Abstract
Background
Coeliac disease is an autoimmune disease triggered by dietary gluten and has been associated with several conditions influencing female and male reproduction. Due to unspecific symptoms, coeliac disease can be unrecognised for years.
Objective
To estimate the prevalence of unrecognised coeliac disease among couples referred to fertility treatment.
Methods
Cross-sectional screening for coeliac disease in men and women referred to fertility treatment using IgA tissue transglutaminase antibodies as a marker of coeliac disease and small-bowel biopsies to confirm the diagnosis. Participants answered a questionnaire on gluten intake, gastrointestinal symptoms and reproductive history.
Results
A total of 893 participants (51% women) were screened and eight were coeliac disease antibody positive. Small-bowel biopsies were obtained from seven antibody positive participants and unrecognised coeliac disease was confirmed in one woman and three men, corresponding to a prevalence of 0.45% (95% confidence interval 0.12–1.14). The total prevalence, combining already diagnosed and unrecognised CD cases, was 0.63% (95% confidence interval 0.29–1.12).
Conclusion
The prevalence of unrecognised coeliac disease in a group of infertile patients was equivalent to that of the Danish general population and low compared with that observed in the majority of other screening studies of infertile patients. Surprisingly, it should be noted that more men than women had coeliac disease. This result does not support a need for routine screening among infertile patients.
Key summary
The established knowledge on this subject
Coeliac disease has been associated with infertility and adverse reproductive outcomes. It has been estimated that only 10–20% of coeliac disease affected patients have been diagnosed. Active case finding and screening of risk-groups is recommended.
What are the significant and/or new findings of this study?
This is the first cross-sectional study reporting on coeliac disease in infertile men and women in Denmark. We find a low prevalence of unrecognised coeliac disease among infertile patients. This finding does not support routine screening for coeliac disease among patients referred to fertility treatment. More men than women had coeliac disease and male infertility and coeliac disease could be an important issue for future research.
Introduction
Coeliac disease is an autoimmune disease where ingestion of gluten proteins from wheat, barley and rye induces T-cell mediated inflammation in the small bowel and an autoimmune response to self-proteins, mainly tissue-transglutaminase. 1 The classical symptoms are diarrhoea and malnutrition. 2 However, most patients suffer from mild or non-classical symptoms and, therefore, the disease often remains undetected for years.3–5 The initial diagnosis is made by serological testing, but the diagnosis can only be confirmed by a small-bowel biopsy, classifying the histological findings. 6 Coeliac disease affects up to 1% of the general population in Europe and the USA with some regional differences. 7 In Denmark, 0.18% of the population had a diagnosis of coeliac disease in 2016, with a female:male ratio of 2:1, 8 but a previous screening study reported a prevalence of 0.48% in the general Danish population, 4 indicating that unrecognised coeliac disease is frequent, and therefore active case-finding is recommended. 9 Coeliac disease has been associated with several conditions influencing female and male reproduction. Reviewing nine studies, Ozgör and Selimoğlu reported a prevalence of coeliac disease of between 0.8% and 8% among infertile women. A meta-analysis by Singh et al., including five studies, found that women with infertility had 3.5 times higher odds of having coeliac disease (95% confidence interval (CI) 1.3–9.0) compared with controls.10,11 Several studies found a higher risk of delayed menarche, early menopause, endometriosis, pregnancy loss and disturbance in male gonadal function and impotence in individuals with coeliac disease.12–17 However, other studies reported no association between coeliac disease and reproductive outcomes18,19 and not all studies found a higher prevalence of unrecognised coeliac disease when screening a population of infertile men and women. 20 Besides negative effects of malnutrition, the possible mechanisms of coeliac disease affecting reproduction are not well understood. Ludvigsson et al. reported a lower placental weight in offspring to mothers who were later diagnosed with coeliac disease compared with non-coeliac mothers. 21 In addition, studies have demonstrated that transglutaminase antibodies affect the angiogenesis in the endometrium, 22 are involved in apoptosis and delayed injury healing affecting the embryo–maternal interface after implantation.23,24 In male coeliac disease patients, Lohi et al. detected coeliac disease autoantibodies in the seminal fluid. 25 Much uncertainty still exists about the relationship between coeliac disease and infertility; however, routine screening in fertility treatment programmes has been suggested,3,26 although rarely considered in Denmark.
The aim of this study was to estimate the prevalence of unrecognised coeliac disease in a population of infertile men and women referred to a fertility treatment programme.
Materials and methods
We conducted a cross-sectional study, estimating the prevalence of unrecognised coeliac disease in a population referred to fertility treatment at two Danish public fertility clinics.
Study population
Heterosexual couples with a Danish address were considered potentially eligible. By the medical records, we identified participants who already had a confirmed diagnosis of coeliac disease (using the ICD-8 code 269.00 and ICD-10 code K90.0) and they were excluded from the study. Both the man and the woman in each couple had to consent for themselves and be able to read the Danish language. If only one of the couple consented they were included on an individual basis.
Questionnaires
At the clinic participants answered a self-administered questionnaire about gastrointestinal function (based on the Gastrointestinal Symptom Rating Scale (GSRS) 27 ), reproductive history and co-morbidity. The GSRS questionnaire is a validated questionnaire that includes 15 questions, which assess severity of GI symptoms using a seven-point Likert scale (one point ‘no problems’ to seven points ‘very severe problems’) in five domains: indigestion, diarrhoea, constipation, abdominal pain and reflux. A total GSRS score is calculated as a mean value of all five domains. The questionnaire also included the question ‘Do you avoid gluten intake in your daily diet?’ to allow identification of participants where coeliac disease antibody screening could be potentially ineffective.
Serological tests
Blood samples were analysed for IgA tissue transglutaminase antibodies (IgA TGA) using a fluoro enzyme immunoassay auto-analyser (Phadia®, ImmunoCap 250). Because IgA TGA is invalid in IgA deficiency, all blood samples were tested for total levels of IgA, and if levels were ≤ 0.80 g/l they were tested for IgG deamidated gliadin peptide antibodies (IgG DGP) as another marker for coeliac disease, also using ImmunoCap 250. The cut-off values for referral to small-bowel biopsies were IgA TGA ≥ 7 U/ml or IgG DGP ≥ 3 U/ml.
Small-bowel biopsies
We took four to five biopsies from the duodenum of each coeliac disease antibody positive patient. The ‘Marsh–Oberhuber criteria’ were used to classify the histology. A Marsh stage 3 is the classic lesions of coeliac disease with villous atrophy. 28 Participants with a Marsh stage 3 were considered as having coeliac disease and patients with a Marsh stage 1–2 with corresponding clinical symptoms of coeliac disease, identified at the clinical examination, were also considered as having coeliac disease.
Information on causes of infertility
Information on the causes of infertility identified in each couple was retrieved from the medical records after the first clinical examination. This information was categorised in ‘explained’ or ‘unexplained’ infertility based on the diagnoses in the medical records.
Statistical analysis
A description of flow of participants in the study and analyses of the baseline characteristics of the population were performed. Mean age was reported with standard deviation (SD). Prevalence proportions were calculated with 95% CIs. The reporting of this study adheres to the STROBE statement of cross-sectional studies. Analyses were performed using Stata version 14 (Stata Corporation, College Station, Texas, USA).
Results
Participants
Between January 2015 and September 2016, 1644 potential participants (822 heterosexual couples) were assessed for eligibility. Three individuals (0.18%) were identified with a coeliac disease diagnosis in their medical records and excluded with their partner, leaving 1638 eligible study participants. A total of 906 individuals (442 infertile couples and 20 women and two men without partner participation), were consecutively enrolled in the study (Figure 1). The rate of participation (55.3%) was approximately the same in both clinics.
Flow of participants in the study.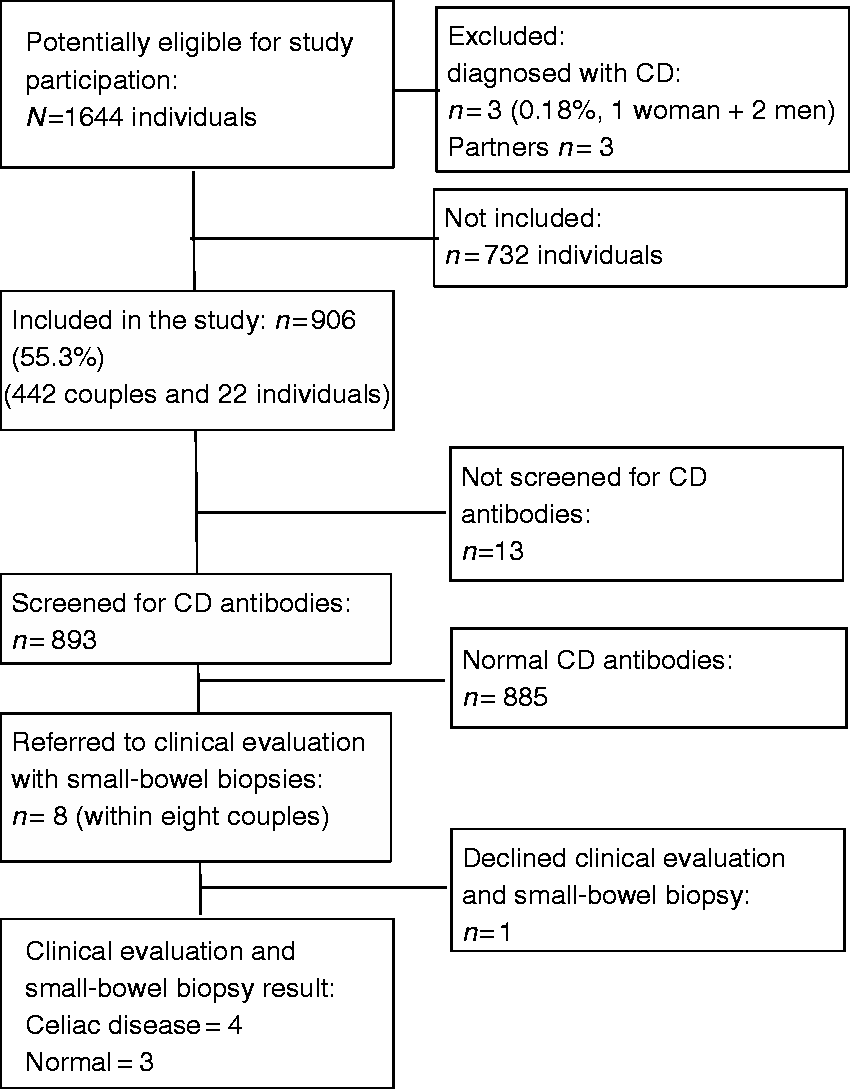
Prevalence of coeliac disease
Characteristics of participants screened for coeliac disease antibodies (IgA transglutaminase antibodies or IgG deamidated gliadin peptide antibodies) and antibody positive participants and confirmed coeliac disease, N = 893.
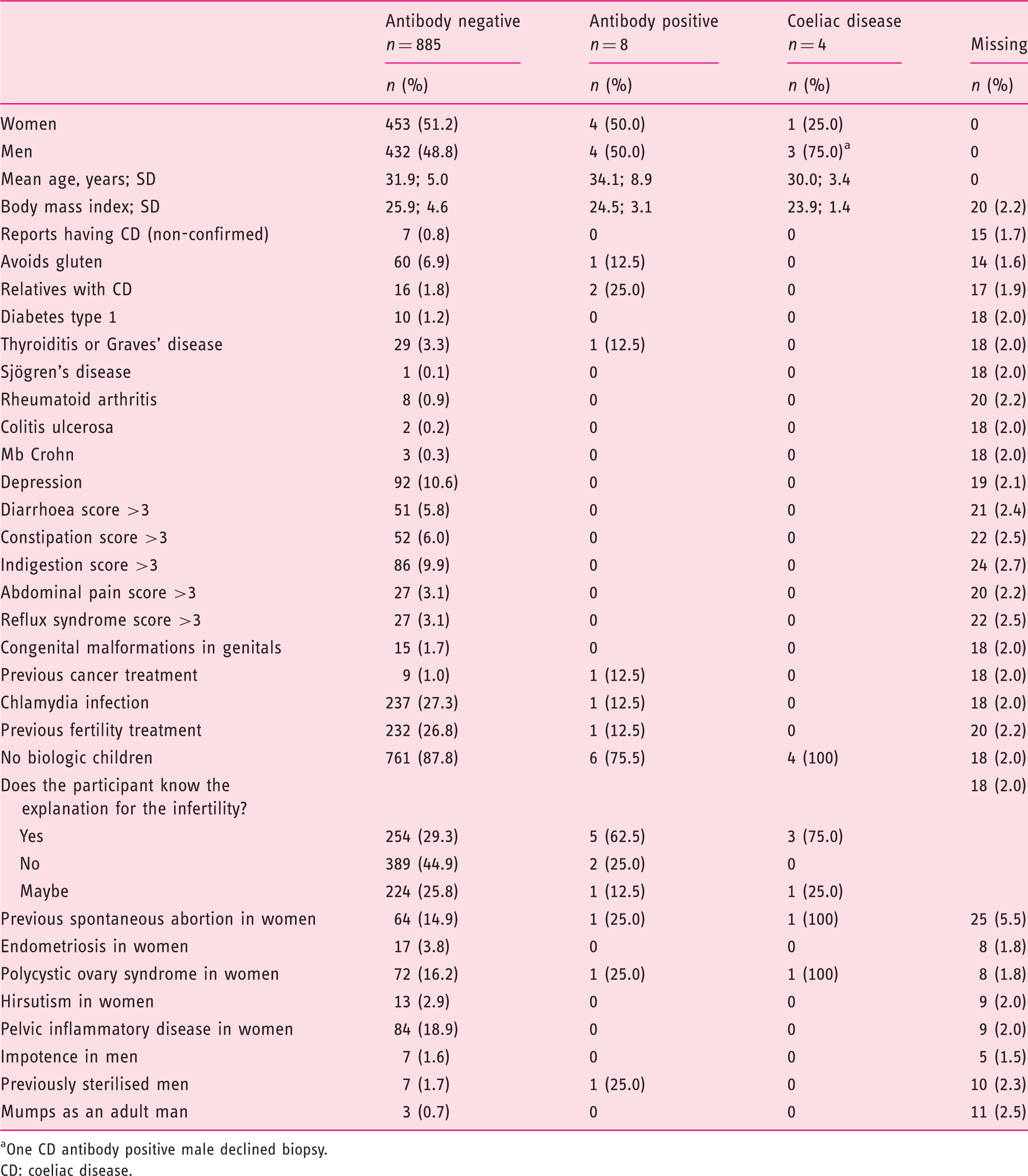
One CD antibody positive male declined biopsy.
CD: coeliac disease.
Symptoms and comorbidities
Of 879 patients both answering the question on gluten intake and being screened for antibodies, 61 patients (6.9%) reported that they were avoiding dietary gluten. One of these participants was positive for coeliac disease antibodies, but coeliac disease was not histologically confirmed. None of the eight participants with positive serology reported a GSRS score > 3 in any of the five symptom domains. Participants reporting that they avoid gluten were more likely to report gastrointestinal symptoms, with 18 out of 61 (29.5%) reporting a GSRS score >3 in at least one of the symptom domains, compared with 150 out of 818 (18.3%) participants not avoiding gluten. Among all participants, 16 (1.8%) reported that they have relatives with coeliac disease and two of them were coeliac disease antibody positive but none had histologically confirmed coeliac disease. Likewise, one coeliac disease antibody positive suffered from thyroid disease, but none of the participants diagnosed with coeliac disease suffered from any of the selected autoimmune comorbidities (Table 1).
Fertility history
Overall, 257 out of 355 couples were categorised with explained infertility (72.3%) and 98 couples had unexplained infertility (27.6%) (data were missing in 23.2% of couples). In two male coeliac disease cases the couples’ infertility was categorised as explained by a male factor and in one male case as unexplained as no cause was identified, neither in the male nor the female. In the female coeliac disease case, the infertility was categorised as explained by a male factor in the partner. This results in 1.5 fewer coeliac disease cases per 1000 subjects (95% CI 2.24–2.53) in the unexplained group compared with the explained group. None of the participants diagnosed with coeliac disease had biological children or undergone fertility treatment prior to referral to the clinic (Table 1).
Discussion
This is the first study reporting on prevalence of unrecognised coeliac disease in men and women referred to fertility treatment in Denmark and we found a histologically confirmed prevalence of 0.45% (95% CI 0.12–1.14%). The prevalence in the Danish general population has recently been reported to be 0.48% (95% CI 0.20–0.76%), including both previously unrecognised and already diagnosed coeliac disease. 4 Among the 1644 potentially eligible participants 0.18% already had a diagnosis of coeliac disease, which corresponded to the recently reported prevalence of diagnosed coeliac disease in Denmark. 8 The estimated total prevalence of coeliac disease (0.63%) among infertile men and women referred to fertility treatment was only marginally higher compared with the prevalence reported from the general population and with overlapping 95% CI. The two study populations differed in age and sex mix; therefore, caution must be applied when comparing the two prevalence estimates directly. In general, clinical prevalence studies are prone to selection bias if participation in the study somehow is dependent on the outcome of interest. Large variations in the prevalence of coeliac disease in the general populations in different western countries have been reported with a range of from 0.2% up to 2.0%, depending on diagnostic criteria, age and sex distribution. 29 The prevalence reported from the general population in Denmark seems to be in the lower range and this must be expected to have a relative influence on the prevalence of coeliac disease in subgroups of the general population.
The prevalence of unrecognised coeliac disease observed in this study is lower than in the majority of comparable screening studies performed in infertile populations. In an Italian study by Tiboni et al., the prevalence was 2.0% of previously unrecognised coeliac disease among women treated in a fertility programme and 1.0% among a comparison group of known fertile women. 3 Choi et al. reported a prevalence of 2.1% (95% CI 0.8–5.4) of previously unrecognised coeliac disease among a group of infertile American women 30 and Collin et al. reported a prevalence of 2.7% of previously unrecognised coeliac disease among 150 Finnish infertile women. 31 In contrast, a large study by Esch et al. reported a low prevalence of unrecognised coeliac disease in an unselected group of 2078 infertile patients attending a fertility clinic in The Netherlands. The prevalence was 0.48% screening sera for coeliac disease antibodies, which potentially could be even lower if biopsies had been performed. 20 The low prevalence observed in our study compared with that of other studies of infertile patients could be related to several factors. Again, due to differences in the populations of patients undergoing fertility treatment in different countries and selection of participants into the studies, direct comparison of studies should be performed with caution. In Denmark, infertile couples are offered 100% reimbursement of three treatment cycles in public fertility clinics if they have 12 months of infertility and if they do not have any mutual biological children. Whether the treatment is free, partly or fully self-paid varies across the world. 32 It is likely that difference in costs leads to differences in patients undergoing treatment in the clinics. In addition, different subgroups of infertile patients may have different risks of having coeliac disease. However, as coeliac disease has been associated with a range of conditions reported as causes of infertility, such as endometriosis and disturbed male gonadal function, unrecognised coeliac disease may still contribute to the infertility as an underlying factor, even if another cause has been diagnosed. In addition, if an explanation is identified in one part of a couple, the other part may still be unexplained infertile. We screened an unselected group of infertile couples, and found no statistically significant difference in the prevalence of coeliac disease comparing the unexplained and explained group. Hence, we could not confirm the findings of previous studies, which have suggested that unrecognised coeliac disease is more prevalent in unexplained infertility compared with explained infertility. A note of caution is due here since information was missing about causes of infertility in 23.2% of couples and the number of coeliac disease cases was low. Surprisingly, the screening revealed coeliac disease in more men than women and among the already diagnosed two out of three were men. This supports some earlier findings of an association between coeliac disease and male infertility. Previously, a few studies have investigated coeliac disease and male infertility. In 1975, Baker and Read reported two cases in which male infertility resolved after taking a gluten free diet.33 In 1982, Farthing et al. found decreased sperm motility in 12 of 16 males with coeliac disease; however, sperm motility did not improve after gluten withdrawal. 34 Further studies on the effect of coeliac disease and gluten withdrawal on male gonadal function and semen quality are needed in order to draw conclusions. Moreover, there are still many unanswered questions regarding the effects of coeliac disease and gluten withdrawal on female fertility, and the identification of subgroups of infertile patients for whom screening for coeliac disease may be relevant.
In the present study, none of the eight participants with coeliac disease antibody positive serology reported a GSRS score > 3 in any of the five domain, whereas up to 9.9% of the coeliac disease antibody negative reported to have GSRS score > 3 in any of the gastrointestinal domains. This supports the findings of other studies which found that coeliac disease patients often have very mild or no gastrointestinal symptoms.5,35 Likewise, only one coeliac disease antibody positive participant suffered from any of the selected autoimmune comorbidities. This emphasises the need for antibody screening in detecting coeliac disease.
Strengths and limitations
The strengths of this study were that participants were enrolled prospectively, minimising the risk of recall bias on gastrointestinal symptoms and gluten intake at the time of screening. All of the participants were screened before embarking on infertility therapy, leaving no risk of potential influence of the administered hormonal therapy on the immune system or the gastrointestinal symptoms. 36
All eligible patients were invited for participation, but only 55.3% participated in the study. If the risk of unrecognised coeliac disease affects the participation in the study, the risk of selection bias is present. Due to Danish legislation regarding protection of personal data, it was not possible to collect any further data from non-participants and therefore we could not compare participants and non-participants in order to estimate the risk of such bias. Selection bias may have contributed to an overestimation of the prevalence of coeliac disease if individuals experiencing symptoms indicating coeliac disease were more likely to participate. On the other hand, patients experiencing fatigue as a symptom of unrecognised coeliac disease may more often decline participation, leading to an underestimation of the prevalence. However, other studies found that patients with unrecognised coeliac disease often are asymptomatic,3–5 diminishing the possibility that participation was dependent on coeliac disease-status. Still, we cannot reject that selection bias could have affected the prevalence estimate.
As 61 participants reported that they avoided gluten and this could cause false negative coeliac disease antibody serology, this could lead to an underestimation of the prevalence. 6 With a specificity of more than 95% of IgA TGA among adults 6 and using the IgG DGP in IgA deficit, the initial screening method leaves only little risk of false negative tests among patients who did not avoid gluten. The sensitivity of IgA TGA is also more than 95% for the diagnosis of coeliac disease. 6 However, we were able to perform clinical examination and small-bowel biopsies in seven out of eight patients with positive IgA TGA or IgG DGP to confirm the diagnosis. One of the antibody-positive patients avoided gluten and biopsies showed normal histology, thus, we could not rule out that normality was induced by the self-prescribed gluten free diet.
A limitation of our study is the fact that we had no control group. Instead, we compared with results from a Danish study from 2015, screening a sample of the general population. Importantly, participants in that sample were older and their fertility status was unknown, therefore, caution must be applied when comparing the results directly.
Conclusion
The prevalence of unrecognised coeliac disease estimated by screening among men and women attending fertility treatment was low compared with previously reported levels. Although a prevalence of 0.45% of unrecognised coeliac disease was revealed, our findings do not support the idea of routine screening for coeliac disease in all patients presenting in a fertility clinic. Surprisingly, more men than women had coeliac disease, therefore some uncertainty still exists, and male infertility and coeliac disease would be an important issue for future research. The total prevalence estimate was not different from that observed in the Danish general population, reported in a previous study. However, it is important to bear in mind the possibility of selection bias and the wide confidence intervals, especially when comparing with other studies.
Footnotes
Acknowledgements
We thank the Fertility clinics and Department of Biochemistry in Horsens and Skive Regional Hospitals for their valuable assistance. We specially wish to thank project nurse Lisbeth Orth, project nurse Alice Toft Mikkelsen and laboratory technician Birgit Rasmussen.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Health Research Fund of Central Denmark Region (R38-A1465-B121), the Engell Friis Fund Denmark (19-09-2014), Boserup Fund Denmark (20795-24), Tillotts Pharma AG (04-12-2014), Aase and Ejnar Danielsens Fund Denmark (10-001478).
Ethics approval
The study was approved by the Ethics Committee of Central Denmark Region. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki (approval date: 27.11.2014 under J No. 1-10-72-321-14). The study was also approved by the Danish Data Protection Agency (J No. 1-16-02-567-14).
Informed consent
All patients gave written informed concent before study inclusion.
