Abstract
Obesity, type 2 diabetes mellitus (T2DM) and nonalcoholic steatohepatitis are increasing pandemic metabolic disorders. Lifestyle intervention (LSI) is the cornerstone treatment for these but is successful as standard care alone in only a few patients, given the modest weight loss at mid and long term. Conversely, bariatric surgery is the only proven effective treatment for these metabolic disorders, albeit offered only in a small percentage of cases because of its invasiveness and cost.
The so-called endoscopic bariatric and metabolic therapies (EBMTs) include new, less-invasive technologies such as intragastric balloons, aspiration therapy, endoscopic sleeve gastroplasty, diversion devices, and duodenal mucosal resurfacing, currently at various stages of development. EBMTs, as an add-on to LSI, might represent an effective treatment filling the gap between medical and surgical management, taking into account, however, that obesity and its associated comorbidities constitute a chronic disease that needs lifelong therapy. In this review we describe the current scientific evidence surrounding EBMTs as well as future opportunities for such treatments in managing obesity and metabolic disorders.
Keywords
Introduction
The increasing global burden of obesity 1 and its related comorbidities such as type 2 diabetes mellitus (T2DM) 2 and nonalcoholic fatty liver disease 3 represent a major health concern. Obesity is a significant cause of global mortality and morbidity with more than 1 million deaths annually due to T2DM, half a million to diabetes-related chronic kidney disease, and 180,000 due to nonalcoholic steatohepatitis (NASH)-related liver cancer and cirrhosis, worldwide.4,5 Lifestyle intervention (LSI) is usually offered first to these patients, of whom only 10% to 20% achieve the targeted 7% to 10% of body weight loss needed to improve their condition (for example, reduce histological features of NASH 6 ), and only a minority of these can maintain a sustained and long-term weight loss, known to improve outcomes such as cardiovascular events. 7 Additional strategies are obviously needed that, combined with LSI, could offer long-term weight loss to patients with metabolic disorders. Bariatric-metabolic surgery is currently the single treatment for morbid obesity proven effective in the long term with a prolonged benefit on its related comorbidities. It allows patients to achieve an excess weight loss (EWL) of 65% to 80% one to two years after surgery, 8 partial or complete T2DM remission in up to 45% of the cases after five years9–11 and the disappearance of NASH in 85% of patients one year after surgery.12,13 However, these procedures are associated with significant complications 14 and lack widespread applicability because of their higher costs.
Even if endoscopic bariatric and metabolic therapies (EBMTs) are not as effective as their surgical counterparts, these less-invasive procedures might not only reproduce the aforementioned positive metabolic effects but also fill the gap between medical and surgical therapy in a cost-effective manner. In addition, because obesity and associated comorbidities are a chronic disease that requires lifelong therapy, having a first interventional therapy possibly offered after or together with LSI, and that does not render more difficult further surgeries if needed, is clearly an add-on to the therapeutic armamentarium.
This review focuses on the existing scientific evidence regarding the safety and efficacy of EBMTs in the setting of obesity but also in its associated comorbidities: T2DM and nonalcoholic fatty liver disease. Additionally, we discuss future directions as well as the unmet needs to address where EBMT might become beneficial.
Endoscopic bariatric therapies
Weight-loss outcomes commonly used in clinical trials are the following: (a) percentage of total body weight loss (% TWL) defined as [initial weight–postoperative weight/initial weight] × 100, and (b) percentage of EWL (% EWL) defined as [(initial weight)–(postoperative weight)]/[(initial weight) – (ideal weight)] (in which ideal weight is defined by the weight corresponding to a body mass index of 25 kg/m2). Furthermore, primary obesity interventions are defined as stand-alone intervention in combination with LSI and/or behavioral therapy to induce weight loss and improvement in obesity-associated medical comorbidities.15,16
Any new endoscopic bariatric therapy (EBT) should fulfill targeted efficacy thresholds, which have been recently defined by the task force convened by the American Society of Gastrointestinal Endoscopy (ASGE) and the American Society for Metabolic and Bariatric Surgery.15,16 The main outcome measurements or features include the following:
a minimum threshold of 25% EWL measured at 12 months, a mean % EWL difference between an EBT and a control group should be at least 15% EWL and statistically significant, 5% of TWL should be achieved as the absolute minimum threshold for any nonprimary EBT (defined as an intervention to promote weight loss specifically to: a) reduce the risk from a subsequent surgical intervention, for example, cardiovascular surgery, b) bridge to bariatric surgery, or c) act as metabolic therapy), the risk associated with EBT should be less than or equal to 5% (incidence of serious adverse event, SAE), and if a low-risk EBT proves to have a significant impact on one or more obesity-related comorbidities, the threshold for intervention may extend to class I obese individuals (body mass index 30–35 kg/m2).
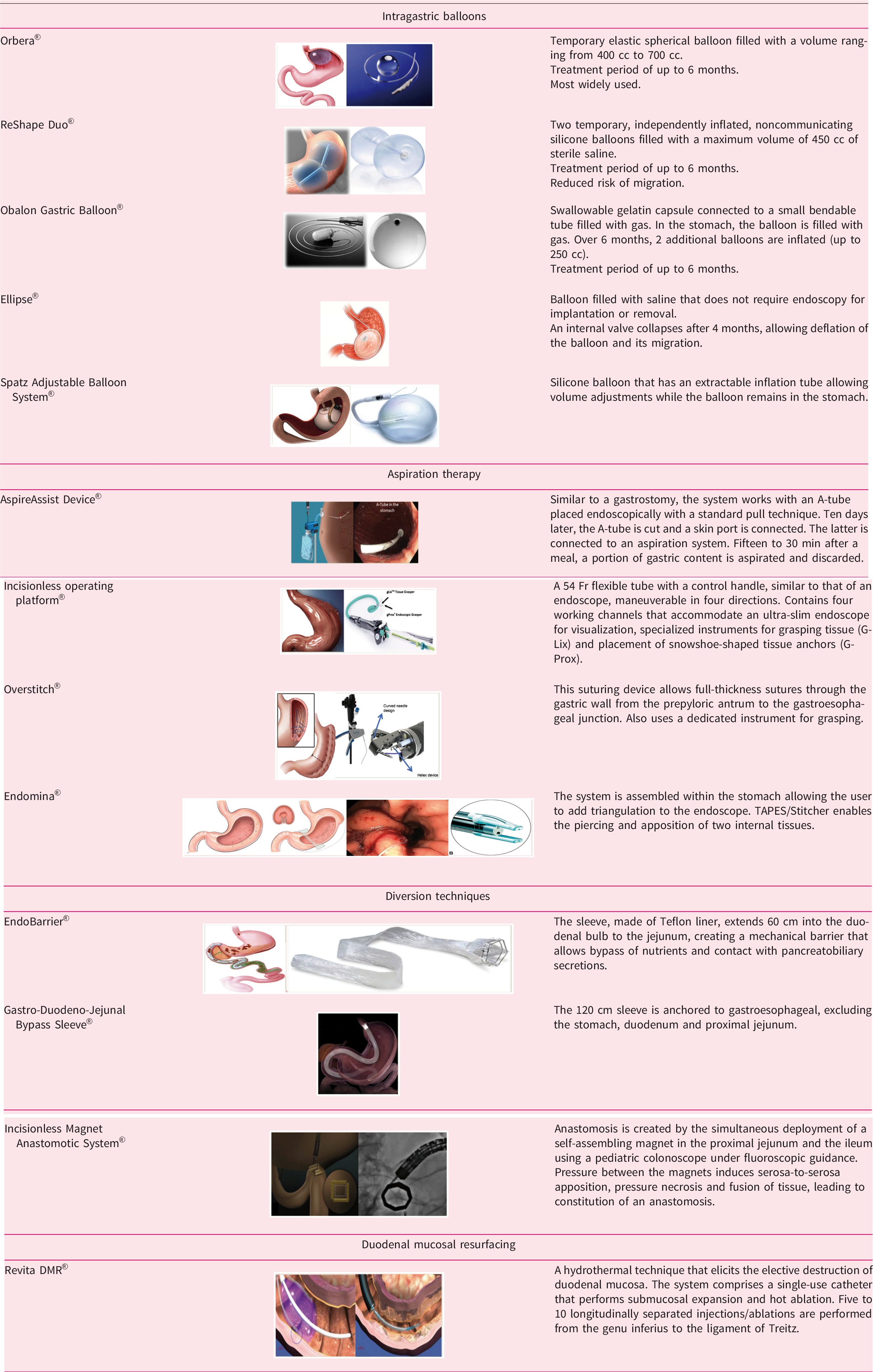
Space-occupying devices
Intragastric balloons (IGBs) are the most widely used space-occupying devices in clinical practice because of their high applicability and reversibility. Numerous IGBs are available in Europe; their main characteristics are summarized in Table 1.
Endoscopic medical devices.
IGBs have been used for several years in Europe, and large retrospective series 17 have suggested their modest but significant efficacy at midterm.
The effectiveness and safety of IGBs have been assessed in several randomized clinical trials (RCTs)18–20 (Table 2). The mean % EWL was more or less 25%, with the most common SAE being digestive intolerance requiring early removal of the balloon. Other SAEs included severe dehydration, one gastric perforation with sepsis, and one gastric outlet obstruction.
Randomized clinical trials in endoscopic and bariatric therapies.
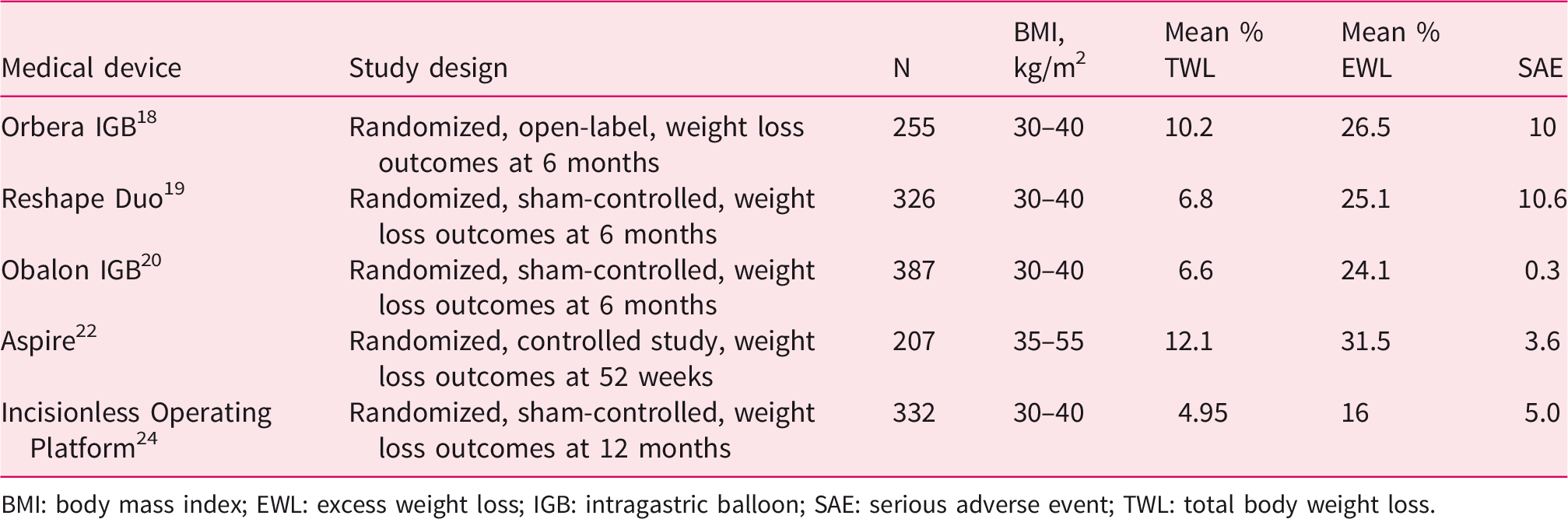
BMI: body mass index; EWL: excess weight loss; IGB: intragastric balloon; SAE: serious adverse event; TWL: total body weight loss.
Overall, these IGBs have met the short-term efficacy thresholds provided by the ASGE task force but the high variability, mainly in terms of study designs, limits the external validity of the results. Recent real-world data 23 regarding Orbera IGB placement has shown that this procedure is safe (the potential risks raised by the US Food and Drug Administration such as acute pancreatitis, spontaneous balloon hyperinflation and unanticipated deaths were not observed in this large cohort), and TWL of 5%, 10% and 15% was achieved by 88%, 62% and 31% of patients, respectively. Nevertheless, weight regain is frequent. In programs associated with adequate nutritional follow-up, the maintenance of weight loss 6 months after removal of the IGB has been observed in two-thirds of patients,18–20 whereas it drops to less than 50% 18 months after IGB placement. 15 Lastly, literature is scarce regarding long-term efficacy data; a few studies have reported that only 25% of patients maintained a TWL of 10% or greater after 2.5 years, 17 whereas only 9.2% of patients exhibited an EWL of more than 20% after five years. 21
Aspiration therapy
Aspiration therapy consists of a gastrostomy-like implant allowing to empty a significant amount of the stomach content after each meal. The Aspire Assist System (AAS) (Aspire Bariatrics, King of Prussia, PA, USA) was developed with this purpose (see Table 1). The effectiveness of this medical device was studied in an RCT 22 (see Table 2) in which 207 patients were enrolled in either AAS plus LSI (N = 137) or LSI alone (N = 70). The coprimary end points were measured at 52 weeks and defined as mean % EWL between the two groups and the proportion of patients achieving a 25% EWL. SAEs were reported in 3.6% of the AAS group, including perioperative peritonitis, intragastric ulceration, and fungal growth of the tube. AAS patients had a mean EWL of 31.5% (vs 9.8% (P < .001) for controls). A total of 58.6% of patients in the AAS group and 15.3% in the control group lost at least 25% of EWL (P < .001). Interestingly, there was no evidence of overusing or worsening of eating disorders such as binge eating. On the contrary, the device somehow reduced binge eating because thorough mastication is required for its successful use.
Suturing devices
Three new endoscopic devices have been designed to induce gastric restriction by performing gastric plicatures whose arrangement, for two of them, justify the term endoscopic sleeve gastroplasty. The idea is to offer a transoral intervention that might become the first surgical option offered to patients for management of this chronic disease. Their main characteristics are summarized in Table 1.
The efficacy and safety of the Incisionless Operating Platform (IOP) (USGI Medical, San Clemente, CA, USA) were simultaneously evaluated in two RCTs. The ESSENTIAL trial 24 was a multicenter, blinded, sham-controlled study (see Table 2) in which patients were randomly assigned to either an IOP group plus LSI (N = 221) or a sham endoscopy plus LSI group (N = 111). At 12 months, the mean TWLs in the active and sham group were 4.95 ± 7.4% and 1.38 ± 5.58%, respectively (P < .001), and the proportion of patients who reached 5% or more TWL were respectively 41.55% and 22.11% (P < .001). SAEs were reported in 5.0% of IOP patients, including one case of hepatic abscess and one case of extraintestinal bleeding. The latter was most likely due to improper plication placement technique. The MILEPOST multicenter RCT 25 was smaller compared to the previous one (N = 44), no SAEs occurred and the 12-month TWL was 13.0%.
The Overstitch (Apollo Endosurgery, Austin, TX, USA) device was up to now studied only in postmarketing reviews. A recent systematic review and meta-analysis 26 based on eight studies including 1772 patients at six months reported mean TWL and EWL of 15.1% and 57.7%, respectively. These results were sustained at 12 months for the minority of patients still followed at that time. The aforementioned meta-analysis presents several limitations: the absence of RCTs, the fact that most of the studies were retrospective, the high percentage of patients lost to follow-up, and, therefore, scarce data regarding long-term efficacy. A randomized, multicentric trial is ongoing (NCT03406975).
Lastly, the Endomina (Endo Tools Therapeutics, Gosselies, Belgium) device was first assessed in a pilot study 27 (N = 11) with short-term results showing 41% of EWL at six months without SAEs. A phase 2 multicentric trial 28 did not show any device-related SAEs either, and the results of EWL and TWL at one year were 29% and 7.4%, respectively. A phase 3 multicentric trial comparing a control group (LSI alone) to a treatment group (Endomina) is ongoing (enrollment completed) (NCT03255005).
Altogether, these procedures are safe and effective, but larger trials are needed to ensure these promising results. Considering the recent evaluation of these procedures, long-term efficacy outcomes are also awaited.
Diversion devices
Based on the experience of bariatric surgery, efforts have been concentrated on the development of medical devices (see Table 1) that reproduce some of the anatomical features of bariatric surgery.
The duodenal-jejunal bypass liner is an endoscopic procedure using the EndoBarrier device (GI Dynamics, Boston, MA, USA), a stent fixed into the duodenum and comprising a liner that avoids the contact of food with the duodenum and proximal jejunum. A meta-analysis 15 has shown a mean EWL of 35.3% at 12 months (three prospective studies, N = 105), whereas the mean EWL difference compared with a control group (four RCTs comparing 12 to 24 weeks of treatment; N = 84 controls compared to EndoBarrier patients, N = 90) was significant at 9.4% but did not meet the efficacy thresholds defined by the ASGE task force 15 of 15%. Moreover, the large, multicenter, double-blinded, sham-controlled ENDO trial conducted in the United States was terminated early (by the Food and Drug Administration) before full enrollment because of serious concerns regarding the safety of the procedure. SAEs occurred in 11.7% of patients in the active group mostly because of the significant incidence of hepatic abscess formation (3.5%), resulting in a high level of early device removal.
Another similar endoluminal medical device has recently been developed, the gastroduodenojejunal bypass sleeve (ValenTx endoluminal bypass, ValenTx Inc, Hopkins, MN, USA) (see Table 1) but it has been evaluated in small trials29,30 that suggest its safety with encouraging initial results (35.9% EWL at 12 months).
To create an anastomosis in the gastrointestinal tract with the purpose of inducing malabsorption, a novel medical device has also been developed: the incisionless magnet anastomotic system (GI Windows, W. Bridgewater, MA, USA). The aim is to create a jejunoileal anastomosis to bypass a great part of the small bowel. A pilot trial 31 has been conducted showing promising results of 10.6% of TWL at six months with few SAEs. The technique remains extremely difficult with the need for upper and lower endoscopy, and laparoscopic assistance was required in the majority of the cases.
Despite encouraging initial results, these last two novel medical techniques still need to be evaluated in larger trials to assess their potential interest.
Endoscopic metabolic therapies
Duodenal mucosal resurfacing (DMR) is a procedure involving the elective destruction of duodenal mucosa, the latter being involved in key metabolic pathways. Indeed, its regeneration might hypothetically restore tissue homeostasis and improve glycemic control in T2DM patients. An initial human monocentric clinical study 32 was performed as a single arm, including 44 patients with poorly controlled T2DM taking oral antidiabetic drugs. Patients underwent a single application of ablation over 3 to 12 cm of the duodenum. The procedure was well tolerated, with the most common side effects being transient abdominal pain and three cases of duodenal stenosis that were successfully treated by a single endoscopic dilation. DMR was, at that time, performed with two separated balloon catheters for submucosal injection and ablation.
Further improvements were made to ensure proper mucosal lifting before ablation to avoid the previously mentioned complications. Hemoglobin A1c (HbA1c) was reduced significantly by 1.2% at six months with modest TWL. Recently, the DMR procedure was performed in a single-arm, open-label, multicenter study 33 in 46 obese patients with T2DM. There were no cases of duodenal stenosis. After 12 months, HbA1c was reduced by a mean of 1.0% ± 0.2% (P ≤ .001), and the homeostatic model assessment of insulin resistance was decreased by a mean of –3.3 ± 0.9 (P = .005). Furthermore, mean alanine aminotransferase decreased significantly from a level at baseline of 40 ± 4 U to 30 ± 3 U (P < .001) 12 months after DMR. There was also a modest reduction of weight (mean 2.4 ± 0.7 kg). A multicentric, randomized, sham-controlled study has terminated full enrollment, and results are expected soon (NCT02879383). There are currently no published clinical trials assessing the impact of available medical devices in the setting of NASH.
Perspectives and conclusions
Data in the literature regarding the safety and efficacy of these novel medical devices are recent, but further development is expected. Although establishment of efficient public health policies is a priority, further advances are needed to better define the place of EBMTs as an add-on to LSI in the clinical management of obesity and its related comorbidities but also their mechanism of action. Moreover, additional aspects need to be explored, such as long-term efficacy and the effect on clinically meaningful outcomes such as cardiovascular events. The key research and societal priorities regarding further development in this field are listed in Figure 1. Addressing these gaps would significantly affect future directions regarding clinical research as well as funding issues.

Research priorities and unmet needs in endoscopic and bariatric therapies (EBMTs).
Even if the clinical relevance remains unclear, numerous mechanisms of action have been proposed to explain the striking weight-independent metabolic effect of bariatric surgery. Although quite challenging to assess in human clinical trials considering several confounding factors such as weight loss, the overcoming of this bias might reside in the direct head-to-head comparison of EBMTs with matched weight-loss cohorts. Depending on the design of EBMTs used, numerous exploratory end points may be studied: orexigenic and anorexigenic gut peptides, composition of bile acids and microbiota, gastrointestinal motility and physiology to better understand the critical role of the gut in the positive metabolic effects observed after EBMTs and whether they are sustained over time (Figure 2), a feature that has currently been observed only with surgical therapies.11,34 In conclusion, EBMTs are safe and effective, and some might become part of the armamentarium in the management of obesity. Temporary endoscopic therapies involving an implant (balloon, sleeve) may vastly fail in the long term but new approaches might be effective for a longer period. Efforts need to be concentrated on the development of large, well-designed, multicenter prospective studies that should be performed at dedicated expert centers (considering that most of these techniques require specific training) to address the current gaps in this field. Overcoming these exciting challenges will likely allow EBMTs to be part of tomorrow’s treatments of obesity and its related illnesses.
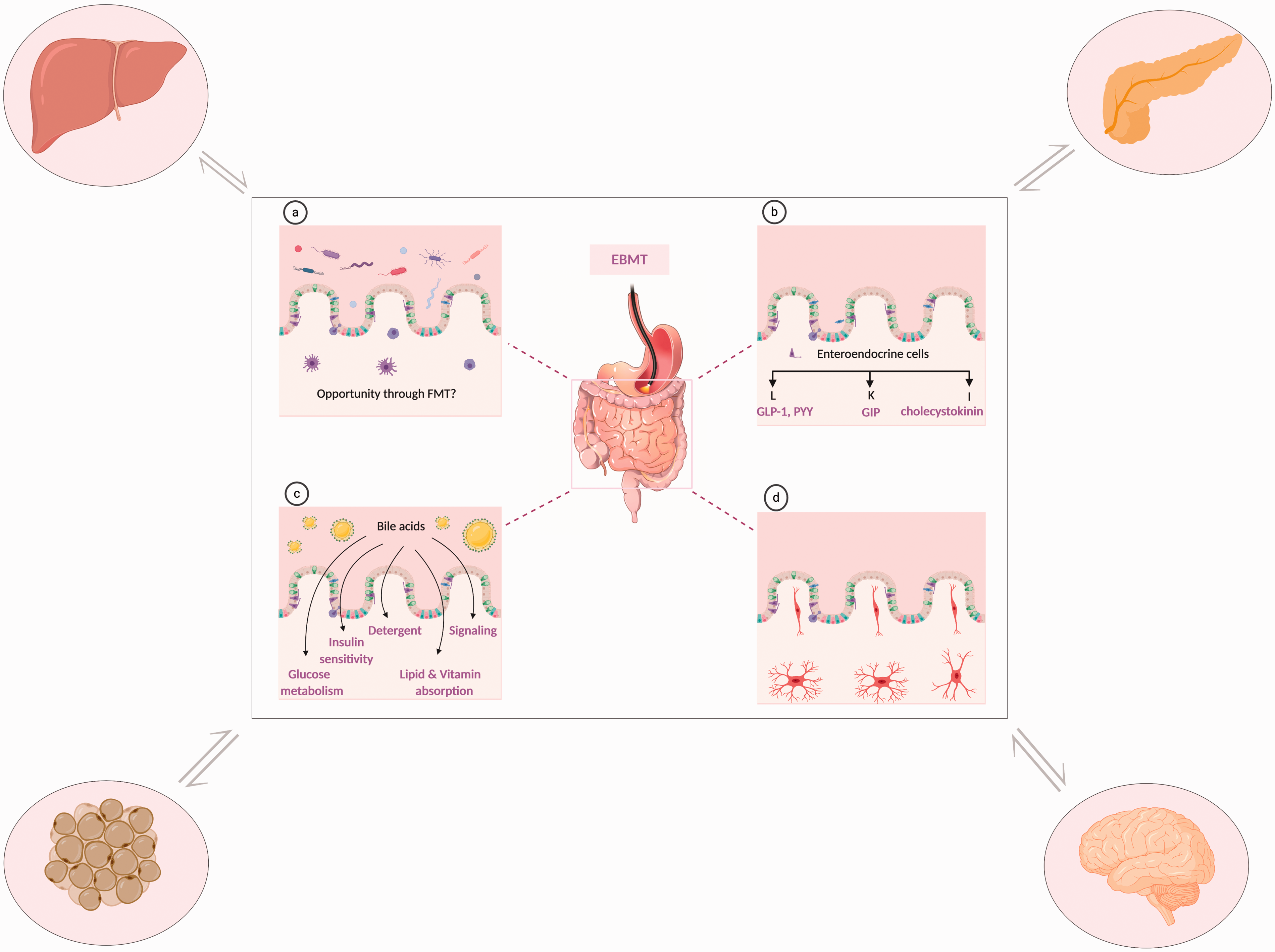
Potential weight-independent mechanisms of action of endoscopic and bariatric therapies (EBMTs) at the level of the gut. (a) Microbiome, opportunity to modify the microbiome through fecal microbiota transplant. (b) Production of gut peptides: orexigen peptides (PYY, peptide YY; cholecystokinin), incretins (GLP-1, glucagon-like peptide-1; GIP, gastric inhibitory peptide). (c) Modification of bile acids affect glucose metabolism and insulin sensitivity. (d) Enteric nervous system. The regulation of blood glucose is complex and necessitates crosstalk between the liver, the endocrine pancreas, the adipose tissue, the central nervous system and the intestine. FMT: fecal microbiota transplant. This figure was created with BioRender (www.biorender.com).
Footnotes
Acknowledgments
Author contributions include the following: writing and original draft preparation: AH; writing, review and editing: AH, MA, VH, JD.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JD has received research support for institutional review board–approved studies from Fractyl Inc of Lexington, Massachusetts, USA, and Endo Tools Therapeutics SA of Gosselies, Belgium, and is a shareholder of Endo Tools. VH has received research support for institutional review board–approved studies from and is a shareholder of Endo Tools. The other authors have nothing to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research grant from the “Fonds Erasme pour la recherche médicale” (doctoral research fellow grant) to AH.
