Abstract
Tuberculosis (Tb) continues to be a dreadful infection worldwide with nearly 1.5 million deaths in 2013. Furthermore multi/extensively drug-resistant Tb (MDR/XDR-Tb) worsens the condition. Recently approved anti-Tb drugs (bedaquiline and delamanid) have the potential to induce arrhythmia and are recommended in patients with MDR-Tb when other alternatives fail. The goal of elimination of Tb by 2050 will not be achieved without an effective new vaccine. The recent advancement in the development of Tb vaccines is the keen focus of this review. To date, Bacille Calmette Guerin (BCG) is the only licensed Tb vaccine in use, however its efficacy in pulmonary Tb is variable in adolescents and adults. There are nearly 15 vaccine candidates in various phases of clinical trials, includes five protein or adjuvant vaccines, four viral-vectored vaccines, three mycobacterial whole cell or extract vaccines, and one each of the recombinant live and the attenuated Mycobacterium tuberculosis (Mtb) vaccine.
Introduction
Tuberculosis (Tb) continues to be a big apprehension and there were nearly 9.0 million people infected with this communicable disease and about 1.5 million deaths in 2013. Of the estimated Tb infected people in 2013, nearly 56% were in South-East Asia and Western Pacific regions and 25% were in Africa. India (24%) and China (11%) alone accounted for 35% of the total Tb cases. In 2013 nearly1.1 million Tb-infected people were detected as human immunodeficiency virus (HIV) positive [WHO, 2014]. An estimated 10% of Tb-infected people developed active Tb in their lifetime [Cox et al. 2009]. Multi/extensively drug-resistant Tb (MDR/XDR-Tb) and totally drug resistant strains of Mycobacterium tuberculosis (Mtb) were other reported complications [WHO, 2014]. After four decades two new chemical entities, bedaquiline (Sirturo, Janssen Pharmaceutical Company, New Jersey, USA) and delamanid (Deltyba, Otsuka Pharmaceuticals, Japan), were approved for the treatment of MDR-Tb. However, both dugs induce arrhythmia, and are recommended only in patients for whom other treatment has failed [Cohen, 2013; Ryan and Lo, 2014] (http://www.ema.europa.eu). However, Bacille Calmette Guerin (BCG) is the only vaccine used in practice for the last 100 years [WHO, 2014]. In the 1990s, the progress of techniques for genetic manipulation of the genome sequence of Mtb invigorated the development of new Tb vaccines [Cole et al. 1998]. The advances in diagnostic and therapeutic strategies are still inadequate to achieve the goal of Tb elimination by 2050 [Ottenhoff and Kaufmann, 2012]. Today newer vaccine development is an active area of research and development (R&D) to limit Tb infection, reactivate latent Tb and tackle the spread of MDR/XDR-Tb. The relationships between vaccines, immunogenic response and protection from Tb would greatly facilitate vaccine development. An effective new Tb vaccine would save millions of lives and is essential to achieve the Millennium Development Goal of eliminating Tb globally by 2050. Introduction of new and more efficacious vaccines would be required for a 95% reduction in Tb mortality and a 90% reduction in Tb incidence by 2035. Furthermore, vaccine candidates need to be safer than BCG and afford protection against pulmonary Tb in newborns that lasts into late adolescence [Kaufmann et al. 2015]. An estimated US$8 billion a year is required to provide Tb treatment and care, which is very high compared with making a new vaccine available in the next 10–15 years at an estimated cost of less than US$800 million, utilizing a highly efficient portfolio management approach (http://www.aeras.org/pdf/TB_RD_Business_Case_Draft_3.pdf). The aim of this review is to present the progress in the development of vaccines for Tb. Tb vaccine development has been the focus of many review articles [Kaufmann et al. 2015; Montangnani et al. 2014; Groschel et al. 2014; Ahsan et al. 2013; Grave and Hokey, 2011;], while antigen-adjuvant interactions was the focus of a review article by Fox and colleagues [Fox et al. 2013].
Tb vaccine development
The variable efficacy of BCG vaccine indicates that Tb vaccines may perhaps be possible and three different approaches could be adopted in vaccine development. The first approach is to develop an improved version of the BCG vaccine that would include safe and long-acting recombinant BCG strains or attenuated Mtb, and the second approach is a prime-boost strategy in which a new vaccine, usually viral vectored or protein adjuvant, is given at a later stage as a booster dose. The third approach is to develop therapeutic vaccines to reduce the duration of Tb therapy and generally includes immunotherapeutic vaccines [Montangnani et al. 2014] (http://i-base.info/htb/21512). The Tb vaccines include protein or adjuvant, viral vectored, mycobacterial whole cell or extract, attenuated Mtb and recombinant live. Different types of Tb vaccines under clinical phases of development and their indications are given in Figure 1. A global portfolio of Tb vaccines represents diverse approaches and strategies that not only include recombinant BCG, rationally attenuated Mtb, viral-vectored platforms, recombinant purified proteins and novel adjuvants, but also novel delivery systems such as RNA or DNA combined with electroporation. It takes almost 11–16 years for a clinical vaccine candidate to progress from preclinical to phase III studies, with expected costs of US$15–25 million per year for clinical studies, US$6–12 million for phase I and IIa studies, US$20–40 million for phase IIb studies and US$115–170 million for phase III studies. But the success rates are nearly 20% for discovery/preclinical studies, 33% each for phase I/IIa and phase IIb studies, and 85% for phase III studies (http://www.aeras.org/pdf/TB_RD_Business_Case_Draft_3.pdf). Clinical trial size and costs can be reduced with the use of molecular testing for blood or other biological samples. The biomarker discovery is increasingly recognized as a vital element in future Tb vaccine R&D [Kaufmann et al. 2015; Weiner et al. 2013; Walzl et al. 2011]. Since 2005, nearly US$600 million global investment allowed more than 15 vaccine candidates to be evaluated in more than 50 human trials [Treatment Action Group, 2012]. Tb vaccine funding was nearly US$95,446,326 in 2011. There are currently 25 plus discovered leads and preclinical candidates, and US$6 million funding was awarded to the National Institute of Biomedical Innovation, Create Vaccine Co., Ltd and Aeras by Global Health Innovative Technology for work on human parainfluenza type 2 (rhPIV2), a preclinical candidate that is being designed to target mucosal immunity to prevent Tb from causing lung disease (www.aeras.org/annualreport2013; http://www.aeras.org/pdf/TB_RD_Business_Case_Draft_3.pdf). Today there are nearly 15 Tb vaccine candidates undergoing clinical trials (Table 1) (http://www.aeras.org/annualreport2013) [WHO, 2014]. There are six vaccines in phase I clinical trials, six vaccines in phase II clinical trials, while there are two vaccines in phase IIb and one vaccine in phase III clinical trials. The vaccines are generally administered with adjuvants which help to generate adequate immune responses. Adjuvants are simply classified as either immunostimulatory molecules or delivery systems, and most adjuvants are a combination of both of these classes [Fox et al. 2013; McKee et al. 2007]. Currently, the National Institute of Allergy and Infectious Diseases is working to bridge basic and clinical research by applying innovative systemic biology, genomics and bioinformatics, animal modelling, and contemporary immunologic and molecular tools to gain maximum knowledge about human immunity to Tb [Sizemore and Fauci, 2012].
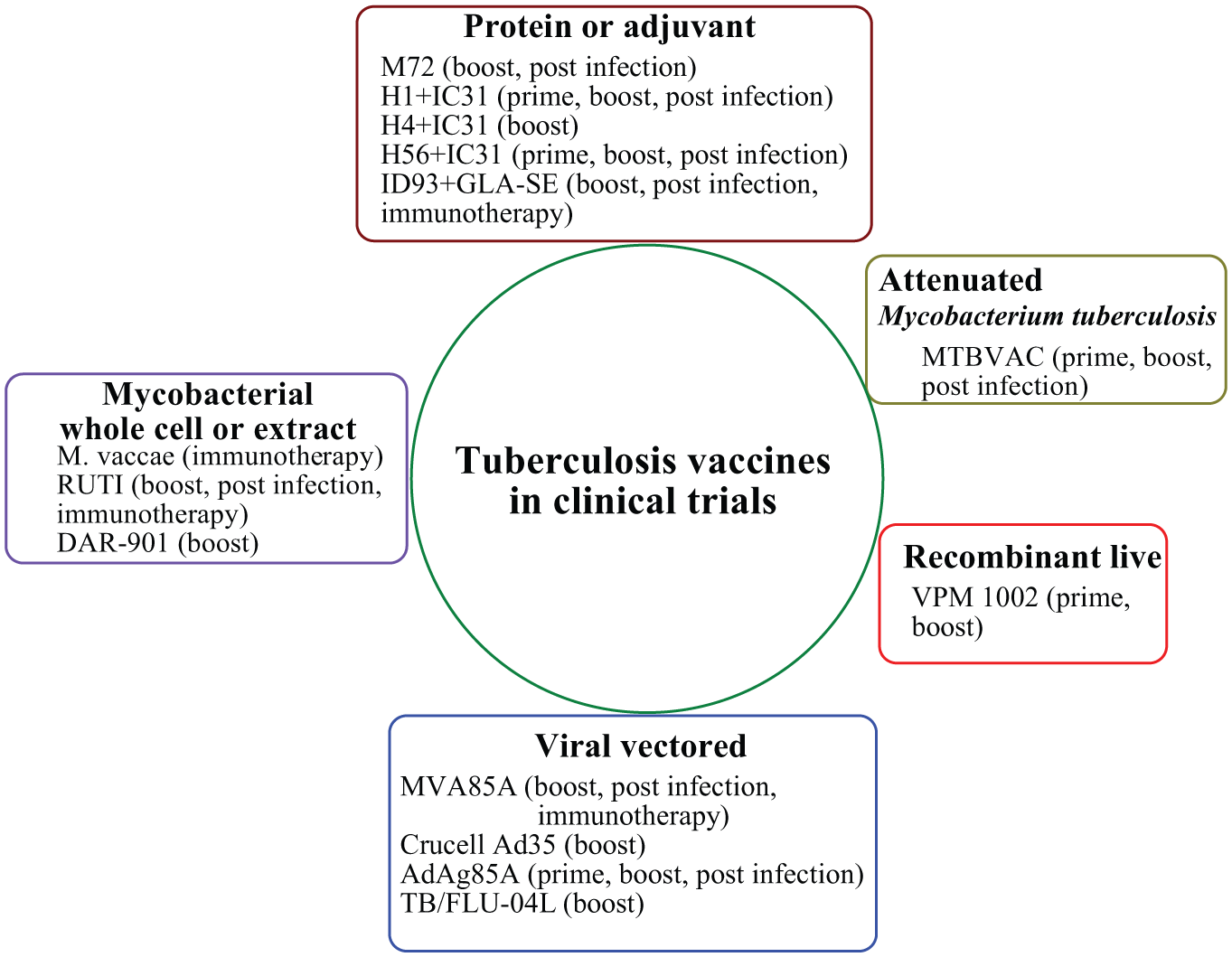
Tuberculosis vaccines in various phases of clinical trials.
The developmental pipeline for new tuberculosis (Tb) vaccines (August 2014).

rBCG, recombinant Bacille Calmette Guerin.
Tb vaccines in clinical trials
There are nearly 15 Tb vaccines in clinical trials. There are six vaccines including AdAg85A, MTBVAC, ID93+GLA-SE, Crucell Ad35/MVA85A, DAR 901 and TB/FLU-04L in phase I trials, six vaccines including VPM 1002, H1/H56/H4+IC3 and Crucell Ad35/AERAS-402 in phase II trials while two vaccines including MVA85A and M72+AS01E are in phase IIb trials and one vaccine M. vaccae (MV) is in a phase III clinical trial.
Phase I
AdAg85A
AdAg85A is a recombinant replication-deficient adenovirus serotype 5 vaccine vector developed by McMaster University (Canada) in collaboration with a Tianjin-based Chinese biotechnology company CanSinoo (China) [WHO, 2014]. The phase I studies showed safe, immunogenic and enhanced protection against virulent Mtb in murine, bovine and guinea pig models [ClinicalTrials.gov identifier: NCT00800670]. In another experiment on goat kids, BCG-AdAg85A vaccinated animals exhibited reduced pathology compared with BCG-vaccinated animals in lungs and in pulmonary lymph nodes. The pathological and bacteriological results showed antigen-specific interferon γ (IFN-γ) and humoral responses as prognostic biomarkers of vaccination [de Val et al. 2012]. Intranasal BCG boosted with AdAg85A is able to significantly enhance the long-term survival of BCG-primed guinea pigs following pulmonary Mtb challenge [Xing et al. 2009]. Researchers are being helped by Aeras (USA) to answer key questions about immune response following two different routes (aerosol and intramuscular) of AdAg85A administration for phase I clinical studies (http://www.aeras.org/pdf/AnnualReport_2013.pdf).
MTBVAC
MTBVAC is a live attenuated Mtb strain with deleted phoP and fadD26 genes developed by TBVI, Netherlands, University of Zaragoza, and Biofabri, Spain. MTBVAC exhibits safety and biodistribution profiles similar to BCG and confers superior protection in preclinical studies [Arbues et al. 2013]. A phase I clinical trial has been completed and phase II studies for MTBVAC vaccine are being planned [WHO, 2014]. Highly attenuated MTBVAC constructed with inactivation of an additional gene generated in exported repeated protein (Erp) could be a potential Tb vaccine candidate for use in a high-risk immunosuppressant population [Solans et al. 2014].
DAR 901
DAR 901 is a heat-inactivated Mycobacterium obuense strain of Mtb developed by investigators at Darmouth University in collaboration with Aeras [WHO, 2014]. Aeras supported the researchers at Darmouth University to design phase I studies to maximize knowledge gain using an immunological assay, use of mRNA expression and growth inhibition of Mtb (http://www.aeras.org/pdf/AnnualReport_2013.pdf). A phase I study was initiated in 77 adults who were HIV negative and HIV positive previously vaccinated with BCG to determine the safety, tolerability and immunogenicity of multiple doses of the vaccine at different dose levels, ranging between 0.1 and 1 mg [ClinicalTrials.gov identifier: NCT02063555]. Phase II studies on adults who are HIV positive and negative in Tanzania will be initiated if the phase I trial is successful [Von Reyn, 2013].
ID93+GLA-SE
The vaccine ID93+GLA-SE consists of the four-antigen Mtb recombinant protein (Rv2608, Rv3619, Rv3620 and Rv1813) in a novel glucopyranosyl lipid adjuvant-stable emulsion (GLA-SE; TLR4 agonist) developed by the Infectious Disease Research Institute, USA, in collaboration with Aeras. The phase I study was carried out to assess safety and immunogenicity [ClinicalTrials.gov identifier: NCT01599897] (WHO, 2014; Orr et al. 2013]. MyD88 and TRIF are necessary for GLA-SE (TLR4 agonist) to induce a polyfunctional T helper type 1 (TH1) immune response characterized by CD4+ T cells producing IFN-γ, tumour necrosis factor (TNF) and interleukin 2 (IL-2) when combined with ID93 [Orr et al. 2013]. The addition of another TLR agonist to ID93+GLA-SE enhances the magnitude of polyfunctional TH1 of the antigen-specific CD4+-cell response characterized by IFN-γ and TNF for the control of Tb [Orr et al. 2014].
Crucell Ad35/MVA85A
Crucell Ad35/AERAS 402, an adenovirus-vectored vaccine expressing three Mtb antigens Ag85A, Ag85B and TB10.4, was developed by Crucell, Netherlands, Aeras and Oxford, United Kingdom [WHO, 2014]. Crucell Ad35 provided CD4+ and CD8+ T-cell response in mice with an emphasized role of IFN-γ [Radosevic et al. 2007]. AERAS-402 also induced a potent CD8+ T-cell response, characterized by cells expressing IFN-γ and TNF-α [Abel et al. 2010]. Several phase I studies for Crucell Ad35/AERAS 402 were completed (https://clinicaltrials.gov/ct2/results?term=aeras+402&Search=Search). Crucell Ad35/AERAS 402 and MVA85A are being tested in early phase trials in combination to drive a balanced CD4+/CD8+ immune response [WHO, 2014] [ClinicalTrials.gov identifier: NCT01683773].
TB/FLU-04L
TB/FLU-04L is a recombinant influenza vaccine candidate developed by Research Institute for Biological Safety Problems and the Research Institute on Influenza, Russia having influenza virus strain A/Puetro Rico/8/34 H1N1 and MTb antigens Ag85A and early secretory antigenic target 6 (ESAT6). A phase I trial was completed and a phase IIa trial is being planned for this vaccine candidate [WHO, 2014] (http://biosafety.kz/index.php?option=com).
Phase II
VPM 1002
VPM 1002 is a recombinant BCG strain having a gene of Listeria monocytogenes coding for the protein listeriolysisn (Hly) integrated into the genome with inactivated urease C gene (to improve immunogenicity) and contains a hygromycin resistance marker developed at the Max Plank Institute of Infection Biology in collaboration with Vakzine Projekt Management, Germany. [WHO, 2014; Grode et al. 2013]. A phase I dose escalation study on safety and immunogenicity revealed that the vaccine candidate was safe and well tolerated up to 5 × 10e5 colony-forming units in health volunteers and induced multifunctional CD4+ and CD8+ T-cell immunogenicity [ClinicalTrials.gov identifier: NCT01113281; NCT00749034] (http://vakzine-manager.de/de/resources/7.-VPM-Tage/Leander-Grode.pdf) [Grode et al. 2013]. A phase II study to evaluate safety and immunogenicity of VPM 1002 in comparison with BCG in newborn infants in South Africa was found to be as safe and well tolerated as that with BCG [ClinicalTrials.gov identifier: NCT01479972] (http://vakzine-manager.de/de/resources/7.-VPM-Tage/Leander-Grode.pdf). A phase II study to evaluate the safety and immunogenicity of VPM1002 in comparison with BCG in HIV-exposed and -unexposed newborn infants in South Africa will be started soon [ClinicalTrials.gov identifier: NCT02391415].
H1:IC31
H1+IC31, a protein subunit adjuvanted vaccine developed by the Statens Serum Institute (SSI) in Copenhagen, Denmark in collaboration with TBVI, Netherlands, Intercell, Austria, and European & Developing Countries Clinical Trials Partnership, Netherlands is the hybrid of ESAT6 and Ag85B with IC31, an adjuvant system composed of the cationic protein polyaminoacid KLK and oligodeoxynucleotide ODN1a [WHO, 2014; Van Dissel et al. 2011] (http://i-base.info/htb/21512). Phase I studies did not cause any local or systemic adverse effects and elicited strong antigen-specific T-cell response (IFN-γ responses) against H1 and both the ESAT6 and Ag85B components which persisted for 2.5 years [Van Dissel et al. 2010; Ottenhoff et al. 2010]. A phase I safety and immunogenicity study of the this adjuvanted vaccine candidate in healthy BCG-vaccinated and prior or latently TB-infected individuals showed a few local or systemic effects and elicited a strong antigen-specific T-cell response against both the ESAT6 and Ag85B components which persisted through 32 weeks of follow up [Van Dissel et al. 2011]. Phase II studies of safety and immunogenicity in adults with HIV infection (CD4+ lymphocyte count greater than 350 cells/mm3) showed that the vaccine candidate was well tolerated, safe and induced a specific and durable TH1 immune response [Reither et al. 2014].
RUTI
RUTI is a nonlive vaccine based on fragmented, detoxified Mtb bacteria encapsulated in liposomes developed by Archivel Pharma, Spain immunotherapeutic vaccine [WHO, 2014]. RUTI was designed to shorten the treatment of latent Tb infection (LTBI) with isoniazid (INH) [Vilaplana et al. 2008]. Reduction of colony-forming units by rifampicin isoniazid therapy given 9–17 weeks post infection was made more pronounced by immunotherapy with RUTI [Cardona et al. 2005]. Treatment with RUTI following chemotherapy was found to be effective in mice and guinea pigs without exhibiting any toxicity [Cardona and Amat, 2006]. Treatment of RUTI and INH in M. caprae infected goats increased IFN-γ production after stimulating the peripheral blood with ESAT6, purified protein derivative and RUTI in vitro, with a temporary increase in rectal temperature and local swelling; however both adverse effects were well tolerated [Domingo et al. 2009]. Phase I studies showed dose-dependent local adverse reactions and elevated the T-cell response in healthy volunteers [Vilaplana et al. 2010]. In another study it was observed that RUTI has the potential for both prophylaxis and immunotherapy for Tb [Vilaplana et al. 2011]. Phase II studies on safety, tolerability and immunogenicity of three different doses (5, 25 and 50 mg) showed it was mild and well tolerated [Nell et al. 2014]. RUTI induced stronger activation of IFN-γ, CD4+ cells and CD8+ cells against tuberculin purified protein derivative, ESAT6 and Ag85B [Guirado et al. 2008]. A phase III trial for this vaccine candidate is being planned.
H56:IC31
H56+IC31, a protein subunit adjuvanted vaccine, contains antigens 85B and ESAT6 as well as AgRv2660c in adjuvant IC31, developed by SSI in Copenhagen, Denmark in collaboration with Aeras and Intercell [WHO, 2014]. H56+IC31 boosting was found to be able to control late-stage infection with Mtb and contains latent Tb [Lin et al. 2012]. H56+IC31 is currently being evaluated in phase II studies in Africa.
H4:IC31
H4+IC31, a protein subunit adjuvanted vaccine, contains antigens Ag85B and TB10.4 and was developed by SSI in collaboration with Sanofi-Pasteur, France, Aeras and Intercell [WHO, 2014]. Several phase I trials to study the safety and immunogenicity of this vaccine candidate have been completed (https://clinicaltrials.gov/ct2/results?term=H4%2BIC31&Search=Search&view=record). H4+IC31 effectively boosted and prolonged immunity induced by BCG, leading to an increased protection against Mtb owing to immune response dominated by IFN-γ, TNF-α, IL-2 or TNF-α, IL-2, CD4+ cell [Billeskov et al. 2012]. H4+IC31 is currently being evaluated in phase II studies in Africa.
Crucell Ad35/AERAS-402
Crucell Ad35/AERAS-402 is an adenovirus-vectored vaccine expressing three antigens: Ag85A, Ag85B and TB10.4 of Mtb [WHO, 2014]. A phase I study to assess the safety and immunogenicity was completed for this vaccine candidate [ClinicalTrials.gov identifier: NCT01683773].
Phase IIb
MVA85A
MVA85A, a viral vector, is a modified recombinant strain of Vacicinia virus Ankara expressing the Mtb antigen 85A [WHO, 2014; McShane et al. 2005]. Phase I studies showed a well tolerated and immunogenic effect in adults with HIV infection in Senegal with minor systemic and local adverse effects [Dieye et al. 2013]. In a study on nonhuman primates, MVA85A showed a high immunogenic effect when delivered by the aerosol route, and suggested evaluation of this route of immunization in humans in clinical trials [White et al. 2013]. Phase IIb studies in 2797 infants showed MVA85A was well tolerated and immunogenic but showed poor protection against Tb infection [Tameris et al. 2013]. MVA85A showed polyfunctional CD4+ T cells expressing IFN-γ, IL-2, TNF-α, IL-17 [Magali et al. 2013, 2014; Satti et al. 2014; Dintwe et al. 2013; Minassian et al. 2011; Scriba et al. 2010]. Safety and immunogenicity of MVA85A prime and BCG booster are being evaluated in phase II studies and phase I studies with protein carrier IMX313 [ClinicalTrials.gov identifier: NCT01650389; NCT01879163].
M72+AS01E
M72+AS01E is a protein subunit containing a fusion protein of the Mtb antigens 32A and 39A in adjuvant. AS01E was developed by GSK, United Kingdom in collaboration with Aeras (http://i-base.info/htb/21512) [WHO, 2014]. Several phase IIa studies were completed and phase IIb studies are being planned for this vaccine candidate. Phase II studies of M72 and the liposome-based AS01 adjuvant system were clinically safe and induced M72-specific CD4+ T-cell and humoral responses [Montoya et al. 2013]. The phase II study of safety and immunogenicity for M72/AS01 was acceptably tolerated in Gambian BCG-vaccinated infants with M72-specific humoral and CD4+ T-cell responses [Idoko et al. 2014]. M72/AS01 has a clinically acceptable safety profile and is highly immunogenic in Mtb-infected and uninfected adults [Day et al. 2013]. The phase II trials on safety and immunogenicity of M72/AS01 in adults with Tb infection and HIV are ongoing [ClinicalTrials.gov identifier: NCT01424501; NCT01755598].
Phase III
M. Vaccae
MV is a whole heat-killed immunotherapeutic agent in Tb developed by Anhui Zhifei Longcom, China. A phase III trial to study efficacy and safety in China is ongoing [WHO, 2014]. MV was well tolerated and no serious adverse effects were reported. It was safe and immunogenic in adults who were HIV positive and induced CD4+ T-cell-expressing IFN-γ and IL-10 responses in cultures from MV-treated mice compared with those from non-MV-treated mice [Yang et al. 2010; Rodríguez-Güell et al. 2008]. Heat-killed MV added to chemotherapy of patients with never-treated Tb showed improved sputum conversion and X-ray appearances [Yang et al. 2011].
Antigen in vaccine candidates
Antigens are proteins on the cell surface of the bacteria that cause Tb (Mtb) and evoke an immune response in infected people. These Tb antigens are not completely understood, which is a great obstacle in finding an effective vaccine against Tb (http://www.crick.ac.uk/news/science-news). There are around 11 antigens that have been used in preparation of Tb vaccines that are currently under various phases of clinical trials. The Mtb antigens used in vaccines under various clinical stages of development are given in Table 2. Five novel immunodominant proteins (Rv1957, Rv1954c, Rv1955, Rv2022c and Rv1471) were identified in research led by Robert Wilkinson that induced similar responses to those of CFP-10 and ESAT6 in both magnitude and frequency [Gideon et al. 2012].
Mycobacterium tuberculosis antigens used in vaccines under various clinical stages of development.

ESAT6, early secretory antigenic target 6. S.No, Serial no. 1,2,3.
Tb vaccines and immune response
Various T cells (CD4+, CD8+ and γ/δ T cells) and T-cell derived cytokines play a pivotal role in the protection against Mtb [Flynn, 2004; Flynn et al. 1993; Deenadayalan et al. 2013]. The therapeutic vaccines for Tb in clinical trials and their immunological responses are summarized in Table 3.
Profile of therapeutic vaccines in clinical trials for tuberculosis (Tb).

IFN-γ, antigen-specific interferon γ; IL, interleukin; TH1, T helper 1; TNF-α, tumour necrosis factor α.
Conclusion
The Tb vaccine developmental pipeline appears to be healthy today, with around 15 vaccines in various phases of clinical trials. All these vaccines may be used as prime, boost or immunotherapeutic vaccines. The ambiguous correlations between immunity and Tb, together with lack of information about Tb antigens, are big obstacles in finding effective vaccines against Tb which need to be overcome. Also, sufficient funding is required to support and sustain the development of a robust preclinical pipeline. The cost of a new vaccine to be developed within the next 10–15 years utilizing a highly efficient portfolio management approach would certainly be less than the annual global financial toll of Tb treatment and care.
Footnotes
Conflict of interest statement
The author reports no financial conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
