Abstract
Introduction
Small rectal neuroendocrine tumours are good candidates for endoscopic resection provided that complete pathological resection (R0) is obtained and their risk of metastatic progression is low. We conducted a large multicentre nationwide study to evaluate the outcomes of the management of non-metastatic rectal neuroendocrine tumours ≤2 cm diagnosed endoscopically.
Patients and methods
The medical records, the endoscopic and pathological findings of patients with non-metastatic rectal neuroendocrine tumours ≤2 cm managed from January 2000–June 2018 in 16 French hospitals, were retrospectively analysed. The primary objective was to describe the proportion of R0 endoscopic resections.
Results
A total of 329 patients with 345 rectal neuroendocrine tumours were included, 330 (96%) tumours were managed by local treatments: 287 by endoscopy only and 43 by endoscopy followed by transanal endoscopic microsurgery. The final endoscopic R0 rate was 134/345 (39%), which improved from the first endoscopy (54/225, 24%), to the second (60/100, 60%) and the third endoscopy (20/26, 77%). R0 was associated with endoscopic technique (90% for advanced techniques, 40% for mucosectomy and 17% for polypectomy), but not with tumour or patient characteristics. Twenty patients had metastatic disease, which was associated with tumour size ≥10 mm (odds ratio: 9.1, 95% confidence interval (3.5–23.5)), tumour grade G2–G3 (odds ratio: 4.2, (1.5–11.7)), the presence of muscular (odds ratio: ∞, (11.9–∞)) and lymphovascular invasion (odds ratio: 57.2, (5.6–578.9)).
Conclusions
The resection of small rectal neuroendocrine tumours often requires multiple procedures. Training of endoscopists is necessary in order to better recognise these tumours and to perform the appropriate resection technique.
Key summary
Summarise the established knowledge on this subject
Small rectal neuroendocrine tumours (r-NETs) are good candidates for endoscopic resection. Previous reports remain limited to Asian studies where the epidemiology of r-NETs and endoscopic expertise (rate of complete pathological resection (R0)) is different to those of Western countries. What are the significant and/or new findings of this study?
In Western countries, r-NETs are not yet sufficiently recognised by endoscopists (18%). The endoscopic R0 is low 134/345 (39%) but improved with endoscopic salvage resection. R0 resection is associated with the type of technique (advanced techniques better than mucosectomy, which was better than polypectomy) but not by patient or tumour characteristics.
Introduction
Rectal neuroendocrine tumours (r-NETs) account for almost one-third of all digestive neuroendocrine tumours (NETs).
1
Most are small (≤2 cm) and diagnosed at a localised stage and are thus discovered incidentally during an endoscopic procedure performed for colorectal cancer screening or other unrelated indication.1,2 Endoscopically, these lesions are usually found as a small single submucosal nodule, generally yellowish, embedded in the rectal wall (Figure 1); r-NETs≤2 cm may be treated by local resection provided that there are no poor prognostic factors such as grade 3 tumour or lymph node (LN), muscular or lymphovascular invasion, while radical resection with LN dissection is required for r-NETs>2 cm or in the presence of the above-mentioned poor prognostic factors.2,3 Thus, the key issue is to recognise such lesions in order to propose local resection with the appropriate technique in order to obtain complete pathological resection (R0). The identification of low-risk lesions (small and grade 1 tumours, without muscular and lymphovascular invasion) is then crucial in order to determine whether local treatment (endoscopic or transanal surgery) is sufficient as it yields lower morbidity than radical surgery (proctectomy with local mesorectal excision).
Endoscopic features of rectal neuroendocrine tumour, with a submucosal appearance ((a) and (c)), a yellowish appearance (b) and after resection (d).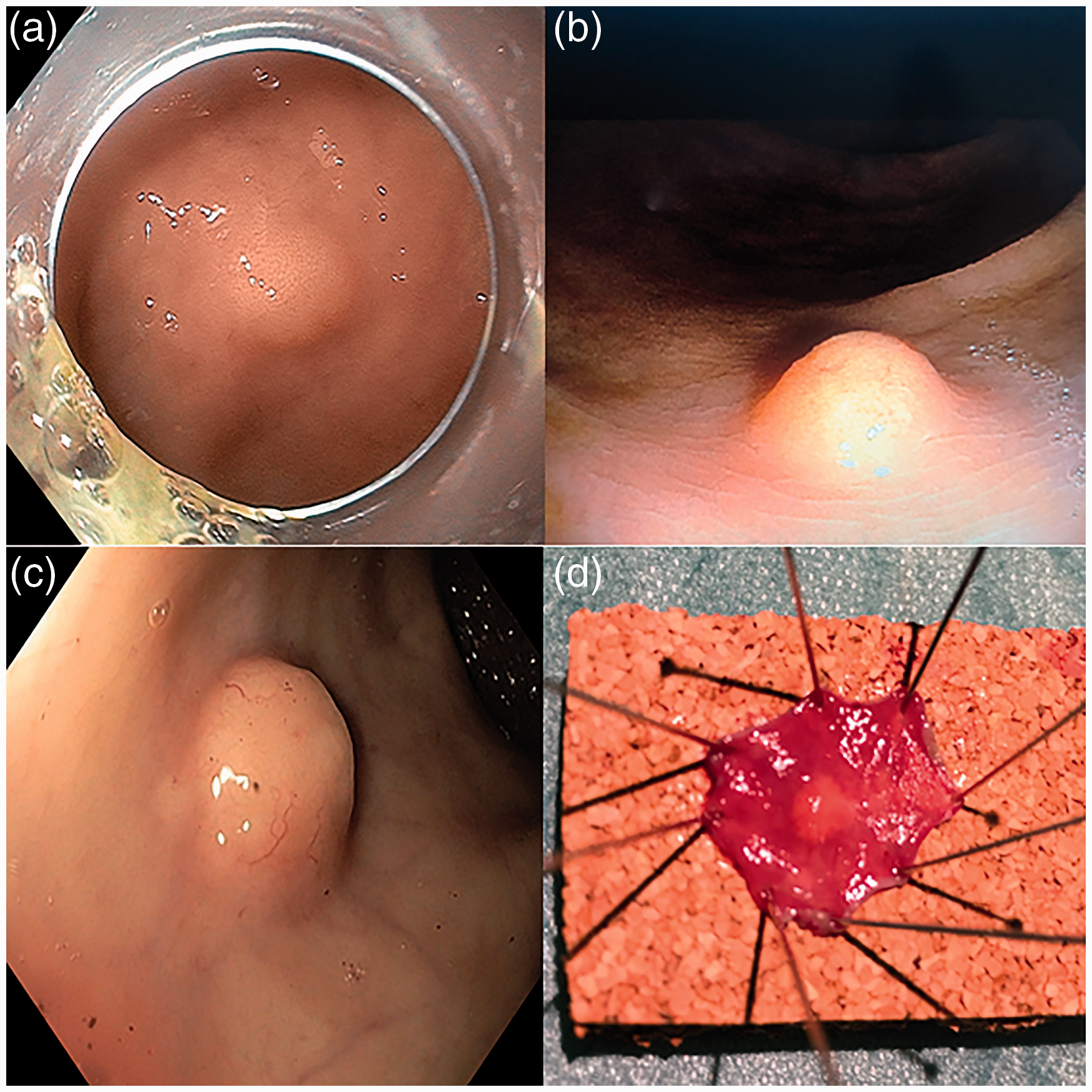
However, endoscopic resections have yet to prove their effectiveness (R0 resection) and safety; previous reports concerning the management of small r-NETs remain limited to several single-institution retrospective studies with a small number of cases, and large Asian studies where the epidemiology of r-NETs, endoscopic expertise and health services are different to those of Western countries.4–11 We therefore conducted a large multicentre nationwide study in France to evaluate the outcomes of non-metastatic r-NETs ≤2 cm in size diagnosed endoscopically. The primary objective was to describe the proportion of R0 resection. Secondary objectives were to describe the characteristics of these small tumours and their management, to identify the factors associated with incomplete resection (R1), and those associated with metastatic spreading (lymph node or distant metastasis).
Methods
Study population
Patients diagnosed from January 2000–June 2018 in 16 French hospitals with non-metastatic r-NETs of endoscopy-estimated sizes ≤2 cm were included. Size was subsequently determined using the pathology sample in case of en-bloc resection or from the endoscopic evaluation when the lesion was removed in several pieces (biopsy samples) precluding precise ulterior determination. r-NETs that were not endoscopically diagnosed were also not included, as were those found incidentally during surgery performed for another indication. Patients were recruited from the RENATEN clinical network (constructed and supported by the Groupe d’étude des tumeurs endocrines (GTE)) and the TENpath pathological network (Réseau national d’expertise pour le diagnostic anatomopathologique des tumeurs neuroendocrines de l’adulte, familiales et sporadiques). This observational study conformed to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by both the Medical Ethics Research Committee and the National Data Protection Commission on 6 November 2015 (Commission nationale de l’informatique et des libertés (CNIL)). Written information was given to each patient included in the study. Their consent is not required by French law, but patients are informed about their right to withdraw their data from the cohort.
Data collection
Medical records were retrospectively reviewed to collect data regarding tumour management (endoscopic or surgical), pathological findings and clinical outcomes. Data collected included: clinical data (age, sex, symptoms); initial endoscopic characteristics (tumour size, localization, presence of ulceration, whether the diagnosis of NET was suspected or not); type of endoscopy resection and/or of surgical procedures (transanal endoscopic microsurgery (TEM) or radical surgery); and any procedure-related complications and their treatment (endoscopy, surgery).
Endoscopic procedures
The type of endoscopic resection was described as follows: polypectomy was defined as a lesion completely removed with biopsy forceps, cold snaring or hot snare resection using a diathermy current but without submucosal injection; endoscopic mucosal resection (EMR) when a submucosal injection was used before resection using hot snare; advanced endoscopic techniques included endoscopic mucosal resection with cap aspiration (EMR-C), endoscopic submucosal resection using a band ligation device (EMR-L), endoscopic submucosal dissection (ESD), and endoscopic full-thickness resection (EFTR) using the full-thickness resection device (FTRD; Ovesco, Tuebingen, Germany).
All types of complications requiring a new procedure or a prolonged/new hospitalization, and their treatment were described, especially the two main procedure-related complications: bleeding related to endoscopy was defined as rectal bleeding requiring transfusion and/or an endoscopic procedure within 14 days after the resection; endoscopic perforation was defined as a transmural wall defect of the rectum diagnosed during the procedure.
Pathological assessment
Pathological data were described by tumour size, quality of resection (piecemeal or not, R0/R1 resection), World Health Organization (WHO) classification including grading, lymphovascular and muscular invasion, and tumor, node, metastases (TNM) stage. A pathological complete resection (R0) was defined as an en-bloc resection of the lesion with both lateral and deep tumour-free margins; when the margins were involved with tumour cells or when their status could not be determined (piece meal resection, multiple biopsy removal), the resection was considered R1 with potential residual tumour on the resection bed. R0 resection could be achieved by the first endoscopic resection or during a subsequent procedure aiming to resect the scar left by the first R1 resection. In the latter case, the first resection was R1 but treatment led to a R0 resection of the tumour bed with free margins. When no residual tumour cells were found in the tumour bed, it was concluded that the resection was R0, except when a simple polypectomy (biopsy forceps or loop) was performed on the scar as, in the absence of submucosal injection, we considered that the superficiality of polypectomy sample cannot provide accuracy in these submucosal lesions.
Follow-up
Follow-up data was collected from patient medical records, eventually completed by a telephone call to the patient’s general practitioner. The last contact with the patient defined the date of the last follow-up. Local recurrence was defined by histological evidence of the tumour at the resection site on a follow-up endoscopy. Regional recurrence was defined by the presence of typical suspect lymph nodes in the mesorectum at morphological/isotopic imaging, confirmed by histology. Metastatic recurrence was defined by the metachronous occurrence of distant metastases, diagnosed on follow-up imaging.
Statistical analysis
Categorical variables are expressed as numbers and percentages and compared using the Chi-squared test or Fisher’s exact test when appropriate. Continuous variables are expressed as medians and ranges, categorised according to median values, and compared using Mann-Whitney U test. Survival was estimated using the Kaplan-Meier method. To analyse lymphovascular invasion, cases with no lymphovascular invasion were pooled with those for which this information was not reported, considering that absence of specific mention on the pathological report reflected its absence. The primary objective was to describe the proportion of endoscopic R0 resections. For the investigation of R0/R1 resection, resections of the tumour bed without residual tumour cells were not considered. To improve analysis of metastatic development we pooled cases with lymph node invasion (LN+) after surgery and patients with recurrent disease (either locoregional or metastatic). Analysis of the predictive factors of R1 resection and LN+/M+(metastases) were carried out by univariate and multivariate analysis using logistic regression after inclusion of all significant variables (p value < 0.1). Associations were expressed as odds ratio (OR) with the 95% confidence interval (CI).
Results
Patient characteristics at diagnosis
From January 2000–June 2018, a total of 329 patients with 345 non-metastatic r-NETs ≤2 cm in size were included; 13 patients had two lesions and one patient had four lesions. The median age was 55.7 years and male/female ratio=1. All r-NETs were asymptomatic and discovered incidentally during colonoscopy for various reasons. The median period of care was 2013.
Endoscopic management
The operator described an aspect of rectal lesion compatible with the diagnosis of r-NET for 85 lesions (submucosal lesion, yellowish nodule, atypical yellow polyp, lipoma), but its suspected neuroendocrine nature was indicated in the report for only 61/345 (18%) lesions. By contrast, r-NET was unsuspected for 284/345 (82%) lesions (Table 1 and Figure 2). When diagnosis was suspected: eight (13%) endoscopists did not perform a biopsy and referred the patient to an expert centre for resection or requested an additional endoscopic ultrasound (due to the sub- mucosal nature of the lesion). In addition, 35 r-NETs that were initially suspected underwent upfront endoscopic resection, among which a higher proportion of EMR (n = 10, 29%) or advanced techniques (n = 5, 14%) was performed than for unsuspected r-NETs. Indeed, when the diagnosis was not suspected: only 4/284 (1%) tumours were not biopsied nor resected and the 190 resected tumours were mostly removed by polypectomy (n = 148, 78%).
Flow-chart of endoscopic management. Tumour characteristics at diagnosis (n = 345). G: tumour grading; NR: not reported; r-NET: rectal neuroendocrine tumour.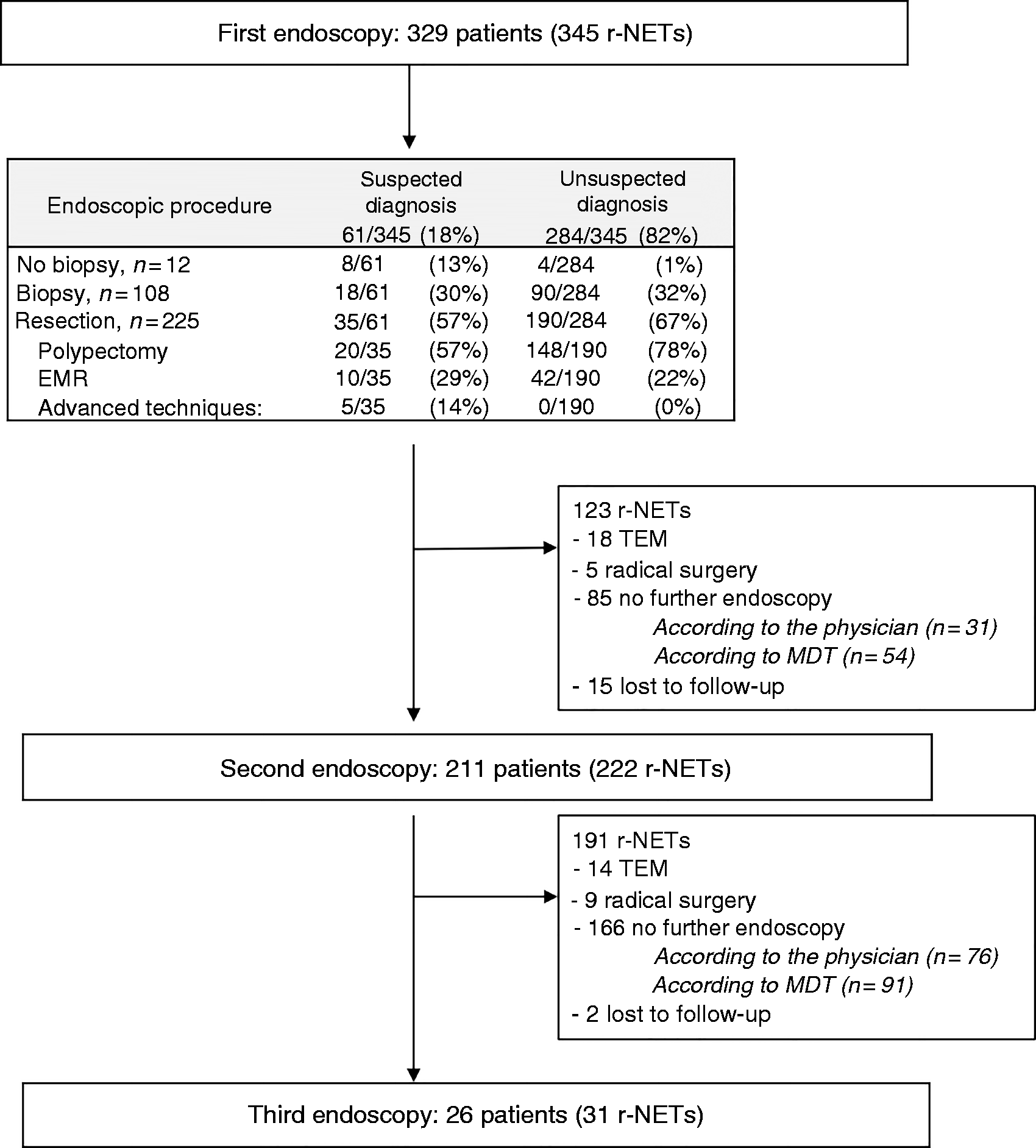

Overall, 100 salvage resection procedures were performed during a 2nd endoscopy within a median 66 (range: 1–215) days and 26 during a 3rd endoscopy within a median 71 (range: 8–420) days (Figure 2). The aspect of the mucosa during the 2nd endoscopy conditioned the endoscopic management, with more salvage resection when a residual tumour was visible (Figure 3). In total, the 329 patients underwent 598 endoscopic procedures and 351 endoscopic resections.
Management during the second endoscopy according to the appearance of the mucosa. r-NET: rectal neuroendocrine tumour.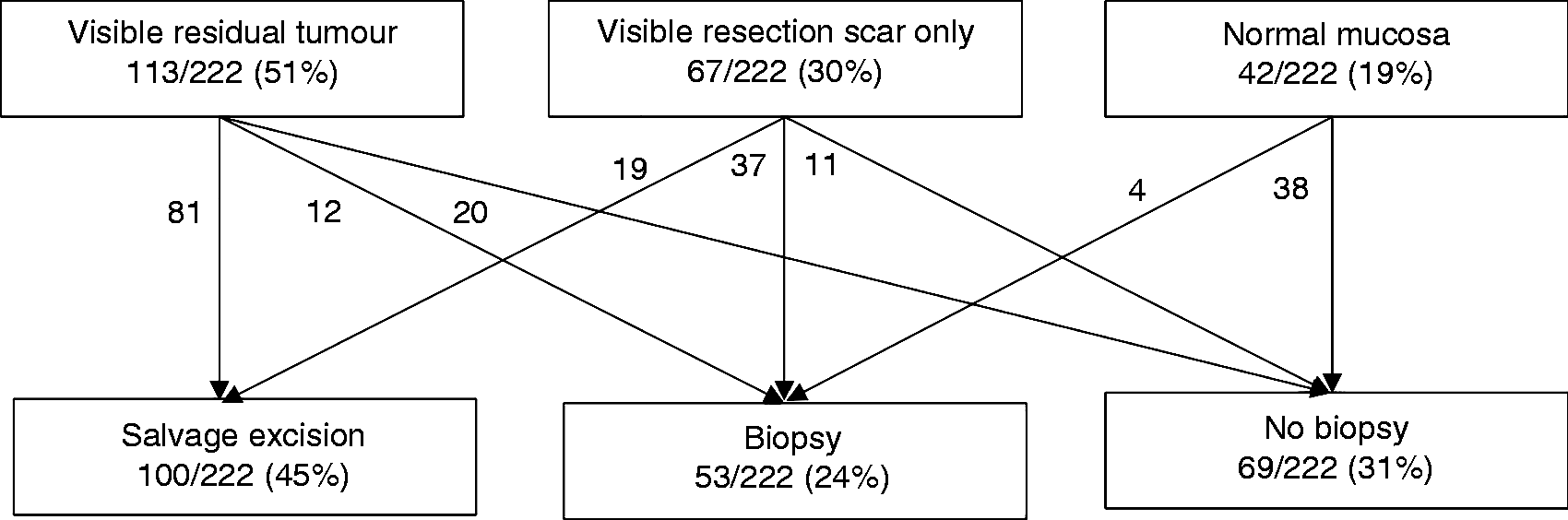
Regarding complications related to the endoscopic procedure, rectal bleeding requiring a new endoscopy occurred for two patients, who were treated by EMR with favourable outcomes. There were six cases of perforation, but all were managed endoscopically during the same procedure: three following ESD, two after EMR-C and one after EMR-L. There were no other complications.
Pathological complete resection after local treatments
Pathological microscopic complete resection (R0) according to procedure and endoscopic technique.
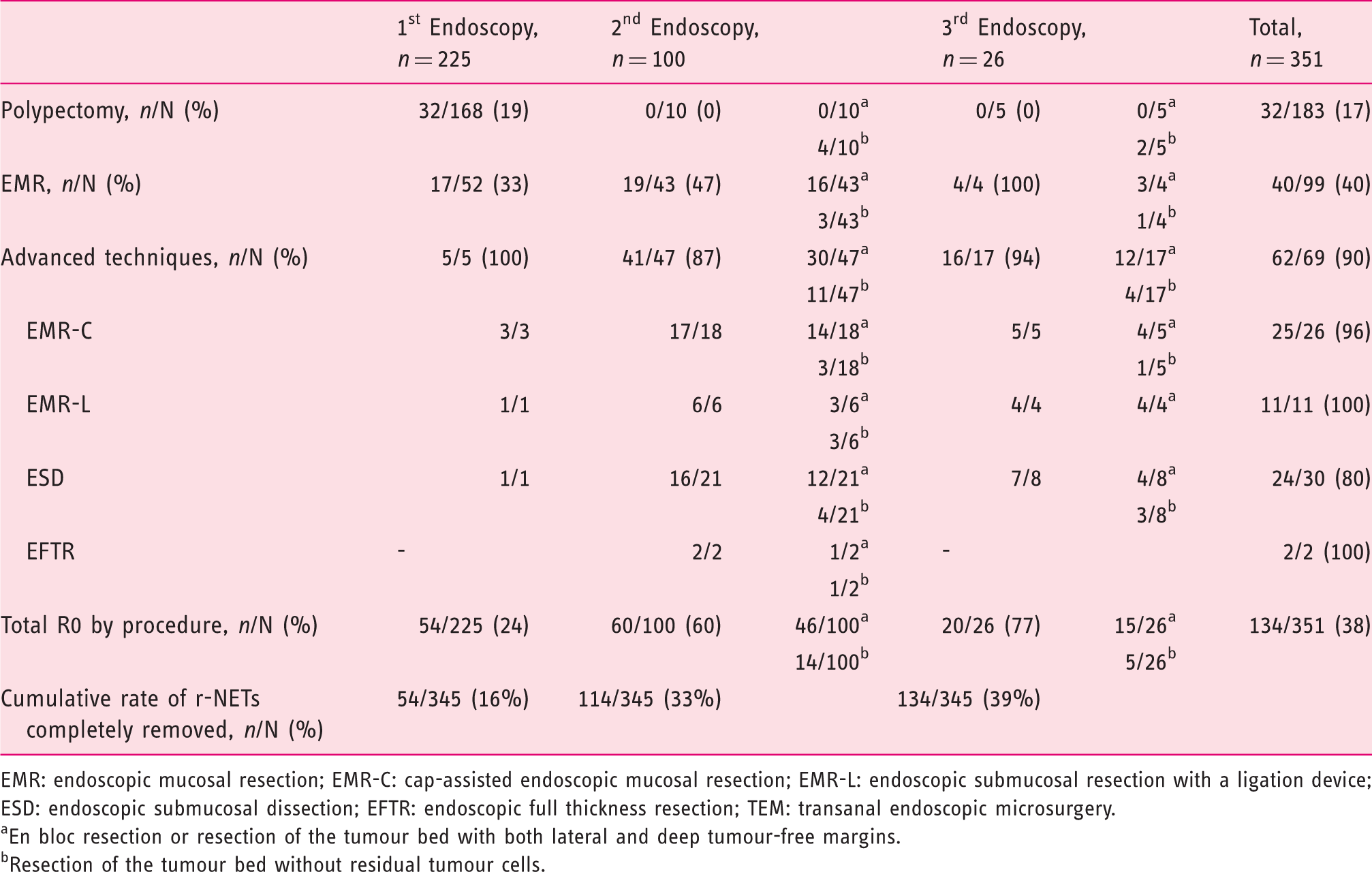
EMR: endoscopic mucosal resection; EMR-C: cap-assisted endoscopic mucosal resection; EMR-L: endoscopic submucosal resection with a ligation device; ESD: endoscopic submucosal dissection; EFTR: endoscopic full thickness resection; TEM: transanal endoscopic microsurgery.
En bloc resection or resection of the tumour bed with both lateral and deep tumour-free margins.
Resection of the tumour bed without residual tumour cells.
Forty-three of the 345 r-NETs (12%) were managed by TEM. For 28/43 (65%) of them, TEM was performed after failure of one (n = 3), two (n = 23) or three (n = 2) endoscopic resections. For 12/43 (35%) r-NETs, the patient was referred directly to the surgeon due to suspected r-NET by endoscopy or after obtaining histological diagnosis. On pathological examination, 27/43 (63%) r-NETs were R0. Eight of the 43 (19%) surgical specimens were free of residual tumour cells, which ensured that the previous endoscopic resection was complete. Eight of the 43 (19%) r-NETs were resected with invaded margins (R1).
Factors associated with involved margin resection (R1) after local procedures.
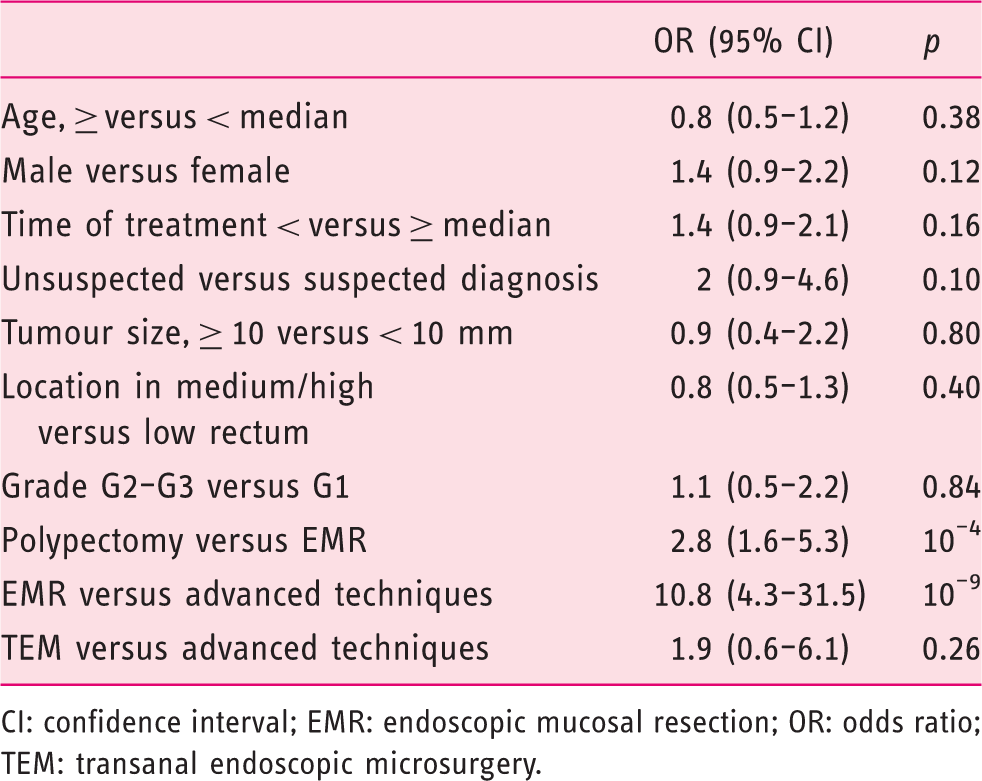
CI: confidence interval; EMR: endoscopic mucosal resection; OR: odds ratio; TEM: transanal endoscopic microsurgery.
Radical surgery
Fifteen r-NETs underwent radical surgery with lymph node dissection (14 total proctectomy with complete mesorectal excision, one abdomino-perineal excision of rectum) within a median 117 (range: 57–274) days. In this group, nine patients underwent a thoraco-abdomino-pelvis computed tomography (CT)-scan before the surgery, eight a pelvic magnetic resonance imaging (MRI), 11 a somatostatin receptor imaging (10 Octreoscan and one 68Gallium-positron emission tomography(PET)) and nine patients underwent a rectal endoscopy ultrasonography (EUS). Ten surgeries were due to suspected LN+ on the extension workup which was confirmed by pathology in all cases; five due to R1 resection after local treatment (one polypectomy, two EMR, one ESD and one TEM). Among the latter, 1/5 r-NET had pLN+ not detected on extension workup (Octreoscan and EUS) and no residual tumour cells (mucosa and lymph nodes) were found in 3/5 surgical specimens; one tumour was removed with free margins. The lymph node ratio (involved lymph nodes/ total lymph nodes excision) was 11/15 in this small group of patients. Among the eight patients with pathological LN+ and having EUS prior to surgery, LN was visible on EUS in four of them (50%). However, among these 15 patients, one had recurrence and no disease-related death occurred.
Clinicopathological data after the end of treatment
Among the 345 r-NETs diagnosed during an initial endoscopy, 15 were managed by radical surgery and 330 (96%) were managed by local treatments (287 by endoscopy only and 43 by endoscopy followed by TEM). The final pathological TNM stage was pT1aN0 for 285 (83%) r-NETs, pT1bN0 (n = 49, 14%), pT1aN1 (n = 1, 0.3%), pT1bN1 (n = 7, 2%), pT2N1 (n = 2, 0.6%), and pT3N1 (n = 1, 0.3%).
Follow-up
The median duration of follow-up was 32 months (range: 1–215). The estimated rate of disease-related overall survival at five years was 100% and at 10 years it was 94%. Sixteen of the 345 r-NETs (5%) recurred: seven locally, one regionally and eight cases with distant metastasis. Seven local recurrences occurred within a median period of 27 months (range: 10–40) after diagnosis, all measured less than 10 mm and were incompletely resected (R1); they were managed locally for five r-NETs (two TEM and three ESD) with R0 resection, one still had R1 resection and data was missing for one of them. One regional recurrence (pararectal LN+) occurred; a total proctectomy was performed which confirmed the pLN+ without residual tumour in the rectal wall. Eight metastatic recurrences occurred (seven in the liver and one in the bone) after a median period of 59 (24–132) months; 7/8 had incomplete resection (R1). The median survival of these eight patients after recurrence was 4.5 years; two of them died. Irrespective of location, the recurrence rate was 2/184 (1%) in patients who had R0 resection and 14/161 (9%) in those who had a R1 resection (p < 0.01).
Factors associated with lymph node invasion and/or metastatic disease.
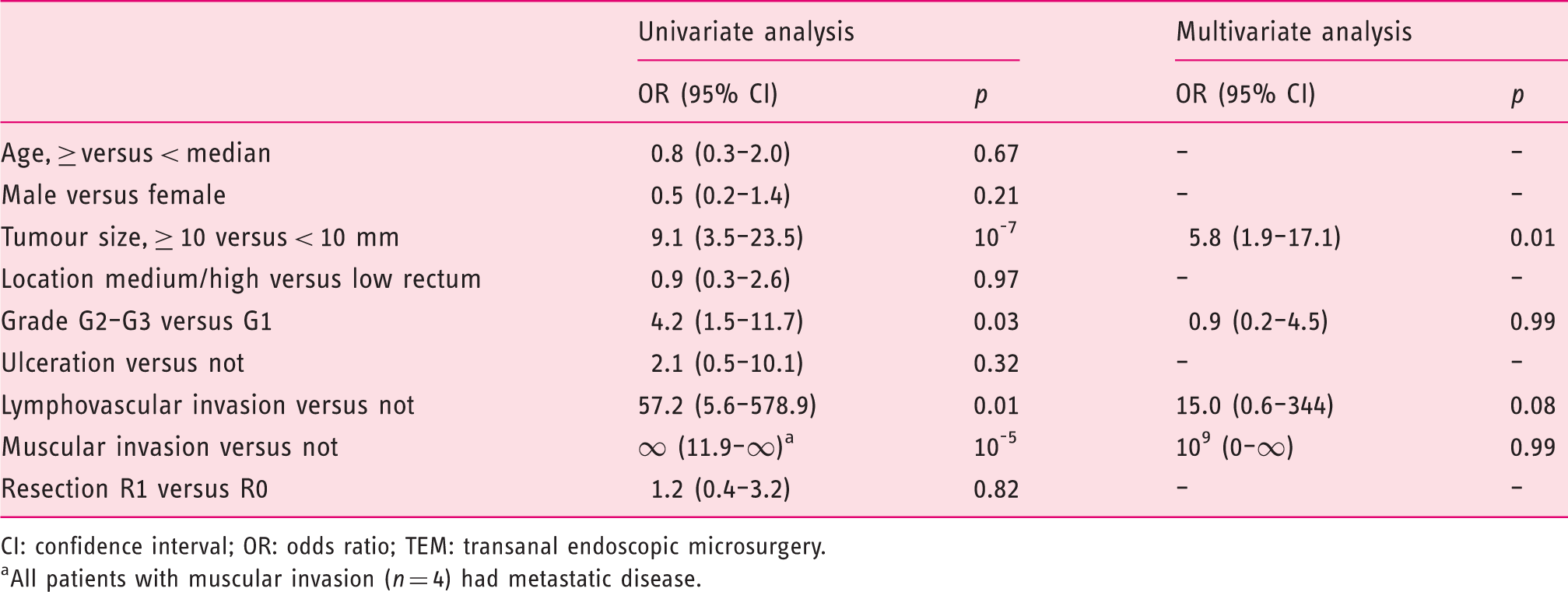
CI: confidence interval; OR: odds ratio; TEM: transanal endoscopic microsurgery.
All patients with muscular invasion (n = 4) had metastatic disease.
Discussion
To the best of our knowledge, we report here the largest multicentre cohort of patients with non-metastatic r-NETs ≤ 2 cm managed by endoscopists in Western countries. The characteristics of included patients were similar to those previously reported4,8,12 (most r-NETs are < 1 cm in size, G1 and discovered incidentally). The first important finding was the low rate of diagnosis suspected on the endoscopic aspect before any pathological finding. Secondly, R0 resection after endoscopic procedures was less frequent than previously reported.4,8,12 This improved with subsequent endoscopic salvage resections and was only influenced by endoscopic techniques (advanced technique >EMR > polypectomy) but not by patient or tumour characteristics. Thirdly, we confirmed the favourable natural history of small r-NETs as few patients had LN+ or metastatic recurrence, and the risk factors of metastatic disease were in accordance with reported data (tumour size, stage, tumour grading, lymphovascular invasion).13–20
Taken together, these results indicate that the initial issue for the treatment of these tumours is a lack of recognition of r-NETs by endoscopists and, as a result, quality resection is rarely offered as a first-line treatment; the overwhelming majority of gastroenterologists did not suspect the correct diagnosis and thus chose polypectomy in more than half of the cases. The proportion of advanced endoscopic techniques (such as EMR-C), as recommended in guidelines,1,3,18,21 was performed in only a fifth of all procedures. It is of note that Son et al. found that the identification of the submucosal feature of the lesion was associated with more frequent R0 resection (90/136, 66%) than when this aspect was not described (8/30, 27%). 8 Thus, an e-learning training programme could be interesting to improve the endoscopic recognition of r-NETs, and their submucosal presentation, in order to either opt directly for an advanced resection technique or to refer the patient to an expert centre.
We chose the proportion of R0 resection as the primary endpoint, which is a familiar endpoint for endoscopists and surgeons. However, in contrast with colorectal adenocarcinoma removed by endoscopy, 22 the true impact of R1 resection for r-NETs on both recurrence-free survival and overall survival remains unclear. For instance, R1 resection is not fully predictive of residual tumour because the destruction of the neighbouring tumour cells by cauterization during hot snare resection or EMR could sterilise the resection site.3,4,8 This is illustrated herein as residual tumour cells after removing the scar were found in only one-third of cases, and by the study reported by Park et al. who found residual tumour cells in only 10% of patients considered as R1 but whose resection appeared complete endoscopically. 4 Furthermore, true R1 resection of an r-NET has not yet been proved to be predictive of recurrence or survival;9,10,20 for instance, Kwak et al. report that two of the 31 patients (6.5%) with r-NET<10 mm only treated by excisional biopsy (without subsequent resection) had local recurrence at eight and 11 years without disease-related death. 10 To determine the true impact of R1 resection in this tumour, prospective studies with a long follow-up (more than 10–30 years) are required, especially for grade 1 r-NETs which have a very favourable natural history.1,18 This has to be kept in mind in order to not over-treat (radical surgery) patients who do not have any other factor of poor prognosis other than R1 resection, especially for elderly patients or those with several comorbidities. 20 However, even though its impact is not yet proven, we believe that, in the current context of fast progress that allows R0 resection to be obtained when patients are addressed to expert centres, we should aim for this result in all r-NET patients. This also has ramifications for the overall management of patients as once R0 is obtained there is no longer a need for follow-up.
The frequency of endoscopic R0 resection was lower than that reported in the Asian studies (59–85%), in which only the results of the first resection attempt performed in tertiary centres were reported.4,8,12 This difference may be explained by a higher proportion of upfront advanced resection techniques in these studies (10%, 4 60% 8 and 100% 12 ) than at first resection herein, as it is known that the rate of R0 is significantly higher after EMR than polypectomy, and after advanced endoscopic techniques than EMR.23–29 Taken together, French endoscopists should be trained in these techniques or refer their patients to the expert centres that perform them. Moreover, when R0 resection is not obtained after the first procedure, this leads to repeated interventions with the risk of complications and a potential source of stress for the patient, a significant economic cost, and a risk of over-treatment. We have to note that some patients herein did not undergo further endoscopy despite having R1 resection, which is not in accordance with the current guidelines.2,3 Further studies are needed as we found that recurrence was less frequent in patients who had R0 resection than those who had a R1 resection.
After endoscopic resection it is essential to identify risk factors of metastatic disease (LN+ or M+) in order to identify high-risk tumours requiring a specific work-up and surgical management with lymphadenectomy. Rectal EUS and MRI seem useful in this setting 11 even if a recent study reported that they do not accurately predict residual disease. 30 Size is an indisputable risk factor for metastatic progression,4,16,17 but with disparities between studies: herein, 8/287 (3%) of r-NETs < 10 mm showed metastatic progression; Soga found a rate of 58/595 (10%), 17 while Park et al. found no metastatic tumours among r-NET < 10 mm. 4 This can be explained in part by the difficulty in estimating the size of small tumours. Anderson et al. have shown that the estimated size in endoscopy was often imprecise and overestimated. 31 The measurement of size in histology is more precise but implies en-bloc resection; this was the case in only a few cases herein which precluded investigation of the tumour size cut-off associated with metastatic disease. We, however, confirm the value of other factors significantly associated with metastatic disease,4,13,16,17,32–34 which are the presence of lymphovascular invasion, the presence of muscular invasion and a high tumour grade. The German guidelines, 35 but not the European neureoendocrine tumor society (ENETS) guidelines, 2 reported that a Ki-Index >10% is a risk factor of LN+, as found herein. De Mestier et al. suggested a management algorithm based on these criteria and how the diagnosis is made (primary resection or primary biopsy). 3 In case of primary resection of a r-NET of < 10 mm with R0 resection and the absence of these risk factors, no further examination is indicated. It is of note, however, that while applying this algorithm to the population of the present study, there was one patient who was LN+. This underlines that not all pejorative factors have been discovered yet; additional information might come from molecular biology in the future.
Due to its retrospective and multicentre approach, the present study has some limitations. First, the indications and endoscopic expertise may differ between centres, but this design did allow the assessment of real-life management of r-NETs. Second, a centralised pathological review of r-NETs was not performed leading to missing data, for instance regarding the presence of lymphovascular invasion, which was associated with LN+ herein and therefore this relationship must be interpreted with caution. To improve this, the French national cancer institute (INCa) now recommend addressing r-NETs to the TENpath pathological network and the French Society of Pathology have also published the minimum items that must be specified on pathological reports. 36 Third, due to a median follow-up of three years, the true impact of R1 resection is difficult to assess; the very good natural history of NET-G1 with very few events requires a much longer follow-up. Despite these limitations, this is, to the best of our knowledge, the largest European study on this subject to date and reinforces the recommendations made on the management of these tumours.
In conclusion, r-NETs are not yet sufficiently recognised by endoscopists and, as a result, quality resection is rarely offered as a first-line treatment. When an advanced mucosectomy or dissection is performed frontline, R0 resection rates are much higher; this avoids repeated endoscopic examinations and long-term monitoring which is potentially a source of complications and stress for the patient, and the associated economic impact.
Footnotes
Acknowledgements
The authors wish to thank Philip Robinson (Hospices Civils de Lyon) for help in manuscript preparation.
Declaration of conflicting interests
None declared.
Funding
This study received financial support from a grant provided by the French group of endocrine tumours (Groupe d’Etude des Tumeurs Endocrines (GTE)) and the French NET patient association (Association de patients avec des tumeurs endocrines diverses (APTED)).
Ethics approval
This observational study was approved by the Medical Ethics Research Committee and the National Data Protection Commission on 6 November 2015 (Commission nationale de l’informatique et des liberte’s (CNIL)).
Informed consent
Written information was given to each patient.
