Abstract
Background
Follow-up of coeliac disease is recommended to prevent complications associated with unsuccessful treatment.
Objective
The objective of this article is to evaluate the implementation and significance of long-term follow-up.
Methods
Medical data were collected from 585 and follow-up questionnaires sent to 559 current adult coeliac disease patients diagnosed in childhood. Diagnostic features and adulthood health outcomes were compared between those with and without adulthood follow-up.
Results
Of paediatric patients, 92% were followed up 6–24 months after diagnosis. A total of 235 adults responded to the questionnaires a median of 18 years after diagnosis, and 25% of them reported regular follow-up. They were diagnosed more recently than those without follow-up (median year 2001 vs 1995, p = 0.001), being otherwise comparable at diagnosis. Those with follow-up were less often smokers (5% vs 16%, p = 0.042) and relatives of coeliac patients (48% vs 66%, p = 0.018), and more often students (48% vs 28%, p = 0.005) and type 1 diabetics (19% vs 4%, p = 0.001). Lack of follow-up was not associated with complications, ongoing symptoms, poorer general health or dietary adherence. All completely non-adherent patients were without follow-up.
Conclusions
Most coeliac disease patients diagnosed in childhood were not followed up according to recommendations in adulthood. The individual effect of this on long-term treatment outcomes varied markedly.
Key Summary
Established knowledge on this subject
Current guidelines recommend regular follow-up of coeliac disease to support patients' dietary adherence and to detect possible complications associated with unsuccessful treatment. Implementation and significance of long-term follow-up are scarcely studied, especially in originally paediatric patients after the transition to adult care.
New findings of this study
Only 25% of patients diagnosed in childhood reported follow-up of coeliac disease in adulthood. Lack of follow-up was not associated with poorer long-term treatment outcomes in general, but all patients not adhering to a gluten-free diet were without follow-up. The results support more personally tailored follow-up of coeliac disease.
Introduction
With a prevalence of approximately 1% to 2%, coeliac disease is one of the most common chronic gastrointestinal diseases.1,2 Achieving optimal treatment outcomes is therefore important both for the patients and for public health. 3 Inadequately treated coeliac disease predisposes to reduced quality of life and possibly severe long-term complications.4,5 The only approved treatment, a strict gluten-free diet, may be demanding to maintain, especially in food-related social situations and because of the expense and limited availability of appropriate products.6,7 These challenges may lead to dietary lapses, highlighting the role of health care in supporting dietary adherence and preventing complications related to unsuccessful treatment.
In children, maintenance of a gluten-free diet is usually the responsibility of parents and other caregivers. The situation changes markedly in adolescence, when patients themselves should take responsibility for the treatment. Unfortunately, this change is realised in turbulent puberty, which increases the risk of poor adherence to an already challenging diet.8–10 Hence, supporting young patients during the transfer to adult care would seem particularly important for strengthening their everyday coping with coeliac disease. 11 Although present guidelines recommend the regular monitoring of coeliac disease,12–15 there is a lack of evidence about the actual implementation and significance of the follow-up of paediatric patients into adulthood. 16
We investigated the prevalence and associated factors of regular follow-up in a large and well-defined cohort of adult coeliac disease patients diagnosed in childhood. Furthermore, the significance of follow-up for long-term treatment outcomes was evaluated.
Materials and methods
Patients and study design
The study was carried out at Tampere University and Tampere University Hospital. The hospital is a tertiary referral centre serving a catchment area of approximately one million people, including 120,000 children. The comprehensive medical data of paediatric coeliac disease patients diagnosed in 1966–2014 were collected from medical records (n = 1070) (Figure 1). Patients now in adulthood (age ≥18 years) with a biopsy-proven diagnosis were recorded (n = 585) and included in the study. After excluding individuals who had died (n = 7) or had missing contact information (n = 19), study questionnaires were sent to 559 patients (Figure 1).
17
The presence of follow-up in childhood 6–24 months, 5–10 years, and more than 10 years after diagnosis was collected from the patient records. Furthermore, the responders were divided into two groups based on the self-reported follow-up of coeliac disease in adulthood as follows: 1) regular follow-up (at least every three years) and 2) no regular follow-up. All relevant information collected from medical records and by questionnaires was compared between these groups based on adulthood follow-up.
Flowchart of the study.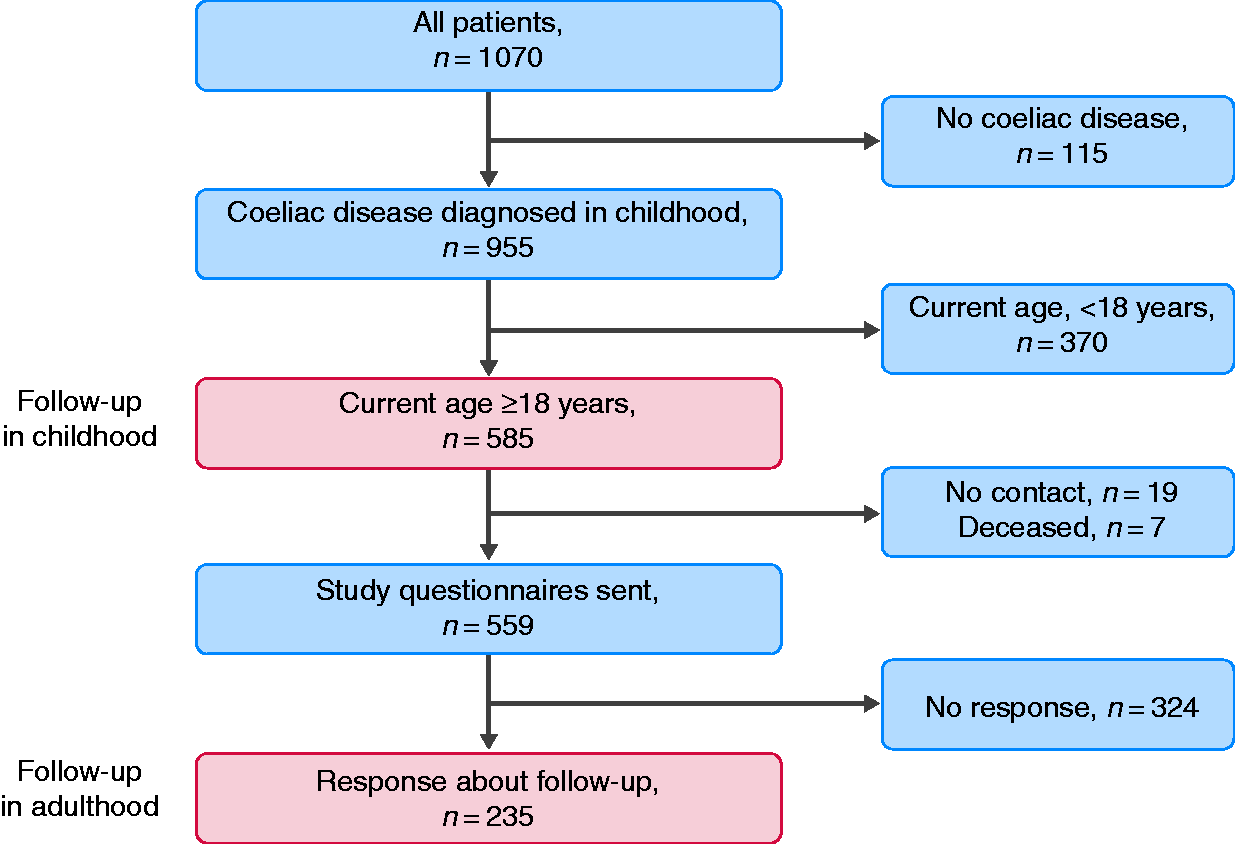
Register-based data
Clinical and histological presentation and haemoglobin and coeliac disease autoantibody values at diagnosis were collected along with adherence and response to the gluten-free diet and follow-up visits to a physician and dietician after diagnosis.
The main reason for suspicion of coeliac disease was classified as (a) gastrointestinal, such as diarrhoea and stomach pain; (b) extra-intestinal, including for example rash, arthralgia and poor growth or (c) at-risk group screening. The presence and severity of symptoms were categorised retrospectively depending on their frequency and burden to daily life as (a) no symptoms, (b) mild (minor/occasional symptoms), (c) moderate (more frequent/distracting symptoms) or (d) severe symptoms causing for example school absence or visits to an emergency room. Poor growth was defined based on Finnish nationwide recommendations. 18
Serum anti-reticulin and endomysium antibodies, which resemble each other closely, 19 have been measured in our setting since the 1980s by indirect immunofluorescence. A titre of 1:5 is considered positive, and it is further diluted up to 1:4000. 20 Blood haemoglobin values (g/l) at diagnosis were collected when available. Anaemia was defined as a haemoglobin value lower than the age- and sex-dependent reference value.
Histology results at the time of coeliac disease diagnosis were collected from the pathology reports. In our clinical routine, at least four representative samples are taken from the duodenum in each esophagogastroduodenoscopy. Before the mid-1980s, the biopsies were taken by the Watson biopsy capsule. Morphological lesion in coeliac disease is classified as partial, subtotal, or total villous atrophy. In children, follow-up biopsies are rarely needed. 21
Adherence to a gluten-free diet approximately one to two years after diagnosis was categorised as (a) strict gluten-free diet, (b) only occasional lapses and (c) non-adherence. Beneficial short-term response to treatment was defined as clinical improvement and a decrease in coeliac disease autoantibodies.
Questionnaires
In addition to follow-up, a specific study questionnaire was used to collect information about patients' current sociodemographic and lifestyle characteristics, such as employment, presence of children, family history of coeliac disease, membership in Finnish coeliac societies, smoking and physical activity. Furthermore, self-reported health and health concerns, ongoing coeliac disease-related symptoms, adherence to a gluten-free diet, and experience of daily life restrictions caused by the diet were surveyed along with the presence of coeliac disease–associated and other chronic comorbidities.
General health was categorised as (a) excellent/good or (b) moderate/poor, and health concerns as (a) none/minor or (b) moderate/severe concerns. Dietary adherence was classified as (a) strict diet, (b) occasional lapses (lapses less than once a month) or (c) frequent lapses or non-adherence (lapses monthly or more frequently). Experience of maintaining the diet was categorised as (a) easy, (b) somewhat difficult or (c) difficult.
The Gastrointestinal Symptom Rating Scale (GSRS) was used to evaluate the presence and severity of gastrointestinal symptoms. Fifteen questions are scored on a Likert scale from 1 to 7, with higher scores indicating more severe symptoms. The total score is calculated as the mean of all answers in addition to five specific subgroup scores – including diarrhoea, indigestion, constipation, pain and reflux – as means of two to four selected answers. 22
The Psychological General Well-Being questionnaire (PGWB) was used to evaluate health-related quality of life. It contains 22 questions scored from 1 to 6, with a higher score indicating better well-being and quality of life. The total score is a sum of all questions, with the range being 22–132. More detailed subgroup scores are calculated as sums of two to four relevant questions for anxiety, depression, positive well-being, self-control, general health and vitality. 23
Statistical analysis
Statistical analyses were performed with SPSS v25.0 (IBM Corp, Armonk, NY, USA). Categorical variables are reported as percentages and numeric variables as medians with lower and upper quartiles. Chi-square and Fisher tests were used to assess significance in comparisons of ordinal or nominal variables, and the Mann-Whitney test was used in comparisons of numeric variables. Binary logistic regression was used to adjust the age difference between the study groups. A p value less than 0.05 was considered significant.
Ethics
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki. The Department of Paediatrics and the Regional Ethics Committee of Tampere University Hospital approved the study design and questionnaire process (ethics committee code R16091, 31 May 2016). All participants answering the questionnaires gave their written informed consent.
Results
Follow-up in childhood
At least one guidance visit after a coeliac disease diagnosis in childhood was arranged for all 585 now-adult patients, and 92% had another follow-up visit 6–24 months later. Thereafter, based on the patient record data, follow-up decreased gradually to 26% after more than 10 years. Correspondingly, 25% of the now-adult patients responding to the study questionnaires reported regular follow-up (Figure 2).
Presence of follow-up in children diagnosed with coeliac disease (n = 585). *Self-reported, data from questionnaires (n = 235).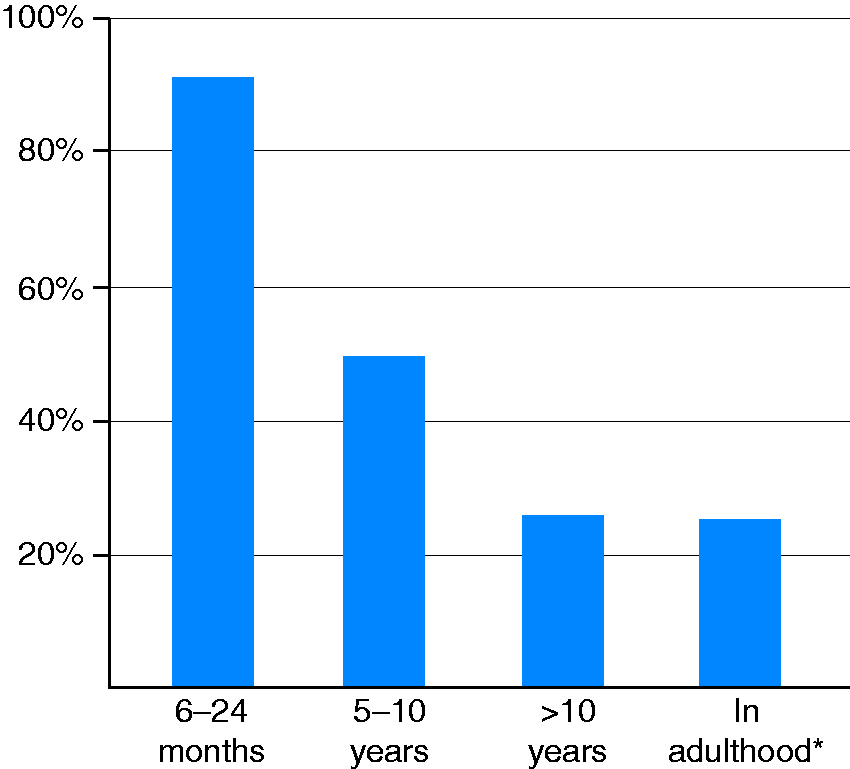
Dietary counselling at the time of diagnosis was given by a dietician in 63% and by another health-care provider in 37% of cases. At 12–24 months after the diagnosis, 85% of the patients reported a strict gluten-free diet, 12% had occasional lapses, and 3% did not adhere to the diet. Despite only partial adherence in some patients, 99% reported a beneficial dietary response.
Follow-up in adulthood
Characteristics at the time of childhood coeliac disease diagnosis in 235 responders with and without regular follow-up in adulthood and in 324 non-responders.
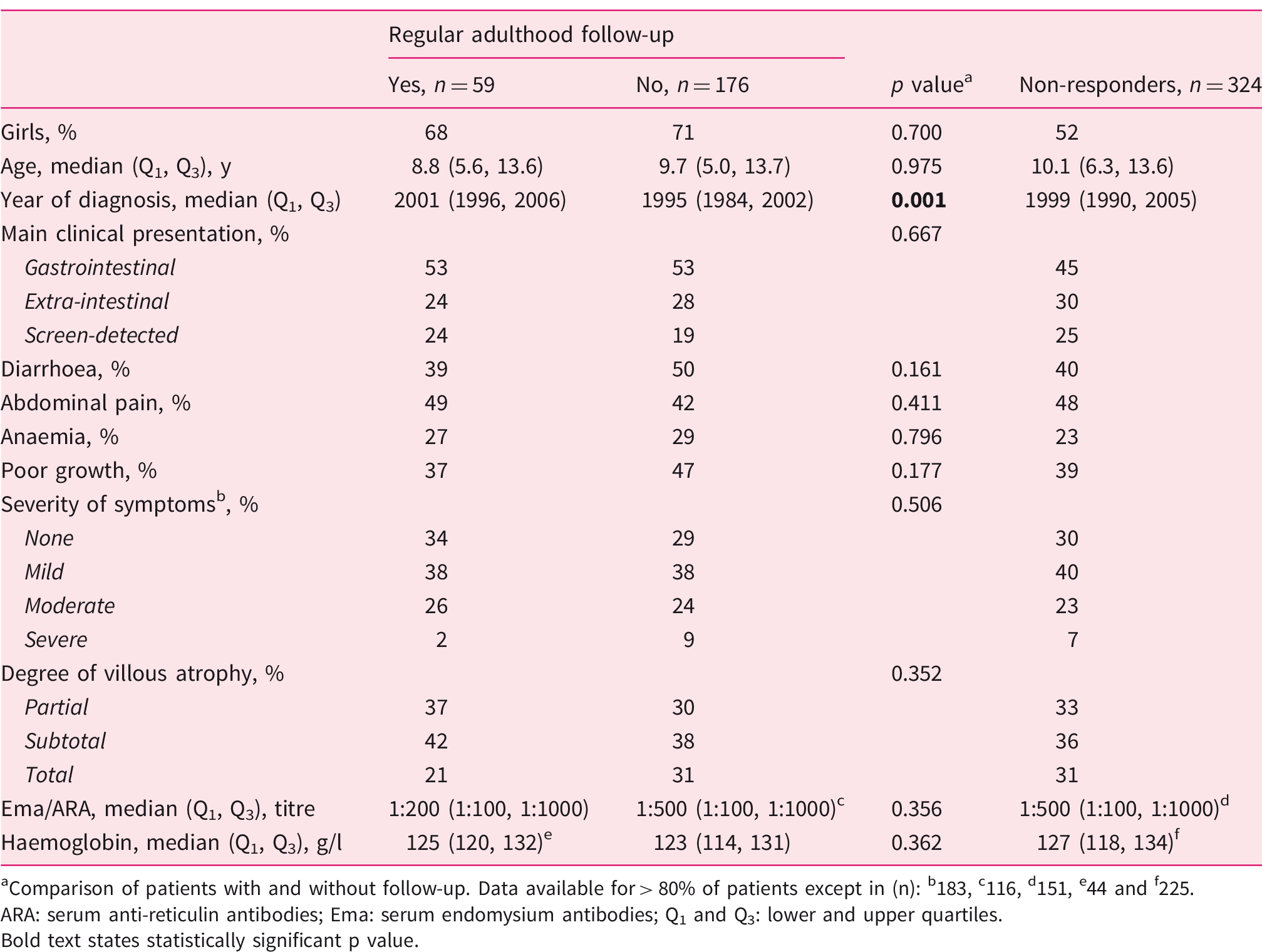
aComparison of patients with and without follow-up. Data available for > 80% of patients except in (n): b183, c116, d151, e44 and f225.
ARA: serum anti-reticulin antibodies; Ema: serum endomysium antibodies; Q1 and Q3: lower and upper quartiles. Bold text states statistically significant p value.
Among the 235 now-adults responding to the questionnaires, the childhood coeliac disease diagnosis had been established more recently in the 59 patients with regular adulthood follow-up, whereas the follow-up and no follow-up groups were comparable with respect to sex, age, clinical presentation, severity of histological damage and laboratory parameters as reported in the patient records (Table 1).
Current sociodemographic and lifestyle characteristics, self-reported health, and gluten-free diet in 235 adult coeliac disease patients diagnosed in childhood and divided into those with and without regular follow-up.
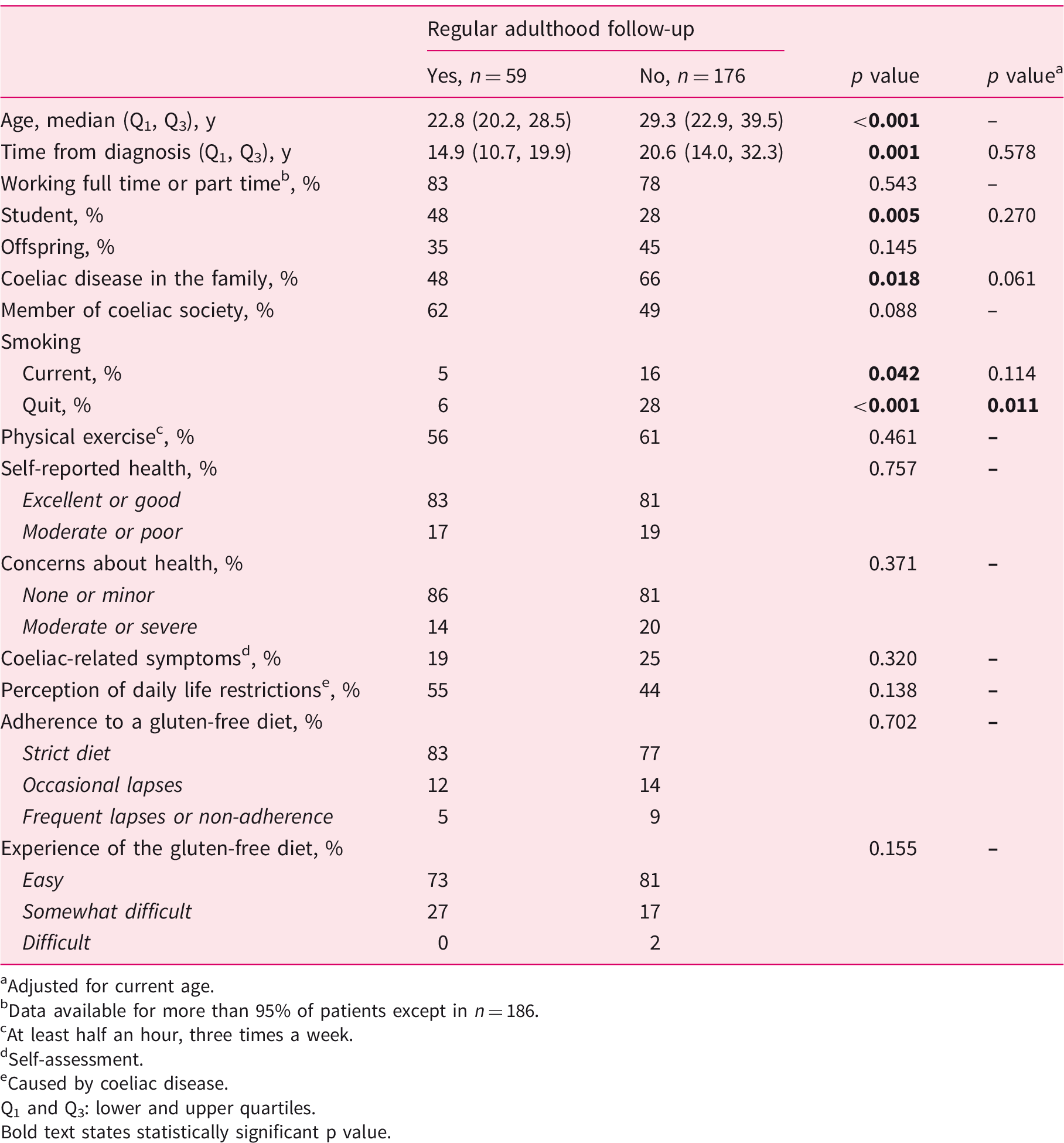
Adjusted for current age.
Data available for more than 95% of patients except in n = 186.
At least half an hour, three times a week.
Self-assessment.
Caused by coeliac disease.
Q1 and Q3: lower and upper quartiles. Bold text states statistically significant p value.
Current symptom and well-being scores in 235 adult coeliac disease patients diagnosed in childhood and divided into those with and without regular follow-up.
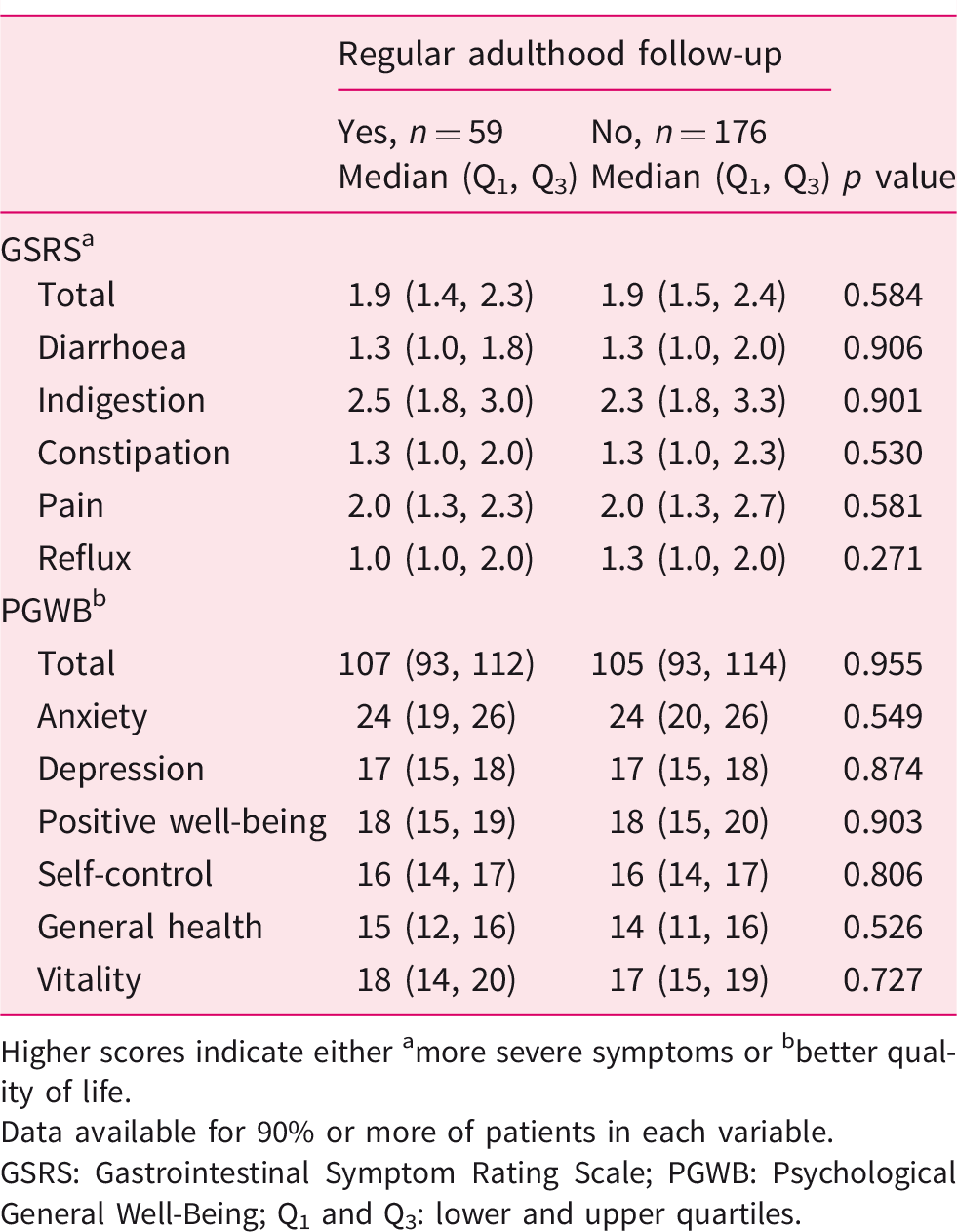
Higher scores indicate either amore severe symptoms or bbetter quality of life.
Data available for 90% or more of patients in each variable.
GSRS: Gastrointestinal Symptom Rating Scale; PGWB: Psychological General Well-Being; Q1 and Q3: lower and upper quartiles.
Comorbidities in 235 adult coeliac disease patients diagnosed in childhood and divided into those with and without regular follow-up.
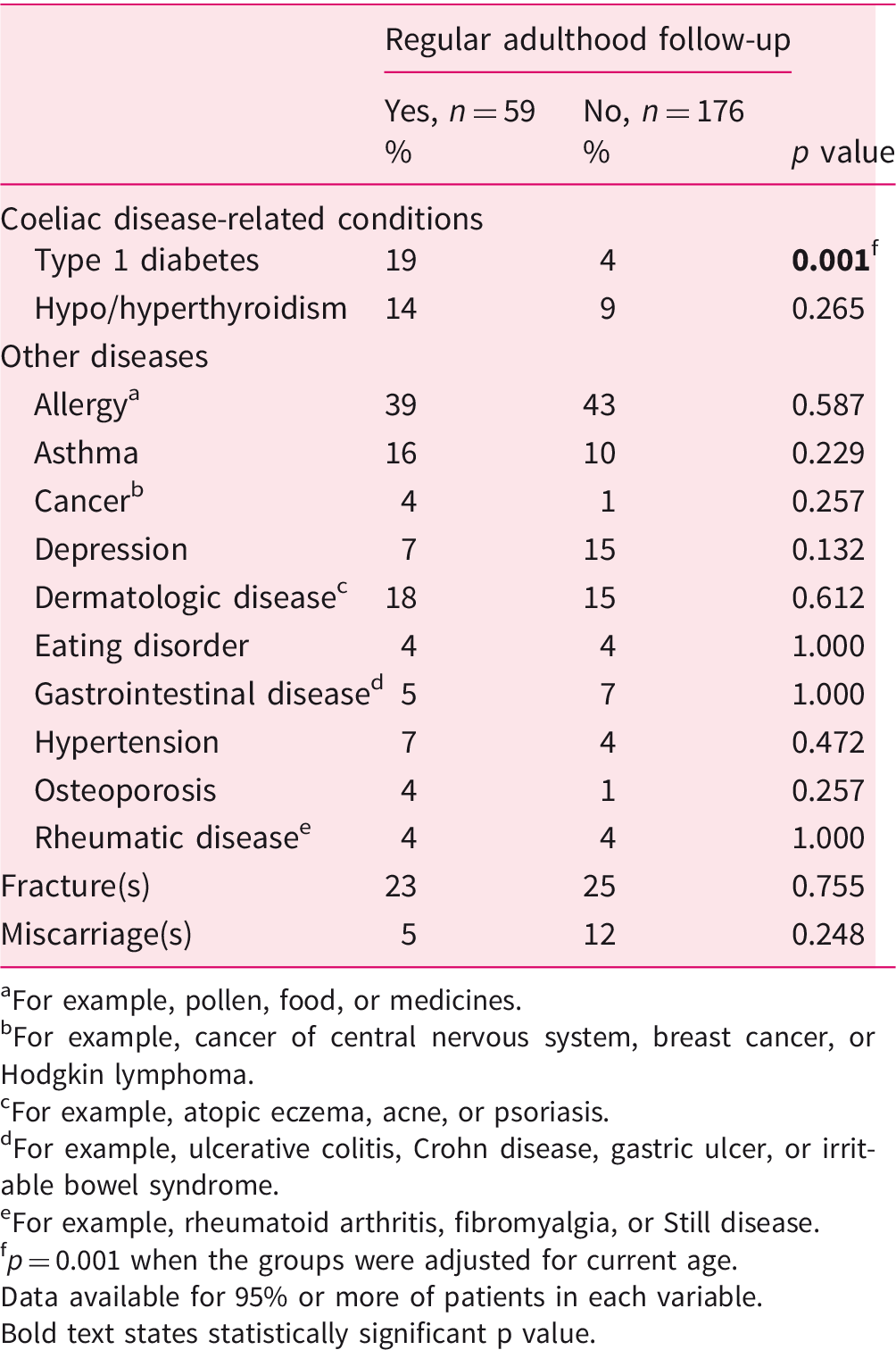
For example, pollen, food, or medicines.
For example, cancer of central nervous system, breast cancer, or Hodgkin lymphoma.
For example, atopic eczema, acne, or psoriasis.
For example, ulcerative colitis, Crohn disease, gastric ulcer, or irritable bowel syndrome.
For example, rheumatoid arthritis, fibromyalgia, or Still disease.
p = 0.001 when the groups were adjusted for current age.
Data available for 95% or more of patients in each variable. Bold text states statistically significant p value.
Subgroup analysis of 59 regularly followed-up adult coeliac disease patients diagnosed in childhood with personal visits to a doctor/nurse and other contact with health care.
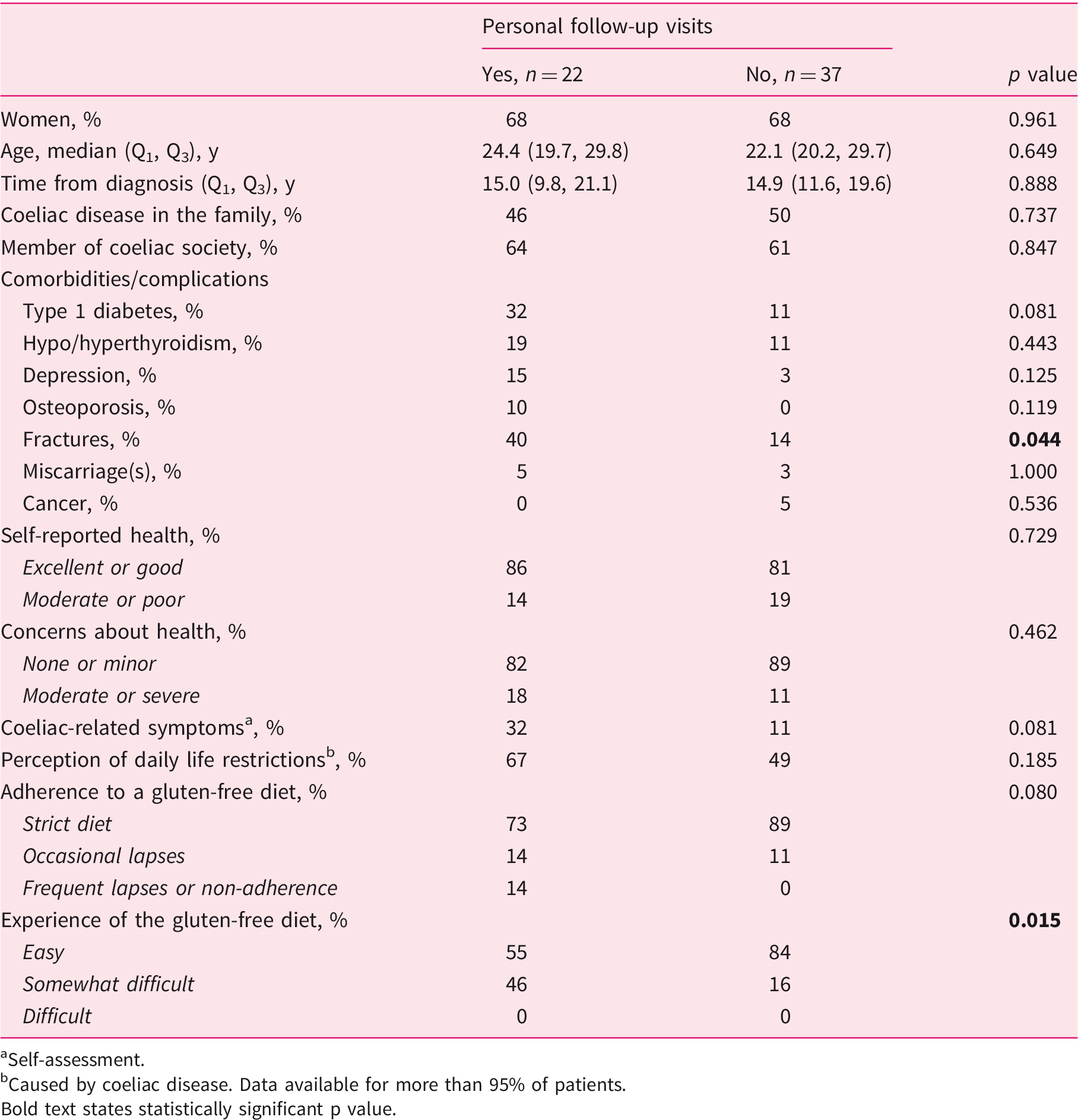
Self-assessment.
Caused by coeliac disease. Data available for more than 95% of patients. Bold text states statistically significant p value.
Discussion
We found that almost all paediatric patients received appropriate short-term monitoring of their coeliac disease, but after this early period the frequency of follow-up declined and was eventually reported only by one-fourth of the sample in adulthood. To the best of our knowledge, there are only a few previous studies on this issue. In accord with us, O'Leary et al. found only 11 (22%) out of 50 coeliac disease patients to attend follow-up after a median of 29 years from the childhood diagnosis. 24 More recently, Norsa et al. reported more positive results, with up to 83% of the patients having regular follow-up 30 years after childhood diagnosis. 25 Studies focusing on patients diagnosed in adulthood have reported long-term follow-up in 15% to 73% of their sample.26–28 Taken together, the follow-up of paediatric and adult coeliac disease does not seem to meet recommendations.12–15
Follow-up was even less frequent when only personal health-care visits were counted, and a parallel finding was reported in a US study investigating short-term follow-up mainly in adults. 28 In the present study, the patients with personal contact reported more fractures and found the gluten-free diet more difficult, despite the comparability of their dietary adherence to those with other forms of health-care contact. In the best-case scenario, follow-up is focussed on those with health issues and challenges with the treatment. Notably, although caution is needed before drawing excessively strong conclusions, the lack of direct health-care contact did not seem to affect long-term treatment outcomes negatively.
Other factors increasing the likelihood of long-term follow-up were being a student and having a more recent coeliac disease diagnosis. The Finnish Student Health Service offers low-threshold health-care services at a reasonable price for those studying at higher education institutions. 29 This easier access to health care could play a significant role in the patients' motivation to attend follow-up. In our previous adult study, most coeliac disease patients wanted regular follow-up, but did not eagerly seek it if not prearranged by their health care provider. 26 In line with this, Hughey and colleagues reported the most common reason for neglecting the follow-up to be the patients' perception that there was no strong need for it. 27 The higher frequency of follow-up in more recently diagnosed patients could be due to better organisation in recent years or because newer patients have not yet been lost to follow-up. Herman et al. 28 reported patients presenting with diarrhoea more often having short-term follow-up, but we did not observe a similar association. It is possible that the classical form of coeliac disease is considered to have a higher risk for complications and therefore the need for follow-up is more intensive. However, evidence on this is lacking; in fact, adherence problems might be even more common among those with mild or atypical presentation. 30
Remarkably, lack of long-term follow-up was associated neither with poor dietary adherence nor with major long-term health problems, such as complications. A similar lack of association between the strictness of the gluten-free diet and the presence of follow-up was observed in our previous studies with different designs.9,26 However, studies from Israel 31 and Croatia 32 have reported poorer adherence among patients not followed up. It might be the case that depending on the local circumstances, factors other than the presence of regular follow-up play a more significant role in the long-term success of the treatment, including for example the availability and prices of gluten-free products, individual knowledge about coeliac disease, and general attitudes toward the importance of a strict diet.8,9,33 Educational training of professionals, membership of coeliac societies and information provided by physicians and dieticians for patients are other factors that may affect compliance with the diet. 34 These country- and patient-related differences are important to consider before implementing our results in clinical practice.
It must be emphasised that although here the lack of follow-up had no major influence on the long-term treatment outcomes, there might be a subgroup of patients who require special attention. In particular, all the participants who reported complete non-adherence to a gluten-free diet were without follow-up, and smoking was more common among individuals who were not followed up, indicating a generally less healthy lifestyle. In fact, the proportion of such patients could be even higher because these poorly motivated individuals might also have been more prone to refuse the study. These patients should be recognised and if possible receive special support with dietary treatment, keeping in mind that long-term poor compliance predisposes them to severe complications such as osteoporosis and even intestinal lymphoma. 4 Patients diagnosed in adulthood may require particular attention because they often have long diagnostic delay, and gluten-free diet is likely less efficient to prevent the complications and comorbidities. 5
It is a challenge to predict which originally paediatric patient needs special support in adulthood and to determine how these patients can be monitored. The transition to adult care could be an opportunity to recognise those at risk for future challenges and to tailor the subsequent follow-up according to personal needs, as well as to ensure sufficient knowledge about coeliac disease before shifting the responsibility for treatment onto the adolescents themselves. 16 Thereafter, those without difficulties could be seen less frequently compared to the more problematic cases. Moreover, as monitoring methods improve, the follow-up could also be made easier, for example, by using practical fingertip tests for antibodies 35 and urine tests for the detection of gluten immunogenic peptides – even at home. 36 Health care could then be obtained when there are compliance problems or other health concerns. Because convenience seems to be an important factor affecting the motivation for follow-up,26,27 such methods could encourage patients to further engage with their own treatment.
Strengths and limitations
The main strength of the present study is the large and well-defined cohort of paediatric coeliac disease patients. We also managed to collect comprehensive adulthood follow-up data, and the use of validated questionnaires for gastrointestinal symptoms and psychological well-being enhances the comparability of the results. Furthermore, the participants represented an unselected population as opposed to selected recruitment, such as via patient organisations. On the other hand, this probably explains the moderate response rate, which may predispose to selection bias. The risk for this was reduced by the similarity of the responders and non-responders as regards the register-based data. The fact that diagnostic and short-term follow-up data were collected retrospectively and were partly incomplete, especially in classification of the severity of symptoms and some laboratory results, is another limitation. Short-term dietary adherence and treatment response were mainly reported by parents of the paediatric patients. Also, we did not have data about the causes of death in seven patients.
Conclusion
We found the long-term follow-up of paediatric coeliac disease patients does not meet current recommendations.12–15 Although in the majority of cases this does not seem to affect dietary adherence or other long-term treatment outcomes, there might be a subgroup of patients who experience significant coping challenges, as all the non-adherent adult patients were without current follow-up. These individuals, as well as those experiencing persistent symptoms or with increased risk for complications and comorbidities, should receive particular attention. Together, these findings support a more personalised approach to the long-term monitoring of paediatric coeliac disease.
Footnotes
Declaration of conflicting interests
Laura Kivelä, Katri Kaukinen and Kalle Kurppa have received personal fees for lectures from the Finnish Coeliac Society outside the submitted work and act as members of the Finnish Coeliac Society’s advisory committee. All other authors declare no conflict of interest.
Ethics approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki. The Department of Paediatrics and the Regional Ethics Committee of Tampere University Hospital approved the study design and questionnaire process (ethics committee code R16091, 31 May 2016).
Funding
This work was supported by the Foundation for Paediatric Research, the Competitive State Research Financing of the Expert Area of Tampere University Hospital, the Maire Rossi Foundation, the Maud Kuistila Foundation, the Mary and Georg Ehrnrooth Foundation, the Paolo Foundation and the Emil Aaltonen Foundation.
Informed consent
All participants answering the questionnaires gave their written informed consent.
