Abstract
Background
The European Society of Gastrointestinal Endoscopy recommends rectal indomethacin or diclofenac before endoscopic retrograde cholangiopancreatography (ERCP) to prevent post-ERCP pancreatitis. However, data on the prophylactic effect in patients with primary sclerosing cholangitis (PSC) are lacking.
Methods
This was a retrospective case-control study. In 2009–2018, a total of 2000 ERCPs were performed in 931 patients with PSC. Case procedures (N = 1000 after November 2013) were performed after administration of rectal diclofenac. Control procedures (N = 1000 before November 2013) were performed with the same indication but without diclofenac. Acute post-ERCP pancreatitis and other ERCP-related adverse events (AEs) were evaluated.
Results
Post-ERCP pancreatitis developed in 49 (4.9%) procedures in the diclofenac group and 62 (6.2%) procedures in the control group (p = 0.241). No difference existed between the groups in terms of the severity of pancreatitis or any other acute AEs. The risk of pancreatitis was elevated in patients with native papilla: 11.4% in the diclofenac group and 8.7% in the control group (p = 0.294). In adjusted logistic regression, diclofenac did not reduce the risk of pancreatitis (odds ratio (OR) = 1.074, 95% confidence interval 0.708–1.629, p = 0.737). However, in generalised estimation equations with the advanced model, diclofenac seemed to diminish the risk of pancreatitis (OR = 0.503) but not significantly (p = 0.110).
Conclusion
In this large patient cohort in a low-risk unit, diclofenac does not seem to reduce the risk of post-ERCP pancreatitis in patients with PSC. The trend in the pancreatitis rate after ERCP is decreasing. The evaluation of the benefits of diclofenac among PSC patients warrants a randomised controlled study targeted to high-risk patients and procedures.
Keywords
Introduction
Endoscopic retrograde cholangiopancreatography (ERCP) is an invasive method to explore and treat diseases of the biliary and pancreatic ducts (PD). Adverse events (AEs) in diagnostic ERCP are rather rare, the most common being post-ERCP pancreatitis (PEP). The risk for PEP in unselected patients is 3.5%. 1
Primary sclerosing cholangitis (PSC) is a chronic cholestatic liver disease characterised by biliary inflammation, fibrosis and consequently strictures and dilatations of the intra- and/or extrahepatic bile ducts. No curative medical treatment is currently available, and the disease may lead to cirrhosis, end-stage liver disease and death or liver transplantation.2–5 ERCP is indicated for PSC patients with symptoms (jaundice, pruritus, elevated liver enzymes,) or suspicion of new or progression of a known dominant stricture (i.e. a stricture <1.5 mm in the common biliary duct or <1 mm in the hilar region within 2 cm from bifurcation) and suspicion of cholangiocarcinoma.3,6 Therapeutic ERCP can reduce symptoms and prevent or treat cholangitis within PSC. 3 The risk for AEs in ERCP has been shown to rise up to 14% when balloon dilatation, endoscopic sphincterotomy and stenting are performed.7,8 PSC is a known risk factor for PEP. The PEP rate among PSC patients varies between 5% and 7.8%,9–11 although rates as high as 21.4% have been reported. 12
The European Society of Gastrointestinal Endoscopy guidelines recommend routine rectal administration of 100 mg diclofenac or indomethacin immediately before or after ERCP as a prevention of PEP in all patients without contraindications. 1 The recommendation is based on meta-analysis where PEP prevention with rectally administrated diclofenac or indomethacin was effective.13–18 Later, another meta-analysis demonstrated the same effect.19,20 However, only two randomised controlled trials (RCTs) also included PSC patients.21,22
Thus, the aim of our study was to detect whether diclofenac has any effect on the risk of PEP in PSC patients. Additionally, we wanted to determine other possible factors affecting PEP risk and AEs of PSC-related ERCP procedures.
Methods
Study design and population
This was a retrospective case-control study. In Helsinki University Hospital (HUS) Meilahti endoscopy unit, 100 mg rectal diclofenac has been administered for PEP prophylaxis since November 2013. We collected 1000 consecutive ERCP procedures after November 2013 as the diclofenac group (DG) and 1000 consecutive ERCP procedures before November 2013 as the control group (GC). Inclusion criteria were suspicion or confirmed diagnosis of PSC and age >16 years. The exclusion criterion for the DG was contraindication for diclofenac, for example renal insufficiency or allergy to non-steroidal anti-inflammatory drugs (NSAIDs). Patients with immunoglobulin G4 (IgG4)-associated cholangitis where excluded. A total of 2000 ERCPs meeting the inclusion criteria were performed for 931 patients between January 2009 and January 2018, and these ERCPs comprised our study groups.
The study was approved by the hospital’s study board, and no further ethical committee approval was needed. The study was retrospective in nature, and the data were based on patient records. Thus, no written consent was needed. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki.
ERCP performance
Common bile duct (CBD) cannulation for PSC patients was performed using a papillotomy knife (Jagtome RX; Boston Scientific, Miami, FL) and a 0.035‐in, 450 cm guide wire (Jagwire; Boston Scientific). Other possible additional advanced cannulation methods included transpancreatic biliary papillotomy (TPBP) or transpancreatic biliary sphincterotomy (TPBS), with additional needle knife (NK), double guidewire (DGW), sphincterotome precut, NK precut or papillectomy. 23 A biliary papillotomy (BP) was performed with the sphincterotome. Among PSC patients, as presented in our previous study, sphincterotomy was performed to prevent papillary oedema and PEP in forthcoming ERCPs. 9 A balloon occlusion technique is used for adequate filling of the intrahepatic bile ducts, and cytological brush samples and deoxyribonucleic acid (DNA) flow cytometry can be taken. All patients without contraindications received a single dose of 500 mg levofloxacin prior to ERCP.
Source and data
We collected the data from the hospital patient records. Data collection included the basic characteristics: age, sex, weight, height, year of PSC diagnosis and co-morbidities – inflammatory bowel diseases (IBDs). American Society of Anaesthesiologists (ASA) grade (physical status classification system), medication and dosages, surgical procedures (cholecystectomy, liver transplantation, appendectomy, other abdominal surgery) and administration of prophylactic 100 mg rectal diclofenac were also collected.
The date, duration of the ERCP and additional procedures during ERCP, number of previous ERCPs, history of BP or TPBP, cannulation methods, biliary stone removal, dilatation of the bile-duct strictures, stenting of the strictures and collected samples (brush cytology, DNA flow cytometry) were collected. The length of hospital stay, laboratory tests (plasma amylase and bilirubin before procedure, plasma amylase 4 and 24 hours after procedure (if patient stayed overnight)) were recorded. ERCP procedures were graded according to the degree of difficulty of the procedures. 24
Grading of PSC
Additionally, the modified Amsterdam score25,26 was collected from the PSC register in HUS. Intra- and extrahepatic scores were determined separately. PSC was determined to be advanced if the ERC score was ≥4 or if biliary tract dilatations were needed during the ERCP. Advanced extrahepatic disease was determined as an ERC score ≥4 from extrahepatic bile ducts and/or a need for extrahepatic bile-duct dilatations of the dominant stricture during ERCP.
Definition of AEs
PEP was defined as plasma amylase levels at least three times the upper limit of normal at 24 hours after ERCP and where then patient suffered from a new or worsening abdominal pain with a need for prolonged hospitalisation for at least two days.1,27
AEs related to the ERCP procedure were evaluated, including symptoms of acute PEP, acute bacterial cholangitis, bowel or CBD perforation, bleeding and other infection, and a 30-day follow-up that considered late-onset acute pancreatitis (AP), death, date and cause of death if available. The severity of AEs was qualified with the consensus criteria of Cotton et al. 27
Statistical methods
For categorical variables, a two-tailed Fisher’s exact test and cross-tabulation were performed. For continuous variables, the Wilcoxon Mann–Whitney U-test was performed. Evaluation of the effect of diclofenac was performed with binary logistic regression analysis. Factors related to PEP were first evaluated as individual risk factors. If a variable was a formerly known risk factor for PEP or the p-value in this binary logistic regression was <0.2, the factor was taken into the adjusted logistic regression analysis. Binary logistic regression analysis with multivariate adjusted values was performed as a combination of the forced model to ensure that diclofenac was in the model and then performed as a forward stepwise conditional model with all suspected or known risk factors for PEP. Results were evaluated as an odds ratio (OR) and 95% confidential interval (CI) with p-value.
To clarify the role of diclofenac and to consider simultaneously the number of patients who underwent several ERCPs during the study period, we developed a generalised estimation equations (GEE) model for logistic regression. This model allowed dependence within clusters, which here was patients with more than one operation, and independence between the clusters. For the model, we imputed the missing values of the variables using a k nearest-neighbour approach. After imputation, we fitted the initial GEE model using three different working correlation structures: independent, exchangeable and first-order autocorrelation structures. From these, the model with the smallest quasi-likelihood information criteria (QIC) statistic was chosen for the analyses. Then, we built several smaller GEE models and compared them using QIC. From these, the model with the smallest QIC was reported. Since our data were unbalanced, that is, the number of operations varied from 1 to 12, we reported sandwich and model-based standard errors.
Statistical analysis of the data was performed with IBM SPSS Statistics for Macintosh v25.0 (IBM Corp., Armonk, NY) and with R v1.2.1335 for Macintosh (R Foundation for Statistical Computing, Vienna, Austria). p-Values of <0.05 were considered statistically significant in all analyses.
Results
Basic characteristics of the patients and ERCP procedures are shown in Table 1. Most of the patients were men in both groups. IBD was more common in the DG (76.9%) than in the CG (63.3%), and most patients had ulcerative colitis (UC; Table 1). The median duration of the procedure was 26 minutes in both groups (range 5–94 minutes in the DG and 5–123 minutes in the CG; p = 0.953). Native papilla cannulations existed more often in the CG (35.8%) than in the DG (19.3%; p < 0.001). The primary cannulation method was the sphincterotome with the guidewire in 99.0% in the DG and in 92.1% in the CG. Additionally, advanced cannulation methods were used: TPBP (with additional NK cut) was performed 40/4.0% (NK 4/0.4%) times in the DG and 80/8.0% (NK 9/0.9%) times in the CG. Moreover, sphincterotome precut was performed eight times (0.8%), NK precut three times (0.3%), DGW twice (0.2%) and papillectomy once (0.1%) in the CG. Only one (0.1%) patient in the DG had a prophylactic pancreatic stent.
Patient and ERCP procedure characteristics.
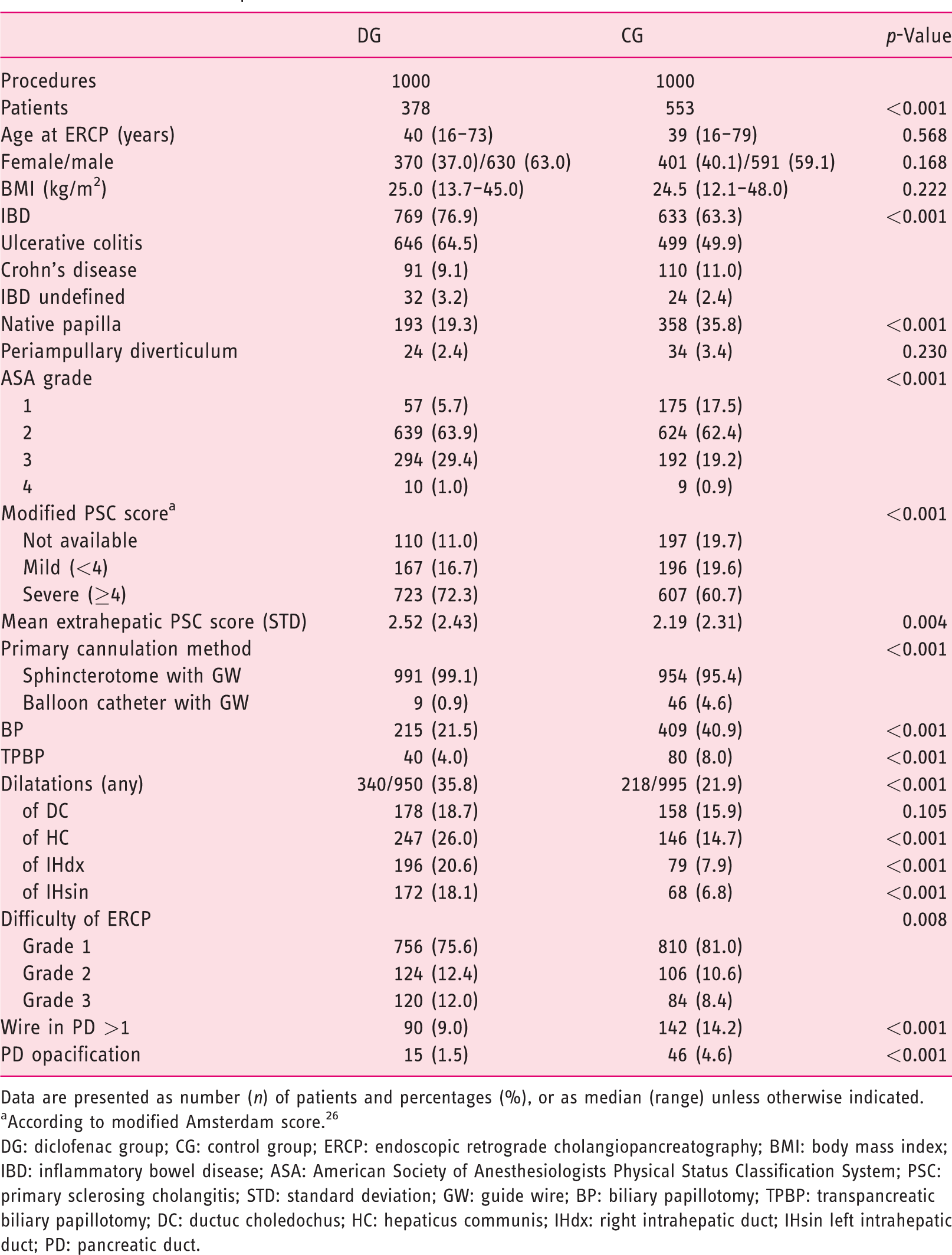
Data are presented as number (n) of patients and percentages (%), or as median (range) unless otherwise indicated.
According to modified Amsterdam score. 26
DG: diclofenac group; CG: control group; ERCP: endoscopic retrograde cholangiopancreatography; BMI: body mass index; IBD: inflammatory bowel disease; ASA: American Society of Anesthesiologists Physical Status Classification System; PSC: primary sclerosing cholangitis; STD: standard deviation; GW: guide wire; BP: biliary papillotomy; TPBP: transpancreatic biliary papillotomy; DC: ductuc choledochus; HC: hepaticus communis; IHdx: right intrahepatic duct; IHsin left intrahepatic duct; PD: pancreatic duct.
The median ERC score was 7 (range 2–16) in the DG, and 6 (range 2–16) in the GC (p < 0.001). Median intrahepatic ERC scores were 5 (range 2–8) in the DG and 4 (range 2–8) in the CG (p < 0.001). Median extrahepatic scores were 2 (range 0–8) in both groups. However, there were 408 patients in the DG and 320 patients in the CG with advanced extrahepatic PSC (p < 0.001).
During the study period, 435 patients underwent one ERCP, 253 patients underwent two ERCPs and 243 patients underwent three or more ERCPs. For the whole study population, a median of two (range 1–12) ERCPs were performed.
Pancreatitis
The PEP rate in this cohort of 2000 procedures was 5.6% (111 patients): 4.9% in the DG and 6.2% in the CG. AEs related to ERCP procedures are listed in Table 2. Most (63%) of the PEP patients suffered from a mild form of PEP. Late-onset AP occurred in three patients in the CG. The PEP rates in the DG and CG, compared to previously known risk factors for PEP, are shown in Table 3.
ERCP procedure AEs.
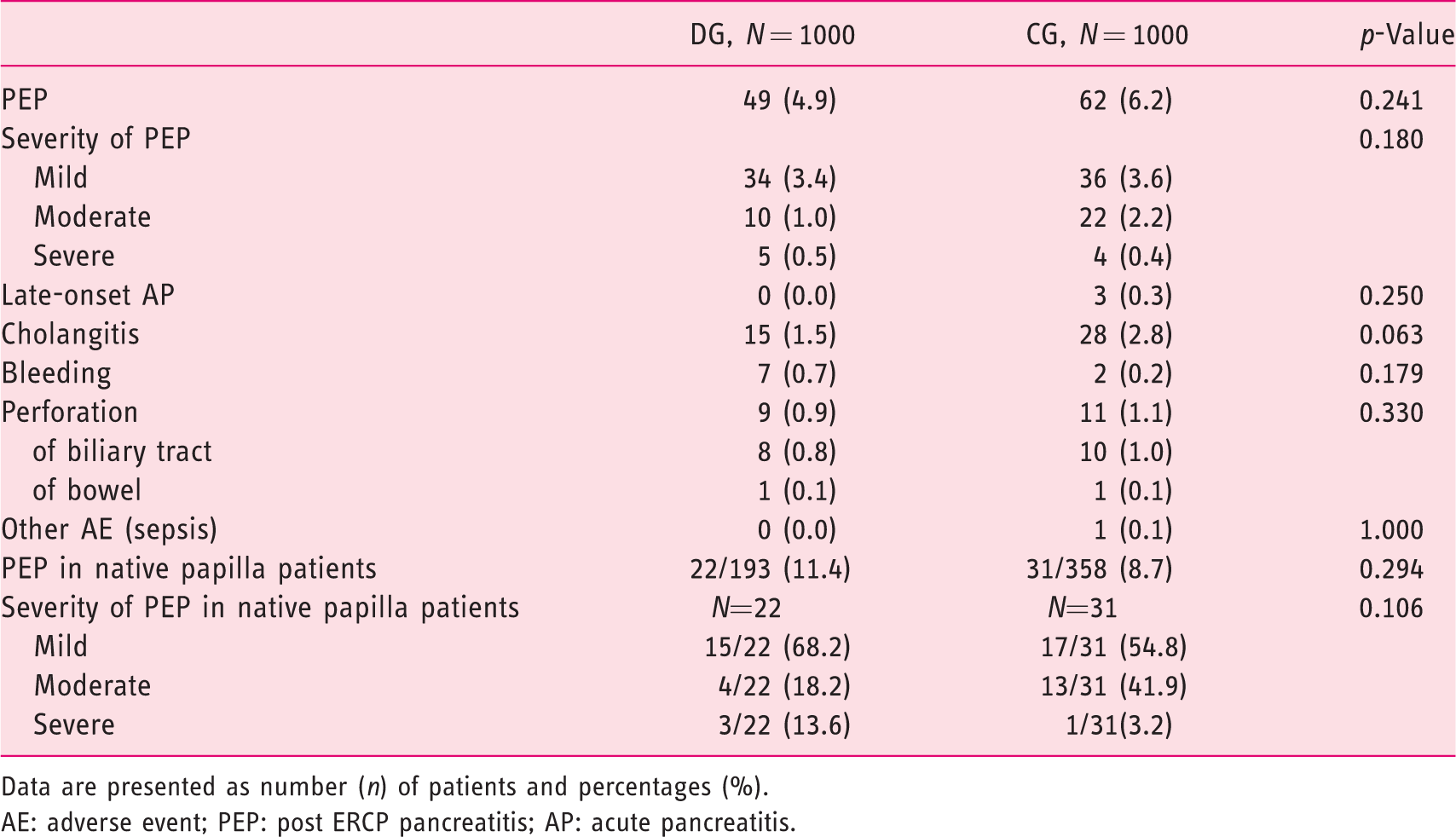
Data are presented as number (n) of patients and percentages (%).
AE: adverse event; PEP: post ERCP pancreatitis; AP: acute pancreatitis.
Incidence of PEP according to risk factors.
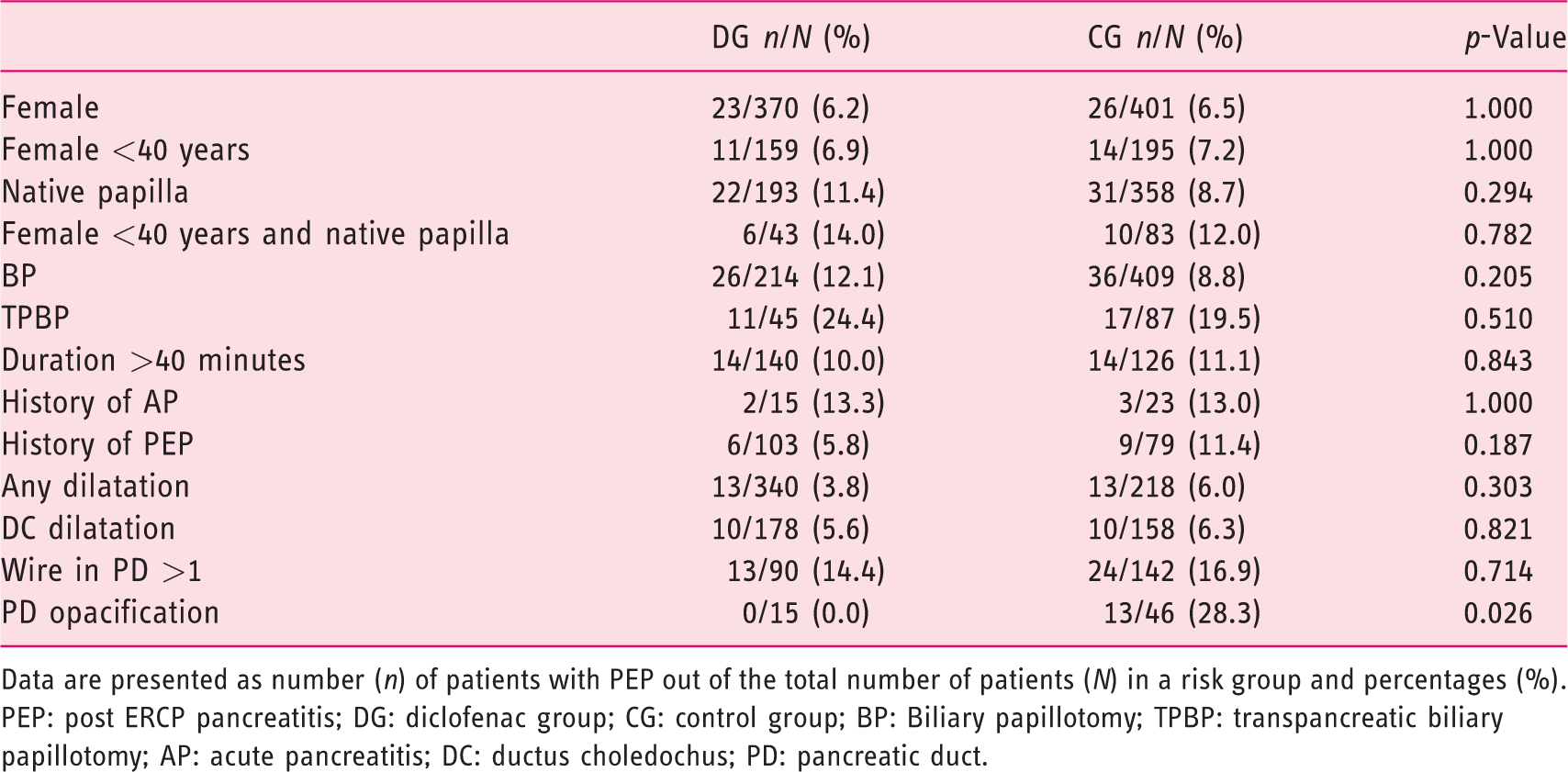
Data are presented as number (n) of patients with PEP out of the total number of patients (N) in a risk group and percentages (%).PEP: post ERCP pancreatitis; DG: diclofenac group; CG: control group; BP: Biliary papillotomy; TPBP: transpancreatic biliary papillotomy; AP: acute pancreatitis; DC: ductus choledochus; PD: pancreatic duct.
Risk factors for PEP were evaluated as an individual risk with binary logistic regression (Table 4). Significant risk factors in this univariate analysis were native papilla, ERCP procedure duration of >40 minutes, periampullary diverticulum, BP, TPBP, PD cannulation and PD injection. Factors that diminished the PEP risk were previously performed ERCP and prior BP.
Risk factors for PEP, univariate analysis.
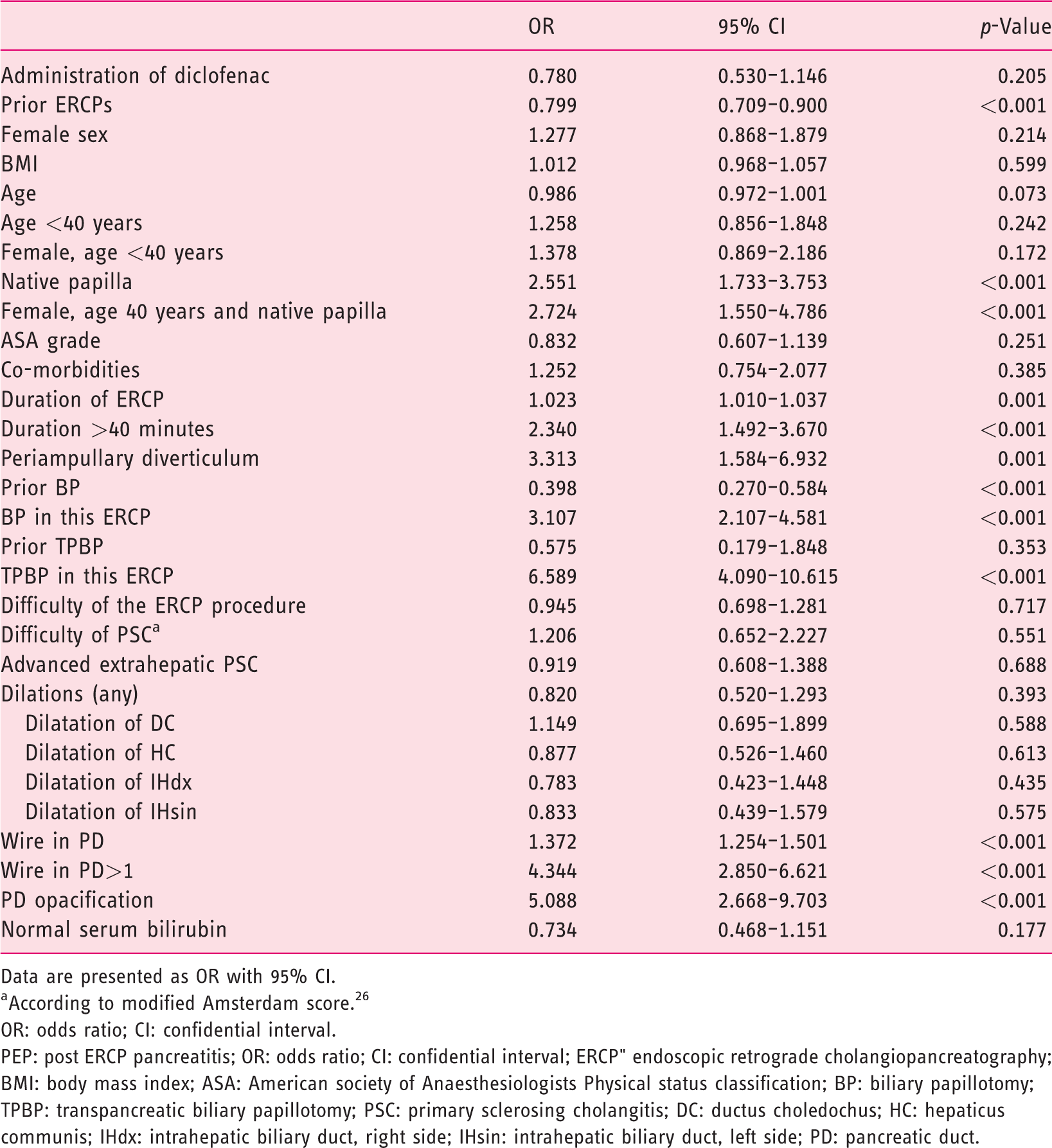
Data are presented as OR with 95% CI.
According to modified Amsterdam score. 26
OR: odds ratio; CI: confidential interval.PEP: post ERCP pancreatitis; OR: odds ratio; CI: confidential interval; ERCP" endoscopic retrograde cholangiopancreatography; BMI: body mass index; ASA: American society of Anaesthesiologists Physical status classification; BP: biliary papillotomy; TPBP: transpancreatic biliary papillotomy; PSC: primary sclerosing cholangitis; DC: ductus choledochus; HC: hepaticus communis; IHdx: intrahepatic biliary duct, right side; IHsin: intrahepatic biliary duct, left side; PD: pancreatic duct.
Results of the adjusted logistic regression model are shown in Table 5. A total of 1978 patients were included in the model; 22 patients were not included due to missing data for the variables involved in the analysis. Statistically significant factors were periampullary diverticulum, procedure duration >40 minutes, TPBP, PD passages >1 and PD opacification.
Multivariate logistic regression analysis with known or suspected risk factors related to PEP, adjusted.
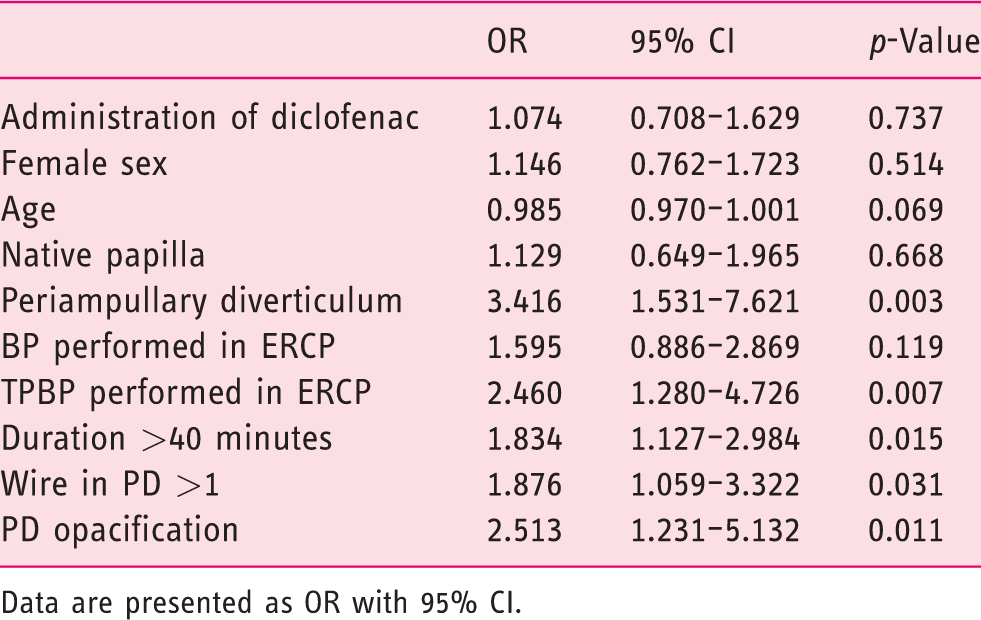
Data are presented as OR with 95% CI.
Generalised estimating equations (GEE) analysis and effect of diclofenac on post-ERCP pancreatitis, basic model.
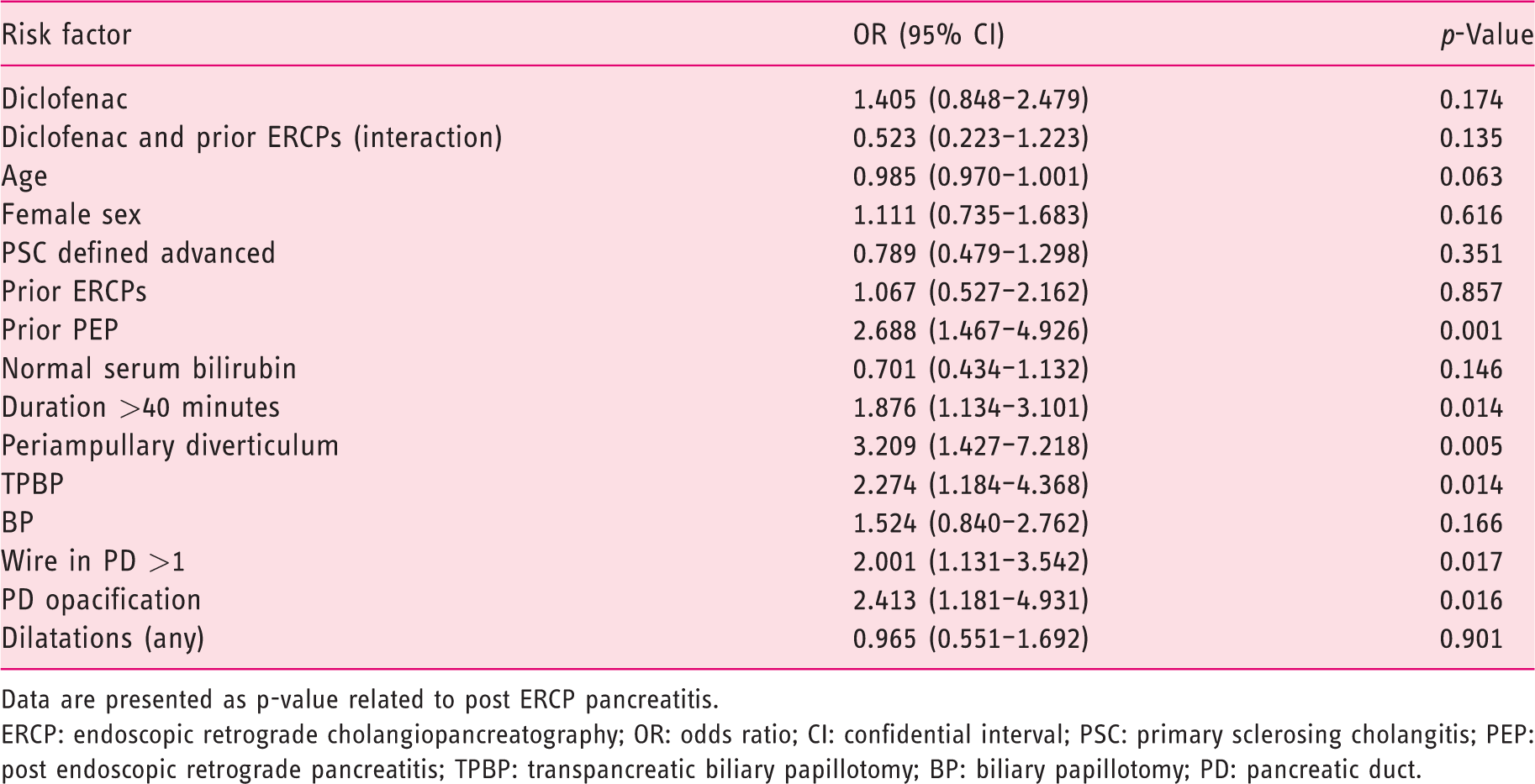
Data are presented as p-value related to post ERCP pancreatitis.ERCP: endoscopic retrograde cholangiopancreatography; OR: odds ratio; CI: confidential interval; PSC: primary sclerosing cholangitis; PEP: post endoscopic retrograde pancreatitis; TPBP: transpancreatic biliary papillotomy; BP: biliary papillotomy; PD: pancreatic duct.
In addition, a stepwise forward model with all suspected or known risk factors for PEP was performed. These factors were diclofenac, periampullary diverticulum, BP, TPBP, PD injection, PD passages >1, ERCP duration >40 minutes, female sex, native papilla, dilatations in biliary tract and PSC categorised as difficult. Statistically significant risk factors for PEP were periampullary diverticulum (OR = 2.861, 95% CI 1.253–6.541, p = 0.013), BP (OR = 1.819, 95% CI 1.146–2.887, p = 0.011), TPBP (OR = 2.306, 95% CI 1.205–4.411, p = 0.012), PD opacification (OR =2.260, 95% CI 1.141–4.885, p = 0.021), PD passages >1 (OR = 1.860, 95% CI 1.052–3.288, p = 0.033) and duration >40 minutes (OR = 1.779, 95% CI 1.098–2.882, p = 0.019). A total of 1939 patients were included in the model; 61 patients were not included due to missing data for the variables involved in the analysis.
In the GEE model, among analysed variables related to risk of PEP, part of the data for the following variables were missing: duration of ERCP procedure, body mass index (BMI) and preoperative serum bilirubin. Missing were 0–2.5% of the data, and these missing data were imputed: duration of ERCP procedure (21/2000; 1.1%), BMI (49/2000; 2.5%) and preoperative serum bilirubin (41/2000; 2.1%). Independent working correlation structure had the smallest QIC, and it was used in the analyses. Sandwich and model-based standard errors did not differ substantially, suggesting that there was no serious problem regarding the chosen working correlation structure. In our basic model, the variables were diclofenac, interaction term ‘diclofenac and prior ERCPs’, age, female sex, normal serum bilirubin, ERCP procedure duration >40 minutes, diverticulum, BP, TPBP, wire in PD >1, PD opacification and PSC defined as advanced (Table 6). The DG had a lower expected probability for PEP when considering the interaction with diclofenac and prior ERCPs (OR = 0.523), which was not statistically significant (p = 0.135).
In the advanced GEE model, we selected seven factors together with diclofenac and prior ERCPs. We found BP (OR = 1.754, 95% CI 1.026–2.999, p = 0.040), TPBP (OR = 2.473, 95% CI 1.309–4.672, p = 0.005), PD passages >1 (OR = 2.031, 95% CI 1.158–3.560, p = 0.013), PD opacification (OR =2.702, 95% CI 1.339–5.452, p = 0.006), periampullary diverticulum (OR = 2.752, 95% CI 1.247–6.075, p = 0.012) and prior PEP (OR = 2.576, 95% CI 1.422–4.666, p = 0.002) as significant factors related to PEP on the study population. The OR for diclofenac and prior ERCPs was 0.503 (95% CI 0.216–1.168, p = 0.110), and for diclofenac only 1.500 (95% CI 0.888–2.535, p = 0.130).
Patients with advanced extrahepatic PSC
In a subgroup analysis of patients with the advanced extrahepatic form of PSC (modified Amsterdam score ≥4 and/or dilatations of extrahepatic biliary strictures), the PEP rate was 17/408 (4.2%) in the DG and 20/320 (6.3%) in the CG (p = 0.235). In addition, there was no significant difference between the groups in perforations: 4/408 (1.0%) in the DG versus 3/320 (0.9%) in the CG (p = 0.602). In the DG, all four perforations happened during papillotomy. In the CG, two perforations were in the biliary duct (one during bile-duct dilatation, one by cytological brush) and one during papillotomy. Bleeding occurred in 2/408 (0.5%) cases in the DG compared to 2/320 (0.6%) in the CG (p = 1.000), and cholangitis occurred in 8/408 (2.0%) in the DG compared to 13/320 (4.1%) in the CG (p = 0.118). Diclofenac did not have an effect on PEP in this subgroup in univariate binary logistic regression (OR = 0.652, 95% CI 0.336–1.267, p = 0.207).
Other AEs
Other ERCP-related AEs are listed in Table 2.
Deaths
Of the 931 patients in the whole cohort, 28 (3.0%) died during the study period. Of these, 11 (1.2%) died of cholangiocarcinoma (five of them also had liver hilar (Klatskin) tumour), three (0.3%) of hepatocellular carcinoma/cholangiocarcinoma, two (0.2%) of metastatic colorectal carcinoma, one (0.1%) of pancreatic carcinoma, one (0.1%) of severe acute PEP and one (0.1%) of acute subarachnoid bleeding. The cause of death was not available for nine (1.0%) patients.
Discussion
Our main target was to evaluate the PEP rate among patients with PSC or suspected PSC and the effect of rectal diclofenac to prevent PEP. We found no significant difference between the diclofenac and control groups in the rate or severity of PEP. The number of other ERCP AEs (cholangitis, bleeding or perforations) also did not differ between the two groups. In logistic regression, administration of diclofenac did not show any statistically significant decrease in PEP rates. When adjusting the confounding factors and prior known risk factors for PEP in multivariate binary logistic regression, the OR for diclofenac was 1.074 (95% CI 0.708–1.629, p = 0.737; Table 5).
ERCP is the gold standard for diagnosis of PSC, but high-quality magnetic resonance cholangiopancreatography (MRCP) has replaced it as a primary diagnostic screening procedure.3,6 However, early-stage PSC, especially intrahepatic form, is very difficult to diagnose with MRCP alone.2,9 Moreover, in our previous study comparing ERCP and MRCP, we demonstrated that the agreement between ERCP and MRCP in scoring bile-duct changes for disease severity was only moderate (intra-hepatic bile ducts: weighted kappa =0.437, 95% CI 0.211–0.644; extra-hepatic bile ducts: weighted kappa = 0.512, 95% CI 0.303–0.720). 26
According to European guidelines, 1 rectal administration of diclofenac immediately before ERCP is used as a PEP prophylaxis in our unit. There are several meta-analyses showing the effect of diclofenac or indomethacin in the prevention of PEP.13–20 These meta-analyses are partly overlapping and based mainly on the same 13 RCTs in which the drug is administrated rectally. According to those meta-analyses, it seems that PEP prevention with NSAIDs is effective, at least in high-risk patient groups.
Controversial results have been reported in a few studies. Levenic et al. demonstrated no effect of rectal indomethacin in PEP prevention in consecutive, unselected patients. 21 In a randomised open-label prospective trial, the administration of rectal diclofenac in PEP prevention was ineffective. 28 In a previous study from HUS, 29 Rainio et al. did not find any positive effect in PEP in a retrospective study among consecutive unselected ERCP-patients, but not including PSC patients. In addition, these meta-analyses included only two RCTs that also included PSC patients.21,22 Subgroup analysis or separate results of PSC patients only were not presented.
In therapeutic ERCPs and in a difficult CBD cannulation in PSC, biliary sphincterotomy (BS) or BP is defined as an independent risk factor for short-term AEs such as PEP, cholangitis or post-procedural bleeding.9,30 PSC is one of the risk factors for PEP, and the PEP rate was 5–21.4% in different studies.9–12 Other patient-related risk factors include female sex, previous pancreatitis or PEP, suspected sphincter of Oddi dysfunction, young age, non-dilated extrahepatic bile ducts, absence of chronic pancreatitis and normal serum bilirubin.1,6,23 ERCP procedure-related risk factors include PD passages with guidewire >1, unintended PD injection, cannulation attempts lasting more than 5–10 minutes, biliary balloon cannulation, TPBS and a native papilla.1,6,23 Therapeutic ERCP can reduce symptoms and prevent or treat cholangitis within PSC. 3
In an open-label trial of PSC patients with advanced disease (N = 65) with dominant stricture and randomisation to either balloon dilatation or a short-term stent placement, the overall PEP rate was 13.6%. 31 In our study, patients in the DG had a more advanced form of PSC. This has a possible impact on the difficulty of the ERCP procedure and duration of the procedure due to the dilatations of the strictures. In addition, patients with IBDs, especially UC, seemed to have a more advanced form of PSC. 32 In our study, in the DG, patients had more IBDs. This can potentially affect the severity of PSC and thus supports our findings that diclofenac does not seem to be effective in a high-risk group of patients. Median durations, however, were similar (26 minutes) in both groups. Diclofenac did not show any significant decrease in the PEP rate in a subgroup analysis of patients with advanced extrahepatic PSC. When considering the rate of BP and TPBP, there were significantly fewer procedures in the DG (Table 1). In the DG, most patients (79.7%) had prior BP. BS was shown to be a protective factor for PEP in the study by Ismail et al. 9
ERCP-related risk factors for PSC patients have been evaluated in several studies. These previously known risk factors (PD injection, PD passage more than once, long-lasting ERCP procedure and TPBP/TPBS) were also statistically significant factors in our study. In addition, periampullary diverticulum and performing BP in ERCP appeared to be significant risk factors for PEP in our study. Interestingly, native papilla did not seem to be a risk factor for PEP in multivariate analysis. Most of the PSC patients are men, 33 and thus we did not find female sex to be a significant factor.
The GEE-model was built to help ignore the confounding factors related to prior ERCPs and prior BP or TPBP. In this model, however, diclofenac did not reduce the PEP rate significantly, although the trend was decreasing.
In a retrospective analysis of 657 ERCPs performed for PSC patients, the PEP rate was only 1.2%, the lowest prevalence reported. 30 In our unit, the PEP rate among PSC patients decreased from 7.0% 9 to the present 5.6%. In the study by Ismail et al., PSC patients and ERCP procedure-related risk factors between 2007 and 2009 were evaluated. Prior BS was the main effect of the decreasing PEP rate among PSC patients. 9
According to our results, diclofenac did not show any statistically significant effect on decreasing the risk of PEP.
Strengths and limitations of the study
Our study consisted of a large patient population with 2000 ERCPs, but the data collection was done retrospectively. However, all the ERCPs were performed by the same endoscopists during the whole study period, so the retrospective nature is unlikely the explanation for the lack of effect. In addition, ERCPs were not performed for 2000 individuals but for 931 patients. The individual risk for PEP theoretically varied among patients due to previous ERCPs and, for example, previous papillotomies. The GEE model tried to solve this problem in a group basis analysis.
Conclusion
In conclusion, in our retrospective study of 2000 ERCP procedures among 931 PSC patients, diclofenac did not show any statistically significant effect in decreasing the rate of PEP. This result is in line with another HUS study which compared the effect of diclofenac in patients other than those with PSC. 29 In a low-risk PEP unit such as HUS, PEP prophylaxis with diclofenac seems to be ineffective, and its routine use does not seem to be justified anymore.
However, GEE analysis showed the trend in the risk reduction, and we suggest that further prospective targeted RCTs are warranted in those high-risk patients identified in this study, that is, females <40 years old and patients with native papilla.
Footnotes
Acknowledgements
Vilja Koskensalo (V.K.) would like to thank Pasi Aronen (University of Helsinki) for his contribution on statistical analyses, especially on analyses of the generalised estimating equations. V.K. would also like to thank the Maud Kuistila Memorial Foundation (2019-0366F) and the Finnish Medical Foundation (1862/2018) for the working grants. The results of this article were demonstrated in an abstract (as a poster) at UEG Week 2019 in Barcelona.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Ethics approval
The study was approved by hospital's study board and no further ethical committee approval was needed. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
The study was performed retrospective and the data was based on patient records. Thus, no written consent was needed.
