Abstract
Background
Gastrointestinal bleeding is a rare but severe complication of pancreatic ductal adenocarcinoma.
Objective
The purpose of this study was to describe the causes and treatments of non-postoperative gastrointestinal bleeding in patients with pancreatic ductal adenocarcinoma, and explore the parameters associated with therapeutic effectiveness.
Methods
This was a single-centre observational retrospective study (2000–2017) with data collected from the prospectively coded diagnostic hospital’s database system including patients with pancreatic ductal adenocarcinoma who had a gastrointestinal bleeding episode. Effectiveness of haemostatic treatment was assessed according to transfusion requirements and immediate and long-term haemostatic efficacy; the latter defined as no bleeding recurrence.
Results
The population included 72 patients with pancreatic ductal adenocarcinoma who had 94 episodes of gastrointestinal bleeding. The main causes of gastrointestinal bleeding were gastroduodenal tumour invasion (56.4%) and oesophageal variceal bleeding due to left-sided portal hypertension (19.1%). In cases of gastrointestinal bleeding caused by tumour invasion, the main treatment was therapeutic endoscopy (41.5%). Among patients who had gastrointestinal bleeding by tumour invasion treated by endoscopy or radiation therapy, haemostatic immediate efficacy rates were 70.6% and 100%, respectively. Bleeding recurrence rates were 35.3% and 25.0%, for patients treated by endoscopy or radiation therapy, respectively, for a first episode of gastrointestinal bleeding by tumour invasion. Transfusion requirements, before and after treatment, were not different in patients treated by haemostatic radiation therapy for gastrointestinal bleeding by tumour invasion compared to other treatments (odds ratio 0.3, 95% CI (0.06–1.59); p = 0.16). The median survival after all-cause gastrointestinal bleeding was 2.72 months (1.43–4.01).
Conclusion
Gastroduodenal tumour invasion was the main cause of gastrointestinal bleeding in patients with pancreatic ductal adenocarcinoma; haemostatic radiation therapy is a potential interesting option for gastrointestinal bleeding treatment in this context.
Keywords
Key summary
Summarise the established knowledge on this subject
Non-postoperative gastrointestinal bleeding in patients with pancreatic ductal adenocarcinoma seems infrequent, but is a complication associated with a very poor short-term prognosis.
What are the significant and/or new findings of this study?
The three main causes of digestive bleeding in this context are gastroduodenal tumour invasion, oesophageal variceal bleeding due to left-sided portal hypertension, and radiation-induced gastritis. One-third of patients having a gastrointestinal bleeding episode – caused by tumour invasion – have a recurrent bleeding episode after a first therapeutic endoscopy. Haemostatic radiation therapy is a non-invasive therapeutic option to be discussed in selected patients with gastrointestinal bleeding by tumour invasion not treatable with endoscopic treatments.
Introduction
Although non-postoperative gastrointestinal bleeding (GIB) in patients with pancreatic ductal adenocarcinoma (PDAC) seems infrequent, this complication is associated with a very poor short-term prognosis.1,2 Limited data exist on causes, prognosis and specific management of GIB in this context.
Digestive endoscopy remains the cornerstone for the diagnosis of GIB, to identify its cause, to stratify the risk of recurrence and for its treatment.3–6 However, when the bleeding surface is large, especially in cases of tumour invasion, endoscopic haemostasis is generally impossible or ineffective. Therefore, haemostatic radiation therapy (HRT) can be a therapeutic option in GIB due to gastroduodenal tumour invasion, although its effectiveness has been poorly described in this context.7–10
The aims of this study were to describe the characteristics, causes and treatments of GIB in patients with PDAC. Additionally, we paid a particular attention in describing the different haemostatic treatments used for GIB caused by tumour invasion, by assessing haemostatic therapeutic effectiveness according to transfusion requirements, immediate haemostatic efficacy, GIB recurrence rate and 30-day mortality rate, particularly after HRT.
Materials and methods
Patients
We conducted a retrospective single-centre observational study. Data were collected from the prospectively coded diagnostic database system of the Beaujon Hospital (Clichy, France), between 1 January 2000–31 December 2017. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki. However, as a retrospective study, it is exempt of ethical committee approval.
The study population included all patients with a diagnosis of PDAC who had one or more GIB episodes reported in the hospital’s diagnostic database (Supplementary Material Table 1), during this period. Patients with one of the following conditions were excluded from the study: primary duodenal tumour, ampullary tumour, cholangiocarcinoma, pancreatic tumours other than PDAC or without proven GIB, or with insufficient data on the characteristics of GIB in their medical record. Patients with GIB during the early postoperative period (within 30 days following pancreatic surgery), lower and upper GIB unrelated to the tumour or its prior treatment, were also excluded. Patient data were collected from medical records on DxCare medical software. Tumour progression at the time of GIB was also collected from the most recent follow-up computed tomography (CT) scan and defined according to Response evaluation criteria in solid tumours (RECIST) v 1.1 criteria.
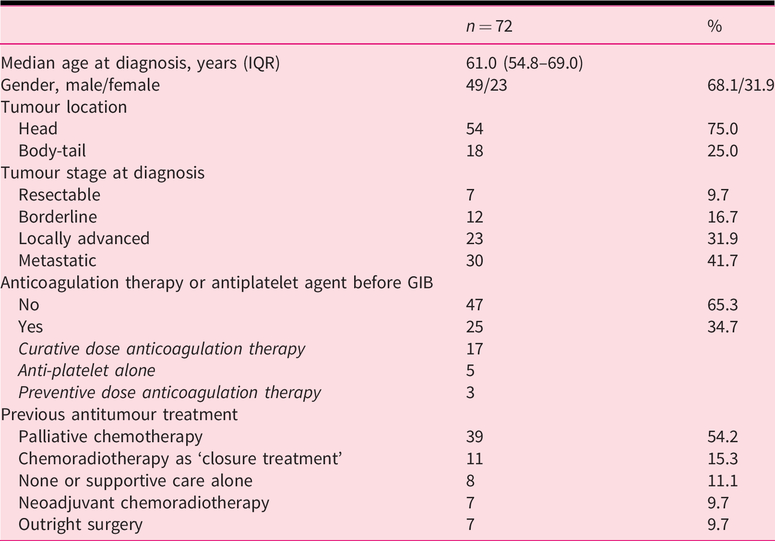
Characteristics of 72 patients with pancreatic adenocarcinoma who had at least one episode of gastrointestinal bleeding (GIB).
GIB description
The following data were recorded: date of occurrence, the lowest haemoglobin level at diagnosis of GIB and clinical signs of GIB. The biological sign of GIB was defined as the rapid loss of at least two points in haemoglobin level in 24 h. Additionally, the cause of GIB (based on the endoscopic report), anatomical bleeding location, patient’s hospitalization unit (intensive care unit (ICU) or regular hospital unit) and haemostatic treatments used, were also recorded. Red blood cell unit transfusion requirements during the GIB period were collected, according to the French blood transfusion establishment (EFS) records. Date and cause of death were obtained from hospitalization reports, if recorded, and from community records.
Immediate haemostatic efficacy was defined as the absence of additional haemostatic treatment within 14 days from the first treatment. In the same way, long-term efficacy was defined as the absence of additional haemostatic treatment more than 14 days after the initial treatment. Recurrent bleeding was defined as a new episode of GIB more than 14 days from the first episode. Causes of GIB and their treatment were determined from the total number of episodes of GIB in the population.
Analysis of parameters associated with treatment effectiveness in case of tumour invasion
We then focused on GIB caused by tumour invasion. In these cases, haemostatic treatments were described: therapeutic endoscopy, proton pump inhibitors (PPIs) only-therapy and specific treatments such as HRT or angiographic arterial embolization.
We calculated the difference in blood unit (BU) transfusion requirements before and after haemostatic treatment in GIB caused by tumour invasion, as a means of assessing effectiveness, in addition to the recurrence rate. Day 0 was considered as the day of GIB diagnosis. A haemostatic treatment could suggest effectiveness if post-therapeutic transfusion requirements (from day 3 to day 14) were less than the pre-therapeutic transfusion requirements (from day –5 to day 2). Mortality rate within the 30 days from the bleeding onset was calculated for each treatment type. We explored potential factors related to the effectiveness of haemostatic treatment of GIB caused by tumour invasion.
Subgroup receiving HRT
The usual indications of HRT were the presence of large ulcerated digestive tract surface caused by tumour invasion, not accessible to therapeutic endoscopy and/or failure of therapeutic endoscopy. The characteristics of the radiation therapy delivered (fractionation schedules and total dose delivered) were collected from the medical records. Population and GIB characteristics were studied to define factors that may be associated with the choice of radiation therapy in this population.
Survival analysis and prognostic factors
Overall survival was calculated from the date of the first GIB episode to the date of death. Data were censored at the date of the last recorded follow-up, if no death event had occurred. Overall survival was assessed in relation to the cause of GIB. GIB and patient characteristics were analysed to assess their impact on patient outcome.
Statistical analysis
Statistical analyses were performed with SPSS software (version 20, IBM). Univariate and multivariate analyses of logistic regression were used. Overall survival was calculated using the Kaplan Meier method, and was expressed as a median and its 95% confidence interval (CI), or six-month overall survival rate and its standard deviation. Values of p < 0.05 were considered to be statistically significant. All statistical tests were bilateral.
Results
A total of 4605 patients were identified with a ‘malignant pancreatic tumour’ from a search of a coded diagnostic database between January 2000–December 2017. Twenty-six of these patients (0.6%) were not included due to an estimated coding error (Figure 1). One hundred fifty-three of the remaining 4579 patients with PDAC (3.3%) had an associated coded diagnosis of GIB. Sixty-seven patients (43.7%) were excluded (Figure 1). A total of 72 patients (1.6%) accounting for 94 GIB episodes fulfilled the inclusion criteria and were included in the study.
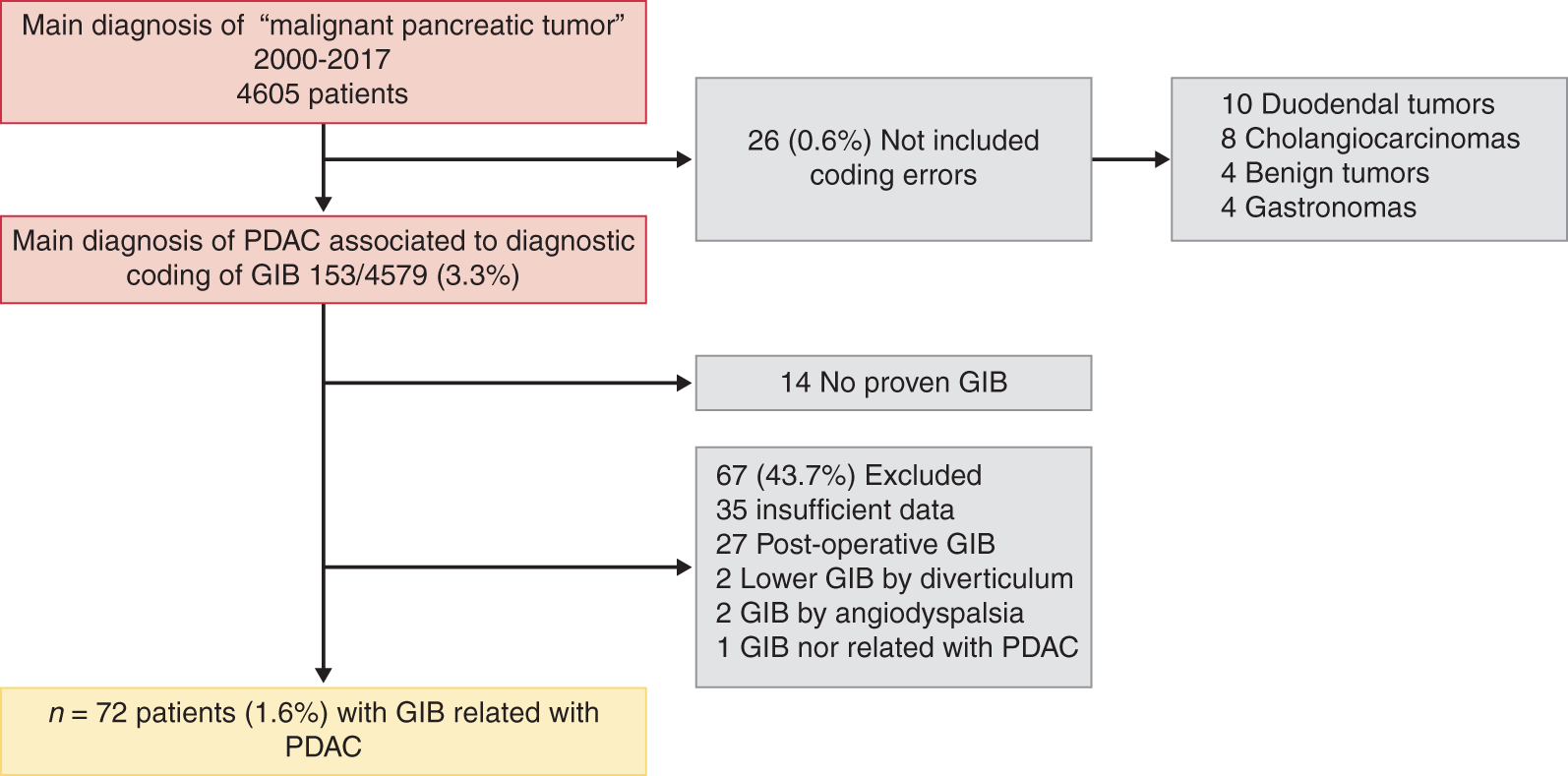
Population flowchart of patients with pancreatic ductal adenocarcinoma (PDAC) and gastrointestinal bleeding (GIB).
Patient characteristics
The median age at diagnosis of PDAC was 61 years (interquartile range (IQR) 54.8–69.0) and 68.1% of patients were male. Tumours were mainly located in the pancreatic head (75%). Seven patients (9.7%) had undergone pancreatic surgery (all by pancreatico-duodenectomy) more than one month before the GIB. One-quarter of patients (25%) had received chemoradiotherapy. Other patients’ characteristics are described in Table 1. The median time from diagnosis of PDAC to the first episode of GIB was 7 months (IQR 1–15). GIB was the revealing event leading to the diagnosis of PDAC in 16 patients (22.2%). Ten of these had metastatic disease at diagnosis; six had a locally advanced tumour and none had borderline or resectable PDAC. Twenty-five patients (34.7%) were receiving anticoagulation medication or anti-platelet agent (APA) when GIB occurred.
GIB characteristics
Melena and/or haematochezia was the first clinical manifestation in 48/94 episodes (51.1%) and haematemesis in 22/94 episodes (23.4%). An initial drop in the haemoglobin level suggested GIB in 21/94 episodes (22.3%). Low blood pressure was the first and single sign for three episodes (3.2%). The median haemoglobin level at the time of GIB was of 7.1 g/dl (IQR 6.4–7.9). Among the 94 episodes, 34 patients (36.2%) were admitted into the ICU.
The disease status when GIB occurred was tumour progression in 56/94 episodes (59.6%), stable disease in 17/94 (18.1%) and persistent remission after curative surgical treatment in three episodes (3.2%).
Causes of GIB and haemostatic treatments
Tumour invasion, mainly of the duodenum wall, was the primary cause of GIB recorded for 53/94 episodes (56.4%). Non-cirrhotic left-sided portal hypertension (LSPHT) causing rupture of oesophageal, gastric or duodenal varices, was the second most frequent cause recorded for 18/94 episodes (19.1%) (Figure 2). Twelve of these patients were treated by endoscopic band ligation, five patients were treated by somatostatin analogues (then non-selective beta-blockers without endoscopic treatment), and one had an angiographic embolization of a duodenal varix.
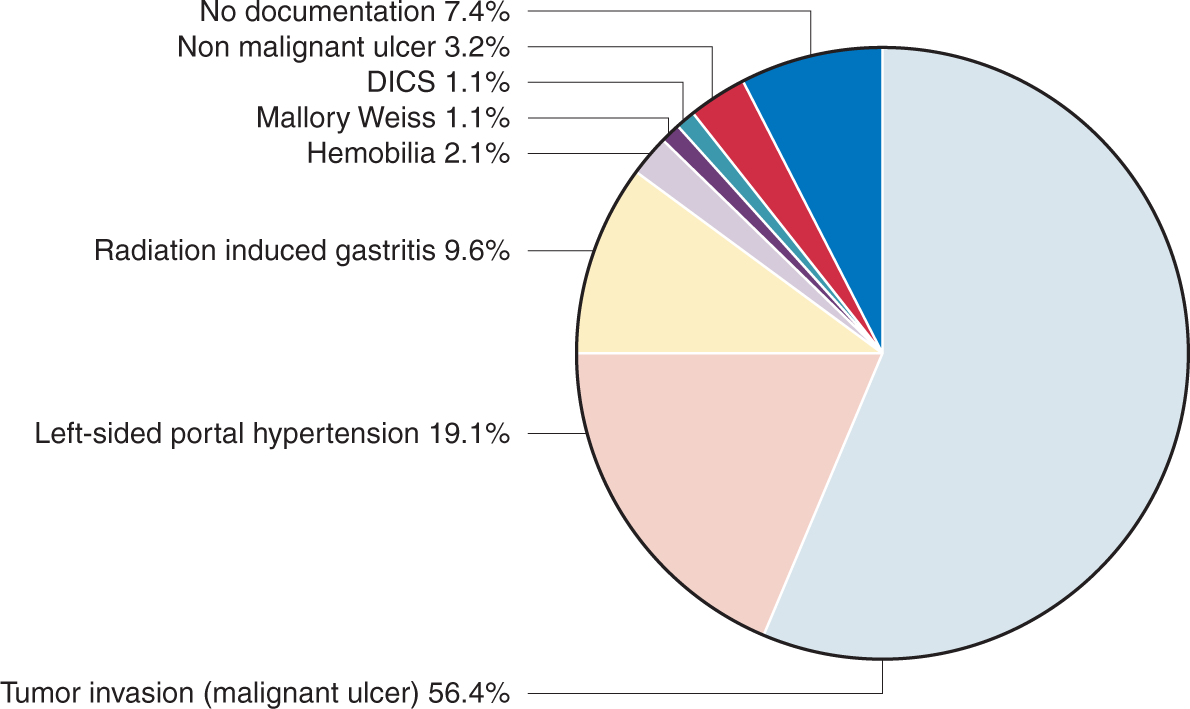
Bleeding causes in patients with pancreatic ductal adenocarcinoma (n = 94 episodes).
Radiation-induced gastritis, following previous PDAC chemoradiotherapy, was the cause of bleeding for 9/94 episodes (9.4%). These nine patients were all treated with PPI therapy and with endoscopic plasma argon coagulation. The median time from PDAC diagnosis to GIB for these nine patients was of 13 months (IQR 12–15). Details on the anatomical bleeding sites and other causes are reported in Supplementary Material Table 2.
Immediate haemostatic efficacy rate, long-term bleeding recurrence rate and 30-day mortality rate in case of tumour invasion depending on haemostatic treatment, n = 41 patients.
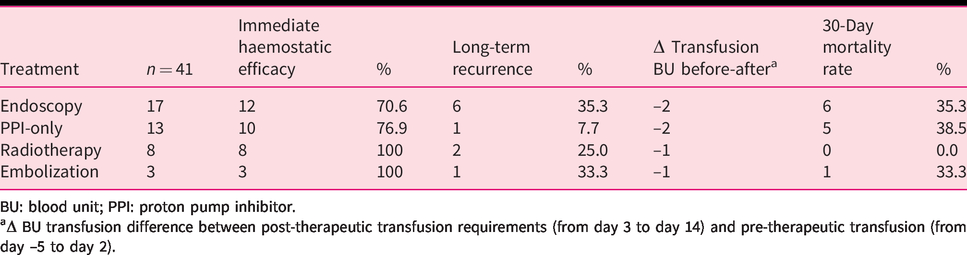
BU: blood unit; PPI: proton pump inhibitor.
aΔ BU transfusion difference between post-therapeutic transfusion requirements (from day 3 to day 14) and pre-therapeutic transfusion (from day –5 to day 2).
Immediate and long-term haemostatic efficacy and mortality rate in cases of tumour invasion
GIB recurrence rate was 22/72 (30.5%). GIB recurrence occurred after a median of 43 days (IQR 29–117) after the initial episode. Among the 72 patients, 41 patients (56.9%) had a first GIB episode caused by tumour invasion. Among these, 17 (41.5%) had therapeutic endoscopy, including eight having haemostatic powder (Hemospray), seven having epinephrine injection therapy, one having a combined technique by injection therapy and clip and one having the section of a duodenal stent that was causing mucosal erosion into the tumour. Thirteen patients (31.7%) received PPI-only therapy, 8/41 (19.5%) received HRT and 3/41 (7.3%) had angiographic embolization (Figure 3). Immediate and long-term haemostatic treatment efficacy rates are described in Table 2. The median difference of BU transfused (before and after) was of –2 BU (IQR –4 to +2) in the case of endoscopic therapy and –1 BU (IQR –2.5 to 0) in the case of HRT. Mortality rate within the 30 days following the haemostatic treatment was 6/17 (35.3%) after therapeutic endoscopy, and 0% after HRT.
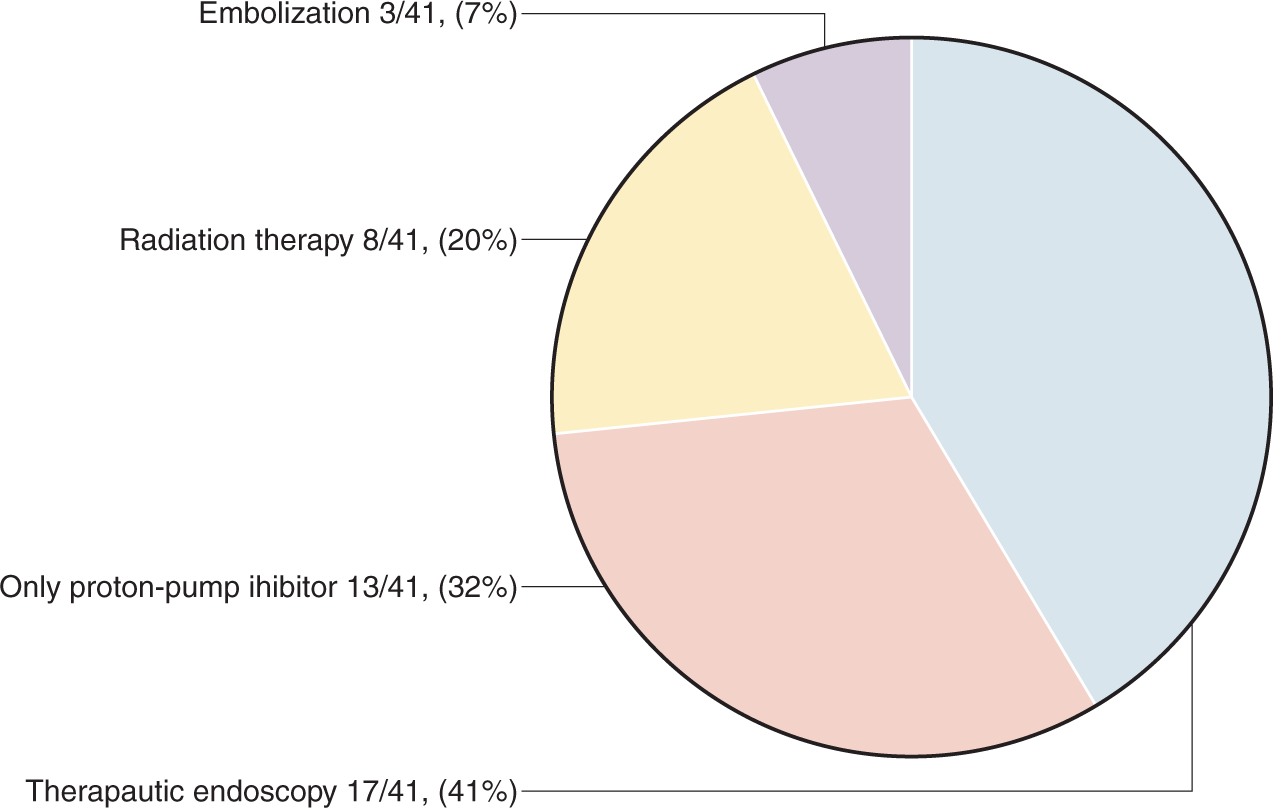
Haemostatic first-line treatments for the first episode of gastrointestinal bleeding by tumour invasion cause (n = 41).
Treatment effectiveness in case of tumour invasion
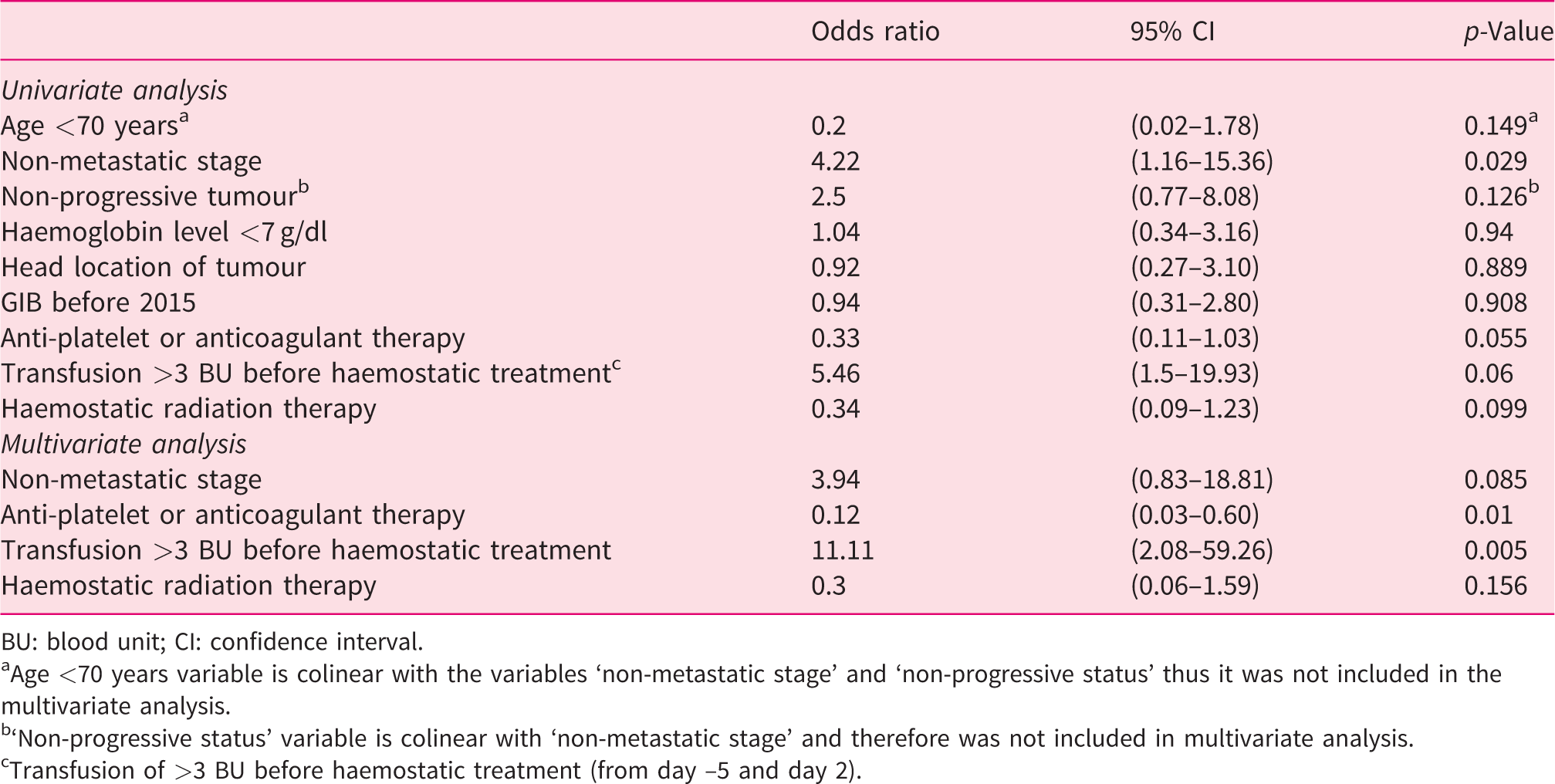
Multivariate analysis showed that the treatment of GIB was significantly less effective in patients receiving APA or anticoagulant therapy at the time of GIB (odds ratio (OR) 0.12; 95% CI (0.03–0.6); p = 0.01) and more effective in cases of transfusion of more than 3 BU before the haemostatic treatment (OR 11.11; 95% CI (2.08–59.26); p = 0.005) (Table 3).
Factors associated with the effectiveness of haemostatic treatment for episodes of gastrointestinal bleeding (GIB) by tumour invasion.
BU: blood unit; CI: confidence interval.
aAge <70 years variable is colinear with the variables ‘non-metastatic stage’ and ‘non-progressive status’ thus it was not included in the multivariate analysis.
b‘Non-progressive status’ variable is colinear with ‘non-metastatic stage’ and therefore was not included in multivariate analysis.
cTransfusion of >3 BU before haemostatic treatment (from day –5 and day 2).
HRT
A total of 17 patients with tumour invasion underwent HRT: as first-line treatment for their first episode (8/17), as a second-line treatment for their first episode (4/17) or as first-line treatment for their second episode (5/17). PDAC was mainly located in the pancreatic head in 11/17 patients and was metastatic in 14/17 patients. The median time between initial PDAC diagnosis and the first GIB episode was four months (IQR 1–9). The median time from GIB to the beginning of HRT was of 12 days (IQR 10–17). All of these patients received PPI therapy until HRT was performed.
When GIB occurred, the disease was progressive for 10/17 patients. The median total radiation dose and the median dose per session were 20 Gy (IQR 20–25) and 5 Gy (IQR 5–5), respectively; the median number of fractions was four (IQR 4–5). The median time to death after radiation therapy was 63 days (IQR 25–147). Two patients did not complete the expected radiation schedule because of rapid degradation of physical condition.
Patients with non-metastatic PDAC at diagnosis had a higher probability of not receiving HRT on univariate analysis (OR 0.1, 95% IC (0.01–0.86), p = 0.036). No other factor was significantly associated with performing radiation therapy in case of GIB, making it impossible to perform multivariate analysis.
Survival
Median overall survival from the GIB episode in the population of 72 patients was 2.7 months (95% CI (1.4–4)), with a six-month overall survival rate of 35% (±5.8). Non-progressive disease at the time of GIB (hazard ratio (HR) 0.5; 95% CI (0.29–0.86); p = 0.012) and any cause of GIB other than tumour invasion (HR 0.55; 95% CI (0.32–0.94); p = 0.03) were independently associated with a significantly better prognosis on multivariate analysis (Supplementary Material Table 3). Median overall survival from the onset of GIB by tumour invasion was 1.9 months (95% CI (0.87–2.87)), compared to 3.7 months (95% CI (0–7.7)) in patients bleeding from other causes (p = 0.027) (Figure 4). Of the 42/72 causes of death documented, 21/42 deaths (50%) were directly related to GIB.
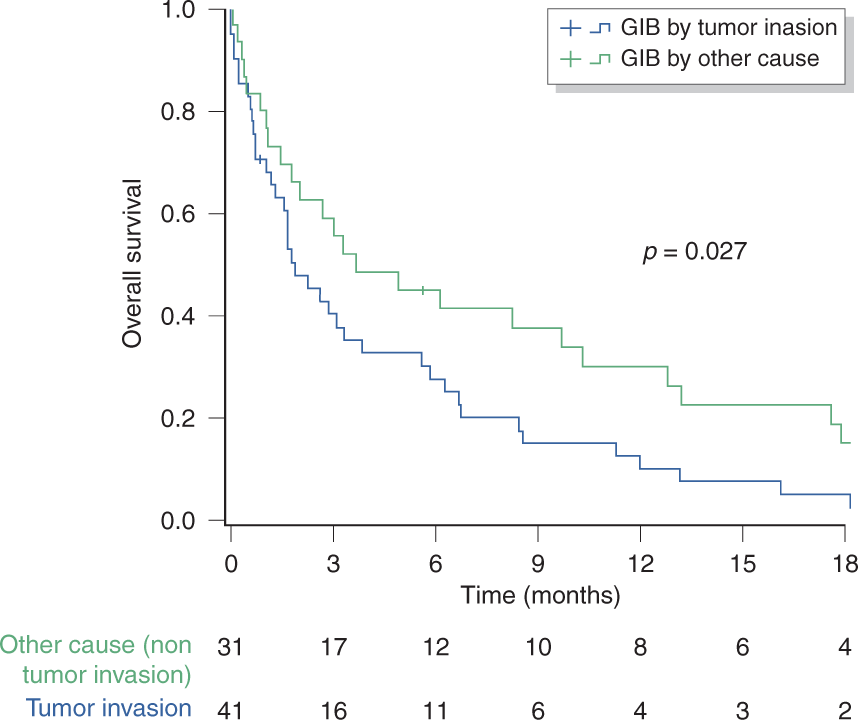
Survival analysis according to the cause of gastrointestinal bleeding (GIB): tumour invasion vs other causes.
Discussion
The frequency of non-postoperative GIB in this population of 4579 patients with PDAC was 1.6%. In the retrospective series by Wang et al., 11 the prevalence of GIB was of 13%, but for 13/32 patients the cause of GIB was gastric or duodenal non-specific ‘ulcer’ and for 16/32 patients it was unknown or not specified. Thus, the GIB rate that we observed might be underestimated because of the retrospective data collection and the strict inclusion criteria applied.
Furthermore, in the present study, GIB was the revealing event of the PDAC in 22.2% of patients. This figure may seem surprising but has been reported in other series.1,12,13 When PDAC is diagnosed late and at an advanced stage, as often happens, parietal and vascular invasion can be the origin of a severe and revealing GIB. Similarly, 73.6% of the patients in our series had metastatic or locally advanced stage disease at diagnosis of GIB. This is consistent with the results of Wang et al. 11 which showed that the primary risk factor of GIB was stage IV (metastatic) disease. Moreover, we also observed that GIB occurred when the tumour was progressive in 59.6% of cases, suggesting that GIB marks a turning point in the disease requiring early evaluation by CT scan.
There are very few studies evaluating the cause of GIB in PDAC patients. In our study, tumour invasion of the digestive tract was the most frequent one (56.4%). Most PDACs were located in the head (75%), so it is not surprising that tumour invasion was the main cause due to close contact with the duodenum.
The number of cases of GIB caused by radiation-induced gastritis seem sizeable (9.4%). Fortunately, chemoradiotherapy techniques for locally advanced PDAC have evolved within recent years with the advent of intensity modulated radiotherapy (IMRT) significantly lowering gastrointestinal toxicity nowadays, but its common use in France was not until 2016, so most of the patients in our series may not have benefited from this technique.14,15 Moreover, these events happening more than 6 months after prior chemoradiotherapy, they are considered as a late gastrointestinal toxicity. It has already been suggested that as patients with PDAC live longer, they have a higher risk of developing late toxicity over time, however exceptionally grade 3 or 4 toxicity. 16
Another important finding of our study, was that transfusion of three or more BU before haemostatic treatment increased therapeutic efficacy, as shown on multivariate analysis, in patients with tumour invasion-induced GIB. This attitude is coherent with the international guidelines for upper GIB management recommending that haemoglobin levels be >7 g/dl before endoscopy. 3 Indeed, reducing tissue hypoxia and obtaining haemodynamic stability before interventional haemostatic treatments, such as endoscopy, probably help to improve control of GIB.
In our series, all types of treatments seemed able to reduce blood transfusion requirements in GIB due to tumour invasion. Nonetheless, a third of patients still did not have immediate haemostatic efficacy or had bleeding recurrence after therapeutic endoscopy or PPI-only therapy. The rate of GIB recurrence (>14 days after the first episode) that we observed was 30.5%, with a median delay of 43 days from the first episode. Yet the definition of GIB recurrence is not clearly described in the literature, making comparisons difficult, but this rate is still consistent with that observed in other similar GIB series.2,17 In fact, the definition of recurrence, in terms of time to rebleed, varies in different studies from 5–30 days.2,18 However, although the absence of recurrent GIB may be a sign of effective haemostatic treatment, it may also be linked to an outcome caused by another short-term complication.
HRT in our series did not seem to be more efficient compared to other treatments in patients with GIB by tumour invasion. However, patients were not comparable in the treatment groups in terms of GIB severity, as well as in the ulcerative surface causing GIB. The median time from GIB to the beginning of HRT was of 12 days, which seems long in the case of GIB. Actually, this time delay was due to organization of HRT. The latter was considered to be a relative semi-urgent treatment to perform as the bleeding was chronic and moderate. This observation and the differences in mortality before day 30 depending on treatment received, reflect the heterogeneity of the population in terms of severity of GIB presentation.
Our study has several limitations. The retrospective nature of the study explains why some results were incomplete. It is not excluded that episodes of GIB were missed during the collection of data by using a hospital coding database. In addition, the severity of GIB was not precisely defined in all patients, making the population too heterogenous to identify the patients who received HRT and thus, may have had benefit from it. Moreover, the exact cause of death was not documented in 30/72 patients.
To our opinion, HRT as a therapeutic option in this context, merits further evaluation in patients with GIB from tumour invasion of a large parietal surface that is not treatable in endoscopy. HRT could be more beneficial when proposed early, for example as a prophylactic complement of upfront therapeutic endoscopy. It might thus improve patients’ quality of life in an early palliative setting.9,19 Nonetheless, endoscopy remains indispensable for the diagnosis of the cause of GIB and eventually for haemostatic treatment for other causes (43.6%), mainly ruptured oesophageal varices.
In our series, the median survival after GIB from all causes was 2.7 months while it was 1.1 months in the Wang et al. 11 series. Progress in the treatment of PDAC, and improvement of GIB management and therapeutic endoscopic techniques may explain this. Although overall survival from GIB in our series was different depending on the cause of bleeding (tumour invasion vs other causes), the six-month overall survival rate was only 35% after GIB. This confirms the severity of this complication, which marks a turning point in the natural history of PDAC.
In conclusion, GIB in patients with PDAC is a rare but severe complication with poor prognosis and it is mainly due to digestive tract tumour invasion. HRT is a non-invasive therapeutic option to be discussed in patients in which a large surface of tumour invasion is not treatable with endoscopic treatments or for patients who have short and long-term bleeding recurrence after other haemostatic treatments.
Supplemental Material
sj-pdf-1-ueg-10.1177_2050640620939788 - Supplemental material for Gastrointestinal bleeding in patients with pancreatic cancer: Causes and haemostatic treatments
Supplemental material, sj-pdf-1-ueg-10.1177_2050640620939788 for Gastrointestinal bleeding in patients with pancreatic cancer: Causes and haemostatic treatments by Lina Aguilera Munoz, Louis de Mestier, Hanah Lamallem, Bénédicte Jaïs, Frédérique Maire, Philippe Lévy, Vinciane Rebours and Pascal Hammel in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The authors declared no conflict of interest.
Ethics approval
This type of study is exempt from ethics approval.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
