Abstract
Background
Impaired intestinal epithelial barrier is highly affected in inflammatory bowel disease. Transmembrane collagens connecting the epithelial cells to the extracellular matrix have an important role in epithelial cell homeostasis. Thus, we sought to determine whether the transmembrane type 23 collagen could serve as a surrogate marker for disease activity in patients with Crohn’s disease and ulcerative colitis.
Methods
We developed an enzyme-linked immunosorbent assay to detect the ectodomain of type 23 collagen (PRO-C23) in serum, followed by evaluation of its levels in both acute and chronic dextran sulfate sodium colitis models in rats and human inflammatory bowel disease cohorts. Serum from 44 Crohn’s disease and 29 ulcerative colitis patients with active and inactive disease was included.
Results
In the acute and chronic dextran sulfate sodium-induced rat colitis model, the PRO-C23 serum levels were significantly increased after colitis and returned to normal levels after disease remission. Serum levels of PRO-C23 were elevated in Crohn’s disease (p < 0.05) and ulcerative colitis (p < 0.001) patients with active disease compared to healthy donors. PRO-C23 differentiated healthy donors from ulcerative colitis (area under the curve: 0.81, p = 0.0009) and Crohn’s disease (area under the curve: 0.70, p = 0.0124). PRO-C23 differentiated ulcerative colitis patients with active disease from those in remission (Area under the curve: 0.75, p = 0.0219) and Crohn’s disease patients with active disease from those in remission (area under the curve: 0.68, p = 0.05).
Conclusion
PRO-C23 was elevated in rats with active colitis, and inflammatory bowel disease patients with active disease. Therefore, PRO-C23 may be used as a surrogate marker for monitoring disease activity in ulcerative colitis and Crohn’s disease.
Key summary
Summarise the established knowledge on this subject
Intestinal permeability in Crohn’s disease (CD) and ulcerative colitis (UC) is impaired, which results in the invasion of numerous bacteria, followed by immune cell infiltration to the gut. Increased tissue remodelling and loss of epithelial integrity is related to flares in CD and UC. Type 23 collagen is a transmembrane collagen and is expressed by intestinal epithelial cells and is therefore a marker of epithelium integrity.
What was the significant and/or new findings of this study?
A novel type 23 collagen marker was developed, quantifying the ectodomain of type 23 collagen (PRO-C23) in serum. Elevated serum levels of PRO-C23 was demonstrated to be associated with dextran sulfate sodium (DSS) colitis rats (acute and chronic) and CD and UC patients with active disease. PRO-C23 may potentially be used as a non-invasive surrogate of disease activity in CD and UC patients and thus aid in diagnosing and monitoring patients.
Introduction
The epithelium is important to maintain the health of the gut. However, in both Crohn’s disease (CD) and ulcerative colitis (UC), intestinal permeability is impaired, which results in the invasion of numerous bacteria, followed by immune cell infiltration to the gut1–3 and triggering of bowel symptoms in inflammatory bowel disease (IBD) patients. 4 Type 23 collagen, a member of type II transmembrane protein family expressed by epithelial cells, was first identified in 2003 by Banyard et al. in rat metastatic tumour cells. 5 Type 23 collagen consists of a short cytoplasmic domain, a membrane-spanning domain and a long ectodomain. 5 The newly synthesised type 23 collagen can be transported to the cell surface as a transmembrane protein, or cleaved intracellularly by furin, triggering release of the ectodomain into the extracellular matrix (ECM) 6 (Figure 1).
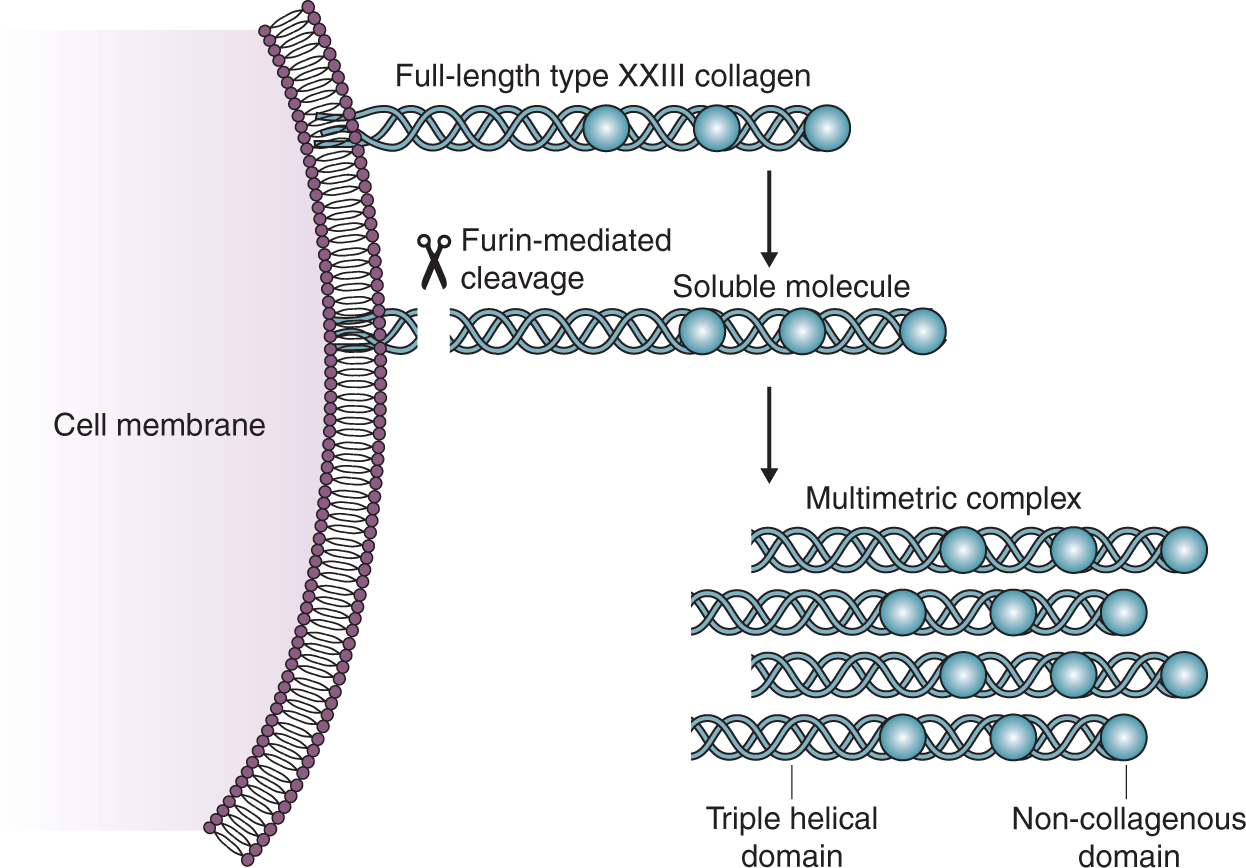
The tertiary structure of type XXIII collagen and furin mediated shedding of type XXIII collagen. Type XXIII is a transmembrane collagen expressed by epithelial cells but also exists as a soluble protein after furin-mediated shedding. Type XXIII collagen consists of a transmembrane domain, three collagenous domains (triple helical domains), and three non-collagenous domains. The soluble form of type XXIII may also form multimeric complexes. Adapted from Karsdal et al. 27
Many studies have shown that some of the junction proteins were significantly down-regulated in IBD inflamed intestinal tissue,7–10 and increased ECM remodeling was highly affected in both UC and CD, including remodelling of the basement membrane which was also related to epithelial cells driven by increased protease activity.11–21 Based on this background it was likely that type 23 collagen was also affected by the ECM remodelling in UC and CD.
Type 23 collagen was found to be related to cell adhesion and metastasis in vitro. 22 , 23 Knockout type 23 collagen in such cell lines resulted in altered expression of cell adhesion molecules and impaired cell adhesion, 22 , 23 which indicated that type 23 collagen might be a regulator of cell adhesion.
Since type 23 collagen is expressed in the intestinal epithelium, 15 we hypothesised that it may be cleaved from the cell surface during epithelial damage in IBD and could be used as a potential biomarker to monitor disease activity. Therefore, we developed an enzyme-linked immunosorbent assay (ELISA) to detect the ectodomain of type 23 collagen (PRO-C23) in serum, followed by an evaluation of its levels in both dextran sulfate sodium (DSS) colitis models in rats and human IBD cohorts.
Materials and methods
Antibody development for PRO-C23
We used the last 10 amino acids of the type 23 collagen α1 chain (531’GLPVPGCWHK’540, Genscript, USA) as the immunogenic peptide to generate specific monoclonal antibodies (mAb). The sequence homology of the peptide was 100% between human and rats. Four- to six-week-old Balb/C mice were immunised subcutaneously with 100 µg of the immunogen (KLH-CGG-GLPVPGCWHK) emulsified with Stimmune adjuvant (Thermo Fisher, USA). Consecutive immunizations were performed at two-week intervals. Mouse spleen cells were fused with SP2/0 myeloma cells to form hybridomas. The hybridomas were raised in 96-well plates and cultured in Iscove’s Modified Dulbecco’s Medium (IMDM)+10% fetal bovine serum (FBS) medium in the CO2 incubator. Hybridoma cells specific to the selection peptide and without cross-reactivity to elongated peptide (GLPVPGCWHKA), truncated peptide (GLPVPGCWH) or to deselection peptides (GLPVQGCWNK, type XIII collagen, GLPMPGCWQK, type XXV collagen) (Genscript, USA) were selected and subcloned. In the end, the supernatant was purified using an immunoglobulin G (IgG) column (GE health, USA). Briefly, the IgG column was washed with 10 column volume of 20 mM phosphate buffered saline (PBS). The supernatant was applied in 4°C with 1 ml/min speed. After supernatant ran through the column, the column was washed with 10 column volume of 20 mM PBS to remove unspecific binding proteins. Antibodies were eluted with 0.1 M Glycine pH 2.7, and dialysis with PBS buffer was subsequently performed.
PRO-C23 assay and technical evaluation
ELISA plates used for the assay development were Streptavidin-coated from Roche (cat.: 11940279). All ELISA plates were analysed with the ELISA reader from Molecular Devices, SpectraMax M, (California, USA). We labelled the selected (mAb) with horseradish peroxidase (HRP) using the Lightning link HRP labelling kit according to the instructions of the manufacturer (Innovabioscience, Babraham, Cambridge, UK). A 96-well streptavidin plate was coated with biotin-GLPVPGCWHK (Genscript, USA) and incubated 30 minutes at 20°C. Twenty µl of standard peptide (standard A had the highest concentration, and was diluted two-fold) or samples were added to appropriate wells, followed by 100 µl of HRP conjugated (mAb) 10F6, and incubated 20 h at 4°C. Finally, 100 µl tetramethylbenzidine (TMB) (Kem-En-Tec cat. 438OH) was added, and the plate was incubated 15 min at 20°C in the dark. The above incubation steps included shaking at 300 rpm. After each incubation step, the plate was washed five times. The TMB reaction was stopped by adding 100 µl of stopping solution (1% H2SO4) and measured at 450 nm with 650 nm as the reference.
The lower limit of detection (LLOD) was determined from 21 zero samples (i.e. buffer) and calculated as the mean+3×standard deviation (SD). Upper limit of detection (ULOD) was determined as the mean–3×SD of 10 measurements of standard A. The intra-assay and inter-assay variations were the mean variations of 10 quality control (QC) samples run 10 independent times in duplicate. Dilution recovery was determined in four serum samples and four plasma samples and was calculated as a percentage of recovery of diluted samples from the 100% sample. Correlation between the PRO-C23 levels in healthy subjects with matched samples from serum -and plasma (heparin, citrate and ethylenediaminetetraacetic acid (EDTA)) was determined in 16 samples (Innovative Research, USA). No additional information for these samples was available.
Western blotting with recombinant human type 23 collagen
Recombinant human type 23 collagen (R&D system, 4165-CL) was diluted in sample buffer containing 80 mM dithiothreitol (DTT) and run on a 10% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) gel, and subsequently transferred onto a nitrocellulose membrane. The nitrocellulose membranes were then blocked for non-specific binding by incubation for 1 h at room temperature in tris-buffered saline Tween 20 (TBS-T) buffer containing 5% skim milk powder. This was followed by incubation with 1 µg/ml 10F6 or commercial type 23 collagen antibody (Ab) (R&D system, MAB4165) diluted in TBS-T milk for overnight. The recombinant type 23 collagen for both commercial Ab and 10F6 were prepared together. Half volume of recombinant protein was loaded for commercial Ab incubation. The other half volume was loaded for 10F6 incubation. The loading and transfer were done using the same gel, which ensures the equal transfer time. The recombinant protein demonstrated a 95% purity, which made it unnecessary to normalise total protein expression Then the membranes were washed in TBS-T three times, followed by incubation in the secondary peroxidase-conjugated antibody. The secondary Ab (Jackson, 315-035-045, 1:5000 dilution) was incubated at room temperature for 1 h. Finally, the membranes were washed in TBS-T three times, and the results were visualised using the enhanced chemiluminescence (ECL) system (GE healthcare, cat. RPN2109).
DSS rat model
Male Sprague–Dawley rats, 12 weeks of age, were used for both the acute DSS colitis study and the chronic DSS colitis model. The rats were divided into two groups: 6% DSS group (n = 12) and a water control group (n = 9) for the acute DSS colitis model. Acute DSS colitis was induced by administration of 6% DSS in the drinking water for 5 days, while control rats received regular drinking water. After 5 days of DSS administration, DSS was withdrawn, and regular drinking water was administered until the end of study at day 16. Six DSS rats and three control rats were sacrificed on day 6. The rats were fasted overnight before blood was drawn from the tail vein on day 0 (n = 21), 6 (n = 9), 7 (n = 12) and 16 (n = 12). The rats were also divided into two groups: 5% DSS group (n = 36) and water control group (n = 12) for the chronic DSS colitis model. Chronic DSS colitis was induced by administrating 5% DSS in the drinking water for four cycles for 7 days with 7 days recovery period with drinking water without DSS. The rats were fasted over-night before blood was drawn from the tail vein on day 0 (n = 48), 7 (n = 48), 14 (n = 42), 21 (n = 39), 28 (n = 36), 35 (n = 33), 42 (n = 30), 49 (n = 27), 56 (n = 24).
The disease progression for both acute and chronic DSS colitis models was evaluated using the Disease Activity Index (DAI), which was scored each day of the study and has been described previously. 14 The DSS in vivo study’s ethical guidelines were followed in accordance with the legislation and under ethical approval of the ‘Dyreforsøgstilsynet’ (agreement number 2017-15-0201-01171).
IBD cohorts
Three different cohorts were measured to evaluate the biological relevance of the PRO-C23 assay. Cohort 1 was used in assay development to evaluate the biological relevance of PRO-C23 in IBD, while cohort 2 and 3 were included to validate the findings in cohort 1 and further assess the applicability of PRO-C23 regarding disease activity in IBD. Serum samples were collected after informed signed consent and approval by the local Ethics Committee. In cohort 1, serum from CD (n = 10) and UC (n = 10) patients was obtained from commercial vendor Reprocell/Valley Biomedical in Table 1. Serum from healthy subjects (HSs) was also obtained from vendor Reprocell/Valley Biomedical (Table 1). Serum samples from CD patients (n = 44) in cohort 2 were obtained from Pavia, Italy, and serum samples from UC patients (n = 29) (cohort 3) were obtained from Zagreb, Croatia, and an additional 29 healthy donors were purchased from vendor Reprocell/Valley Biomedical (Table 2). For CD patients, disease activity was assessed by Crohn’s disease Activity Index (CDAI). Patients with scores below 150 were classified as being in remission. In UC patients, disease activity was assessed according to the partial Mayo score (pMayo) for UC. Clinical remission was defined as a score below two.
Patient demographics of cohort 1.
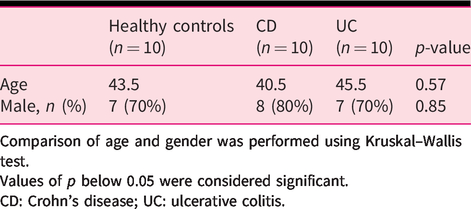
Comparison of age and gender was performed using Kruskal–Wallis test.
Values of p below 0.05 were considered significant.
CD: Crohn’s disease; UC: ulcerative colitis.
Patient demographics of cohorts 2 and 3.
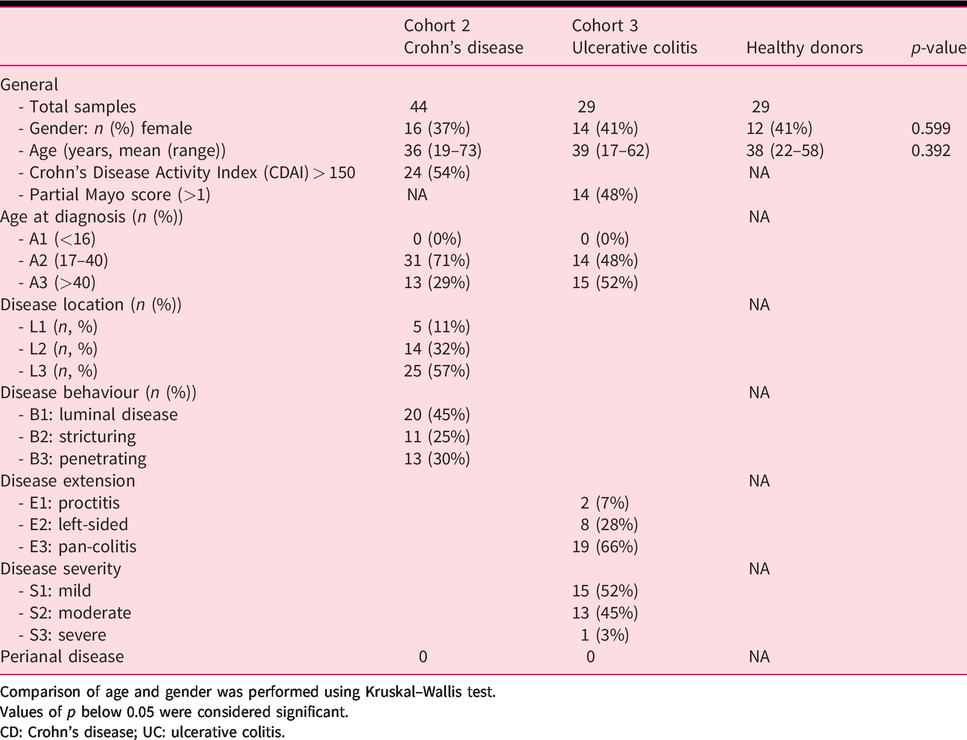
Comparison of age and gender was performed using Kruskal–Wallis test.
Values of p below 0.05 were considered significant.
CD: Crohn’s disease; UC: ulcerative colitis.
Statistics
Statistical analysis was performed using MedCalc version 14 and GraphPad Prism version 7. The biomarker levels were presented as mean values and standard error of the mean (SEM). Key data was represented as Tukey plots with interquartile range (IQR). Mixed-effects analysis with Sidak’s test for multiple comparisons was applied for testing the differences in changes of PRO-C23 levels between DSS rats and controls and for testing differences in the DAI between DSS rats and controls. Pearson r correlation was applied for testing the association between serum PRO-C23 and DAI in DSS rats and controls. In human cohorts, age and gender were compared using a Kruskal–Wallis test. The differences of PRO-C23 between patients and healthy controls were determined by Kruskal–Wallis one-way analysis of variance (ANOVA) test, Dunn’s multiple comparisons test. The diagnostic power of biomarkers was investigated by the area under the curve (AUC) of the receiver-operating characteristics (ROC) curve with 95% confidence interval (CI). Sensitivity and specificity were determined for appropriate cut-off values based on the ROC curves. The significance threshold was set at p < 0.05.
Results
Characterization of PRO-C23 assay
Like type 23 collagen, type XIII and XXV collagens are also transmembrane collagens and share highly similar sequences in their C-terminus (Figure 2(a)). Western blot of recombinant type 23 collagen ectodomain (4165-CL, R&D system) showed that the chosen antibody 10F6 recognised type 23 collagen ectodomain around 60kD, while the reference commercial antibody (MAB4165, R&D system) was also shown (Figure 2(b)). 10F6 specifically recognised the last 10 amino acids of the C-terminus of type 23 collagen 531’GLPVPGCWHK’540, but did not recognise the truncated peptide GLPVPGCWH, type XIII collagen C-terminal peptide GLPVQGCWNK or type XXV collagen C-terminal peptide GLPMPGCWQK. It only weakly recognised elongated peptide GLPVPGCWHKA (Figure 2(c)).
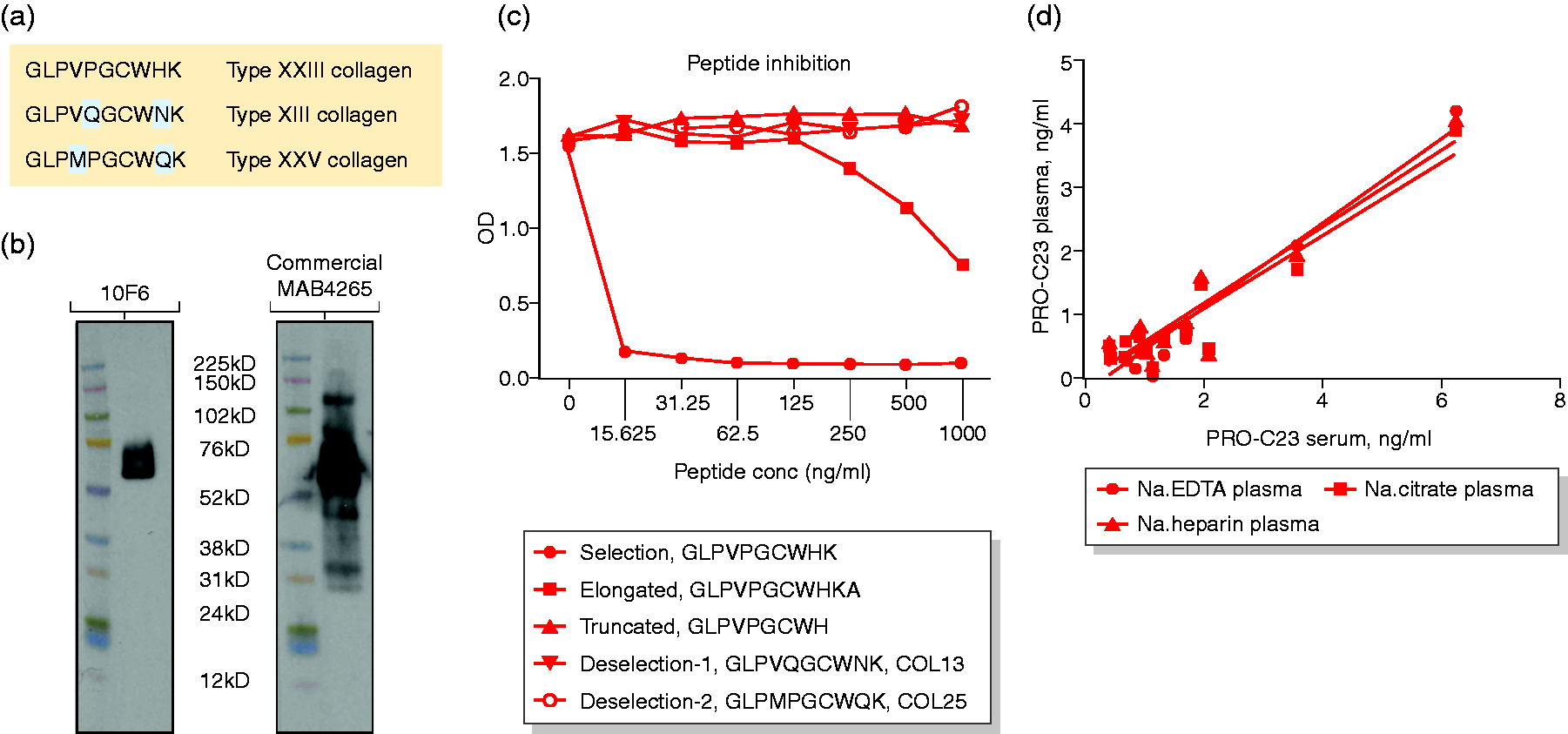
Specificity assessment of the PRO-C23 antibody 10F6. (a) Sequence alignment for C-terminus of type XIII, XXIII, and XXV collagens. The antibody recognises the residues from 531 to 540 of type 23 collagen. (b) Western blot results of recombinant type 23 collagen using 10F6 as a primary antibody. (c) PRO-C23 antibody specificity towards different peptides. Reactivity to the type XXIII collagen selection peptide (GLPVPGCWHK), the elongated peptide (GLPVPGCWHKA), the truncated peptide (GLPVPGCWH), a peptide from type XIII collagen (GLPVQGCWNK) and peptide from type XXV collagen (GLPMPGCWQK) was tested in the PRO-C23 assay. (d) Correlations of PRO-C23 levels serum levels with the levels of the EDTA, heparin and citrate plasma (n = 16). PRO-C23: biomarker of the ectodomain of type 23 collagen; OD: optical density; EDTA: ethylenediaminetetraacetic acid.
PRO-C23 competitive ELISA provided a measurement range from 0.38 ng/ml (LLOD) to 18.73 ng/ml (ULOD). The inter- and intra-assay variabilities were 8.1% and 3.5%, respectively. The dilution recovery and spiking recovery in human serum are shown in Supplementary Material Table 1. There was a significant correlation between human serum PRO-C23 values and EDTA values, heparin and citrate plasma values (EDTA; r = 0.95, heparin; r = 0.93, citrate; r = 0.94, p < 0.0001, Figure 2(d)), showing that PRO-C23 levels were independent of the blood preparation method.
PRO-C23 biomarker in DSS rat model
A rat model of DSS-induced colitis was used to investigate the biological relevance of the PRO-C23 assay. Compared to control rats, DSS rats had significantly higher DAI scores from day 2–11 in the acute DSS model (Figure 3(a)) and from day 5–56 in the chronic DSS model (Figure 3(d)), indicating colitis was successfully induced. The percentage change in serum PRO-C23 relative to baseline was significantly increased in DSS rats compared to controls at day 7 (p = 0.027, Figure 3(b)), which returned to normal at day 16. Serum PRO-C23 and DAI was positively correlated at day 6 and 7 (Pearson r = 0.50, p = 0.04, Figure 3(c)). PRO-C23 was also modulated in the chronic DSS-induced colitis model, which was demonstrated to be significantly different from baseline at blood sampling every day (p < 0.001), except at day 56. This statistical difference from baseline, however, was only seen in the DSS group and not in the control group (Figure 3(e)). Furthermore, serum PRO-C23 and DAI were positively correlated only at day 21 (Pearson r = 0.33, p = 0.02, Figure 3(f)) and 56 (Pearson r = 0.45, p = 0.04, Figure 3(g)).
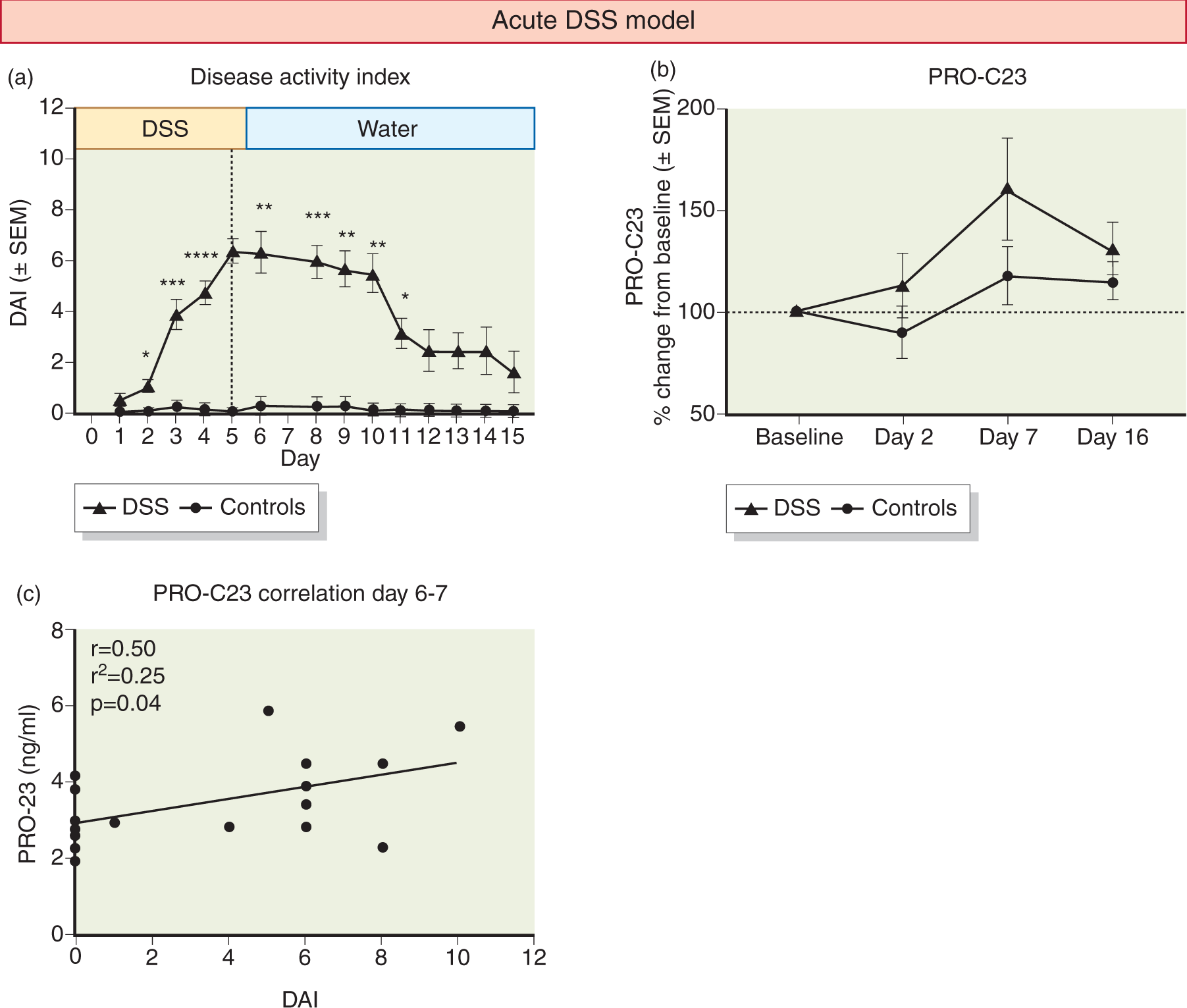
PRO-C23 serum levels during models of acute dextran sulfate sodium (DSS) colitis ((a)–(c)) and chronic DSS colitis ((d)–(g)). (a) Disease Activity Index (DAI) for DSS rats and controls during and after DSS administration in the acute DSS model; (b) percentage PRO-C23 serum levels relative to baseline in DSS rats and controls in the acute DSS model; (c) correlation between PRO-C23 serum levels and DAI at day 6 and 7; (d) DAI for DSS rats and controls during and after DSS administration in the chronic DSS model; (e) percentage change of PRO-C23 serum levels relative to baseline in DSS rats and controls in the chronic DSS model; (f) correlation between PRO-C23 serum levels and DAI at day 21 and (g) day 56.
Patient demographics
There were no statistical differences between the patient demographics (gender and age) of healthy donors, CD and UC patients in all cohorts (Tables 1 and 2).
PRO-C23 biomarker in human IBD cohorts
PRO-C23 was measured in serum from three independent human cohorts. In cohort 1, PRO-C23 was quantified in 10 CD (IQR: 2.107–4.181 ng/mL) and 10 UC (IQR: 1.914–3.004 ng/mL) patients, together with 10 age-matched healthy donors (IQR: 1.159–2.391 ng/mL ng/ml). The results showed that CD (AUC = 0.80; p = 0.023) and UC (AUC: 0.80; p = 0.023) patients have significantly higher levels of PRO-C23 compared to healthy donors (Figure 4).
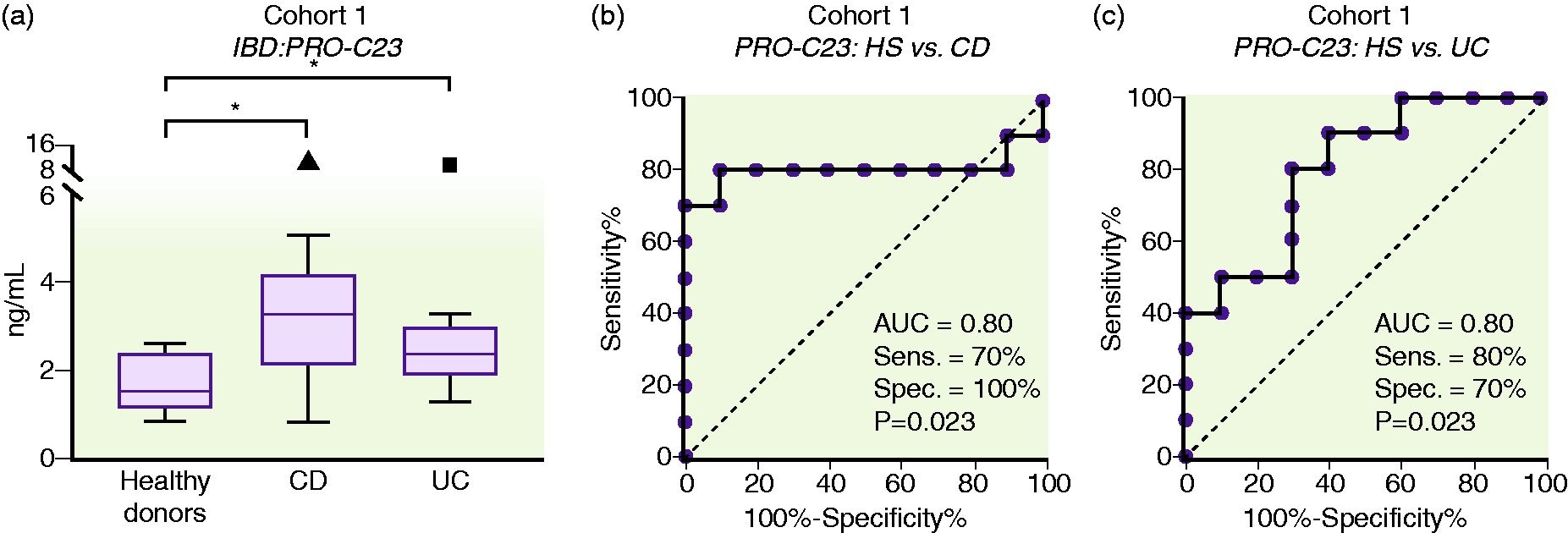
PRO-C23 serum levels in cohort 1 for healthy subject (HS), UC and CD groups ((a)), and its discriminative power to differentiate between HS and CD/UC patients ((b) and (c)).
PRO-C23 levels, in cohorts 2 and 3, were elevated in active CD patients (IQR: 1.999–4.287 ng/mL ng/ml) and active UC patients (IQR: 2.830–5.700 ng/mL) compared to healthy donors (IQR: 1.775–2.795 ng/mL) (CD: AUC = 0.70, p < 0.05; UC: AUC = 0.81, p < 0.01; Figure 5). PRO-C23 serum levels were significantly elevated in patients with active disease compared to inactive disease for CD and UC. (CD: AUC = 0.68, p = 0.05; UC: AUC = 0.75, p < 0.05; Figure 5).
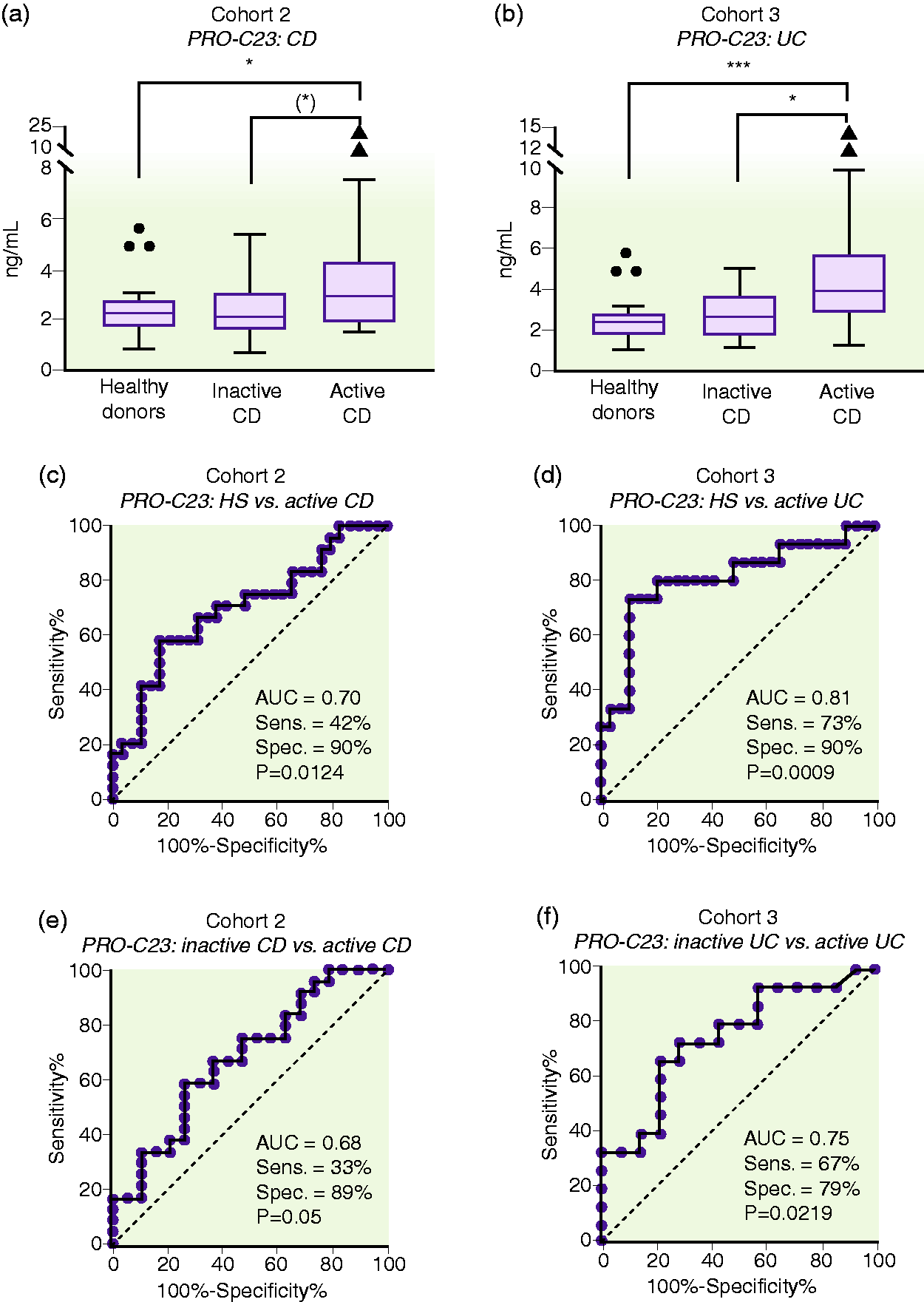
PRO-C23 serum levels in cohort 2 (Crohn's disease (CD)), cohort 3 (ulcerative colitis (UC)) and healthy subjects (HSs) ((a) and (b)), and its discriminative power to differentiate between HSs and CD and UC patients ((c) and (d)) and between active disease vs inactive disease in CD and UC patients ((e) and (f)).
Discussion
IBD patients with active disease have increased intestinal permeability and mucosal damage, including loss of epithelial integrity. 24 The tight junction/adhesion proteins, such as E-cadherin and β-catenin, are dramatically down-regulated in the inflamed tissue of IBD patients. 7 Therefore, proteins related to the intestinal epithelium for assessing intestinal permeability/epithelial integrity and intestinal tissue homeostasis may be used to evaluate the disease burden. 1 , 11 ,13–15,25–27
The antibody in the PRO-C23 ELISA only recognised the C-terminus sequence of the ectodomain of type 23 collagen and had no cross-reaction with the C-terminus of type XIII and XXV collagen, which have similar sequences. This data confirmed the specificity of the antibody. Our data demonstrated that the PRO-C23 could be detected in the circulation by the PRO-C23 competitive ELISA in serum and plasma samples.
We found that type 23 collagen was significantly elevated in DSS rats, and it weakly correlated with disease activity in both the acute (day 6 and 7, Figure 3) and chronic DSS colitis model (day 21 and 56, Figure 3). This finding indicated that the ectodomain of type 23 collagen found in circulation related to disease activity of DSS rats, and these data were in agreement with the study by Lindholm et al. which demonstrated that markers of type III collagen remodelling (interstitial matrix) and markers of type IV collagen remodeling (basement membrane) were also elevated in the DSS model. 14 Furthermore, the continuous elevated levels of PRO-C23 in the chronic DSS colitis model indicate the remodeling of type 23 collagen is ongoing during induction of inflammation and in the healing phases. This can be explained by the involvement of type 23 collagen in cell migration, which is necessary for epithelial restitution.
To further validate the PRO-C23 biomarker, it was measured in two additional human cohorts. PRO-C23 was found elevated in human CD patients (cohort 2) and UC patients (cohort 3) with active disease, and it was also able to differentiate between UC and CD vs healthy subjects and active disease vs inactive disease for both UC and CD. These findings suggest that the release of the PRO-C23 was reinforced in the active intestinal damage, which was consistent with the animal model results.
It is believed that type 23 collagen facilitates cell-cell adhesion and cell-matrix adhesion. 22 Silencing type 23 collagen in lung cancer and clear cell renal cell lines showed altered adhesion protein expressions and less ability on cell adhesion and migration. 22 , 23 However, type 23 collagen is also present in other tissues, and the function and use in other diseases are yet unknown. To our knowledge, this is the first study showing that type 23 collagen level is modulated in IBD. Our results indicate that PRO-C23 is not specific for either UC or CD, but more related to the process of intestinal mucosal remodelling, affecting the epithelium equally in UC and CD (Figures 4 and 5). Furthermore, since PRO-C23 measures the shedding of type 23 collagen from epithelial cells, this assay may be used as a monitoring tool to evaluate the integrity of the intestinal mucosal epithelium. Whether increased levels of PRO-C23 is solely a consequence of inflammation or also an contributor to the pathogenesis in IBD patients is unknown. If type 23 collagen is essential for restitution of intestinal epithelial cells, it is possible the underlying dysregulation of the collagen may adversely affect healing of the inflamed intestine.
There are several limitations to this study. Firstly, we cannot exclude that PRO-C23 may be released from other tissues than the intestines; however, the DSS colitis models confirm that PRO-C23 is a product of the intestinal mucosa remodeling, suggesting that PRO-C23 at least is derived from the intestines. Secondly, the number of patients in this study is low. The fact that the IBD patients were recruited at different medical institutes may introduce discrepancies e.g. in sample handling and disease activity scoring. The PRO-C23 assay, however, was still able to obtain similar results in the IBD cohorts included. While PRO-C23 only showed a weak correlation to DAI in the DSS models, PRO-C23 was demonstrated to be elevated in IBD patients with active disease. Therefore, more clinical studies in comprehensive cohorts are needed to evaluate this biomarker further.
Conclusion
This is the first study showing elevated serum levels of type 23 collagen in IBD, and consequently, that transmembrane collagens and the basement membrane axis is essential for the pathology of IBD. PRO-C23 was found elevated in both the acute and chronic rat DSS colitis model and patients with active CD and UC. This indicates that PRO-C23 is associated with a compromised interstitial mucosa and epithelial cell dysfunction. PRO-C23 may potentially be used as a non-invasive surrogate of disease activity in CD and UC patients and thus aid in the diagnosis and monitoring of patients.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620977371 - Supplemental material for Elevated ectodomain of type 23 collagen is a novel biomarker of the intestinal epithelium to monitor disease activity in ulcerative colitis and Crohn’s disease
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620977371 for Elevated ectodomain of type 23 collagen is a novel biomarker of the intestinal epithelium to monitor disease activity in ulcerative colitis and Crohn’s disease by T Manon-Jensen, S Sun, M Lindholm, V Domislović, P Giuffrida, M Brinar, G Mazza, M Pinzani, Ž Krznarić, A Di Sabatino, MA Karsdal and JH Mortensen in United European Gastroenterology Journal
Footnotes
Acknowledgement
The authors would like to thank Mie Andersen for great technical assistance.
Declaration of conflicting interests
S Sun, JH Mortensen, M Lindholm, MA Karsdal and T Manon-Jensen are employed at Nordic Bioscience A/S which is a company involved in the discovery and development of biochemical biomarkers. MA Karsdal and T Manon-Jensen own stocks at Nordic Bioscience.
Ethics approval
Production of monoclonal antibodies performed in mice was approved by the National Authority (The Animal Experiments Inspectorate) under approval number 2013-15-2934-00956. All animals were treated according to the guidelines for animal welfare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The biomarker portion of the study was supported by the Danish Research Foundation (den Danske Forskningsfond).
Informed consent
Informed consent and approval by the local Ethics Committee were obtained before sample collection and the studies were performed in compliance with the Helsinki Declaration of 1975.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
