Abstract
Lower gastrointestinal bleeding is common and occurs often in elderly patients. In rare cases it is associated with hemorrhagic shock. A large number of such bleedings, which are often caused by colon diverticula, subside spontaneously. Alternatively they can be treated by endoscopic procedures successfully. Given the aging population of our society, the rising incidence of lower gastrointestinal tract bleeding and new anticoagulant therapies, some of the bleedings tend to be severe. Colonoscopy is the established standard procedure for the diagnosis and treatment of lower gastrointestinal bleeding. However, a small number of patients experience re-bleeding or shock; their bleeding does not resolve spontaneously and cannot be treated successfully by endoscopic procedures. In such patients, interventional radiology is very useful for the detection of bleeding and the achievement of hemostasis. Against this background we performed a literature search using PubMed to identify all relevant studies focused on the endoscopic and radiological management of lower gastrointestinal bleeding and present recent conclusions on the subject.
Introduction
Diverticular bleeding is the most common cause of lower gastrointestinal bleeding (LGIB). Severe hematochezia is caused by diverticular bleeding in approximately 40% of cases (Figure 1). In 70%–80% of cases the bleeding ceases spontaneously without any intervention, but some patients require endoscopic, surgical or angiographic treatment. Although endoscopic hemostasis for diverticular bleeding appears to be effective and is the therapeutic option of choice, the optimal technique remains to be determined.
1
The most challenging and complex factors concerning the emergency management of an LGIB are the large surface area of the colon and residual stool.
2
It is still a matter of debate whether an endoscopic approach should be performed immediately after admission, or electively after hemodynamic stabilization. Given these challenges and the optimal time point of diagnostic endoscopy, a timely colonoscopy performed 6–12 h after hospitalization has been reported to identify the site of bleeding in 42%. In contrast, the rate of identification was only 22% when an elective colonoscopy was performed 72 h after admission.3,4 Furthermore, an aggressive bowel preparation regimen 3–4 h before emergency colonoscopy was able to identify bleeding more often and permitted hemostasis in the colon more frequently. However, it remains unclear whether other strategies should be used for the primary management of LGIB in the future in order to improve the high rates of early and late re-bleeding associated with this condition. The “endoscopy first” approach has not been proven superior to the “angiography first” strategy. In a randomized controlled trial by Green et al., 50 patients were randomized to urgent purge preparation, followed immediately by colonoscopy or an angiographic intervention without colonoscopy. The authors found no difference in outcomes between the two groups, including mortality rates, hospital stay, intensive care unit stay, transfusion requirement, early re-bleeding, surgery, and late re-bleeding.
4
A 66-year-old man with severe hematochezia and hemorrhagic shock. Endoscopy was performed immediately after admission to the hospital without bowel preparation. The endoscopic finding at the right colon was a spurting bleeding from a large diverticular that failed endoscopic therapy.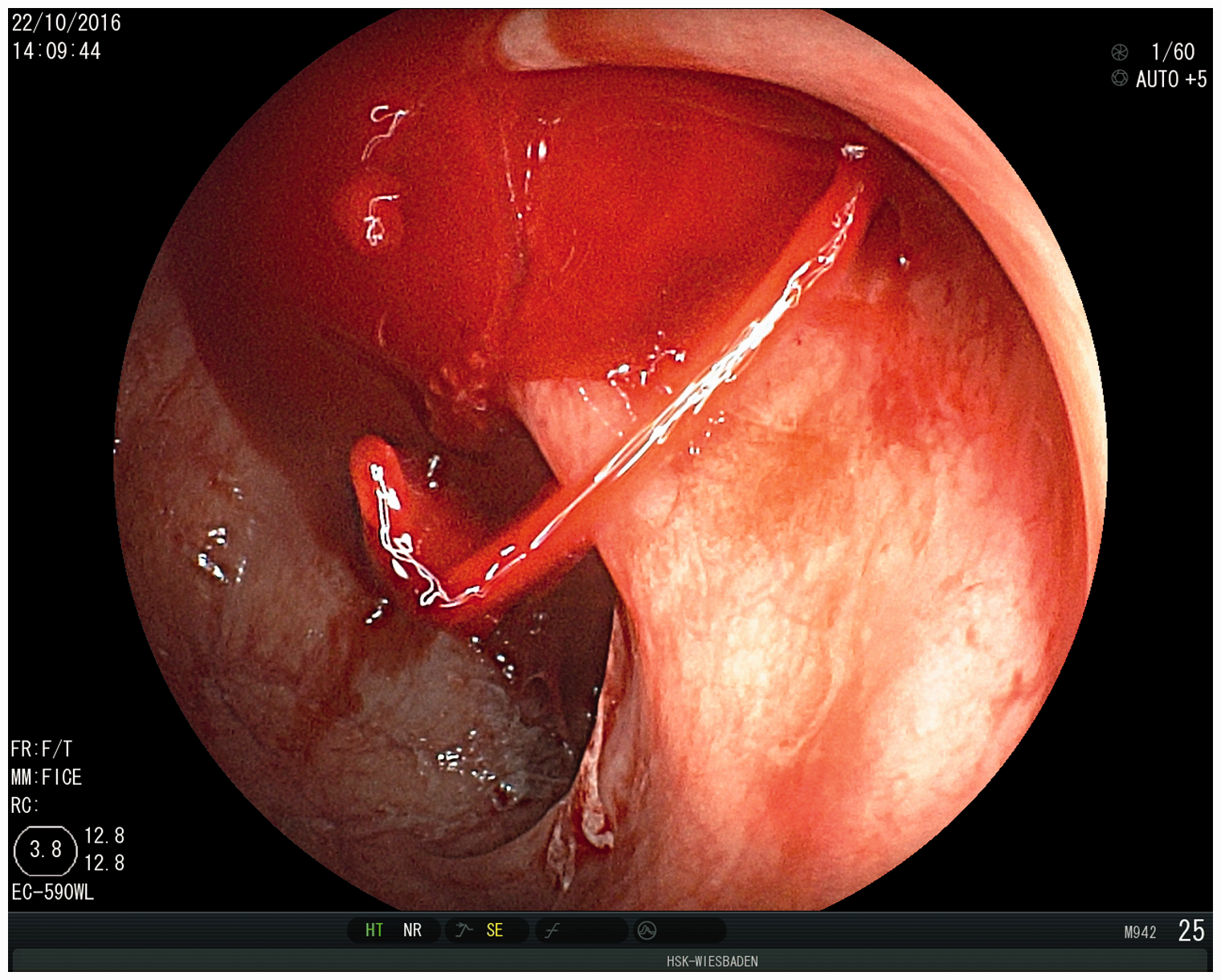
Methods
A comprehensive literature search was performed to identify all relevant studies in which endoscopy and angiography were performed for LGIB. Through PubMed the medical terms “lower gastrointestinal bleeding,” “endoscopic management” and “angiography” were used in the search. We also searched the reference lists of the screened articles to identify any additional studies that might be relevant.
Clinical management
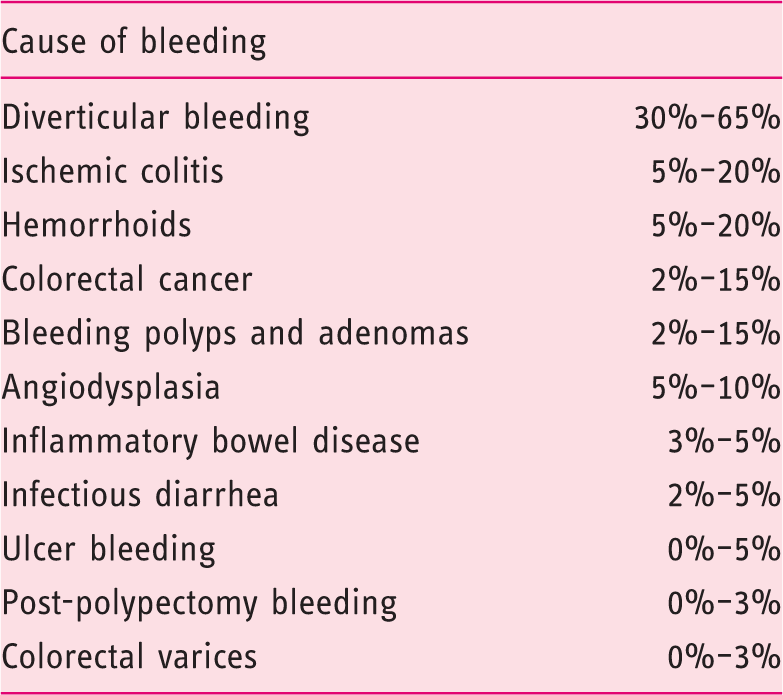
Causes of lower gastrointestinal bleeding.
Endoscopic management
Gastrointestinal endoscopy is the gold standard for the diagnostic investigation and treatment of LGIB. The sequence of gastroscopy or colonoscopy depends on the patient’s clinical symptoms.9–11 Although an urgent colonoscopy performed within 6 h is able to identify the source of bleeding more frequently in patients with LGIB, the urgent colonoscopy does not differ from elective colonoscopy in terms of mortality rates, the duration of hospital stay, the need for transfusions, early and late re-bleeding rates, and the need for surgical treatment. 4 Eleven percent of upper gastrointestinal bleedings are manifested clinically by hematochezia, therefore a gastroscopy should proceed in severe hematochezia to rule out upper gastrointestinal bleeding. 12 Complication rates after urgent colonoscopies (0.6%) are two-fold higher than those after elective colonoscopy. 2 In unstable patients an urgent endoscopy should be performed by an experienced investigator. 13 Nevertheless, successful endoscopic therapy depends on the visualization and identification of an actively bleeding diverticulum. This is rendered especially difficult in patients with diverticulosis of the entire large bowel and inadequate bowel preparation. Since the majority of bleedings tend to resolve spontaneously, the bleeding may not be detected on endoscopy either. Therefore, a rapid endoscopic diagnostic investigation, possibly with aggressive bowel preparation over three to four hours, may be advisable to maximize the diagnostic and therapeutic yield. 14
Endoscopic therapy
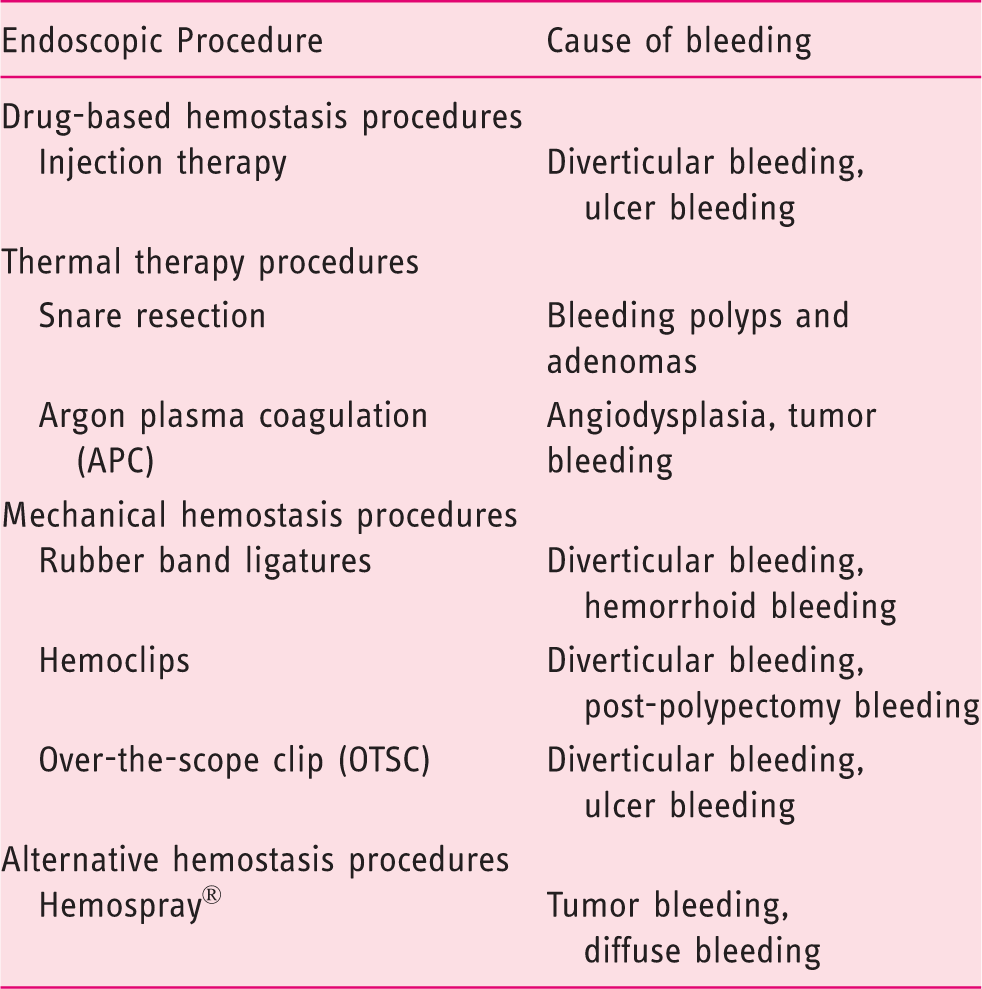
Procedures for achieving endoscopic hemostasis in lower gastrointestinal bleeding dependent on the cause of bleeding.
Radiological management
The radiological management of gastrointestinal bleeding is well investigated and has become increasingly significant in the last few years. The major advantage of angiography—similar to endoscopy—is the possibility of simultaneous diagnostic investigation and therapy. Nevertheless a computed tomography angiography (CTA) should be performed prior to conventional angiography in order to detect the source of bleeding and to map the vessel anatomy.
2
The American College of Gastroenterology (ACG) recommends in their guidelines that radiographic interventions should be considered in patients with high-risk clinical features and ongoing bleeding who do not respond adequately to hemodynamic resuscitation efforts and are therefore unlikely to tolerate bowel preparation and urgent colonoscopy. In general, surgery for acute LGIB should be considered after all other therapeutic options have failed. In contrast, in patients without high-risk clinical features or serious comorbid disease or without signs or symptoms of ongoing bleeding, colonoscopy should be performed next available after a colon purge and they should not undergo radiographic intervention. However, these recommendations have very low-quality evidence but strong or conditional recommendation levels.
18
Taking these recommendations into account radiological therapy is a less invasive complementary option for the diagnosis and therapy of LGIB, especially before a surgical procedure is chosen. Radiographic intervention could be considered especially in all patients with ongoing bleeding or when endoscopic therapy has failed
19
(Figure 2).
Flowchart for the management of lower gastrointestinal bleeding. Patients with high-risk indicators should be considered as instable. It is still a matter of debate whether and how often patients should receive endoscopic diagnostic and if computed tomography angiography (CTA) is useful before angiography. Hb: hemoglobin.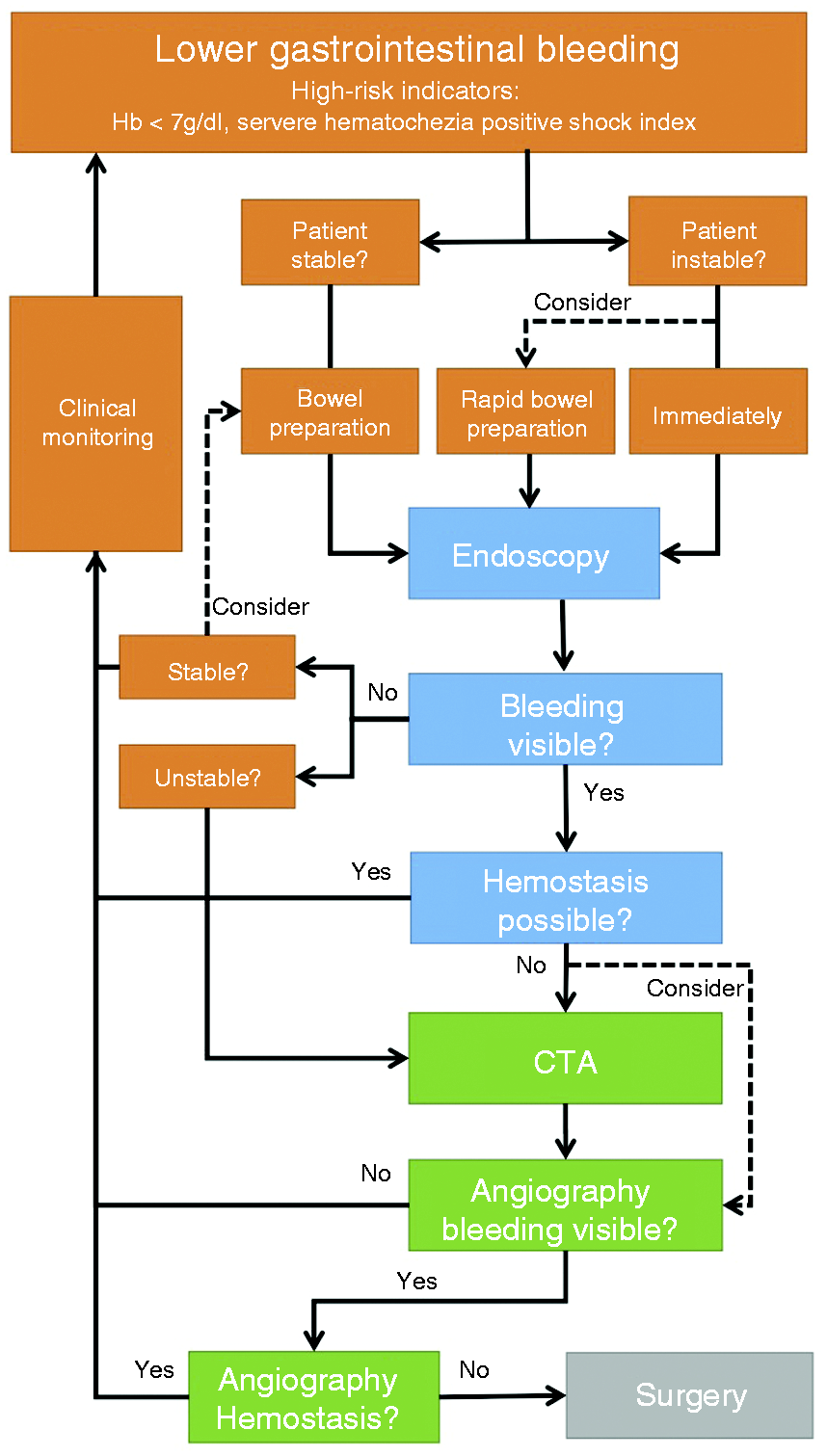
Diagnostic radiology
CTA is able to detect gastrointestinal bleeding from a flow rate of 0.3 ml/minute onward; its sensitivity is 50%–86% and its specificity 92%–95%. Some series even report sensitivities of 100% and specificities of 96%. 20 Furthermore, CTA is able to isolate the site of bleeding and permit the investigator to express differential diagnoses. 21 In a meta-analysis of 124 patients with gastrointestinal bleeding who underwent CTA, extravasations of contrast medium were seen in 74 patients (60%). However, it is still unclear whether CTA performed prior to angiography has an impact on hard endpoints like re-bleeding rates, the need for surgery, or mortality rates. 2 Nagata et al. showed that CTA performed in addition to colonoscopy provided additional information about the vascular situation in 15% of cases. In contrast, angiography after CT is able to show extravasations in 52% of cases; the probability is even higher when the angiography is performed within 90 min after the CT. 22 A pioneering study conducted by Kennedy et al. showed, in 74 patients with gastrointestinal bleeding of uncertain origin whose CTA failed to reveal bleeding, that angiography performed 24 h after CTA also failed to demonstrate bleeding. Twenty-two patients had positive CTA, which was confirmed by angiography in 86% of cases. Sixty-four patients with no evidence of bleeding on CTA needed no subsequent treatment in 92% of cases. Thus, the positive predictive value of CTA was 86% and its negative predictive value 92%. 23 Although these cases are very rare, diagnostic nuclear medicine could play an important role in hemodynamically stable patients with bleeding that escapes detection by endoscopy and radiological investigation. 24
Diagnostic angiography
Diagnostic angiography is able to detect the source of bleeding at a bleeding rate of 0.5–1 ml/min (Figure 3). Its positive predictive value is 100% and its negative predictive value 24%.
21
In cases of anticipated LGIB, the superior mesenteric artery and the inferior mesenteric artery are first visualized on angiography in order to rule out the two main causes of LGIB—angiodysplasia and diverticula of the colon. When no bleeding is found, the internal iliac artery and the middle and inferior rectal arteries are visualized.
21
Extravasations of contrast medium are a reliable sign of active bleeding.
25
When no active bleeding is seen, a repeat investigation of the same vessels is recommended in order to avoid false-negative findings. When endoscopic treatment has been attempted earlier, the previous colonoscopy and the findings obtained by the endoscopist may be useful. In patients with an initially negative angiography, the previous diagnostic CT investigation and interdisciplinary planning are decisive steps for the localization of bleeding and optimal therapy planning.21,26
Radiological image during catheter angiography of the same patient with superselective visualization of a side branch of the middle colic artery and active extravasation.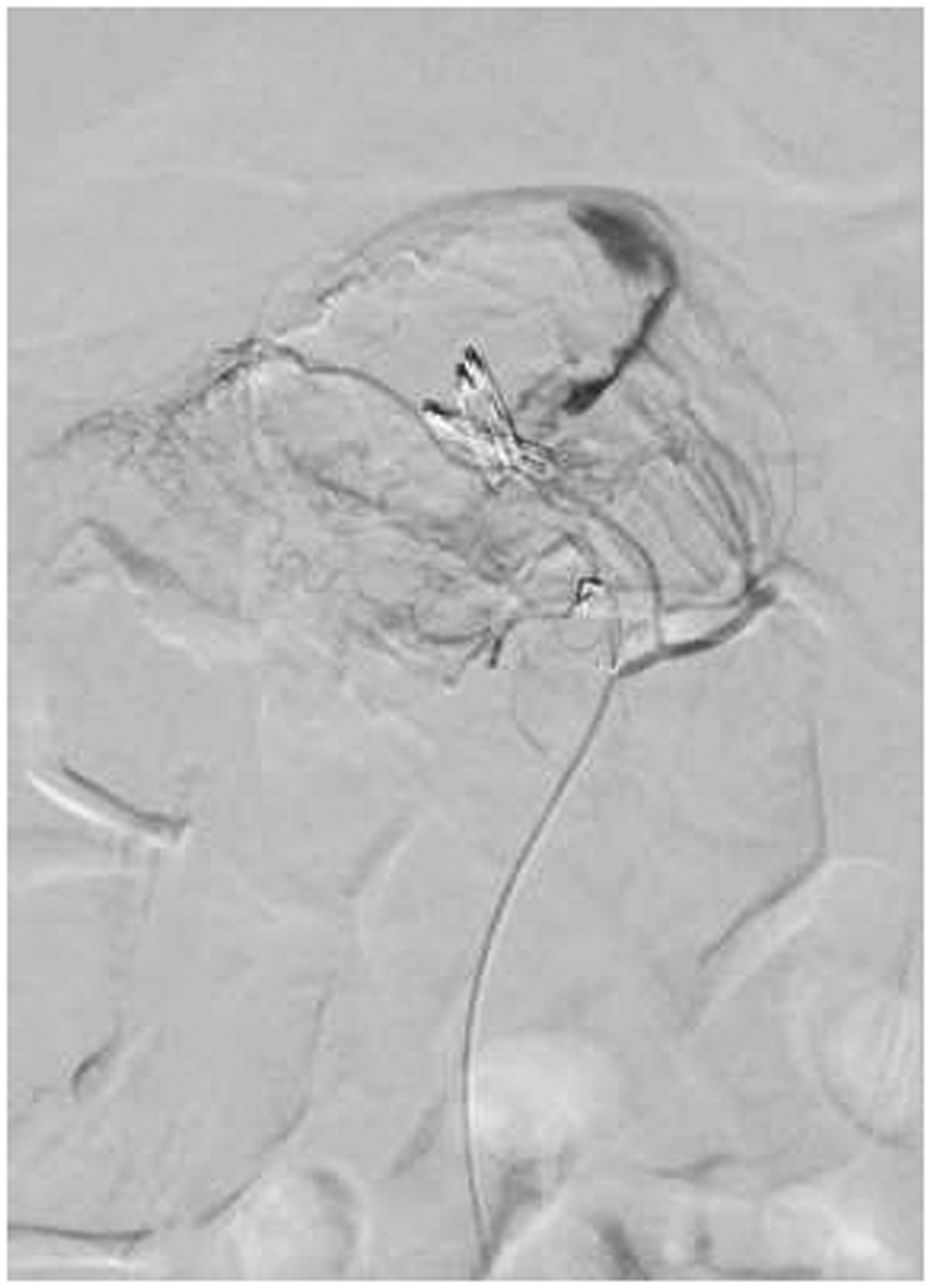
Radiologic therapy
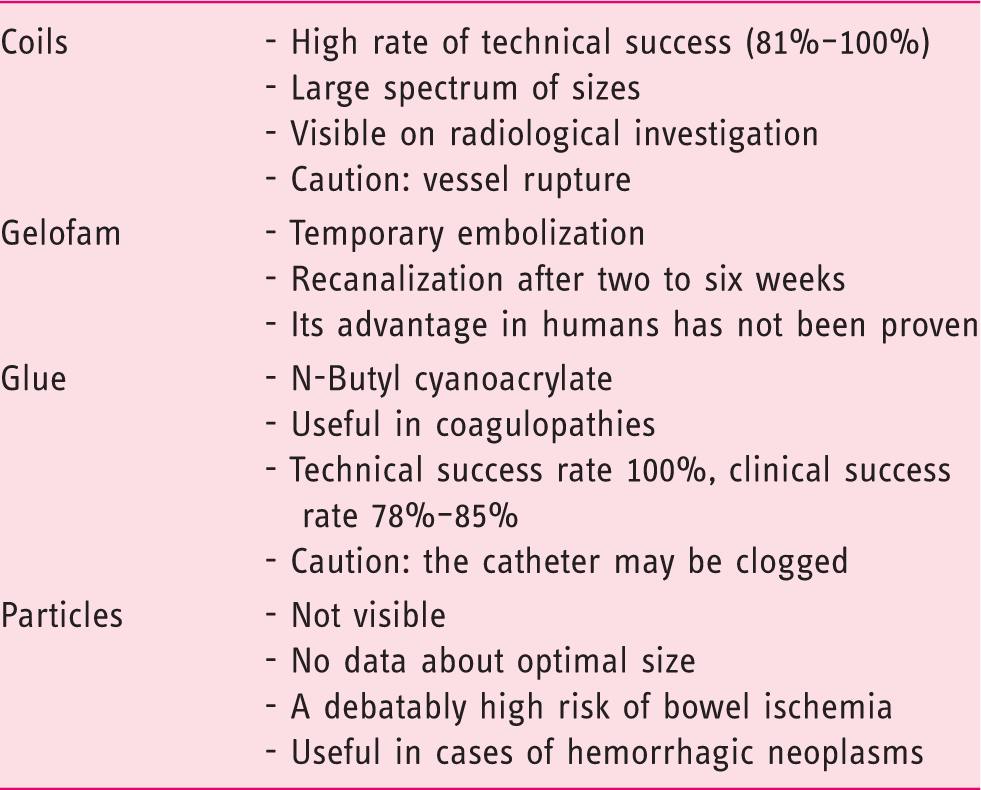
Hemostasis procedures by interventional radiology.
Conclusions
A paradigm shift has occurred during the last few years in the treatment of patients with LGIB when endoscopy therapy has failed. In these cases radiological treatment should be considered before surgery. Therefore a CTA prior to angiography is useful, especially when the source of bleeding is unclear. A CTA is also useful in order to detect anatomical variants or obtain secondary findings, in addition to localizing the possible source of bleeding. Selective investigation of the most likely area of bleeding should be performed. Coils or PVA particles are especially suitable for embolization in LGIB.
Footnotes
Declaration of Conflicting Interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This review conforms to the ethical guidelines of the 1975 Declaration of Helsinki.
Informed consent
Informed consent was obtained from all participants.
