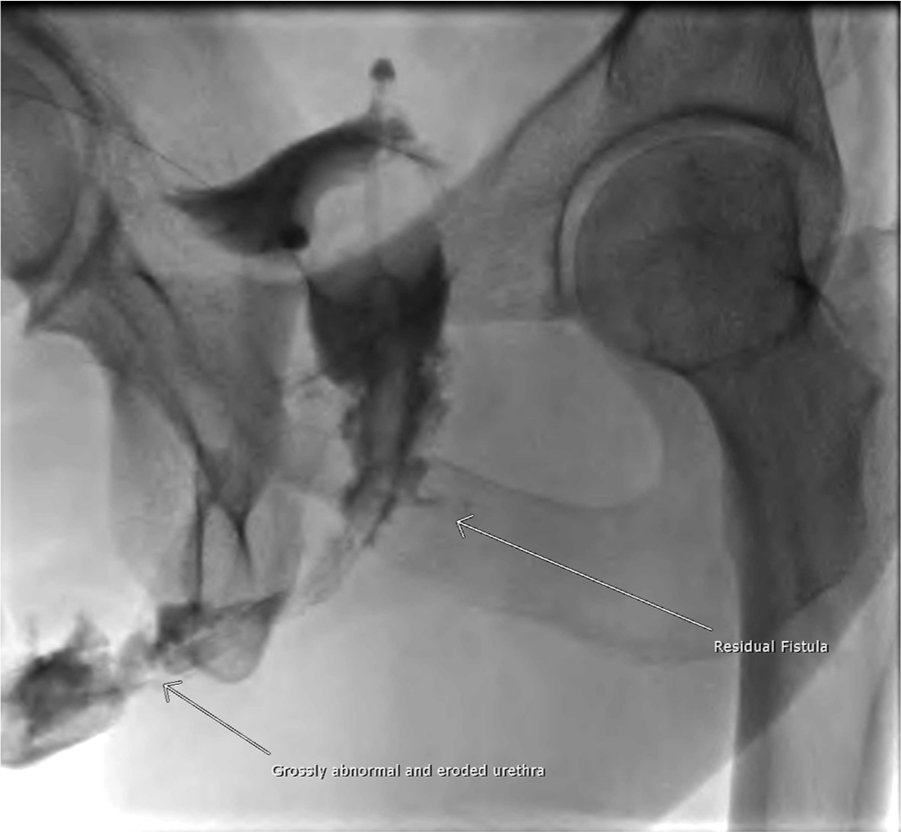
Plain x-ray cystourethrogram using Onnipaque 300 contrast – grossly abnormal urethra with residual fistula seen.
The patient was later commenced on rituximab infusions along with long-term antibiotic prophylaxis in order to maintain sterile urine, a pre-requisite for the safe administration of disease-modifying anti-rheumatic drugs (DMARDs). The patient made a gradual improvement and successfully voided after removal of his urethral catheter at the next attempt.
Discussion
GPA can affect multiple organ systems; however, the literature available on urethral and prostatic vasculitis is limited to a few case reports. Cases have been shown to very rarely cause urinary retention and prostatitis. 1 In our literature review, this is the first case of prostatic GPA causing urinary retention and prostate abscess, along with anorectal–urethral fistula and cavitating lung lesions.
This case highlights the importance of early accurate diagnosis to aid timely treatment with DMARDs. Our patient had prostatic and peri-anal abscess which required surgical treatment, which may have been avoidable if systemic therapies had been started earlier. Literature review does identify a clear link with GPA and prostatitis and prostatic abscess.2,3 These cases also failed to resolve with typical supportive antimicrobial or operative treatment and did require immunosuppression to gain remission. 4 The cases similarly identify the delay in diagnosis due to the rarity of the link between GPA and inflammatory prostatitis or abscess.
The peri-anal abscess and urethral fistula, to the best of our knowledge, have not been identified as linked to GPA in the literature. Recto-urethral fistulae (RUF) are often secondary to trauma, radiation, post pelvic/prostate surgery and chronic inflammation. 5 In this circumstance, his symptoms developed post TURP, and the fistula itself was between the peri-anal and rectal region connecting to the bulbar urethra. The fistula was effectively managed conservatively. Literature review of the topic identified no standard of care for RUF, but did advocate that smaller, less symptomatic fistulae did not necessitate invasive surgery such as faecal or urinary diversion. 6 One study of post prostatectomy patients advocated conservative RUF management with catheter drainage, parenteral and limited enteral feeding alongside antibiotics. Fistulae were not evident on RGU at 4 weeks post treatment. 7 For Mr X, DMARDs and antibiotic usage to maintain sterile urine, as well as diverting urine away from the site of the fistula with catheters, encouraged the spontaneous healing and closure in roughly the same timeframe.
There is a documented link between GPA and bladder outflow obstruction. The cause appears multifactorial, likely pain associated with the inflammatory urethritis and prostatitis, as well as an element of bladder outflow obstruction from prostatic swelling and inflammation. 8 Urinary retention may develop, which responds to DMARDs and rituximab, but this does appear rare and the extent of the literature on treatment length is not clear. 9
Conclusion
GPA is a complex multi-system vasculitis that rarely involves the prostate and urethra. We conclude it is important to maintain a clinical suspicion of vasculitis in the context of inflammation and infection without obvious underlying cause. This report underlines the complicated nature of GPA involving a fistulating disease between ano-rectum and urethra, which was managed conservatively with success. RUF in the context of GPA is unique and so this paper should prompt further inquiry into management options and further research. We also document bladder outflow obstruction in this case which may resolve, as per the available evidence, once GPA remission is achieved with immunosuppression. We therefore emphasise the importance of close correspondence with urology and rheumatology in these difficult cases.
Footnotes
Acknowledgements
We would like to thank Dr Majid Rashid (Histopathology Consultant, Royal Gwent Hospital) with assistance in gathering the pictured histology for this paper.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Royal Gwent Hospital does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
Guarantor
A.C.O. is guarantor of this article.
Contributorship
M.R. wrote the initial and final drafts. A.C.O. reviewed the final manuscript.
