Abstract
Type-C hepatic encephalopathy is a complex neurological syndrome, characteristic of patients with liver disease, causing a wide and complex spectrum of nonspecific neurological and psychiatric manifestations, ranging from a subclinical entity, minimal hepatic encephalopathy, to a deep form in which a complete alteration of consciousness can be observed: overt hepatic encephalopathy. Overt hepatic encephalopathy occurs in 30–40% of patients. According to the time course, hepatic encephalopathy is subdivided into episodic, recurrent and persistent. Diagnostic strategies range from simple clinical scales to more complex psychometric and neurophysiological tools. Therapeutic options may vary between episodic hepatic encephalopathy, in which it is important to define and treat the precipitating factor and hepatic encephalopathy and secondary prophylaxis, where the standard of care is non-absorbable disaccharides and rifaximin. Grey areas and future needs remain the therapeutic approach to minimal hepatic encephalopathy and issues in the design of therapeutic studies for hepatic encephalopathy.
Keywords
Brief clinical case
A 57-year-old male with nonalcoholic steatohepatitis-related cirrhosis, Child-Turcotte-Pugh class C and a Model for End-Stage Liver Disease score of 18 was hospitalized due to an alteration of consciousness and fever. A diagnosis of stage 2 episodic type C hepatic encephalopathy (HE) precipitated by a urinary tract infection was made and the patient started empirical antibiotic therapy and lactulose per os and enemas with a prompt resolution both of infection and HE. After discharge, the patient continued lactulose as secondary prophylaxis without experiencing other bouts of HE.
Definition and burden of type C HE
Type-C HE is a complex neurological syndrome, characteristic of patients with advanced liver disease, causing a variety of nonspecific neurological and psychiatric manifestations, ranging from a subclinical entity, minimal HE (MHE), to a most severe form characterized by a complete alteration of consciousness, clinically overt HE (OHE). HE is a harmful condition that is the result of severe hepatocellular failure or the presence of large portal-systemic shunts. The real epidemiology of HE is not easy to estimate and depends on symptom variability, the diagnostic tools and the variability of these data from published studies. OHE occurs in 30–40% of patients with cirrhosis during the natural history of their disease.
1
Usually the first episode of OHE is reported up to 25% of patients within 5 years of diagnosis, in relation to the presence of precipitants. After a first episode, the risk of further bouts increases and is scarcely responsive to treatment.
1
Moreover, prevalence rates of HE may be much higher in transjugular intrahepatic portosystemic shunt (TIPS),
2
as well as spontaneous3,4 or surgical shunting carriers.
5
In its mild expression, MHE, this condition is ‘apparently’ lacking any clinical evidence, by altering (only) the performance of psychometric evaluations or electrophysiological and other functional brain measures. MHE prevalence is debated but is considered very frequent, in fact 20–80% of patients suffer from this condition, depending on the diagnostic tools used. Nevertheless, MHE is clinically relevant and is related to patients’ falls, fitness to drive, working ability, sarcopenia and prognosis6–8 and worsens patients’ and caregivers’ lives by altering their quality of life and even their socioeconomic status. As grade I HE is difficult to define, the term ‘covert’ has been recently proposed combining MHE and Grade I HE. The aim is to simplify the clinical pattern thereby easily and uniformly diagnosing the presence of OHE. ‘Covert’ refers to a condition that is not unapparent, but also not overt. MHE and covert HE (CHE) are risk factors for developing OHE, with 5–25% of patients developing OHE within 5 years after cirrhosis diagnosis.9,10 According to its time course, HE is subdivided into episodic, if precipitated; recurrent if bouts occur with a time interval of 6 months or less; and persistent when there is a pattern of alterations always present, interspersed with relapses of overt HE.1–5,11 The burden of HE goes well beyond morbidity and mortality and the overall costs for the healthcare system should also include indirect costs for patients, families and caregivers, namely, the inability to work or risk of unemployment, poor quality of life and unfitness to drive.1,6 Figure 1 illustrates HE classifications according to worsening of cognitive function and time course.
Classification of hepatic encephalopathy (HE) according to worsening of cognitive function (a) and time course (b), aimed to simplify the clinical pattern and easily and uniformly diagnose the presence of minimal, covert and overt HE.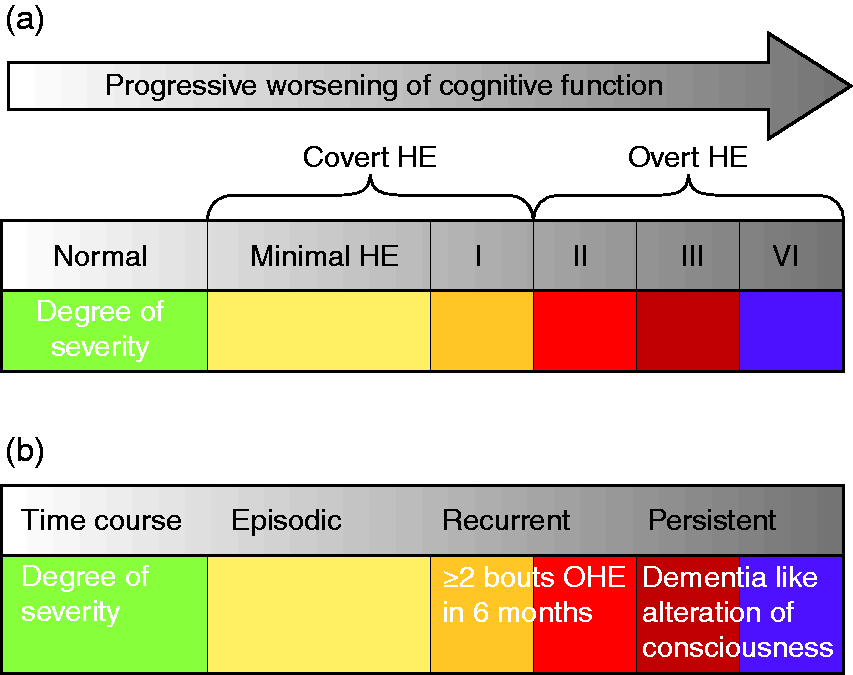
Diagnosis of type C HE
When and in which patients should type C HE presence be investigated?
There is a suspicion of type C OHE when personality changes (apathy, irritability and disinhibition, or obvious alterations in consciousness and motor function) occur in a cirrhotic patient. Moreover, alterations of the sleep-wake cycle with excessive daytime sleepiness can be frequently observed and asterixis may be present. Patients may also develop progressive disorientation to time and space, inappropriate behaviour and an acute confused state with agitation or somnolence, stupor and, finally, coma. The patient may experience a progressive impairment of consciousness from a mild to heavy degree or fall directly into a deeper stage of HE.
Precipitants and surroundings: an etiological look at HE
Episodic HE is often characterized by the presence of a precipitating event that should be identified and treated. Finding the precipitating event, both new or superimposed, is therefore mandatory when patients present with OHE. In some cases, multiple precipitating events may coexist in the same patient and the failure to identify and correct all the precipitating factors can worsen disease management.
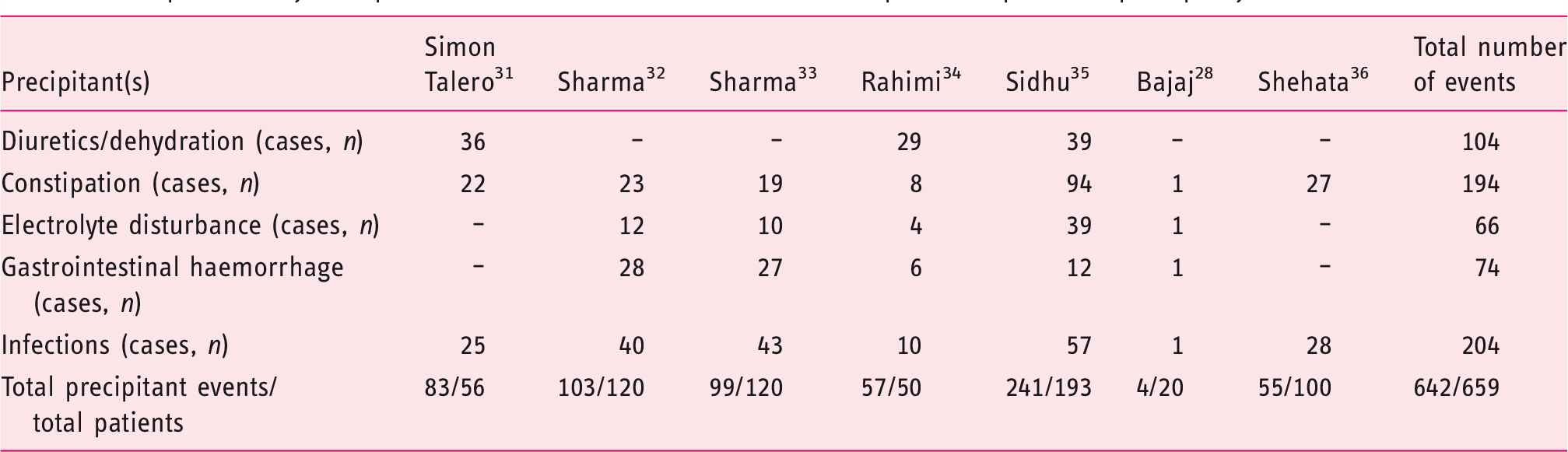
12
Table 1 summarizes the characteristics of the randomized controlled trials (RCTs) published on episodic precipitant-induced HE treatment. Infections represent the most common precipitating event (31%), followed by constipation (29.4%), dehydration and hypokalaemia and/or hyponatremia (17.7% and 11.2% respectively) or gastrointestinal bleeding (12.6%). If used, psychoactive drugs (opioids or benzodiazepines) should be discontinued. The differential diagnosis for patients not responding to the standard pharmacological approach should exclude the presence of alcohol withdrawal and neurologic alterations.
13
Brain imaging may therefore play a role in differential diagnosis of type C HE. In fact, cirrhotic patients admitted into hospital with HE may need to be evaluated by imaging techniques when the diagnosis between different etiologies of neuropsychiatric impairment remains difficult. For example, intracerebral haemorrhage, ischaemic brain injury, Wernicke’s encephalopathy and meningoencephalitis can be excluded using appropriate imaging tools. Some patients may present chronic HE, refractory to the conventional medical therapy, often without evident precipitating events. The presence of unrecognized large spontaneous portal-systemic shunts (SPSSs) may cause the chronic course of HE. Around 46–70% of cirrhotic patients with refractory HE show SPSSs at radiological imaging.3,4,14–16 Our group demonstrated the presence of SPSSs in 71% of cirrhotic patients with chronic HE, refractory to the standard medical treatment
3
(Figure 2) and recently published data suggest a total cross-sectional SPSS area >83mm
2
increases the risk for overt HE and mortality in liver cirrhosis.
14
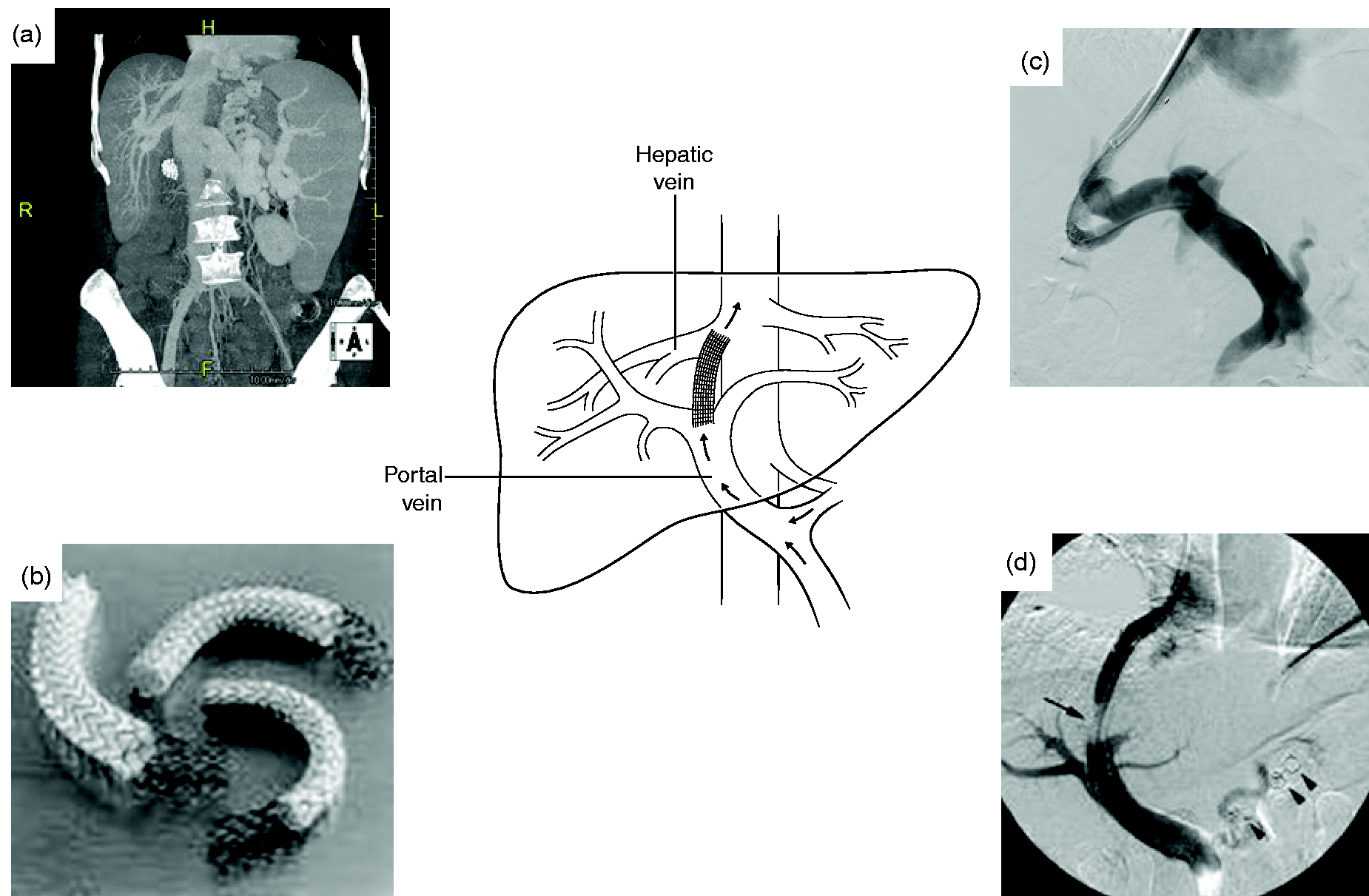
TIPS opens an artificial link between the portal and hepatic veins, shifting the blood from the splanchnic circulation into the systemic vascular bed. The aim of the procedure is to decompress the portal system and avoid the major complications of portal hypertension (Figure 3). The wide adoption of the polytetrafluoroethylene (PTFE)-covered stents significantly reduces the incidence of shunt insufficiency
17
but is unfortunately counterbalanced by the development of OHE.
18
Nevertheless, the incidence and gravity of HE are still high in the first month after TIPS placement then decrease progressively because the diameter of the shunt tends to reduce spontaneously;
18
however, they range from 10–50%.
Computed tomography (CT) scan and three-dimensional (3D) reconstruction of a large splenorenal shunt, frequently found in patients with persistent/recurrent hepatic encephalopathy (HE). Transjugular intrahepatic portosystemic shunt with polytetrafluoroethylene (PTFE)-covered placement: (a) computed tomography (CT) scan imaging of the spleno-portal vein system, (b) PTFE-covered stents, (c) and (d) angiography imaging of the procedure. Precipitant analysis of published randomized controlled trials on episodic hepatic encephalopathy treatment.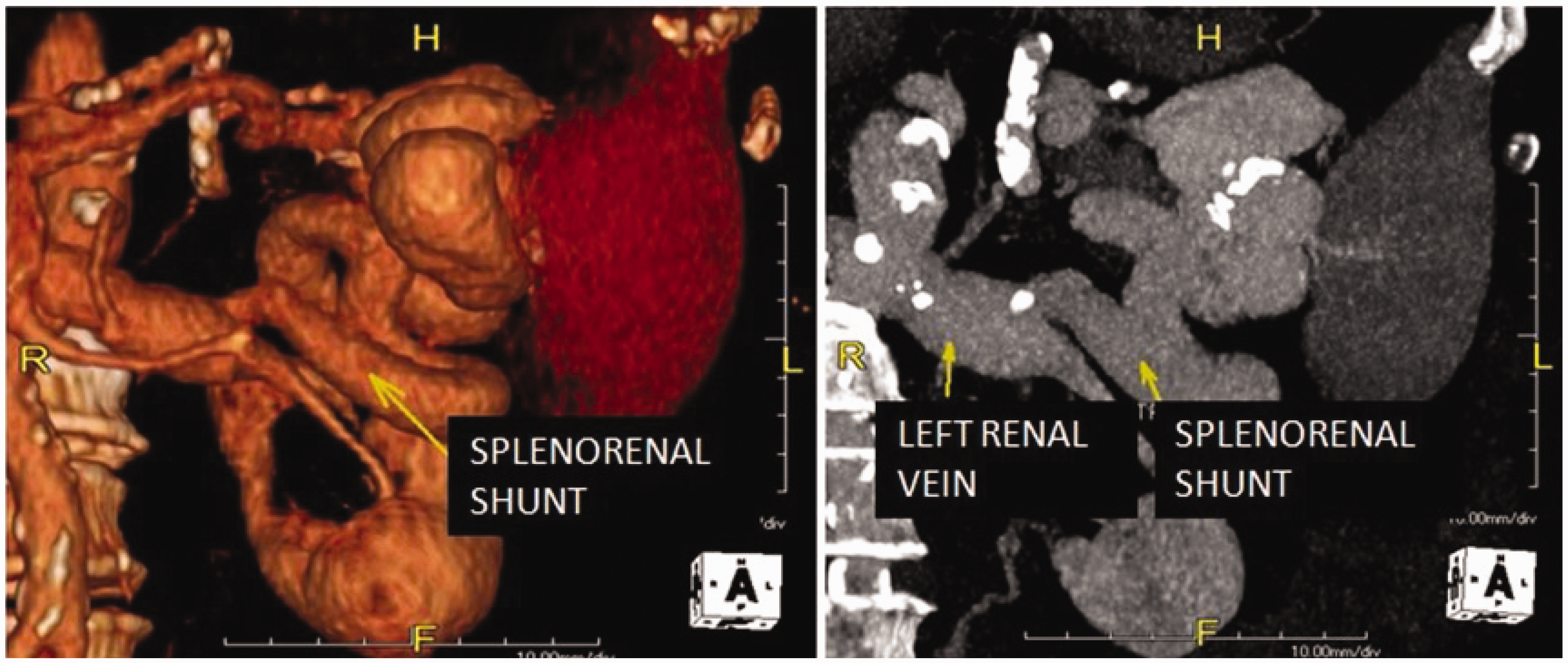
How can I diagnose the presence of HE?
Algorithm for OHE grading.11
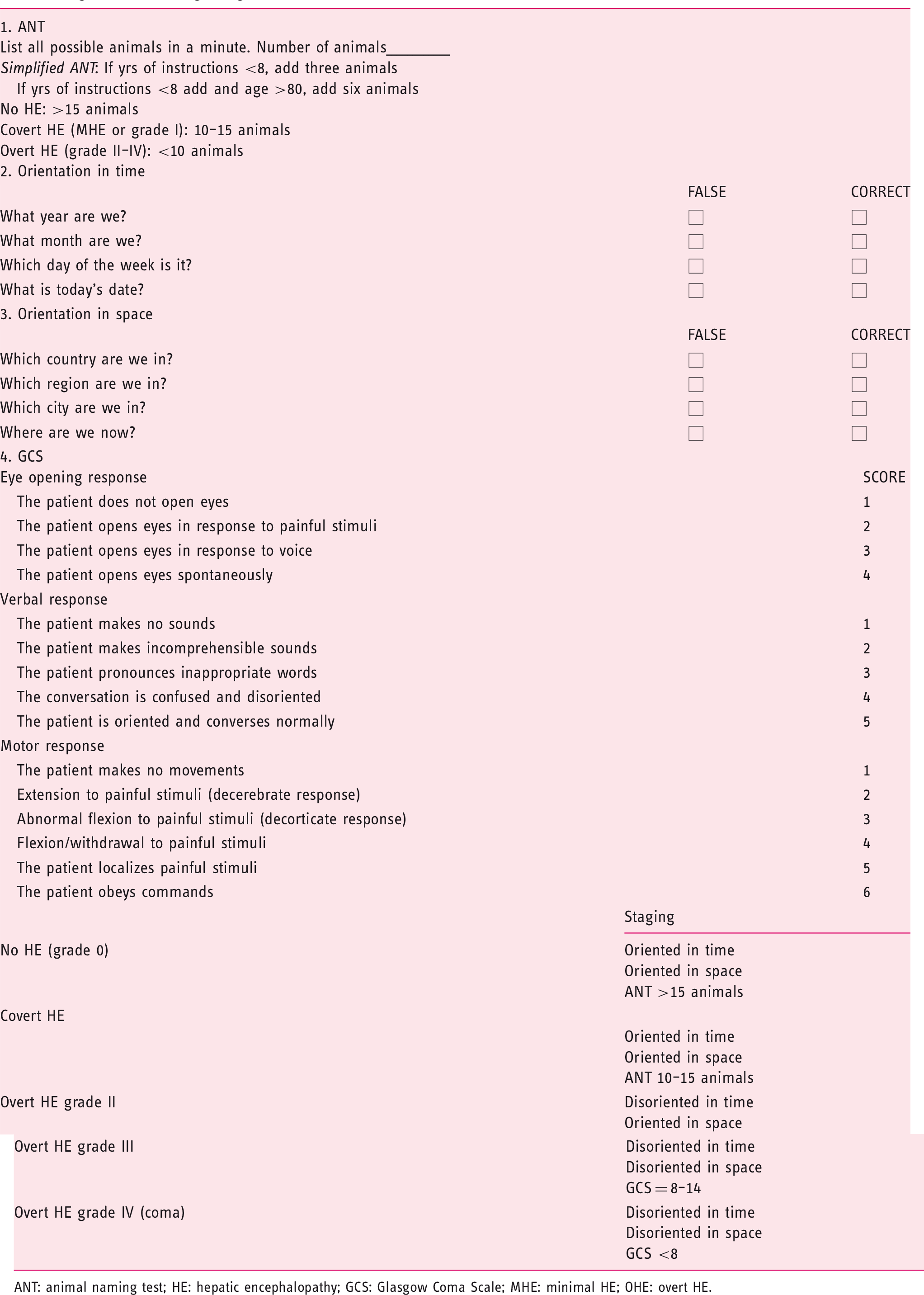
ANT: animal naming test; HE: hepatic encephalopathy; GCS: Glasgow Coma Scale; MHE: minimal HE; OHE: overt HE.
How should I treat type C OHE?
The cornerstones of managing OHE are: (a) prompt start of care of hospitalized patients with an altered mental status; (b) identify and eventually treat alternative and co-existing causes; (c) identify and correct precipitating factors; (d) start empirical ammonia-lowering treatment. Patients with grades III and IV OHE 19 at risk of not being able to protect their airways should ideally be managed in an intensive care setting. The most widely empirical pharmacological approaches are non-absorbable antibiotics (rifaximin) and non-absorbable disaccharides (lactulose, lactitol per os and enemas). Other agents (branched-chain amino acids, probiotics other antibiotics or intravenous L-ornithine L-aspartate) are available, but the evidence supporting its efficacy remains lacking.1,11 Precipitant-induced HE will benefit both from the prompt recognition and elimination of the precipitating agent, and from the specific HE therapies. In many cases it will unfortunately not be possible to understand which of the different approaches has had the decisive role. Supplementary Table 2 summarizes the results of the main RCTs published on the topic. Primary prophylaxis of OHE is not generally recommended, apart after an upper gastrointestinal bleed using drugs able to remove the blood from the gastrointestinal tract.20,21 Among the treatments proposed, rifaximin has also been shown to be as effective as lactulose in preventing OHE after and upper gastrointestinal bleed. 22 The management of gastrointestinal hemorrhage could be also improved by a sedation of patients that undergo to diagnostic and therapeutic endoscopy. The use of propofol is safe, effective and now widely used. Secondary prophylaxis of HE represents a real therapeutic challenge and means the likelihood of further episodes is high after the first bout of OHE. Secondary OHE prophylaxis should start with non-absorbable disaccharides.1,11 The overuse of lactulose may be dangerous and lead to complications such as dehydration, which can precipitate new bouts of HE. If OHE becomes recurrent, the addition of the non-absorbable antibiotic rifaximin has been demonstrated to be useful and safe in maintaining remission. 23 To date, there is scarce evidence on the role of a pharmacological prophylaxis of HE in TIPS carriers; 24 only Bureau et al. showed preliminary data about the role of rifaximin on preventing HE post TIPS placement (the abstract was presented in 2019 at the Annual Meeting of the French Society of Hepatology). The use of shunts with different diameters may be considered 25 and recently Schepis and colleagues demonstrated that under-dilation of the stent during the TIPS placement appears to be feasible and effective and is associated with lower rates of HE during follow-up. 26 Moreover, new controlled expansion of PTFE-covered stents (VCX) has been proposed with promising results. 27 Supplementary Table 3 shows the summarized results of RCTs on HE secondary prophylaxis. In the case of recurrent HE, a therapeutic option in patients that is not associated with TIPS or shunts may be fecal microbiota transplantation (FMT) because the alteration of gut microbiota, partly due to a frequent antibiotic and lactulose use, may be correct. To support this hypothesis, Bajaj et al. recently demonstrated the efficacy and safety of FMT. 28 Recurrent HE in TIPS carriers may benefit from shunt revision when a causal relationship between the shunt and HE is supposed, namely, if HE occurs soon after the TIPS, or when the procedure leads to a significant reduction of the portal-systemic gradient. The decision to revise the stent requires caution due to a possible recurrence of complications in portal hypertension (ascites or varices) after shunt reduction. 18 Recurrent or persistent HE is more frequent in patients with a splenorenal shunt. Therefore, detection of SPSS in patients with advanced liver disease is recommended to prevent, treat or identify causes of recurrent HE. Recently, new radiological techniques such as plug-assisted retrograde transvenous obliteration or coil-assisted retrograde transvenous obliteration have been proposed to manage recurrent or persistent HE. 16 At this regard, a large European multicentre study showed that embolization of SPSSs in 37 patients with chronic HE significantly improved neurological symptoms and reduced the incidence of HE after the procedure. Considering secondary parameters of success, defined as either improved autonomy, or decreased number of hospitalizations or severity of the worst HE episode after embolization, an improvement was observed in three-quarters of the patients. 29 Finally, liver transplantation remains the last therapeutic chance for persistent or highly recurrent HE and for patients with prominent HE-related motor dysfunction. 11 Therefore, it is important to adequately weight the prognostic impact of these conditions for patient on a waiting list for transplantation. This issue is still under debate and a quantitative or clinical HE parameter could be proposed to improve the available scoring systems.
Grey areas and future directions
Should MHE be treated?
Despite their subclinical nature, MHE and CHE alter daily life because of an impairment of driving skills or work performance, poor quality of life or cognitive complaints. Therefore, the indication to treat the patient may prevail. However, because of various concerns on available data and on the design of RCTs on MHE treatment, recently published guidelines state that treating MHE and CHE is not routinely recommended unless on a case-by-case basis.1,11 Supplementary Table 4 summarizes most of the studies published on the argument.
Unmet issues on the design of RCTs in HE
Designing the ‘optimal’ RCT in HE remains a debated argument. For episodic HE, a precise standard-of-care algorithm must be agreed before and monitored carefully during the study. Patients should not be enrolled into trials if the alteration of mental states persists, a placebo comparator is strongly recommended allowing an evaluation of the trial treatment as an adjuvant to standard therapy, and a strong clinical endpoint(s) should be used in multicentre trials. Trials for secondary prophylaxis for HE should be randomized and controlled by placebo. Moreover, outpatients with normal mental status and a previous history of HE should be enrolled. The sample size calculation should take into account the incidence of HE among the at-risk population and inclusion of a ‘no-treatment’ or a ‘placebo’ group is recommended. Occurrence of one or more episodes of overt HE (at least of grade II) is considered a strong primary endpoint, whereas hospitalization, survival, socioeconomic burden analysis and health-related quality of life evaluation and safety should be chosen as secondary endpoints, as treatments continue for a very long period of time. Concerning MHE, changes of psychometric performance are considered a useful tool to enroll comparable patients but should not be the main endpoint of the study. The calculation of sample size should consider clinically relevant endpoints (safety due to prolonged use, quality of life or the occurrence of OHE during the follow-up). Finally, large multicentre studies should be designed and considered in parallel with a placebo or a no-treatment arm. 30
Supplemental Material
UEG909675 Supplemental file - Supplemental material for Clinical management of type C hepatic encephalopathy
Supplemental material, UEG909675 Supplemental file for Clinical management of type C hepatic encephalopathy by Lorenzo Ridola, Oliviero Riggio, Stefania Gioia, Jessica Faccioli and Silvia Nardelli in United European Gastroenterology Journal
Footnotes
Acknowledgements
Dr Giuseppe Pelle, Interventional Radiology Unit, S.M. Goretti Hospital-Latina for the radiological images.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
Not applicable
Informed consent
Not applicable
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
