Abstract
Background
Fecal microbiota transplantation is an emerging therapeutic option, particularly for the treatment of recurrent Clostridioides difficile infection. Stool banks that organise recruitment and screening of feces donors are being embedded within the regulatory frameworks described in the European Union Tissue and Cells Directive and the technical guide to the quality and safety of tissue and cells for human application, published by the European Council.
Objective
Several European and international consensus statements concerning fecal microbiota transplantation have been issued. While these documents provide overall guidance, we aim to provide a detailed description of all processes that relate to the collection, handling and clinical application of human donor stool in this document.
Methods
Collaborative subgroups of experts on stool banking drafted concepts for all domains pertaining to stool banking. During a working group meeting in the United European Gastroenterology Week 2019 in Barcelona, these concepts were discussed and finalised to be included in our overall guidance document about fecal microbiota transplantation.
Results
A guidance document for all domains pertaining to stool banking was created. This document includes standard operating manuals for several processes involved with stool banking, such as handling of donor material, storage and donor screening.
Conclusion
The implementation of fecal microbiota transplantation by stool banks in concordance with our guidance document will enable quality assurance and guarantee the availability of donor feces preparations for patients.
Introduction
Fecal microbiota transplantation (FMT) has become an established treatment for recurrent Clostridioides difficile infection (CDI),1–4 and it appears promising as a treatment modality for other disorders. 5 In order to ensure a safe, accessible and cost-effective implementation of FMT, stool banks that provide ready-to-use donor feces preparations are required. Such stool banks may operate at an institutional, national or international level and are currently being set up in different European countries.6,7 To date, FMT and stool banking protocols vary significantly between institutions, mostly due to the novelty of this treatment approach and the scarcity of guidelines addressing FMT and stool banking. A recent international consensus meeting addressed FMT and stool banking, 8 and a British guideline concerning the use of FMT was published in 2018. 9 Based on: (a) available consensus reports; (b) previous experiences;6,7,10,11 and (c) lessons learned from blood banks, 7 an attempt was made to define a standardised model for stool banks in Europe. In addition, the regulatory boundaries that are required for safe and cost-effective FMT are outlined. This resulted in a practice-oriented consensus report including templates for standard operational procedures and questionnaires (included in the Supplementary Appendix) that may help to standardise the working plans of stool banks, and facilitate further implementation and regulation of FMT. In addition, our report will support clinicians who want to offer this treatment to their patients.
The statements made throughout this paper are supported by all working group members and describe minimum requirements. In addition, country-specific regulations need to be taken into account to complement the statements.
Methods
A multidisciplinary working group was formed with experts from mainly European countries. Authors of previously published consensus reports8,9 were invited to participate to prevent inconsistencies. Based on the working process of stool banks as previously described,5,6 and the clinical application of FMT, topics to be addressed were subdivided into five groups. Subgroups were formed based on the expertise of subgroup members. Subgroup-specific literature searches were performed prior to submission of concept documents addressing the previously defined issues/questions, and statements were phrased. During a working group meeting at the United European Gastroenterology Week in Barcelona, 23 October 2019, the concepts were discussed in depth by the entire working group. Although the aim of the working group was to provide a manual for stool banking in Europe, and not a guideline, an attempt was made to grade the evidence to support statements. The GRADE system (Grades of Recommendation Assessment, Development and Evaluation) was used to grade the strength of evidence (high/moderate/low/very low) and strength of recommendation (strong/weak). 12 Statements addressing organisational aspects of stool banking were based on expert opinion or law governed. Of note, based on the lack of available evidence, most statements are based on expert opinion. An aggregate document was prepared based on the individual working group’s concept documents and expert input during the meeting. After additional comments from experts, the document was finalised and is presented here as a summary. Additional information, containing questions for subgroups, statements discussed by the workgroup, practical standard operating procedure (SOP) templates and questionnaires are available as supplementary files.
Definitions
The definitions used in this paper are mainly based on practice recommendations by the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and by the Infectious Diseases Society of America (IDSA),1,2 and on the guide to the quality and safety of tissues and cells for human application (tissue guide) of the European Council. 13
Diarrhoea
Diarrhoea is loose stools at a frequency of three or more per 24 or fewer consecutive hours. 1
CDI
CDI is defined as a combination of compatible signs and symptoms (diarrhoea, ileus, toxic megacolon) confirmed by microbiological evidence of C. difficile toxin and C. difficile in stools without reasonable evidence of another cause of diarrhoea or by findings demonstrating pseudomembranous colitis on endoscopy or histology. 1
Treatment response
Treatment response in CDI is present when stool frequency decreases to less than three per 24 hours (or return to the pre-existent defecation frequency) over a period of at least 72 hours in combination with improvement of parameters of disease severity (clinical, laboratory, radiological).
Recurrent CDI
Recurrence is present when CDI re-occurs after the resolution of symptoms of the previous episode. Recurrence within the first 8 weeks after the onset of the previous episode is mostly due to relapse, whereas renewed symptoms during long-term follow-up may be due to relapse or reinfection. 1
Refractory CDI
Lack of response to medication after 5 days or more of treatment.
Severe CDI
Severe CDI is defined by CDI in combination with either a white blood cell count of 15 × 103/μl or greater or a creatinine level 1.5 times or greater of the baseline. 2
Complicated CDI
CDI is complicated if at least one of the following signs or symptoms are present and related to CDI (2): hypotension requiring vasopressors, intensive care unit (ICU) admission for a complication of CDI, ileus leading to placement of nasogastric tube, toxic megacolon, colonic perforation, colectomy or colostomy.
Adverse event
Any untoward occurrence associated with the procurement, testing, processing, storage or distribution of fecal preparations.
Adverse reaction
Any unintended response, including a communicable disease, in the donor or the recipient that is associated with the procurement or human application of fecal preparations.
Regulatory framework and organisational plan
Classification of FMT
FMT is regarded as a medical treatment that is carried out by registered specialists.9,14,15 Although FMT is not yet uniformly regulated within Europe, it seems obvious that the voluntarily donated material (feces) should be collected, handled and used according to the standards defined by the EU Commission in the EU Tissue and Cells Directive (EUTCD) (2004/23/ec).16,17 Feces should be regarded as a substance of human origin and may thus be considered equal to tissue, as long as no additional modifications other than those necessary for conservation and/or administration are made. 18 Unfortunately, some countries within the EU have currently classified donor feces preparations as a drug, even though only modifications necessary for conservation and/or administration are applied. Although classification as a drug may enhance the commercial environment for microbiome therapeutic drug product development, classification as a drug will inevitably result in time-consuming and costly registration processes, sharp and unjustified rise in costs, and will negatively impact availability and innovation. 17
Differentiation between stool banks and FMT services
In general, stool banks need to be differentiated from FMT services. While stool banks are responsible for the manufacture and distribution of FMT preparations, FMT services are responsible for patient treatment by the use of FMT preparations. While it is not mandatory, the organisation of an FMT service may be integrated into that of a stool bank. In relation to FMT administration, we suggest that the responsible person should be qualified to treat patients and have a specialist regristration. Centres offering FMT should accommodate fully equipped treatment facilities to handle side effects and complications of FMT procedures.
Organisation of stool banks
A stool bank should comply with the following standard requirements.16,19
Organisational description
The EUTCD requires a documented organisational plan for all intended activities and the institutional appointment of an overall responsible person. 16 This responsible person should be a registered medical specialist. The stool bank coordinator should be in charge of the dedicated personnel, the overall quality control and the reporting and handling of adverse events and reactions. The organisational plan should feature a list or a chart of primary implicated partners, their assigned responsibilities, a description of required personnel, validation and qualification concepts, audit schedules, equipment and facilities for each of the three divisions; that is: donor recruitment, screening and manufacture.
Quality management
Stool banks should implement a quality system in accordance with the requirements described in detail in the guide to the quality and safety of tissues and cells for human application (tissue guide) of the European Council. 13 Although the EU Guidelines for Good Manufacturing Practices (GMP) specifically guide the preparation of medicinal products, their content contains relevant aspects for tissue and stool banks as well. Therefore, the recently released 4th edition of the tissue guide contains good practice guidelines for tissue establishments including relevant elements of GMP.
For all activities, SOPs, guidelines, reference manuals, reporting forms and procedural protocols must be present, and should be updated continuously. Any substantial change to activities requires prior written approval from the responsible specialist. Any processes which may critically affect the quality or the safety of donated material must be identified and lead to implementation of precautionary measures. This includes describing responsible personnel, SOPs, logging and referencing manuals. All activities related to the preparation and distribution of donated material should be logged.
Incidents occuring during donor screening, the laboratory processing of donated feces, or distribution which may potentially impact patient outcome should be handled as adverse events and result in re-evaluation of (adherence to) protocols. Auditing by local or preferably (in the future) national authorities should be part of the quality management of a stool bank. Such audits should ensure that data are entered and maintained properly, and evaluate the working processes and quality assurance programme of the stool bank.
Personnel
All personnel should have clearly defined and documented work descriptions. To ensure competencies, the personnel should be trained in relevant hygiene measures, safety procedures, technical protocols and the organisation before conducting any activities.
Activity
A stool bank should maintain a registry of its activity and should be able to collect and report data on the patients treated with the preparations it distributes. Reporting should include the number of patients, the treatment modality used (colonoscopy, enema, gastric or duodenal tube, gastroscopy, capsules, etc.), as well as follow-up data including safety and outcome of FMT. In the near future, these data should also be entered into a national or international registry.
Distribution
The distribution of fecal preparations from a stool bank to an FMT service requires a (written) agreement between the stool bank and the FMT service in which each party’s responsibilities are stated. Prior to distribution, the third party must confirm that it is in possession of sufficient facilities to conduct FMT safely. It is the duty of the stool bank to evaluate and select third parties. Clinical application and patient follow-up is the responsibility of the treating clinician at the FMT service. The FMT service has to report these data back to the stool bank as described above (in Activity). SOPs are required for requests of FMT preparations by third party institutions, and clear instructions on how to handle the preparations should be available. Return policies of preparations from third party institutions should be handled according to a shared SOP.
Adverse reaction documentation
Adverse events, adverse reactions (ARs) and serious ARs as defined by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) harmonised tripartite guidelines 20 should be documented. The stool bank should be able to communicate data related to performance, outcome and safety in an annual report for national health authorities or other auditing authorities. Quality auditing should be organised in collaboration with local or national authorities. Complications and ARs of FMT are generally underreported.21,22 Expected ARs following FMT include short-term (including among others abdominal pain, dyspepsia, diarrhoea, fever, constipation) and long-term (including post-infectious irritable bowel syndrome) reactions. 22 In addition, unexpected ARs may occur and careful monitoring is required. A serious AR may be procedure related (such as aspiration, perforation), or not clearly related to the procedure. A distinction between procedure-related and unrelated complications should be made according to established guidelines.23,24 Each stool bank or FMT service should have a SOP to describe how to handle serious ARs. In the case of any serious AR that is deemed related to FMT, the unit is obligated to notify the appropriate authority immediately. The stool bank is responsible for ensuring notification to the health authorities and confirming that sufficient actions and adaptations are being made. All relevant personnel and divisions should be notified in the case of a serious AR to facilitate traceability and control of quality and safety. Effective and rapid procedures for recall and disposal must be in place in the case of a serious AR.
Data management
Anonymity
All data regarding donors and recipients should be anonymised to maintain a principle of voluntary altruism between donor and recipients without any revealing elements. This also applies to third party data access. All steps of the donation should therefore be uniquely coded and linked using specific donor identifiers, donation identifiers and donation product identifiers.
Traceability
An efficient and well protected data storage system for logging should be in place, allowing accurate tracing of the entire process from donor to recipient. Both donor and processing records should be kept. All data necessary to map the process and handling should be kept for a minimum of 30 years.
Storage
Paper-based documentation systems are acceptable, but secure electronic systems that are auditable, traceable and retrievable are preferred. Storage must be in accordance with local data protection regulations.
Donor recruitment, selection and screening
Donor recruitment
Donation of stool is a voluntary act and should not become a subject of trade. The use of unpaid donors reduces the risk that applicants withhold information during the screening process. Reimbursement of expenses for travel to the donation centre is reasonable. Universal donors are preferred over patient-selected donors. In individual cases, a patient-selected donor may be accepted, if (s)he fulfils all criteria determined in the screening process for universal donors. Healthcare workers may be approved as donors if they fulfill all necessary requirements, although some stool banks choose to exclude them because of a potential risk of multi-drug resistant organism (MDRO) carriership.
Donor informed consent
All donors should be informed about the associated risks and benefits and provide written informed consent that covers the provision of personal information, the screening processes, the provision of multiple donations, the storage of donor data in a donor registry and future unscheduled contacts by the stool bank in case of adverse events or for research purposes. A proposed template for an informed consent form for healthy donors is included in the Supplementary Appendix.
Donor screening
Donor screening should be conducted in accordance with a locally approved set of SOPs and performed by dedicated staff. Meetings between the donor screening staff and the rest of the stool bank team need to be scheduled on a regular basis, in order to identify problems with respect to screening, or the availability of donations well in advance. The collected data should be documented on pre-specified forms that facilitate the structured display of results obtained during the screening process (interviews/questionnaires and microbiological testing) and include documentation of donor clearance.
Initial questionnaire
During the first step of the screening process, the medical history and risk behaviour of potential donors should be assessed by the use of a dedicated questionnaire. The results should be evaluated by a clinician. Objective evaluation should include a document clearly identifying the consequence of a specific response for the screening process. The topics and items that should be addressed in this questionnaire are listed in Table 1. A proposed questionnaire with consequences for positive answers is added to the Supplementary Appendix.
Items to be assessed during screening of donors before approval for FMT.

Exclusion criteria related to each item are presented in Supplementary File 3.
Some stool banks accept donors with a BMI >18 and <30 kg/m2.
BMI: body mass index; FMT: fecal microbiota transplantation.
Microbiological testing
Once a potential donor has been found suitable for further evaluation based on the donor questionnaire and the physical examination, he or she should undergo blood and feces screening for transmissible pathogens listed in Table 1.
Stool donation, quarantine and retesting of donors
Once a donor has been approved, (s)he should complete a second short questionnaire before each donation, assessing the occurrence of any event that may have occurred between donor approval and donation. Complete donor screening based on blood and fecal analyses should be repeated a minimum of every 3 months. A donation period should have a defined maximum duration, not exceeding 3 months, and should start and end with a complete donor screening. Blood tests pertaining to the close-out screening should be performed a minimum of 4 weeks after the last donation to allow for a lag time of serology or polymerase chain reaction (PCR)-based testing for certain pathogens. FMT products prepared during a donation period should be put under quarantine until the repeat donor screening results are available.
If donor screening is performed as recommended in this document, direct testing of each preparation is not mandatory. It may, however, be required by local regulations.
Donor screening for treatment of patients with severe immunosuppression
Additional screening tests are advised for the use of FMT preparations in severely immunocompromised patients (see Table 2, and Supplementary File 2).
Blood and feces tests for donor screening before approval for FMT.
Enteropathogenic Escherichia coli (EPEC) testing may be considered in some countries.
Covid-19 (SARS-CoV-2) testing of asymptomatic donors combining serology and stool testing requires further validation.
If residing in or foreign travel to tropical country within the past 6 months.
Colonisation with Blastocystis hominis is not considered an exclusion criterium. However, the working group advises to monitor for the effects of transmission.
Several stool banks also include HTLV 1 and 2 testing.
Additional screening tests advised for immunocompromised patients (see Supplementary File 8):
Stool: Plesiomonas shigelloides, adenovirus, parechovirus, astrovirus, enterovirus, sapovirus, microsporidia.
Blood: CMV, EBV, toxoplasmosis.
ALT: alanine aminotransferase; CMV: cytomegalovirus; CRP: C-reactive protein; EBV: Epstein–Barr virus; FMT: fecal microbiota transplantation; PCR: polymerase chain reaction.
FMT products should be put under quarantine until the donor has been found acceptable in a repeat screen. For severely immunocompromised patients (see paragraph below about the clinical application of FMT), the prepared FMT preparation itself should undergo a quality control that includes (re)screening for potential pathogens as listed in Table 2.
Processing and storage of donor feces preparations
Donor feces collection and preparation for FMT should follow a standard protocol to ensure that products are consistently produced and controlled with respect to the quality standards appropriate to their intended use. The process described below is based on experiences in different centres and has not been tested rigorously. There are no reported studies comparing different preparation protocols of fecal preparations. A possible protocol is presented in Table 3. Following a standard protocol enables the comparison of outcomes between institutions, facilitating quality assurance. The described protocol has proved effective for FMT treatment of recurrent CDI; for other potential indications, adjustment of the protocol may be required in the future.
Validated standardised conditions for processing of FMT suspensions and treatment of patients with recurrent Clostridioides difficile infection.
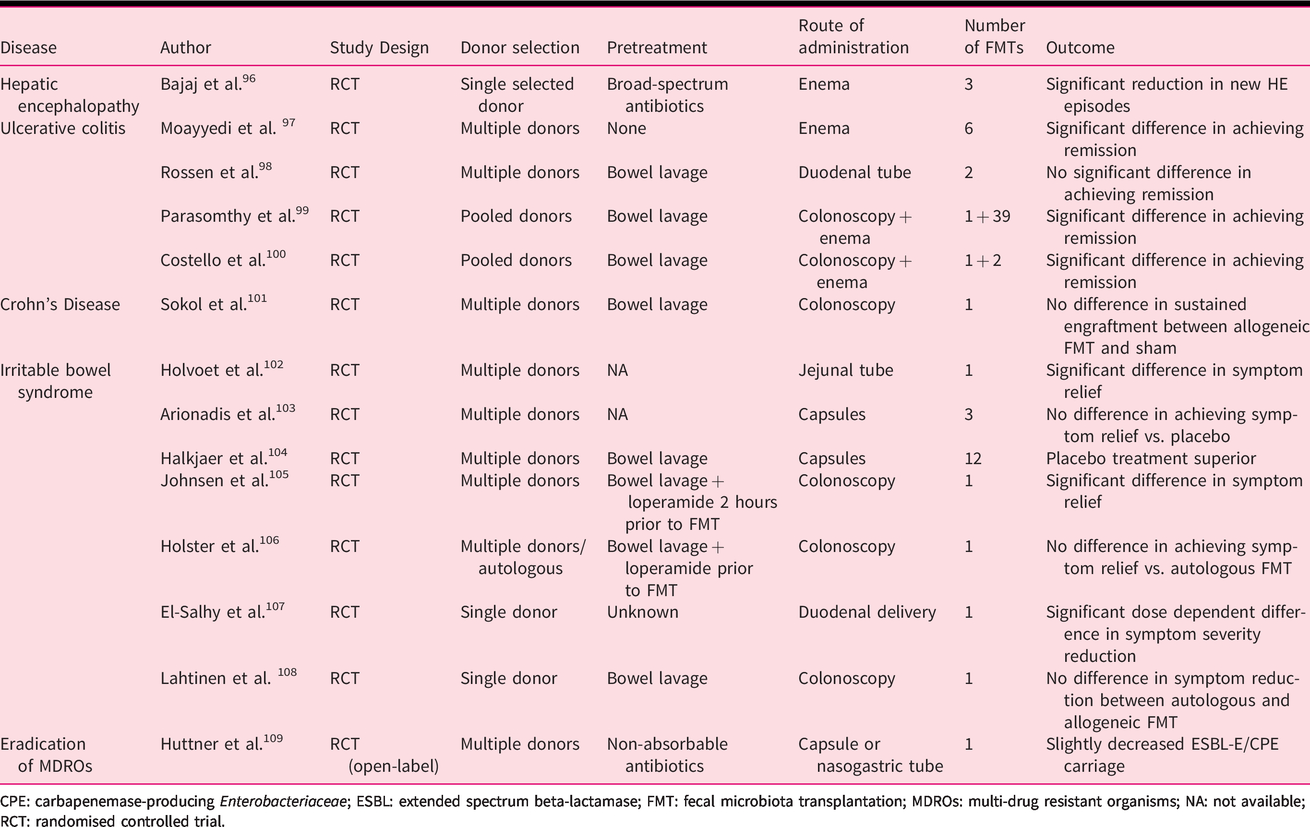
For upper gastrointestinal delivery for treatment of CDI, bowel lavage could be limited to 50% of the advised dose for colonoscopy cleansing.
CDI: Clostridioides difficile infection; FMT: fecal microbiota transplantation.
Collection of donor stool
To promote standardised practice and a safe and effective product, clear written instructions should be provided to the donor for feces collection and delivery procedures. To prevent environmental/cross-contamination, feces is collected by the donor in a fecal container for single use only (e.g. fecotainer).
Donors should hand in their feces within 2 hours after defecation; some stool banks prefer on-site donation. After defecation and until further processing, the stool sample can be stored at room temperature. If it takes more than 30 minutes to deliver the collected feces to the stool bank, temporary storage in a cooler bag or refrigerator is preferred because fecal storage without stabilisation buffer significantly changes taxa abundances from 30 minutes onwards.25–28
Processing of preparations
In most studies that performed FMT for recurrent CDI, donor feces was processed within 6 hours of defecation.4,11,29–48 Studies in which a longer interval between defecation and processing was allowed (24–48 hours) report a somewhat lower cure rate of FMT (approximately 75%).49–52 In addition, it is hypothesised that a high viability of bacteria in donor stool increases the efficacy of FMT. Subsequently, this could allow a reduced amount of donor feces used per suspension. To minimise sample degradation and alteration, donor feces should be processed as soon as possible as most fecal bacteria are anaerobic.
The preparation of fecal preparations under aerobic conditions is considered suitable for FMT treatment of recurrent CDI, as anaerobic processing does not increase cure rate.53–55 This observation may be explained by the fact that a considerable part of the bacterial genera can tolerate oxygen for a limited amount of time or produce resilient spores that allow transfer of at least a proportion of anaerobic intestinal bacteria. 56
Pooling (mixing) of multiple donor feces during processing is not recommended because: (a) it hampers the traceability of the fecal preparation to the individual donor; (b) the risk of transmissible disease may be increased; and (c) the principle of transfusing a balanced microbiota preparation may be lost.
It is preferable to use 50 g or more of stool to prepare a FMT suspension for recurrent CDI treatment. Systematic reviews suggest that the use of 50 g or more of feces is more effective than less than 50 g.57,58 However, the strength of this recommendation is impaired by the absence of comparative studies, limited numbers of recent studies and variation in delivery routes. Several experts report (mostly unpublished) positive results with less than 50 g of donor feces. 59 Reducing the amount to 25 g (or even lower) could therefore be considered but should be validated.
In general, the fecal suspension is made with preservative-free sterile 0.9% saline as diluent.4,11,29,31,33–36,38–42,45–47,50–55,60–75 The suspension should not be too viscous for infusion through a naso-duodenal tube or biopsy channel of a gastroscope or colonoscope.
The suspension can be homogenised by a variety of methods such as blenders,29,33–35,40,41,44,46,50,53,55,60,61,65,67,69,73,74 stomachers,31,51,71 with mortar and pestle, 6 or wooden spatulas.42,49,76 Of utmost importance is the use of clean consumables, which implies that materials should be decontaminated using a validated method if reused or should be disposable. To prevent clogging of the tube/biopsy channel during the administration procedure the fecal suspension should be filtered. Filtration can be performed by a gauze, filter paper, strainers or sieves. To prevent external contamination either a closed system or an open system in a biological safety cabinet is preferred. To combine homogenisation and filtration, a bag mixer appears easy to handle and is efficient.
The infused volume may be reduced using centrifugation,6,55,61,74 but the reported cure rate of 68% after multiple centrifugation and washing steps appears relatively low, 39 and such an approach should be validated.
To prepare frozen preparations, a cryoprotective agent should be added prior to freezing. In general, the cryoprotectant glycerol is used in a final concentration of 10–15%.31,39,42,43,53,55,61,66,69,71,73,74,77 Cryopreservation is a process of preservation of the biological and structural functions of tissues or cells when cooling to sub-zero temperatures. The viability of six representative groups of fecal bacteria after 6 months of storage at –80°C in normal saline with 10% glycerol did not differ from baseline, whereas viability was reduced in suspensions stored without glycerol. 39 A possible side effect of large amounts of glycerol in the bowel is a mild alteration in serum glucose. This is not observed with doses below 0.75 g of glycerol per kg body weight. 78 A feces preparation of 200 cc, containing 10% glycerol (approximately 20 g) is far below this limit.
Encapsulation of glycerol preparations and cryopreservation of encapsulated donor feces is feasible and could reduce the burden of FMT for patients in the future, but requires further validation. 79
Storage of FMT preparations
FMT preparations should be stored in sealed, clean plastic containers, with a unique code ensuring traceability of the sample (see data management). Storage of FMT preparations (intended for treatment) should be accommodated by the stool bank in a dedicated –80°C freezer with connected alarm notification to guarantee continuous registration of the storage conditions. Documentation requires a biobanking information and management system for coding, registrations and tracing of the samples.
All processed FMT preparations should be placed into quarantine and stored in clearly distinguishable sections until all donor screening results are available. The release of products must be documented, and following these quarantine measures, the FMT preparations should be cleared and transferred to another distinct storage section. Only cleared FMT preparations should be used for patient treatment.
Distribution of FMT preparations should be performed on dry ice (or 4°C or room temperature in the case of immediate use after transfer) shipment through a certified courier service. Registration in a ‘biobanking information and management system’ related database for the shipped fecal preparations, including recipient institution and requestor information, should be undertaken so that biovigilance tracing can be performed in case of ARs. Long-term storage of fecal preparations at the recipient centre should be discouraged.
To ensure the maximum safety and quality of the FMT preparation, it is mandatory to specify a maximum storage time with an expiry date. The association between storage conditions and clinical efficacy has not been investigated. Storage at –80°C is preferred, although storage of fecal preparations at –20°C for up to one month (30 days) may not reduce efficacy. 80 In general, long-term storage should be at –80°C or lower to prevent sample degradation. Once preparations are transferred to a ward or an endoscopy unit for administration, temporary storage at –20°C is acceptable. High cure rates have been reported, with frozen FMT suspensions stored up to 10 months at –80°C,6,31,42,43,53,55,73,74 but this could in theory be much longer. OpenBiome and The Netherlands Donor Feces Bank have good experiences with storage up to one and 2 years, respectively. 6,81
A sample of the original donor feces and/or of the processed FMT preparation should be stored for a minimum of 10 years following the application for retrospective quality assessment in case of an AR.
Thawing of donor feces preparations
Thaw times depend on the volume and type of FMT preparations. A 200 cc suspension can be thawed overnight in a 4°C refrigerator, or during 5 hours at room temperature. Warm water baths (37°C) can be used to speed up thawing of FMT suspensions, but are not applicable to the thawing of capsules. However, only clean water baths with fresh water should be used to avoid cross-contamination (especially with water-dwelling bacteria such as Pseudomonas). Thawed fecal suspensions should be infused the same day, and should not be refrozen, because freeze–thaw cycles adversely affect the viability of the microbial communities in the fecal suspension. 82
The fecal suspension should be at room temperature when infused into the recipient, to minimise abdominal discomfort.
Clinical application of FMT
The primary aim of a stool bank is to centralise the process of donor screening and processing and storage of donor feces preparations. Given the novelty of this treatment modality, guidance for treating physicians may be required to optimise the safety and efficacy of large-scale FMT implementation. Stool banks can offer expert consultation at the request of the treating physician, or they can incorporate consultation as part of a standard procedure to verify the correct indication for FMT before delivering FMT preparations. Currently, stool preparations are indicated for recurrent CDI only. In a research or compassionate use setting, other indications could be considered. Stool should not be sold or delivered to patients upon their own request. Finally, the need for documentation and quality improvement requires a strong and well-defined infrastructure.
Consultation
To offer appropriate consultation, an expert panel is required. As indications for FMT are likely to become more numerous in the future, medical specialists in the field pertaining to the added indications should be included in the FMT expert panel of a stool bank. A standardised patient questionnaire with key information about the current episode of CDI, previous episodes of CDI, treatment effects and microbiological testing is required (see Supplementary File 8). The expert panel may assess whether the indication is appropriate and advise about additional diagnostics or an alternative therapy. Based on such a consultation process, The Netherlands Donor Feces Bank has rejected about a quarter of the requests for FMT preparations for treatment of recurrent CDI.6 An even higher rate of inappropriate FMT indications (around 50%) was noticed by a French reference centre (St Antoine Hospital, Paris). If regular consultation is not offered by a stool bank, treating clinicians should receive contact information for consultation in case of: (a) unexpected problems during the FMT treatment; and (b) unexpected ARs.
Patient data should be shared and stored appropriately, in accordance with European General Data Protection Regulation (GDPR) guidance and other local regulations.
Treatment protocol
A standardised treatment protocol for recurrent CDI will enable the comparison of outcomes among FMT centres, and subsequently increase safety and facilitate quality assurance. However, variation of almost all steps of the FMT treatment protocol is conceivable. Therefore, a validated treatment protocol is presented in Table 3.
Pretreatment of patients
In clinical practice, antibiotics are initiated directly after a positive C. difficile test. It is unknown if pretreatment with antibiotics increases the effectiveness of FMT, but most centres perform FMT following at least 4 days of oral vancomycin (125–250 mg qid) or fidaxomicin (200 mg bid). Antibiotics should be stopped the day before FMT (≥24 hours before FMT). Bowel lavage is required before a colonoscopy. In general, (modified) bowel cleansing is also performed one day before donor feces infusion via the upper gastrointestinal route; this could for example include 2 instead of 4 l polyethylene glycol-electrolyte lavage solution. 6 Whether bowel lavage increases clinical efficacy for the treatment of CDI is unknown.
Administration method
In a small study, FMT via a naso-enteric tube (upper gastrointestinal delivery) or colonoscopy (lower gastrointestinal delivery) appears equally effective (P = 0.53). 74 In addition, nasogastric administration is possible after pretreatment with proton pump inhibitors (PPIs). 33 One study suggested that treatment with enemas more often requires repeated infusions, 49 but this study used low volume enemas (50 ml) and no bowel lavage. Capsules containing either lyophilised or glycerol-preserved feces suspensions also appear effective.62,79 The choice for the route of delivery should be based on local preferences and patient characteristics, if applicable. A volume of 200 cc can be safely infused via the upper gastrointestinal route. Slow infusion of the donor feces suspension (for example 10–25 cc/minute) may prevent nausea and regurgitation/aspiration. In patients with impaired gastrointestinal motility, infusion through a colonoscope is preferred to prevent regurgitation/aspiration. Moreover, upper gastrointestinal delivery (including capsules) should be avoided in patients with swallowing disorders, because of the increased risk of aspiration. Small intestinal bacterial overgrowth has not been recognised as an adverse event of upper gastrointestinal delivery of FMT. In frail patients, ileocolonoscopy can be avoided by upper gastrointestinal delivery, rectosigmoidoscopy or enema. The infusion of the donor feces suspension is straightforward and can be performed by any appropriately qualified healthcare professional.
Indications and contraindications for FMT
The development of national and international guidelines for the clinical applications of FMT is a dynamic process, driven by the emerging body of clinical evidence. The indications and contraindications outlined below represent the consensus of the working group.
Indications
FMT should be considered in patients with two or more CDI recurrences. A balance of risks and benefits should guide a joint decision of either FMT or (tapered) antibiotics with or without additional treatment with, for example, monoclonal antibody or non-absorbable antibiotics. Few studies have addressed the effects of FMT in patients with refractory or severe CDI, although FMT as rescue treatment appears encouraging.38,63,83–86 We suggest FMT as an option for patients with refractory or severe CDI. The route of FMT application in severely ill patients should be guided by the complication risk of the individual patient. Toxic megacolon should mainly be treated surgically. In patients with fulminant disease, a diverting ileostomy or other surgical treatment options should be considered. In patients who refuse surgery, or in whom surgery is somehow contraindicated, FMT via the lower or upper gastrointestinal route can be cautiously considered and has resulted in cure.45,53 Because the efficacy of FMT in severe and complicated CDI appears lower, repeated infusions are often required.
Contraindications
Currently, no data from clinical studies or reviews indicate absolute contraindications for FMT. Based on expert opinion, the following contraindications for FMT apply: anatomical abnormalities that prevent safe application of FMT by endoscopy, enema or tube; signs of intestinal perforation; pregnancy and severe food (e.g. peanut) allergy. The latter may be solved by a patient-related donor with strict dietary restrictions before stool donation. Further limitations appear mainly related to the mode of application. Relative contraindications for upper gastrointestinal delivery are increased risk of regurgitation (e.g. known large hiatus hernia, severe gastro-oesophageal reflux disease), swallowing disorders and impaired small bowel motility or obstruction. An increased risk of sedation may be considered a contraindication for lower gastrointestinal delivery by colonoscopy.
FMT for other indications
Both patients and clinicians are currently exploring the opportunities for FMT to alter the course of a large variety of diseases. Although promising, studies that confirm the efficacy of FMT for other disorders besides CDI are lacking, limited, or contradicating, and FMT is not mentioned in treatment guidelines for disorders beyond C. difficile infection. Although randomised trials suggest a beneficial effect in a subset of patients with hepatic encephalopathy and ulcerative colitis, many questions about the optimal use of FMT for those patients remain. For irritable bowel syndrome, the results of randomised studies are less consistent. Until further evidence becomes available, FMT for other indications should preferably be limited to the research setting or, in the absence of alternative therapeutic options, as compassionate use. The results of studies addressing FMT for other indications are summarised in Table 4.96–108
Summary of studies addressing the efficacy of FMT for other disorders.
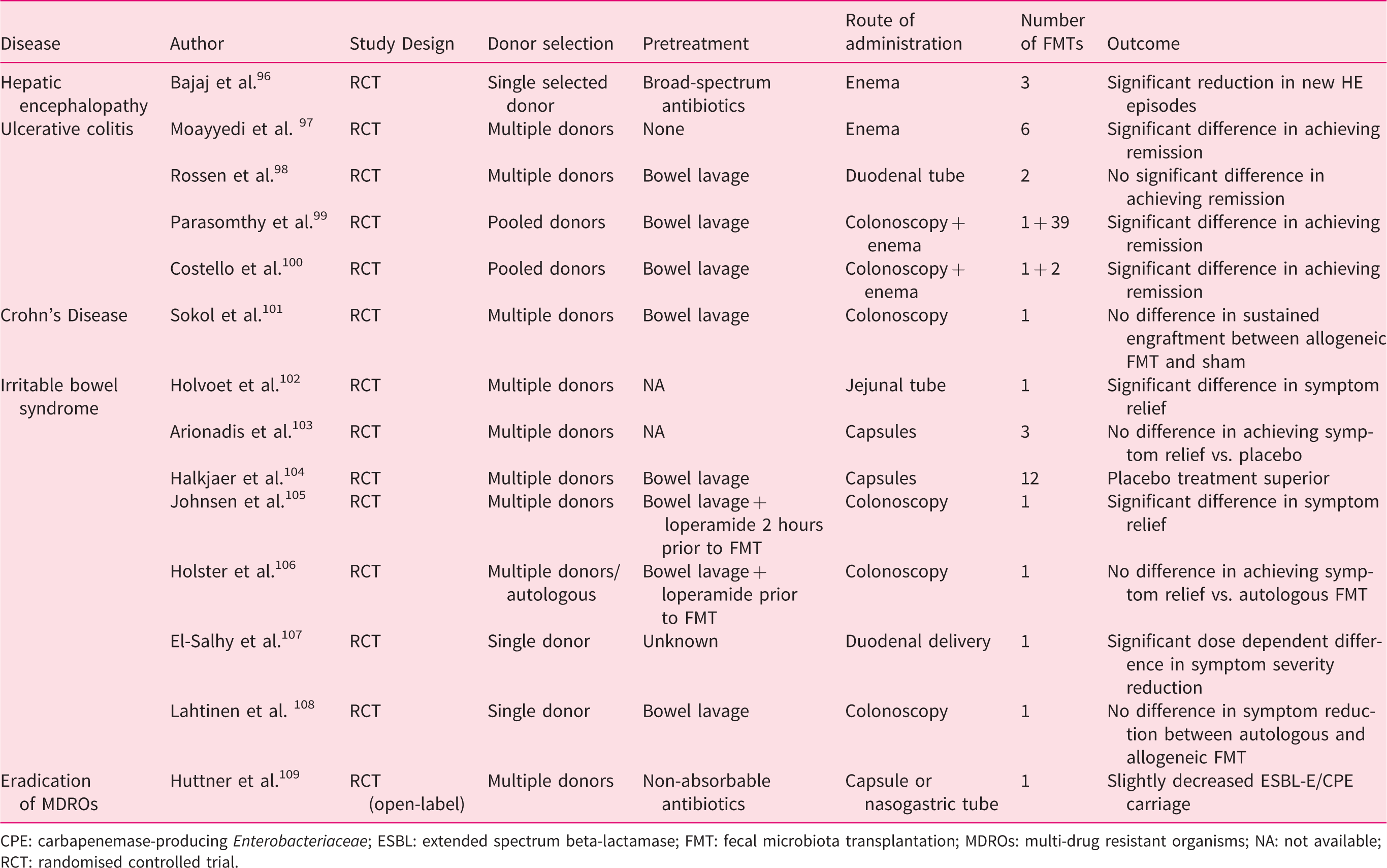
CPE: carbapenemase-producing Enterobacteriaceae; ESBL: extended spectrum beta-lactamase; FMT: fecal microbiota transplantation; MDROs: multi-drug resistant organisms; NA: not available; RCT: randomised controlled trial.
Special situations
Children
Although rare, the use of FMT in children suffering from CDI has been described.87,88 Antibiotics remain the cornerstone of initial CDI treatment, but recent European and American guidelines recommend FMT for children suffering from multiple recurrent CDI.2,89 FMT protocols for children and adults are similar. It is unknown whether age-matched donors should be considered for paediatric use. Donor preparations from adults appear safe, but long-term effects are unknown and careful follow-up is warranted. Particular attention is required because the microbiota rapidly develops during infancy, and maturation of the microbiota in children occurs parallel with the maturation of the immune system; processes which are likely to influence each other. 90 Endoscopy and the instillation of suspensions may be performed under general anaesthesia with intubation.87,88 As for adults, existing guidelines only recommend the use of FMT in children for recurrent CDI. 2
Immunocompromised patients
FMT appears safe in immunocompromised patients. 91 The recent report of a severely immunocompromised patient’s death following transfer of a MDRO calls for particular caution in this patient group. 92 The risk of developing complications after the transfer of potential pathogens appears increased in severely immunocompromised patients, defined as: (a) patients with current or foreseeable neutropenia within the next 14 days (defined as <500 neutrophils/µl); (b) patients scheduled for allogeneic stem cell transplantation (SCT) or having received allogeneic SCT within 100 days; and (c) patients with active graft versus host disease requiring immunosuppressive treatment. For severely immunocompromised patients, we suggest: (a) additional screening tests of donors; and (b) testing of the FMT preparation or donated stool used for treatment of the individual patient, as described in the Supplementary Appendix. For less severely immunocompromised patients (<200 CD4 T cells/µl; or prolonged use of corticosteroids at a mean dose of ≥0.3 mg/kg/day of prednisone equivalent for over 3 weeks; or treatment with other recognised T-cell immunosuppressants, such as cyclosporin, TNF-α blockers, specific monoclonal antibodies (e.g. alemtuzumab), methotrexate or nucleoside analogues during the past 90 days); or inherited severe immunodeficiency (e.g. chronic granuloumatous disease or severe combined immunodeficiency), normal donor screening appears sufficient. More stringent follow-up is justified in all immunocompromised patients, and careful reporting of outcomes and side effects is needed to confirm the safety of FMT in those patients.
Covid 19-pandemic
During the covid-19 pandemic, stool banks have to re-evaluate their working processing to optimise safety for donors, patients and healthcare professionals, as outlined in a recent consensus report. 93 Screening for SARS-CoV-2b should be incorporated in donor screening protocols (Table 2).
Follow-up of patients after FMT
Patients who receive FMT should be registered by the stool bank with support of the treating FMT service, in order to document patient flow, ensure traceability and document treatment outcome and treatment-related complications. A structured patient follow-up should be conducted. This should consist of clinical information and relevant paraclinical tests, depending on the indication for FMT. Short term follow-up includes the documentation of side effects or complications that occur in relation to the treatment or within the first 24 hours after any FMT-related procedure. Long-term follow-up includes the documentation of clinical details and relevant clinical results beyond the first 24 hours. The duration of long-term follow-up depends on the indication, but should cover a minimum of 8 weeks and preferably 6 months. Relevant clinical variables to document during follow-up are general symptoms, disease-specific symptoms, standardised symptom or disease activity scores and patient weight.
FMT appears safe with limited side effects,21,94 although infectious complications have been described in severely immunocompromised recipients underlining the need for careful donor screening. 92 However, some concern about the long-term side effects of FMT has been raised. This may include weight gain, the development or worsening of inflammatory bowel diseases, cancer, autoimmune diseases, allergies, or neurological diseases. 95 Follow-up should include clinical and analytical data, also determined by the indication for FMT and comorbidity of the patient. National or institutional registries that are able to perform long-term follow-up, and collect outcome data after FMT for various conditions are warranted to document the long-term safety of FMT.
Conclusions
FMT is a new, safe and efficacious treatment strategy. The critical step of FMT is the process of donor selection and screening. Stool banks have enabled centralisation of this process, following the model of blood banks. We provide a model for the standardisation of stool banking; key recommendations are summarised in Table 5. Standardisation will result in further improvement of the quality and safety of FMT in Europe. Finally, stool banks will enable quality assurance of FMT and guarantee the availability of donor feces preparations for patients.
Summary of key recommendations regarding FMT and stool banking.

CDI: Clostridioides difficile infection; FMT: fecal microbiota transplantation
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620967898 - Supplemental material for A standardised model for stool banking for faecal microbiota transplantation: a consensus report from a multidisciplinary UEG working group
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620967898 for A standardised model for stool banking for faecal microbiota transplantation: a consensus report from a multidisciplinary UEG working group by Josbert J Keller, Rogier E Ooijevaar, Christian L Hvas, Elisabeth M Terveer, Simone C Lieberknecht, Christoph Högenauer, Perttu Arkkila, Harry Sokol, Oleksiy Gridnyev, Francis Mégraud, Patrizia K Kump, Radislav Nakov, Simon D Goldenberg, Reetta Satokari, Sergiy Tkatch, Maurizio Sanguinetti, Giovanni Cammarota, Andrey Dorofeev, Olena Gubska, Gianluca Ianiro, Eero Mattila, Ramesh P Arasaradnam, Shiv K Sarin, Ajit Sood, Lorenza Putignani, Laurent Alric, Simon MD Baunwall, Juozas Kupcinskas, Alexander Link, Abraham G Goorhuis, Hein W Verspaget, Cyriel Ponsioen, Georgina L Hold, Herbert Tilg, Zain Kassam, Ed J Kuijper, Antonio Gasbarrini, Chris JJ Mulder, Horace RT Williams and Maria JGT Vehreschild in United European Gastroenterology Journal
Footnotes
Acknowledgements
The templates of standard operational procedures are based on SOPs provided by The Netherlands Donor Feces Bank and Aarhus University Hospital, Aarhus, Denmark. The presented informed consent form for healthy donors was provided by the Cologne Stool Bank, Germany. The working group was hosted by the European Helicobacter and Microbiota Study Group (EHMSG), which is a member society of UEG. Delegates of the ESCMID study group for Host and Microbiota Interaction (ESGHAMI) group participated in the working group. The working group was supported by Romana König.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JJK and EJK have received research grants from Vedanta Biosciences.
CP has received grant support from Takeda, speaker’s fees from Takeda, Tillotts and Pfizer, and consultancy fees from Takeda, Shire and Pliant.
RN is in the speaker’s bureau of Takeda and Pfizer.
PA has received a research grant from Vifor Pharma, and speaker’s fee from MSD.
MJGTV has received research grants from 3M, Astellas Pharma, DaVolterra, Gilead Sciences, Heel, Glycom, MaaT Pharma, Merck/MSD, Organobalance, Seres Therapeutics; speaker’s fees from Astellas Pharma, Basilea, Falk Foundation, Gilead Sciences, Merck/MSD, Organobalance, Pfizer and has been a consultant to Alb Fils Kliniken GmbH, Astellas Pharma, BioMérieux, DaVolterra, Ferring, MaaT Pharma, Merck/MSD and Roche.
SDG reports consultancy fees from Astellas, Enterobiotix, Menarini, MSD, Pfizer and Shionogi.
OG reports speaker's fees from Abbott and a research grant from Biocodex.
HS reports board membership, consultancy, or lecture fees from Carenity, Abbvie, Astellas, Danone, Ferring, Biocodex, Mayoly Spindler, MSD, Novartis, Roche, Tillots, Enterome, Maat, BiomX, Biose, Novartis and Takeda; and is a co-founder of Exeliom Biosciences.
ZK is an employee/shareholder of Finch Therapeutics.
AL reports a grant from European Commission European funds for regional development (EFRE) related to microbiome and FMT research.
GLH reports consultancy fees from Ferring and Proctor and Gamble; and scientific board membership of Enterobiotix.
EM has been a scientific advisor and received lecture fees from MSD Finland.
CH receives grant support from Seres Therapeutics and consultancy, or lecture fees from Abbvie, Astellas, Ferring, Falk, Janssen, Merck/MSD, Pfizer, Takeda.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The working group was supported by an UEG standards and guidelines activity grant.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
