Abstract
Abscess of the corpus cavernosum is a rare condition. We describe the case of a 36-year-old male presenting with bilateral corpus cavernousm abscesses, on a background of distal shunt procedures for recurrent priapism, that was managed with a bilateral corporotomy and systemic antibiotic therapy.
Keywords
Case history
A 36-year-old male presented to the emergency department with a four-day history of drug-induced priapism. His medical history included depression and asthma. The patient had no history of diabetes or steroid use. His regular medications consisted of fluoxetine and quetiapine. The patient reported a long-standing history of erectile dysfunction prior to the onset of his recurrent priapism and had taken sildenafil multiple times in the past.
After a failed attempt at cavernosal aspiration with phenylephrine, a Winter’s shunt was performed under penile block. This was successful. However, the patient presented again four days later with recurrent priapism. Cavernosal aspiration failed, and a repeat Winter’s shunt was performed the following day. Intravenous ceftriaxone was administered for each procedure.
The patient absconded from hospital two days later. However, he presented again to the emergency department 14 days after the second Winter shunt. He described a two-week history of penile pain and swelling, with fever developing over the 12 hours prior to presentation. He had been taking pseudoephedrine as instructed at home for his recurrent priapism prior to his third presentation.
On admission, he was tachycardic (heart rate 125 bpm) and had a fever of 38.6°C. Examination revealed a swollen erect penis with erythematous skin. There was an extensive deep mass, which was fluctuant, extending bilaterally to the base of the penis. Testicular and scrotal examinations were normal.
Investigations showed raised inflammatory markers, with a white cell count on presentation of 24.1×109/L and C-reactive protein of 357 mg/L. A bedside ultrasound showed a potential collection within the corpus cavernosum bilaterally. The patient underwent needle aspiration in the emergency department, with a total of 30 mL pus collected for microscopy, culture and sensitivities. He was commenced on intravenous fluids and broad-spectrum antibiotics (ceftriaxone, azithromycin and flucloxacillin in the emergency department and then switched to piperacillin-tazobactam on admission under urology).
Computed tomography revealed a marked fluid distension of the corpora cavernosa bilaterally, with enhancement of the surrounding tunica albuginea. This was associated with a bilobed fluid collection extending across the ventral aspect of the mid-penile shaft suggestive of a bilateral corpus cavernosum abscess (Figure 1).
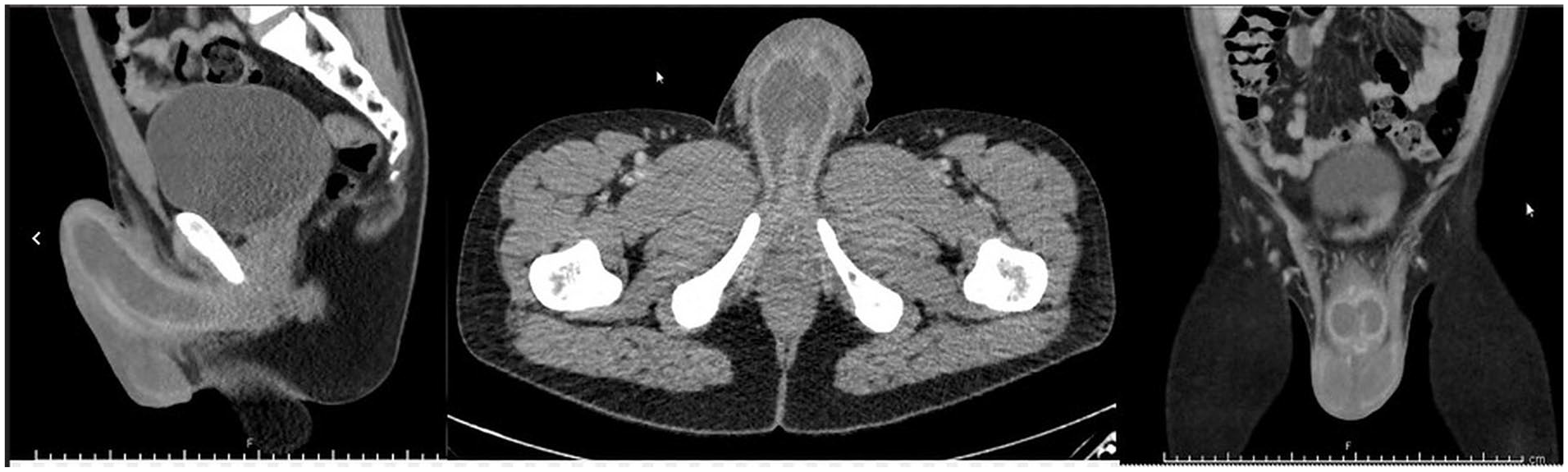
Computed tomography images of corpus cavernosum abscess.
The patient was emergently taken to theatre for a bilateral corporotomy and drainage of the abscess. A longitudinal incision was made near the base of the penis. Artery forceps were inserted to the opposite side, and a separate incision was made over the forceps. Pus was drained, and the corpora were washed out with normal saline. A Penrose drain was inserted and secured using a silk suture and a combine dressing and crepe bandage applied (Figure 2).
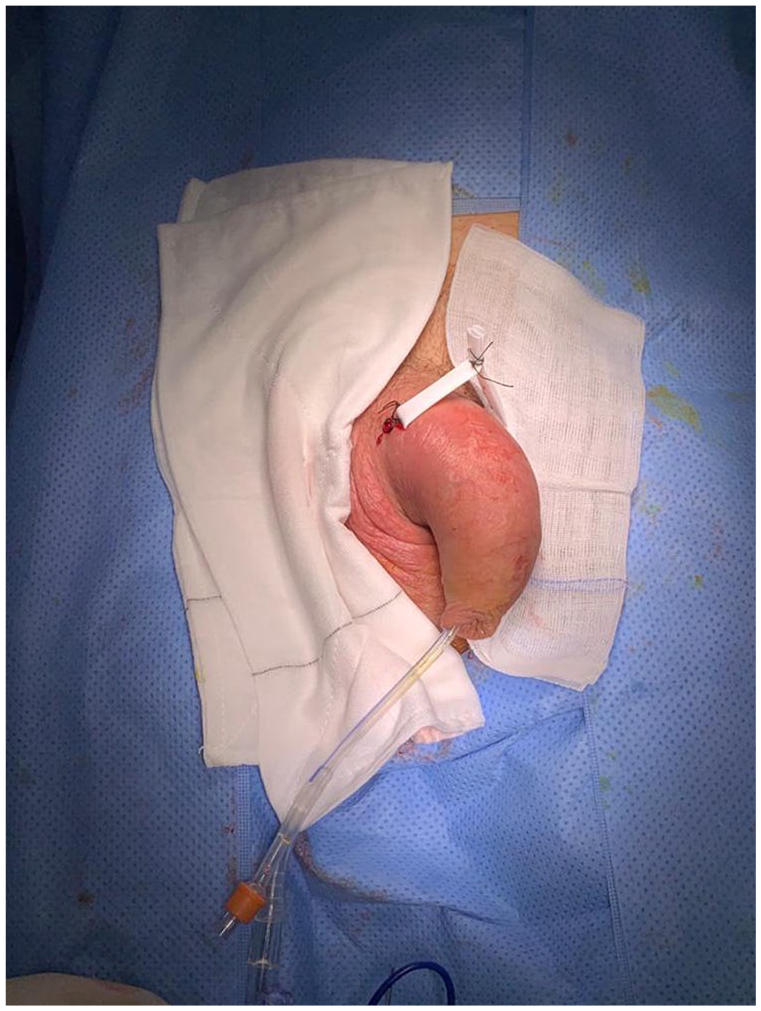
Intraoperative image.
The aspirated pus had Gram-negative bacilli on microscopy and grew a heavy growth of mixed anaerobes on culture. Prevotella bivia was isolated on blood cultures. The patient’s condition improved postoperatively. His tachycardia and fever settled, and his white cell count normalised. He continued on piperacillin-tazobactam for a further four days and BD saline flushes of the Penrose drain until it was removed on postoperative day 4. The patient self-discharged on a four-week course of oral amoxicillin-clavulanic acid and metronidazole. The patient failed to attend any further follow-up.
Discussion
Abscess of the corpus cavernosum is a rare condition. A review of the literature revealed 43 known case reports since 1980 (Appendix 1). They usually present with progressive penile pain and swelling. 1
Cases have been described after trauma, as a complication of cavernosography, as an unusual presentation of gonorrhoea or tuberculosis, following intracorporeal injections, priapism, from local infections or from haematogenous seeding from distant infections, following penile prosthesis placement, as a complication of Winters’ procedure or from indeterminate causes.1–6 Iida et al. proposed corpus cavernosal abscess as an early symptom of penile pyodermal gangrenosum. 7 More than a third of corpus cavernosal abscesses have also been reported as idiopathic. Almost half of the cases reported bilateral corpus cavernosum abscesses, as seen with our patient.
The corpora cavernosa are thought to be resistant to haematogeneous dissemination of infection, the aetiology of which is unclear. 8 However, previous case reports have attributed abscess of the corpus cavernosum to bacteraemia from dental infections. 5 Priapism leads to ischaemia in the corporeal tissue and can make it more prone to infection. 4 As with Paladino et al., pathogens were likely introduced during the multiple procedures performed for priapism.
A number of different pathogens have been the causative organism, perhaps due to the variety of potential aetiologies of corpus cavernosal abscess. Most cases involved Gram-positive cocci. However, a number of cases were caused by Gram-negative bacilli. Staphylococcus aureus, Streptococci, Fusobacteria and Bacteroides are common organisms amongst reported cases. 1 Corpus cavernosum abscesses can also occur from fungal infections 9 as well as Mycobacterium tuberculosis. 6 Three cases of recurrent corpus cavernosum grew MRSA on cultures.1,7,10 P. bivia is an anaerobic Gram-negative bacillus species which resides in the flora of the female genital tract and is occasionally found in the oral flora. 11 It is typically associated with infections of the female urogenital tract and the oral cavity. 12 P. bivia infections have also been found in rare locations such as the bones/joints, nail bed, male external genitalia, endocardium and the chest wall. 11 As the patient admitted to ongoing sexual activity with his recurrent priapism, it is possible that Prevotella became the causative organism as a result of attempted sexual intercourse following the recurrent drainage procedures.
Anaerobic organisms are frequently cultured in suppurative genitourinary tract infections. 13 In our case, ceftriaxone was given prophylactically during the patient’s procedures, which did not provide any anaerobic cover. Greater antibiotic coverage may be required in recurrent priapism with multiple drainages to cover for possible anaerobic infection.
Early surgical intervention with intravenous antibiotics is recommended to prevent serious complications such as necrotising fasciitis and persistent infection requiring a penectomy. Possible complications of surgical drainage can include abscess recurrence, fistulae, poor erectile function and secondary fibrosis leading to penile deviation. 8 Intravenous antibiotics and radiologically guided aspiration could be considered a viable alternative of treating a corpus cavernosum abscess while also reducing the risk of secondary fibrosis associated with corporotomy, with some cases showing successful resolution of the abscess without any complications.14,15 However, given the risk of recurrence with incomplete evacuation of the abscess, surgical drainage remains the mainstay of therapy. 1 Previous case reports have necessitated a penectomy for persistent infections. 10 Previous case reports showed pyoderma gangrenosum as the underlying cause of persistent abscess of corpus cavernosum despite surgical drainage. Initiation of steroid treatment avoided the need for a penectomy. 7
Conclusion
This is only the second case of corpus cavernosal abscess as a result of recurrent priapism and following a Winter’s procedure. Adequate antibiotic coverage, including anaerobic cover, is required with recurrent priapism, requiring multiple drainages to prevent the development of a corpus cavernosal abscess. Early administration of intravenous antibiotics and surgical intervention or radiologically guided aspiration are crucial to avoiding serious complications associated with corpus cavernosal abscess.
Footnotes
Appendix
Summary of corpus cavernous abscess reports in the literature.
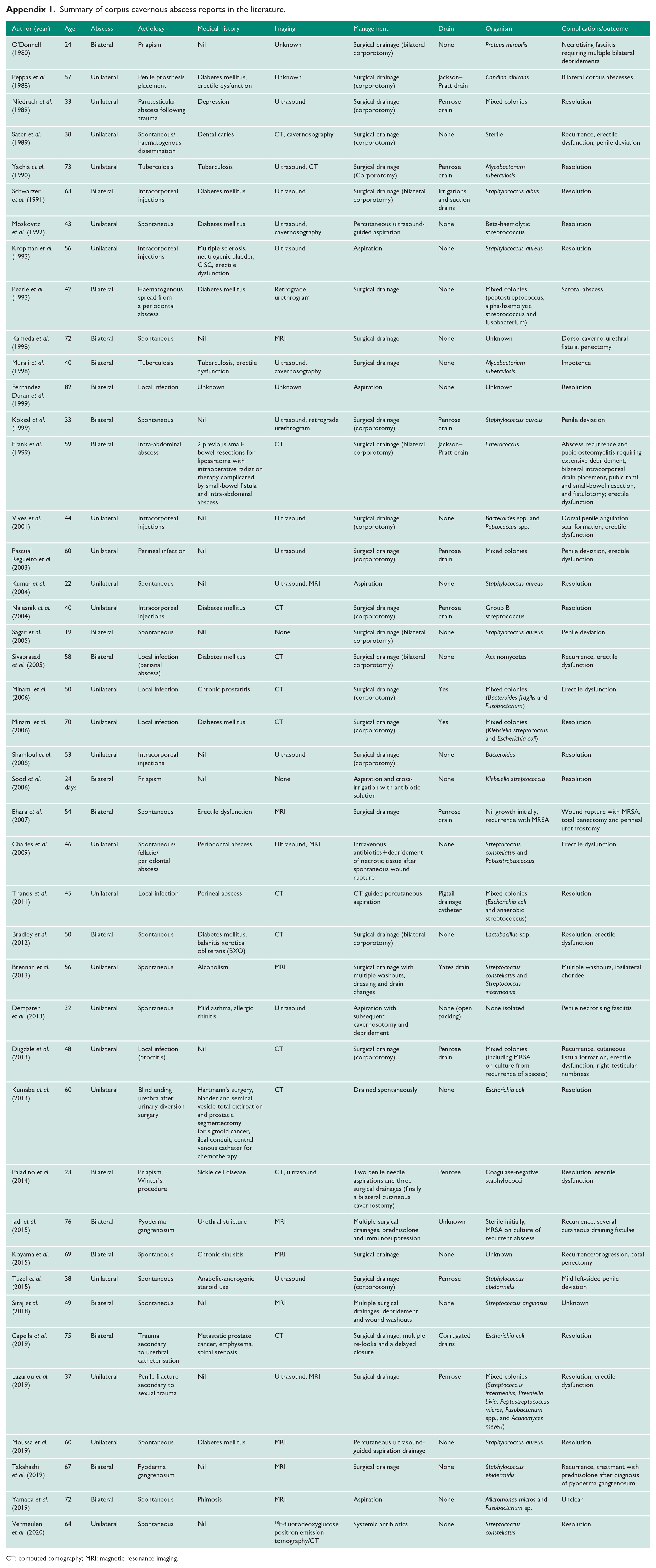
| Author (year) | Age | Abscess | Aetiology | Medical history | Imaging | Management | Drain | Organism | Complications/outcome |
|---|---|---|---|---|---|---|---|---|---|
| O’Donnell (1980) | 24 | Bilateral | Priapism | Nil | Unknown | Surgical drainage (bilateral corporotomy) | None | Proteus mirabilis | Necrotising fasciitis requiring multiple bilateral debridements |
| Peppas et al. (1988) | 57 | Unilateral | Penile prosthesis placement | Diabetes mellitus, erectile dysfunction | Unknown | Surgical drainage (corporotomy) | Jackson–Pratt drain | Candida albicans | Bilateral corpus abscesses |
| Niedrach et al. (1989) | 33 | Unilateral | Paratesticular abscess following trauma | Depression | Ultrasound | Surgical drainage (corporotomy) | Penrose drain | Mixed colonies | Resolution |
| Sater et al. (1989) | 38 | Unilateral | Spontaneous/haematogenous dissemination | Dental caries | CT, cavernosography | Surgical drainage (corporotomy) | None | Sterile | Recurrence, erectile dysfunction, penile deviation |
| Yachia et al. (1990) | 73 | Unilateral | Tuberculosis | Tuberculosis | Ultrasound, CT | Surgical drainage (Corporotomy) | Penrose drain | Mycobacterium tuberculosis | Resolution |
| Schwarzer et al. (1991) | 63 | Bilateral | Intracorporeal injections | Diabetes mellitus | Ultrasound | Surgical drainage (bilateral corporotomy) | Irrigations and suction drains | Staphylococcus albus | Resolution |
| Moskovitz et al. (1992) | 43 | Unilateral | Spontaneous | Diabetes mellitus | Ultrasound, cavernosography | Percutaneous ultrasound-guided aspiration | None | Beta-haemolytic streptococcus | Resolution |
| Kropman et al. (1993) | 56 | Unilateral | Intracorporeal injections | Multiple sclerosis, neutrogenic bladder, CISC, erectile dysfunction | Ultrasound | Aspiration | None | Staphylococcus aureus | Resolution |
| Pearle et al. (1993) | 42 | Bilateral | Haematogenous spread from a periodontal abscess | Diabetes mellitus | Retrograde urethrogram | Surgical drainage | None | Mixed colonies (peptostreptococcus, alpha-haemolytic streptococcus and fusobacterium) | Scrotal abscess |
| Kameda et al. (1998) |
72 | Bilateral | Spontaneous | Nil | MRI | Surgical drainage | None | Unknown | Dorso-caverno-urethral fistula, penectomy |
| Murali et al. (1998) | 40 | Bilateral | Tuberculosis | Tuberculosis, erectile dysfunction | Ultrasound, cavernosography | Surgical drainage | None | Mycobacterium tuberculosis | Impotence |
| Fernandez Duran et al. (1999) | 82 | Bilateral | Local infection | Unknown | Unknown | Aspiration | None | Unknown | Resolution |
| Köksal et al. (1999) | 33 | Bilateral | Spontaneous | Nil | Ultrasound, retrograde urethrogram | Surgical drainage (corporotomy) | Penrose drain | Staphylococcus aureus | Penile deviation |
| Frank et al. (1999) | 59 | Bilateral | Intra-abdominal abscess | 2 previous small-bowel resections for liposarcoma with intraoperative radiation therapy complicated by small-bowel fistula and intra-abdominal abscess | CT | Surgical drainage (bilateral corporotomy) | Jackson–Pratt drain | Enterococcus | Abscess recurrence and pubic osteomyelitis requiring extensive debridement, bilateral intracorporeal drain placement, pubic rami and small-bowel resection, and fistulotomy; erectile dysfunction |
| Vives et al. (2001) | 44 | Unilateral | Intracorporeal injections | Nil | Ultrasound | Surgical drainage (corporotomy) | None | Bacteroides spp. and Peptococcus spp. | Dorsal penile angulation, scar formation, erectile dysfunction |
| Pascual Regueiro et al. (2003) | 60 | Unilateral | Perineal infection | Nil | Ultrasound | Surgical drainage (corporotomy) | Penrose drain | Mixed colonies | Penile deviation, erectile dysfunction |
| Kumar et al. (2004) | 22 | Unilateral | Spontaneous | Nil | Ultrasound, MRI | Aspiration | None | Staphylococcus aureus | Resolution |
| Nalesnik et al. (2004) | 40 | Unilateral | Intracorporeal injections | Diabetes mellitus | CT | Surgical drainage (corporotomy) | Penrose drain | Group B streptococcus | Resolution |
| Sagar et al. (2005) | 19 | Bilateral | Spontaneous | Nil | None | Surgical drainage (bilateral corporotomy) | None | Staphylococcus aureus | Penile deviation |
| Sivaprasad et al. (2005) | 58 | Bilateral | Local infection (perianal abscess) | Diabetes mellitus | CT | Surgical drainage (bilateral corporotomy) | None | Actinomycetes | Recurrence, erectile dysfunction |
| Minami et al. (2006) | 50 | Unilateral | Local infection | Chronic prostatitis | CT | Surgical drainage (corporotomy) | Yes | Mixed colonies (Bacteroides fragilis and Fusobacterium) | Erectile dysfunction |
| Minami et al. (2006) | 70 | Unilateral | Local infection | Diabetes mellitus | CT | Surgical drainage (corporotomy) | Yes | Mixed colonies (Klebsiella streptococcus and Escherichia coli) | Resolution |
| Shamloul et al. (2006) | 53 | Unilateral | Intracorporeal injections | Nil | Ultrasound | Surgical drainage (corporotomy) | None | Bacteroides | Resolution |
| Sood et al. (2006) | 24 days | Bilateral | Priapism | Nil | None | Aspiration and cross-irrigation with antibiotic solution | None | Klebsiella streptococcus | Resolution |
| Ehara et al. (2007) | 54 | Bilateral | Spontaneous | Erectile dysfunction | MRI | Surgical drainage | Penrose drain | Nil growth initially, recurrence with MRSA | Wound rupture with MRSA, total penectomy and perineal urethrostomy |
| Charles et al. (2009) | 46 | Unilateral | Spontaneous/fellatio/periodontal abscess | Periodontal abscess | Ultrasound, MRI | Intravenous antibiotics+debridement of necrotic tissue after spontaneous wound rupture | None | Streptococcus constellatus and Peptostreptococcus | Erectile dysfunction |
| Thanos et al. (2011) | 45 | Unilateral | Local infection | Perineal abscess | CT | CT-guided percutaneous aspiration | Pigtail drainage catheter | Mixed colonies (Escherichia coli and anaerobic streptococcus) | Resolution |
| Bradley et al. (2012) | 50 | Bilateral | Spontaneous | Diabetes mellitus, balanitis xerotica obliterans (BXO) | CT | Surgical drainage (bilateral corporotomy) | None | Lactobacillus spp. | Resolution, erectile dysfunction |
| Brennan et al. (2013) | 56 | Unilateral | Spontaneous | Alcoholism | MRI | Surgical drainage with multiple washouts, dressing and drain changes | Yates drain | Streptococcus constellatus and Streptococcus intermedius | Multiple washouts, ipsilateral chordee |
| Dempster et al. (2013) | 32 | Unilateral | Spontaneous | Mild asthma, allergic rhinitis | Ultrasound | Aspiration with subsequent cavernosotomy and debridement | None (open packing) | None isolated | Penile necrotising fasciitis |
| Dugdale et al. (2013) | 48 | Unilateral | Local infection (proctitis) | Nil | CT | Surgical drainage (corporotomy) | Penrose drain | Mixed colonies (including MRSA on culture from recurrence of abscess) | Recurrence, cutaneous fistula formation, erectile dysfunction, right testicular numbness |
| Kumabe et al. (2013) | 60 | Unilateral | Blind ending urethra after urinary diversion surgery | Hartmann’s surgery, bladder and seminal vesicle total extirpation and prostatic segmentectomy for sigmoid cancer, ileal conduit, central venous catheter for chemotherapy | CT | Drained spontaneously | None | Escherichia coli | Resolution |
| Paladino et al. (2014) | 23 | Bilateral | Priapism, Winter’s procedure | Sickle cell disease | CT, ultrasound | Two penile needle aspirations and three surgical drainages (finally a bilateral cutaneous cavernostomy) | Penrose | Coagulase-negative staphylococci | Resolution, erectile dysfunction |
| Iadi et al. (2015) | 76 | Bilateral | Pyoderma gangrenosum | Urethral stricture | MRI | Multiple surgical drainages, prednisolone and immunosuppression | Unknown | Sterile initially, MRSA on culture of recurrent abscess | Recurrence, several cutaneous draining fistulae |
| Koyama et al. (2015) | 69 | Bilateral | Spontaneous | Chronic sinusitis | MRI | Surgical drainage | None | Unknown | Recurrence/progression, total penectomy |
| Tüzel et al. (2015) | 38 | Unilateral | Spontaneous | Anabolic-androgenic steroid use | Ultrasound | Surgical drainage (corporotomy) | Penrose | Staphylococcus epidermidis | Mild left-sided penile deviation |
| Siraj et al. (2018) | 49 | Bilateral | Spontaneous | Nil | MRI | Multiple surgical drainages, debridement and wound washouts | None | Streptococcus anginosus | Unknown |
| Capella et al. (2019) | 75 | Bilateral | Trauma secondary to urethral catheterisation | Metastatic prostate cancer, emphysema, spinal stenosis | CT | Surgical drainage, multiple re-looks and a delayed closure | Corrugated drains | Escherichia coli | Resolution |
| Lazarou et al. (2019) | 37 | Unilateral | Penile fracture secondary to sexual trauma | Nil | Ultrasound, MRI | Surgical drainage | Penrose | Mixed colonies (Streptococcus intermedius, Prevotella bivia, Peptostreptococcus micros, Fusobacterium spp., and Actinomyces meyeri) | Resolution, erectile dysfunction |
| Moussa et al. (2019) | 60 | Unilateral | Spontaneous | Diabetes mellitus | MRI | Percutaneous ultrasound-guided aspiration drainage | None | Staphylococcus aureus | Resolution |
| Takahashi et al. (2019) | 67 | Bilateral | Pyoderma gangrenosum | Nil | MRI | Surgical drainage | None | Staphylococcus epidermidis | Recurrence, treatment with prednisolone after diagnosis of pyoderma gangrenosum |
| Yamada et al. (2019) | 72 | Bilateral | Spontaneous | Phimosis | MRI | Aspiration | None | Micromonas micros and Fusobacterium sp. | Unclear |
| Vermeulen et al. (2020) | 64 | Unilateral | Spontaneous | Nil | 18F-fluorodeoxyglucose positron emission tomography/CT | Systemic antibiotics | None | Streptococcus constellatus | Resolution |
CT: computed tomography; MRI: magnetic resonance imaging.
Acknowledgements
None.
Conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The Northern Hospital, Epping, VIC 3076, does not require ethical approval for case reports.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
Guarantor
P.S.
Contributorship
F.C. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
