Abstract
Background and aims
Recent adult evidence suggests that infliximab (IFX) trough levels (TL) in patients with severe ulcerative colitis (UC) may be decreased. The aims of our study were to compare post-induction IFX TL of children with severe versus moderate UC and to evaluate short- and long-term outcomes.
Methods
In this single-center retrospective study, children with a diagnosis of UC starting IFX with a Pediatric Ulcerative Colitis Activity Index (PUCAI) ≥35 and with available post-induction TL were recruited. UC characteristics, IFX dosage and interval, primary non-response, IFX failure, and surgery after 24 months were collected. Post induction TL, anti-IFX antibodies, and laboratory evaluations at the time of starting IFX were also acquired.
Results
A total of 90 children were enrolled, of whom 39 (43.3%) were classified as severe UC and 51 (56.6%) as moderate UC. Median post-induction IFX TL were lower in severe UC versus moderate group (5.5 vs 10.3; p = 0.03), despite a more frequently intensified IFX regimen. Children in the higher TL quartiles showed increased rates of clinical, biological, and combined remission (p = 0.04, p < 0.001, and p = 0.01, respectively). In a multivariate analysis, a PUCAI ≥65 and time interval from last IFX infusion were the only predictors associated with IFX TL. At 24 months, children in the higher TL quartiles had a decreased risk of IFX failure (p = 0.002). The severe UC group showed a higher risk of IFX failure at 24 months (16/23 (41%) vs. 11/40 (21.6%); p = 0.05). Kaplan–Meier methods demonstrated a trend toward statistical significance, with a two-year cumulative colectomy rate of 15.38% (95% confidence interval (CI) 8.1–15.6%) in children with severe UC and 3.92% (95% CI 2.9–10.8%) in patients with moderate UC (logrank test p = 0.06).
Conclusions
Children starting IFX with severe UC showed lower post-induction TL and poor disease outcomes. Achieving adequate TL was associated with better efficacy outcomes.
Introduction
Pediatric ulcerative colitis (UC) is characterized by more extensive colonic involvement and more aggressive disease compared to adult-onset UC.1–3 As part of this phenotype, children with UC show a higher likelihood of developing acute severe colitis (ASC) exacerbations, with a rate as high as 28% compared to about 15% of adult cases. 4 ASC is a medical emergency, often requiring hospitalization. 5 Although intravenous corticosteroids (CCS) remain the mainstay of ASC treatment, 6 the rate of CCS refractoriness is higher in children than it is in adults. 7 Even though several studies have demonstrated the short-term efficacy of infliximab (IFX) in avoiding colectomy in patients with ASC,8,9 little is known about long-term efficacy. 10 In a recent study, 50% of patients receiving IFX after failing CCS required a colectomy at the two-year follow-up. 11
It is well established that the clinical response to IFX relies on pharmacokinetic factors. Indeed, higher IFX trough levels (TL) are associated with better clinical and endoscopic outcomes in all pediatric patients with inflammatory bowel disease (IBD), emphasizing the importance of targeting therapeutic TL.12,13 However, specific TL for pediatric UC have not yet been determined. In addition, recent adult evidence suggests that IFX TL may be significantly decreased in patients with more severe disease due to higher fecal loss and severe mucosal damage.14–16
The primary aim of our study was to compare post-induction IFX TL in children affected by severe UC compared to moderate UC. The secondary aims were to evaluate short- and long-term disease outcomes in relation to disease activity and TL in order possibly to define targeted TL associated with a better disease course.
Methods
This was a retrospective study conducted at the Children’s Hospital of Philadelphia. We performed a chart review of children diagnosed with UC undergoing IFX (Remicade®) therapy between August 2012 and June 2018. The inclusion criteria were: a diagnosis of UC; aged >6 years but ≤18 years at diagnosis; Pediatric Ulcerative Colitis Activity Index (PUCAI) ≥35 when starting IFX; available post-induction TL; and a minimum follow-up of six months. UC diagnosis was made on established criteria. 17 Children started IFX with a dose of 5 mg/kg or 10 mg/kg at the discretion of the treating physician on the basis of disease severity. The IFX schedule included induction infusions at week 0, week 2, and week 6, followed by maintenance every eight weeks, although the IFX regimen, including dosing and interval, could be intensified at the discretion of the treating physician based on disease severity or TL measurements. After enrollment, on the basis of the PUCAI score at the start of IFX treatment, children were divided into two groups: severe UC (PUCAI ≥65) and moderate UC (PUCAI ≥35 but <65). 18 The following information was collected: demographic data; disease extent based on the Paris classification; 19 PUCAI at diagnosis, before starting IFX, and at follow-up; therapeutic history; IFX protocol, including dose, time intervals, and intensifications, such as dose escalation or decreased intervals; and disease outcomes, including clinical response, clinical remission, biological remission, primary non-response (PNR), loss of response (LOR), and colectomy. Laboratory data including complete blood count, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), albumin, and fecal calprotectin at diagnosis and before starting IFX therapy were also collected. In addition, IFX TL and antibody anti-IFX (ATI) were noted post induction and at 6 and 12 months, when available. Post-induction TL were defined as levels drawn within 48 hours prior to the first maintenance IFX dose. During the study period, three different assays were used: (a) Anser IFX enzyme-linked immunosorbent second-generation assay (IFX quantitation limit <1.0 μg/mL and ATI concentration <3.1 IU/mL; Prometheus, San Diego, CA); (b) electrochemiluminescence immunoassay (IFX quantitation limit <0.4 μg/mL and ATI quantitation limit <22 ng/mL; Esoterix, Austin, TX); (c) quantitative chemiluminescent immunoassay (IFX quantitation limit 0.65 μg/mL and ATI concentration <0.65 μg/mL; ARUP Laboratories, Salt Lake City, UT). Children were followed until 24 months when the follow-up was available, or until IFX discontinuation or colectomy. The Institutional Review Board of the Children’s Hospital of Philadelphia approved the study protocol (May 9, 2018; number 18-015037). Written informed consent was obtained from all parents and from children, where appropriate. The study complies with the Declaration of Helsinki on research ethics.
Outcomes definitions
PNR was defined as a PUCAI decrease of <20 points in the setting of an IFX level ≥3 μg/mL by the first maintenance infusion leading to drug discontinuation, 20 while LOR was defined as an elevated clinical score accompanied by the decision to stop IFX. 20 Clinical remission was defined as a PUCAI score ≤10, with clinical response described as a PUCAI decrease of ≥20 points without reaching a score of ≤10. Biological remission was defined as CRP ≤5 mg/L and ESR ≤20 mm/h in patients with elevated inflammatory markers when starting of IFX. 20 A combined remission was defined as the achievement of both clinical and biological remission.
Statistical analysis
Variables were screened for their distribution, and appropriate parametric or nonparametric tests were adopted as necessary. The Mann–Whitney test for continuous variables and the chi-square test and Fisher’s exact test for categorical variables were used. Correlations between IFX TL and laboratory parameters were evaluated through the Spearman coefficient. Proportions of patients who met the established outcomes were grouped into quartiles according to the overall median IFX TL per patient, and a linear trend was tested with the Cochran–Armitage test. The receiver operating characteristic (ROC) curve was used to determine the IFX TL threshold that best discriminated between different outcomes. Optimal thresholds points from the ROC curve were calculated using Youden’s J statistic. Multiple ordinary least squares regression tests were performed to find out the contribution of different variables, adjusting for age at diagnosis and sex. The Kaplan–Meier method was used to estimate the cumulative risk of surgery at 24 months. Differences between curves were tested using the log-rank test. Statistical significance was predetermined as p < 0.05. Percentages were rounded to the nearest whole number. IBM SPSS Statistics for Windows v20 (IBM Corp., Armonk, NY) and R v3.6.0 (R Foundation for Statistical Computing, Vienna, Austria) were used for all statistical analyses.
Results
A total of 90 children were included in the final analysis (median age at IFX starting: 14.5 years; range 6.4–21.2 years; male:female = 45:45), of whom 39 (43.3%) were classified as severe UC and 51 (56.6%) as moderate UC. Figure 1 shows the subjects’ progression through the study. Baseline characteristics are reported in Table 1.
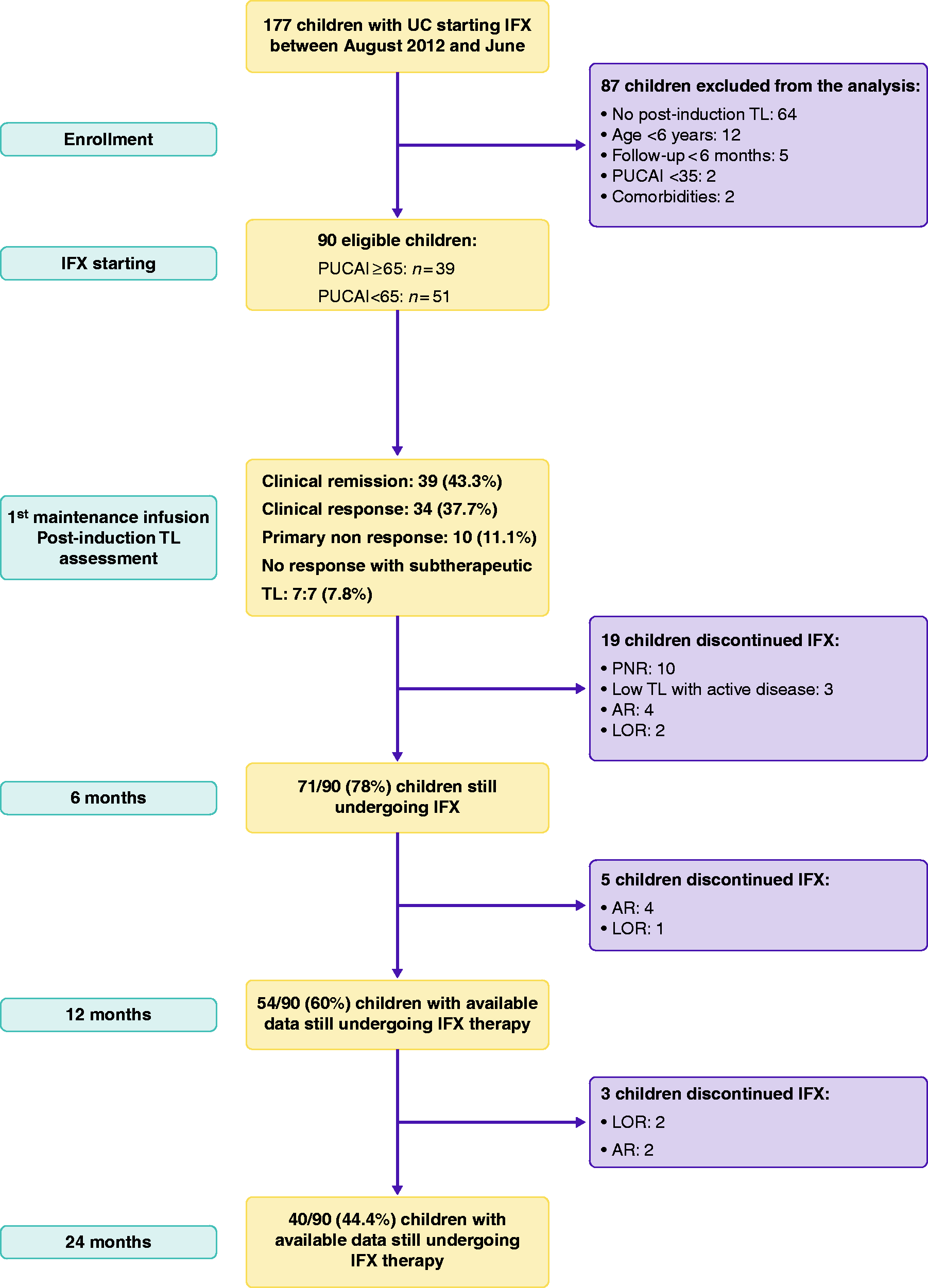
Flow diagram of the subjects’ progression through the study. AR: adverse reaction; IFX: infliximab; LOR: loss of response; PNR: primary non-response; TL: trough levels; UC: ulcerative colitis.
Clinical and laboratory characteristics of 90 children with severe (PUCAI ≥65) or moderate (PUCAI ≥35 but <65) UC starting IFX therapy.
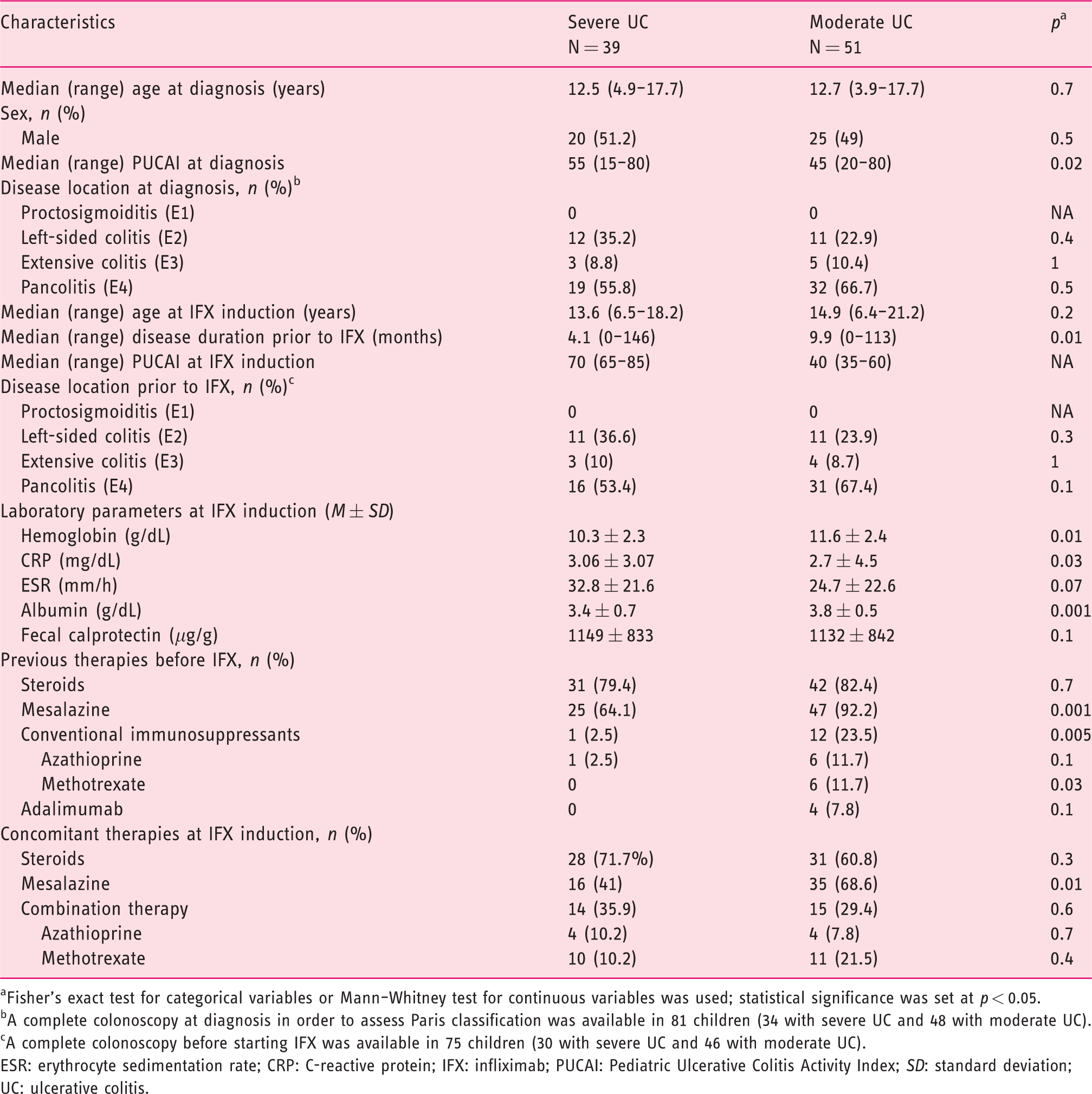
aFisher’s exact test for categorical variables or Mann–Whitney test for continuous variables was used; statistical significance was set at p < 0.05.
bA complete colonoscopy at diagnosis in order to assess Paris classification was available in 81 children (34 with severe UC and 48 with moderate UC).
cA complete colonoscopy before starting IFX was available in 75 children (30 with severe UC and 46 with moderate UC).
ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; IFX: infliximab; PUCAI: Pediatric Ulcerative Colitis Activity Index; SD: standard deviation; UC: ulcerative colitis.
IFX induction protocol
Out of 90 children, 41 (45.6%) started induction with an IFX dose of 5 mg/kg, while 49 (54.4%) used a 10 mg/kg dose. The mean starting dose in the severe UC group was higher than that in the moderate UC children (8.2 ± 2.3 mg/kg vs. 7.2 ± 2.4; p = 0.03). Of 39 children with severe UC, 29 (66.6%) started IFX with a 10 mg/kg dose versus 23/51 (45.1%) children with moderate UC (p = 0.05). The mean time between the first and the second infusions was significantly lower in the severe UC group compared to moderate UC children (1.8 ± 0.3 weeks vs. 2 ± 0.2 weeks; p = 0.01), as well as the mean time between the second and the third infusions (5.3 ± 1.2 weeks vs. 5.8 ± 0.7; p = 0.04). Overall, 13/39 (33.3%) children with severe UC intensified the protocol timing between the second and third infusions versus 8/51 (15.7%) of the moderate group (p = 0.05).
Post-induction TL
Post-induction IFX TL were drawn with a median time of 42 days from the previous IFX infusion without significant differences between children with severe and moderate UC (41 (range 16–63) vs. 47 (range 14–64); p = 0.1). IFX TL ranged from 0 to 40, with a median of 8.7 μg/mL. Based on the therapeutic window of 3–7 μg/mL, 20 47 (52.2%) children showed supra-therapeutic levels (>7.0 μg/mL), while 22 (24.4%) patients had therapeutic TL (3–7 μg/mL) and 21 (23.3%) sub-therapeutic TL (<3μg/mL), including 10 (11.1%) with undetectable levels (<1 μg/mL). In 3/21 (14.2%) children with sub-therapeutic levels, ATI were positive. Median post-induction IFX TL were significantly lower in children with severe UC compared to those with moderate UC (5.5 (range 0–40) vs. 10.3 (range 0–40); p = 0.03), despite more frequent 10 mg/kg dosing in the severe UC group (28/39 (71.8%) vs. 23/51 (45.1%); p = 0.07; Figure 2). Specifically, 14/39 (36.8%) children with severe UC had IFX TL ≤3 compared with 7/51 (13.7%) children with a moderate UC (p = 0.02). Among the children with sub-therapeutic TL, 9/39 (23.1%) with severe UC showed undetectable levels versus 1/51 (1.9%) in the moderate UC group (p = 0.008). IFX TL were positively correlated with hemoglobin (r = 0.310, p = 0.003) and albumin (r = 0.36, p = 0.001). Conversely, IFX levels were inversely correlated with ESR (r = −0.33, p = 0.01) and CRP (r = −0.398, p = 0.0002; Supplemental Figure).
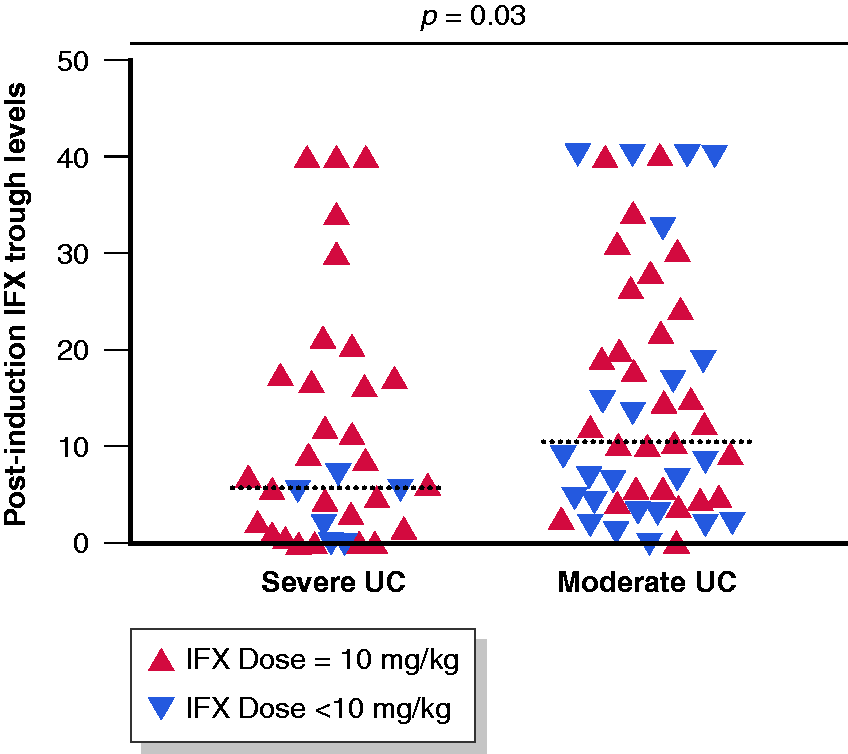
Post-induction IFX TL in children with severe or moderate UC with dose of last infusion specified (<10 mg/kg or 10 mg/kg). Median IFX TL were significantly lower in children with severe UC versus children with moderate UC (5.5 (range 0–40) vs. 10.3 (range 0–40); p = 0.03)). *Mann–Whitney test.
Relationship between TL and efficacy outcomes
After the induction protocol, clinical remission was reported in 39/90 (43.3%) children, while 51/90 (56.7%) had active disease. Clinical response was observed in 34/90 (37.7%) children, while 17/90 (18.8%) patients had no response. Among non-responders, 10 (58.8%) were classified as PNR, while 7 (41.1%) had sub-therapeutic TL. PNR did not differ in children with moderate versus severe UC (8/51 (15.2%) vs. 2/39 (5.1%); p = 0.1)]. Median TL were significantly higher in children with PNR than in the remaining children (37 (range 4.34–40) vs. 6.9 (range 0–40); p < 0.001)). Out of 90 children, 73 (81.1%) had positive inflammatory indexes at the start of IFX, and 37/73 (50.6%) of these achieved biological remission. Combined remission was reached in 22/73 (30.1%) patients.
To assess the relationship between IFX TL and efficacy outcomes, including clinical, biological, and combined remission after the induction, IFX TL were categorized into quartile groups (Q1: ≤3.31 μg/mL; Q2: >3.31 but ≤8.71 μg/mL; Q3: >8.71 but ≤18.91 μg/mL; and Q4: >18.91 μg/mL). We excluded PNR from this analysis. For all the outcomes, higher rates of remission were reported in the two highest quartiles, with a significant Cochran–Armitage trend test (p = 0.04, p < 0.001, and p = 0.01, respectively; Figure 3(a)–(c)). For clinical remission, the ROC curve analysis identified a cutoff of ≥5.29 μg/mL, with an area under the curve (AUC) of 0.70 (95% confidence interval (CI) 0.57–0.82, p = 0.004; Figure 4(a)). For biological remission, the cutoff was ≥4.83 μg/mL, with an AUC of 0.79 (95% CI 0.67–0.90, p < 0.001; Figure 4(b)). When we analyzed the combined remission, the cutoff was ≥5.29 μg/mL, with an AUC of 0.71 (95% CI 0.58–0.83, p = 0.005; Figure 4(c)).
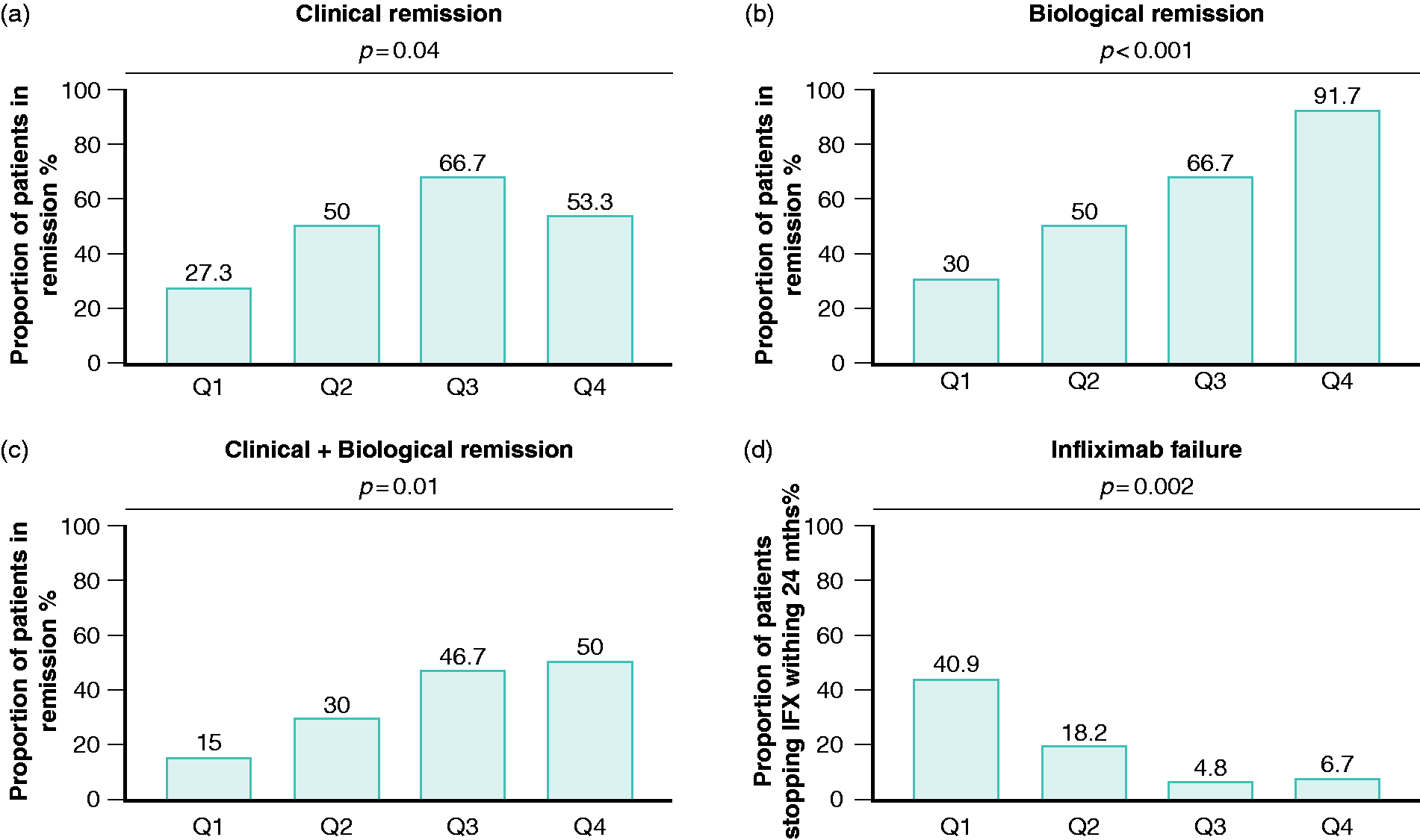
Quartile analysis of the IFX efficacy endpoints divided according to four quartiles of the median IFX TL per patient (Q1–Q4). IFX TL (μg/mL) quartiles were as follows: Q1: ≤3.31 μg/mL; Q2: >3.31 but ≤8.71 μg/mL; Q3: >8.71 but ≤18.91 μg/mL; and Q4: >18.91 μg/mL. The p-values derived from the Cochran–Armitage trend test analysis for the different IFX efficacy endpoints were 0.04, <0.001, 0.01, and 0.002, respectively, for clinical (a), biological (b), combined clinical+biological (c) after the induction and IFX failure within 12 months (d).
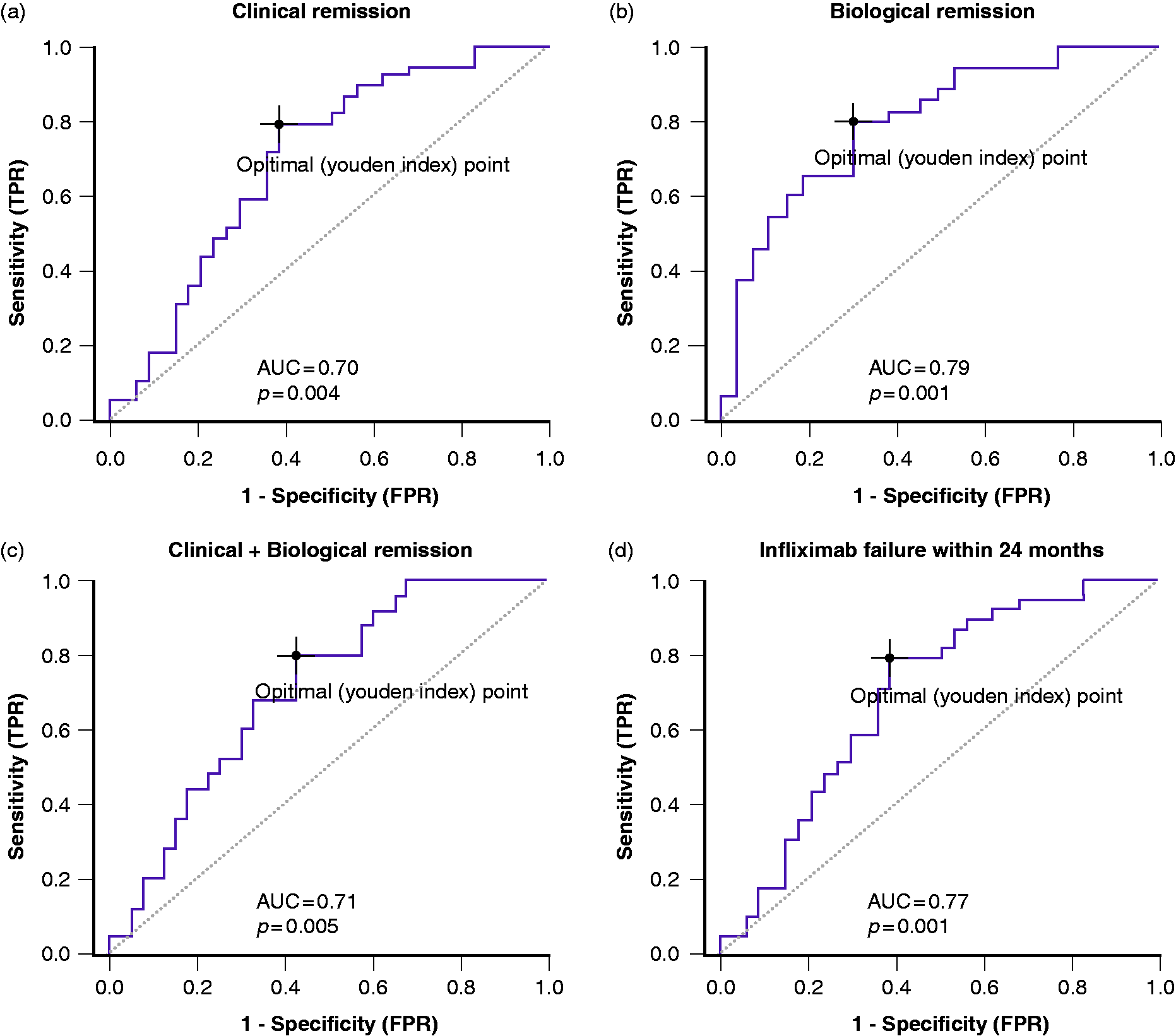
The received operating characteristic curve was used to determine the post-induction IFX TL threshold that best discriminated between different outcomes. Thresholds were calculated using Youden’s J statistic. (a) The area under the curve (AUC) to achieve clinical remission was 0.70 (95% confidence interval (CI) 0.57–0.82, p = 0.004); an optimal cutoff was defined as ≥5.29 μg/mL, with 80% sensitivity and 61.8% specificity. (b) The AUC to achieve biological remission was 0.79 (95% CI 0.67–0.90, p < 0.001); an optimal cutoff was defined as ≥4.83 μg/mL, with 80% sensitivity and 69.3% specificity. (c) The AUC to achieve combined biological and clinical remission was 0.71 (95% CI 0.585–0.83, p = 0.005); an optimal cutoff was defined as ≥5.29 μg/mL, with 80% sensitivity and 57.5% specificity. (d) The AUC to prevent IFX failure within 12 months was 0.77 (95% CI 0.585–0.83, p = 0.001); an optimal cutoff was defined as ≥5.29 μg/mL, with 73% sensitivity and 76.5% specificity.
Multivariate analysis
In order to determine the predictors contributing to post-induction IFX TL, we conducted a multiple ordinary least squares regression. A PUCAI ≥65 at the start of IFX and the time interval from the last IFX infusion resulted in the only predictors contributing to post-induction TL. Specifically, regression coefficients show that a PUCAI ≥65 at the start of IFX decreases the post-induction TL on average by 6.57 μg/mL (p = 0.016), while an everyday increase of the interval from the last infusion reduced TL on average by 0.25 μg/mL (p = 0.017; Table 2).
Multiple ordinary least squares regression using post-induction IFX trough levels as dependent variable.
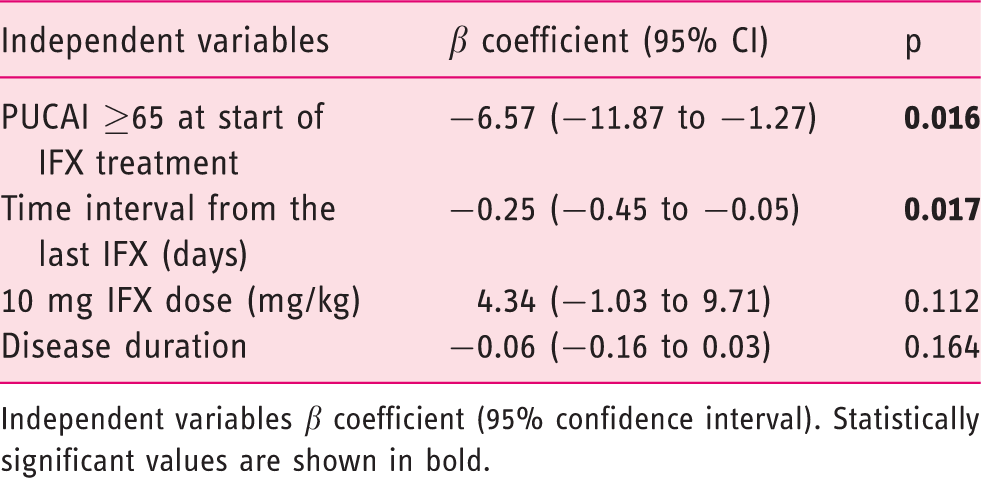
Independent variables β coefficient (95% confidence interval). Statistically significant values are shown in bold.
Follow-up
At six-month follow-up, 71/90 (78.8%) children were still undergoing IFX therapy. Among children stopping IFX, 13 (68.4%) discontinued immediately after the induction (PNR = 10; low TL with active disease = 3) and six (31.5%) during the follow-up (adverse reaction (AR) = 4; LOR = 2). Of the 71 children, 53 (74.6%) were in clinical remission. TL were available in 54/71 (76%) patients, with a median of 11.6 μg/mL (range 0–40 μg/mL). Specifically, 30/54 (55.5%) children showed supra-therapeutic levels, while 20/54 (37%) patients had therapeutic TL and four (7.4%) sub-therapeutic TL, including one (1.8%) with undetectable levels. None of the patients with sub-therapeutic levels showed positive ATI. At 12 months, data from 54 children were still available. Between 6 and 12 months, five children experienced IFX failure (AR = 4; LOR = 1). TL were measured in 42/54 (77.7%) patients, with a median of 11.4 μg/mL (range 1.4–40 μg/mL). Specifically, 28/42 (66.6%) children showed supra-therapeutic levels, 10/42 (23.8%) patients had therapeutic TL and 4/42 (9.5%) sub-therapeutic TL. None of the patients with sub-therapeutic levels showed positive ATI. At 24 months, data from 40 children still undergoing IFX were available. Between 12 and 24 months, three additional patients discontinued IFX (LOR = 2; AR = 1). Overall, 27/90 (28.8%) enrolled children had to stop IFX within 24 months, with a median of 4.3 months (range 1–24 months). After IFX failure, patients were exposed to the following therapies: adalimumab (n = 9; 36%), vedolizumab (n = 9; 36%), colectomy (n = 4; 16%), methotrexate (n = 2; 8%), azathioprine (n = 1; 4%), and steroids (n = 1; 4%). In order to evaluate the relationship between post-induction TL and IFX failure within 24 months, we used quartile groups. We did not include children stopping IFX after PNR in the analysis. A linear trend was identified in patients in the lower two quartiles showing higher rates of IFX failure when compared to the higher two quartiles (p = 0.002; Figure 3(d)). The ROC curve analysis identified a cutoff of ≥5.29 μg/mL, with an AUC of 0.77 (95% CI 0.63–0.89, p = 0.001; Figure 4(d)). With regard to disease activity at the start of IFX treatment, a higher number of children starting therapy with a PUCAI ≥65 experienced IFX failure compared to moderate UC children (16/23 (41%) vs. 11/40 (21.6%); p = 0.05). Specifically, LOR was increased in children with severe versus moderate disease at the start of IFX treatment, with a trend toward statistical significance (5/39 (12.8%) vs. 2/51 (21.6%); p = 0.08).
Surgery at 24 months
Of 90 children, eight (8.8%) underwent subtotal colectomy, with a median time from the start of IFX treatment of 3.35 months (range 1–24 months). A higher number of children with a PUCAI ≥65 at the start of IFX treatment underwent colectomy compared to moderate UC patients, with a trend toward statistical significance (6/39 (15.4%) vs. 2/49 (3.9%); p = 0.07). Kaplan–Meier methods demonstrated a two-year cumulative colectomy rate of 15.38% (95% CI 8.1–15.6%) in children with severe UC and 3.92% (95% CI 2.9–10.8%) in patients with moderate UC (p = 0.06; Figure 5).
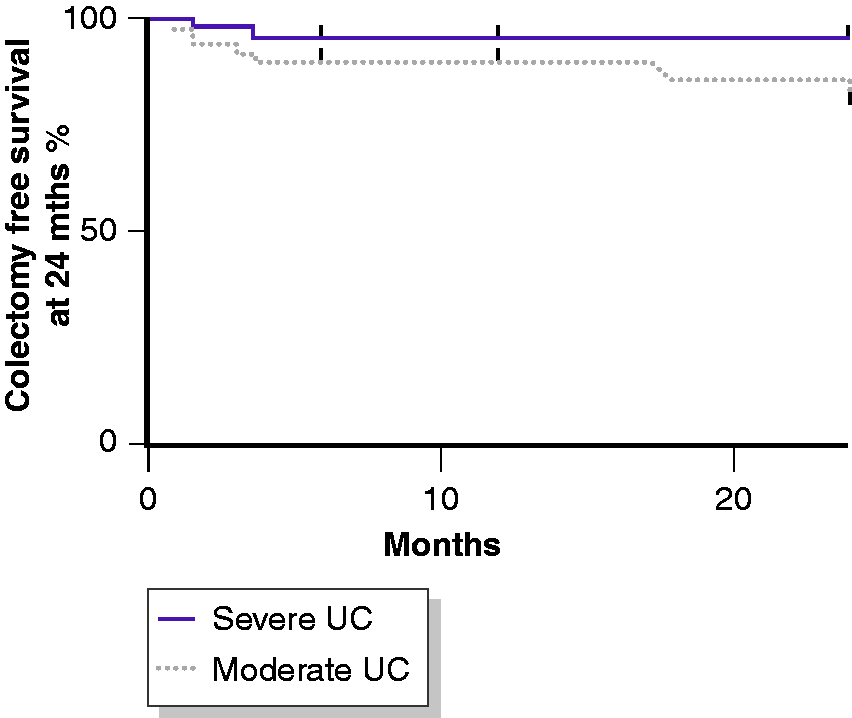
The Kaplan–Meier curve of the proportion of children undergoing colectomy on the basis of Pediatric Ulcerative Colitis Activity Index (PUCAI) score (severe: ≥65; moderate: ≥35 but <65) at IFX initiation. The survival distributions demonstrated a trend toward statistical significance (log-rank test p = 0.06) with a two-year cumulative colectomy rate of 15.38% (95% CI 8.1–15.6%) in children with severe UC versus 3.92% (95% CI 2.9–10.8%) in patients with moderate UC.
Discussion
To the best of our knowledge, this is the largest pediatric investigation aiming to compare post-induction IFX TL in children with severe UC versus moderate UC. Our data demonstrate that children starting IFX with a PUCAI score ≥65 have decreased TL and show a higher risk of IFX failure and subtotal colectomy at 24 months. Conversely, children reaching adequate post-induction TL experience higher rates of remission and a decreased rate of IFX failure at 24 months.
Over the last two decades, IBD therapeutic strategies have profoundly evolved, leading to an increased use of biologics, particularly anti-tumor necrosis factors (TNFs). 21 However, only recently, the use of therapeutic drug monitoring (TDM) has allowed for the optimization of biologic dosing and administration. 22 The need for TDM comes from a large body of literature, including two meta-analyses, demonstrating that higher TL are associated with clinical remission, mucosal healing, and decreased incidence of LOR.22–24 Most of these data are based on adult populations, while pediatric papers have mostly focused on Crohn’s disease (CD) rather than UC.12,13,20,22–25 Our study provides evidence that reaching an adequate post-induction TL is also associated with better efficacy outcomes in pediatric UC. Supporting this hypothesis, we found that children in the higher TL quartiles showed increased rates of clinical, biological, and combined remission, as well as a decreased rate of IFX failure at 24 months. These data were corroborated by the ROC curve analyses, which detected a value ≥4.83 μg/mL for biological remission and ≥5.29 μg/mL for clinical and combined remission. IFX optimization strategies are particularly needed in the setting of ASC. Recently, Krauthammer et al. reported the follow-up of a multicenter cohort of children admitted for an ASC flare in the biologics era. 26 Colectomy-free rates were 71.3%, 66.4%, and 63.6% after one, three, and five years, respectively, demonstrating that despite the introduction of anti-TNFs, the clinical outcomes of children with ASC are still poor. 26 In our cohort, children with severe UC at IFX initiation developed poorer long-term outcomes, including a higher rate of IFX failure and a higher colectomy rate, than those with moderate UC. Specifically, the Kaplan–Meier curve detected a two-year cumulative colectomy rate of 15.38% in children with severe UC versus 3.92% in patients with moderate UC. IFX pharmacokinetics seems the most conceivable reason. The ATLAS study elegantly demonstrated that anti-TNF tissue levels correlate with TL, inflammatory markers, and TNF in the tissue. 15 The authors hypothesized that local inflammation with high TNF levels may serve as a sink for anti-TNFs. 15 More interestingly, at a certain level of mucosal injury, there will be increased protein clearance, including stool losses. 14 In line with this hypothesis, our study confirms that children starting IFX with a PUCAI ≥65 are more likely to have sub-therapeutic and even undetectable levels, despite receiving an intensified IFX regimen more frequently. As previously reported, 20 TL were directly correlated with hemoglobin and albumin and inversely correlated with CRP and ESR. Not surprisingly, children with severe UC showed lower hemoglobin and albumin values and increased CRP and ESR.
Despite being used for more than 15 years, the optimal IFX dose strategy in ASC is unknown due to the difficulty in performing randomized controlled trials (RCTs). 6 Intensification of IFX therapy during induction, including an escalating dose at 10 mg/kg and/or shortening intervals between infusions, are now more common practice. 6 In a recent North American survey, 76% of respondents indicated the preferred use of an intensified dosing regimen for ASC. 27 However, whether an intensified IFX regimen may affect the response rate is still unclear. In the Acute Colitis Trials 1 and 2, no differences in term of efficacy were observed between the 10 mg/kg and 5 mg/kg IFX induction strategies. 28 Nevertheless, a post hoc analysis suggested a higher long-term colectomy-free rate in the 10 mg/kg dose regimen. 29 More recently, Nalagatla et al. reported no differences in hospital rates or short- or long-term colectomy in adult patients with ASC receiving an accelerated versus standard induction protocol. 30 Conversely, Choy et al. found that multiple doses of IFX 5 mg/kg are superior to a single-dose salvage. 31 In our cohort, children with severe UC were more often exposed to an intensified IFX regimen. However, in a multivariate model, the time interval from the last infusion and disease severity, rather than IFX dose, were the factors that significantly predicted TL. Overall, these findings highlight that complex factors determine the IFX dose–exposure relationship. We speculate that proactive TDM, including TL frequent measurements and guided administration of additional doses, may have a significant impact on the response rate rather than an a priori intensified dose regimen alone. Going along with this hypothesis, Assa et al. recently demonstrated in the first pediatric RCT that ADA TL proactive monitoring was associated with higher remission rates than reactive monitoring in pediatric CD. 32 This approach may perhaps also be extrapolated in severe UC.
PNR has been reported as one of the main causes of IFX failure in IBD, 33 with rates widely ranging from 10% to 42% based on the different study designs and definitions. 34 In the current study, we did not include within PNR patients with sub-therapeutic levels. In that way, we were able to exclude children with a clear explanation for the lack of response (low TL or high ATI). Following this strategy, we found that children with PNR showed higher median TL than the remaining children. Our interpretation is that the strict PNR criteria allowed us to identify a cluster of patients with different underlying inflammatory pathways, which would not benefit from an intensified IFX regimen or a switch to a different anti-TNF.
It is acknowledged that the present study has several limitations. The main drawbacks are related to the retrospective nature and the possibility of recall bias, which need to be taken into account. Another possible limitation is that due to the evolution of laboratory technology over time, as three different therapeutic drug-monitoring assays were used. However, all the assays are extensively validated, 34 and TL with these tests have been shown to be largely comparable. 35 Finally, due to the lack of endoscopic evaluations and FC assessment at the same time points, it was not possible to include mucosal healing within the outcomes.
Conclusion
This large cohort study demonstrated that children affected by UC starting IFX with a PUCAI ≥65 tend to have lower TL and poor disease outcomes, despite a more frequently intensified IFX regimen. On the other hand, children reaching an adequate post-induction TL have higher rates of clinical, biological, and combined remission, together with a significantly reduced risk of IFX failure at 24 months. Thus, protocol intensification after TL proactive monitoring during induction may be necessary to increase the efficacy, especially in patients with severe disease. Further well-designed RCTs are urgently required to identify the optimal induction and maintenance strategy in children with UC.
Supplemental Material
UEG912877 Supplemental Material - Supplemental material for Post-induction infliximab trough levels and disease activity in the clinical evolution of pediatric ulcerative colitis
Supplemental material, UEG912877 Supplemental Material for Post-induction infliximab trough levels and disease activity in the clinical evolution of pediatric ulcerative colitis by Hillary Moore, Pasquale Dolce, Nina Devas, Robert Baldassano and Massimo Martinelli in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
The authors declare no conflict of interest to disclose regards to this paper.
Ethics approval
The study protocol was in accordance with the ethical guide-lines of the 1975 Declaration of Helsinki and was approved by the local ethics committee.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all patients.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
