Abstract
Background and objective
The objective of this article is to externally validate and update a recently published score chart for chronic mesenteric ischemia (CMI).
Methods
A multicenter prospective cohort analysis was conducted of 666 CMI-suspected patients referred to two Dutch specialized CMI centers. Multidisciplinary consultation resulted in expert-based consensus diagnosis after which CMI consensus patients were treated. A definitive diagnosis of CMI was established if successful treatment resulted in durable symptom relief. The absolute CMI risk was calculated and discriminative ability of the original chart was assessed by the c-statistic in the validation cohort. Thereafter the original score chart was updated based on the performance in the combined original and validation cohort with inclusion of celiac artery (CA) stenosis cause.
Results
In 8% of low-risk patients, 39% of intermediate-risk patients and 94% of high-risk patients of the validation cohort, CMI was diagnosed. Discriminative ability of the original model was acceptable (c-statistic 0.79). The total score of the updated chart ranged from 0 to 28 points (low risk 19% absolute CMI risk, intermediate risk 45%, and high risk 92%). The discriminative ability of the updated chart was slightly better (c-statistic 0.80).
Conclusion
The CMI prediction model performs and discriminates well in the validation cohort. The updated score chart has excellent discriminative ability and is useful in clinical decision making.
Keywords
Key summary
What is the current knowledge?
Chronic mesenteric ischemia (CMI) is the result of insufficient mucosal perfusion of the gastrointestinal tract. The diagnosis of CMI is established by multidisciplinary consensus and confirmed if successful therapy results in symptom relief. A recently published prediction model identifies low-risk, intermediate-risk and high-risk patients for CMI. What is new here?
Validation of the previously published prediction model in a new multicenter cohort shows good performance with acceptable discriminative ability. An updated score chart has been designed with the following predictors: presence of weight loss, presence of cardiovascular disease, the degree of celiac artery (CA) stenosis combined with the cause of CA stenosis and the degree of superior mesenteric artery stenosis. The updated score chart performs well with excellent discriminative ability and defines a low-risk group with absolute CMI risk of 19%, an intermediate-risk group with a 45% CMI risk and a high-risk group with a 92% CMI risk. The updated score chart is a useful tool to be used in clinical practice to stratify CMI-suspected patients in three groups: 1. wait-and-see policy justified, 2. additional functional testing indicated, and 3. immediate vascular intervention justified.
Introduction
Chronic mesenteric ischemia (CMI) is defined as ischemic symptoms caused by insufficient blood supply of the gastrointestinal tract. 1 The most common cause is atherosclerotic stenosis of one or more supplying gastrointestinal arteries.1,2 Other common causes for mesenteric artery stenosis are vasculitis, and the most common cause of isolated celiac artery (CA) stenosis is compression of the CA by the median arcuate ligament (median arcuate ligament syndrome (MALS)). Chronic nonocclusive mesenteric ischemia (NOMI) is caused by hypoperfusion or hypooxygenation as can be seen in underlying conditions such as cardiac or pulmonary disease, shunting, microvascular occlusion, and autonomic dysfunction. 3
The exact incidence of CMI is unknown, since population-based studies are lacking. However, the number of CMI patients undergoing revascularization procedures is increasing significantly according to data from the United States (1.8 per million in 2000 to 5.6 per million in 2012 (p < 0.01)). 4 Considering the aging population and the increased prevalence of cardiovascular disease (CVD), the incidence of CMI is expected to increase in upcoming years.
Diagnosing CMI is important since untreated CMI may develop into acute-on-chronic mesenteric ischemia, which is associated with high morbidity and mortality. Since no gold-standard test for CMI is currently available, the diagnostic work-up consists of symptom assessment and radiological evaluation of the mesenteric arteries. 3 Symptoms alone are associated with a low predictive value for CMI.2,5,6 If available, a functional test to assess mucosal ischemia such as gastric-jejunal tonometry7–9 or visible light spectroscopy (VLS)10,11 can enhance diagnostic accuracy. A consensus diagnosis is established in a multidisciplinary meeting, 1 an accepted method in the absence of one specific test. 12 A definitive diagnosis of CMI is established when revascularization for occlusive mesenteric ischemia or medical therapy for chronic NOMI results in durable symptom relief.
The diagnostic work-up for CMI is cumbersome and time consuming since multiple tests are required. This exposes patients to invasive diagnostic interventions and because of the need for successive investigations may lead to a delay in treatment for patients with CMI. This underlines the need for an easy-to-use tool to promptly and reliably assess the risk of CMI in patients suspected of having this diagnosis to guide clinical decision making: 1) patients with low risk of CMI for whom a wait-and-see policy is justified to save them from unnecessary diagnostics, 2) patients with intermediate risk of CMI for whom further testing to assess mucosal oxygenation such as VLS or tonometry is indicated to establish the diagnosis and 3) patients with high risk of CMI for whom no additional test is indicated and who require intermediate vascular intervention.
Score chart for the prediction of CMI and the absolute CMI risk from Harki et al. 13
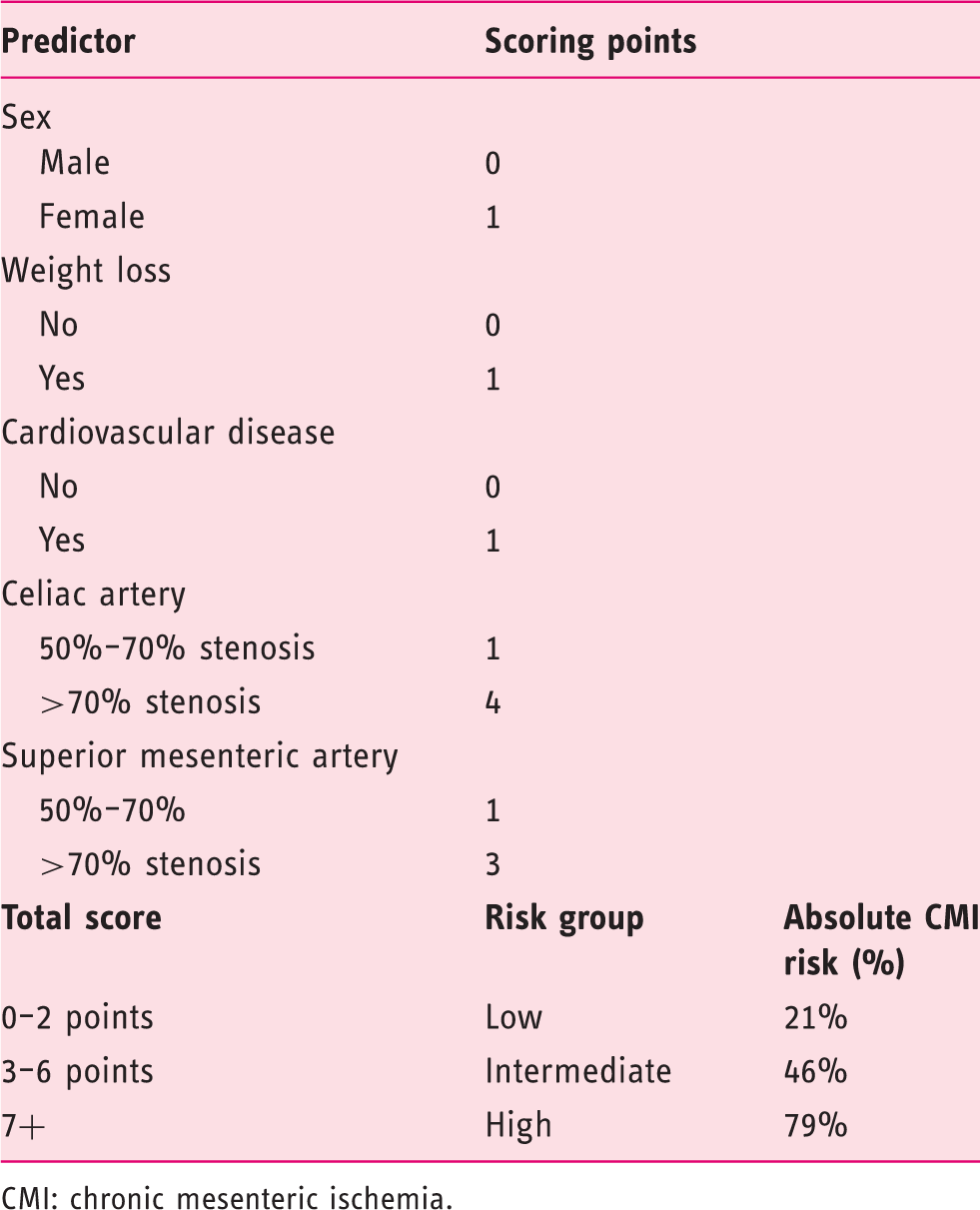
CMI: chronic mesenteric ischemia.
The current diagnostic work-up for CMI is extensive and cumbersome. A validated tool is much needed to stratify patients suspected of having CMI in distinct risk groups to guide clinical decision making. Such a tool and corresponding strategy should result in a more optimal identification of patients with CMI who may profit from treatment using tailored diagnostics and should lead to a decrease in patient burden and reduced health costs. We aimed to externally validate the previously published CMI prediction model in a new multicenter cohort. Furthermore, we aimed to update the score chart based on the performance of the model by combining the original and validation cohort and by including the CA stenosis cause.
Material and methods
Study design and setting of validation cohort
A multicenter, prospective cohort study was conducted for all consecutive patients suspected of CMI referred to two Dutch centers specializing in functional testing for mucosal ischemia: Erasmus MC University Medical Center Rotterdam (inclusion January 2014 to July 2016) and Medisch Spectrum Twente Enschede (inclusion May 2015 to January 2016). Patients were suspected of having CMI based on the criteria for the original cohort of Harki et al. 13 Patients without radiological evaluation of the mesenteric arteries with computed tomography angiography (CTA) or magnetic resonance angiography (MRA) were excluded.
The medical research ethics committee of Erasmus MC University Medical Center approved that the Medical Research Involving Human Subjects Act does not apply to this study and that no informed consent was required according to local directives (MEC-2013-317). The study complies with the Declaration of Helsinki on research ethics. To enhance transparency this article is written according to the Strengthening the Reporting of Observational Studies in Epidemiology checklist for cohort studies. 14
Data sources
All data were retrieved from the hospital records in the context of standard clinical care. Follow-up of patients was by means of outpatient clinic contact or by phone. Standard protocol visits were scheduled 1, 3, 6, 12, and 24 months after revascularization during which recurrent symptoms (i.e. similar to presenting symptoms before therapy) were assessed and body weight was documented.
Participants
All included patients underwent a standardized diagnostic work-up for CMI at baseline. This work-up consists of symptom assessment, physical examination, imaging of the mesenteric arteries with either CTA and MRA, and a functional test for mucosal ischemia detection with either 24-hour gastric-jejunal tonometry, gastric exercise tonometry or VLS.6–8,10,15–17
All cases were discussed in a multidisciplinary meeting attended by gastroenterologists, vascular surgeons and interventional radiologists, all specializing in CMI. An expert-based consensus diagnosis of CMI was established if two of the following three criteria were met: 1) typical clinical presentation of CMI (postprandial abdominal pain, weight loss, or diarrhea); 2) significant stenosis of at least the CA or SMA (≥50% diameter reduction18–21) on CTA or MRA and/or conventional catheter angiography; 3) mucosal ischemia as determined by 24-hour gastric-jejunal tonometry, gastric exercise tonometry or VLS.7,10
The degree of stenosis of the mesenteric arteries was calculated on CTA or MRA using interactive vessel segmentation software. The stenosis degree was classified as <50% stenosis, 50%–70% stenosis or ≥70% stenosis.
Definitive diagnosis of CMI
Patients with a consensus diagnosis of CMI based on occlusive disease were scheduled for either endovascular or surgical revascularization. Patients with stenosis of one or more mesenteric arteries based on vascular disease were scheduled first for endovascular revascularization: percutaneous mesenteric artery stenting (PMAS). If patients were not eligible for PMAS or if patients had recurrent episodes of restenosis, they were treated with open surgical mesenteric artery repair (OSMAR).
In patients with a CA stenosis, the diagnosis of MALS was established if CTA demonstrated focal narrowing of the proximal CA ≥50% with poststenotic dilatation and eccentric indentation on the superior aspect of the CA, creating a hook-shaped contour of the CA. This characteristic kinking dependent on the respiratory cycle in the absence of calcifications distinguishes this condition from other causes of CA stenosis such as atherosclerosis. 22 When CTA did not clarify the cause of CA stenosis, an additional catheter angiography of the CA in inspiration and expiration was performed. Patients with CA stenosis based on MALS were planned for surgical release (open or endoscopic) of the median arcuate ligament.
Patients with a consensus diagnosis of chronic NOMI were treated with vasodilatory medical therapy: oral nitrates (isosorbide mononitrate or isosorbide dinitrate). First, a low dose was prescribed (10–20 mg twice per day) and if symptoms persisted with the absence of side effects the dose was increased (to 40 mg twice per day). Nitrates were replaced by Ketanserin (selective α1-receptor antagonist) if side effects occurred and/or clinical improvement was absent, starting with 10–20 mg twice per day and increased to 40 mg twice per day.
The treated patients were evaluated during follow-up visits. A definitive diagnosis of CMI was established if the patient reported relief of initial symptoms after successful therapy. This patient-reported outcome of symptom relief was classified into two groups: no or minimal symptom relief and major or complete symptom relief. A definitive diagnosis of no CMI was established when consensus diagnosis was no CMI or when symptom relief did not occur after technically successful treatment. Patients with a consensus diagnosis of no CMI were discharged without further follow-up.
Variables
Patient characteristics that were collected included age, sex, past medical history, presenting symptoms such as abdominal pain, postprandial pain, diarrhea, nausea, weight loss (in kg, defined as >5% loss of body weight), body mass index (BMI) at presentation and cardiovascular risk factors. Vascular lesions were specified for localization (CA, SMA or inferior mesenteric artery (IMA)) and cause (vascular disease, MALS, NOMI).
The primary outcome was definitive diagnosis: CMI or no CMI. The definitive diagnosis was compared with the total score of the score chart to validate this score chart. Secondary outcome was the cause of CMI (vascular disease vs MALS).
Score chart
The score chart (Table 1) was applied for each included patient. Based on the total score each patient was classified in one of three risk groups: low-risk (0–2 points), intermediate-risk (3–6 points) and high–risk (≥7 points).
Study size
There was a requirement to include minimally 215 patients suspected of having CMI assuming a CMI diagnosis rate of 47% 13 according to literature recommendations on sample size considerations for the external validation of a multivariable prognostic model.23,24
Statistical methods
Baseline characteristics were described for the validation cohort either as numbers and percentages for dichotomous variables, or as means and standard deviations or medians and interquartile ranges (IQR) for continuous variables. Multiple imputation (10 times) was used to impute missing values of the predictors from the score chart and cause of CA stenosis. Univariable and multivariable associations were estimated with logistic regression analysis as odds ratios (ORs) with a 95% confidence interval (CI). The added value of cause of the stenosis of the CA (vascular disease vs MALS) was assessed by including the variable in the multivariable logistic regression.
Performance of the original score chart was studied by comparing the definitive diagnosis with the total score of the score chart. Discriminative ability of the score chart was assessed with the c-statistic. This measure of concordance is identical to the area under the receiver-operating characteristics (ROC) curve. C-statistic of 0.5 suggests no discrimination, c-statistic of 0.7–0.8 is considered as acceptable discrimination, c-statistic of 0.8–0.9 as excellent discrimination and c-statistic ≥0.9 as outstanding discrimination. 25 Furthermore, calibration was assessed graphically with a calibration plot: a plot with the predicted risk of CMI on the X-axis and the observed proportion of CMI on the Y-axis. After validating the score chart in the validation cohort, the data of the validation cohort was combined with the data of the original cohort described by Harki et al. 13 The original score chart was updated based on the performance of the score chart in the combined original cohort and validation cohort and with inclusion of the cause of the CA stenosis based on expert view. Scores for the updated score chart were calculated by dividing the regression coefficients of the predictors by 0.17. Calibration and discriminative ability were also tested for the updated score chart.
Results
Patient characteristics of validation cohort
Patient characteristics and presenting symptoms of validation cohort.
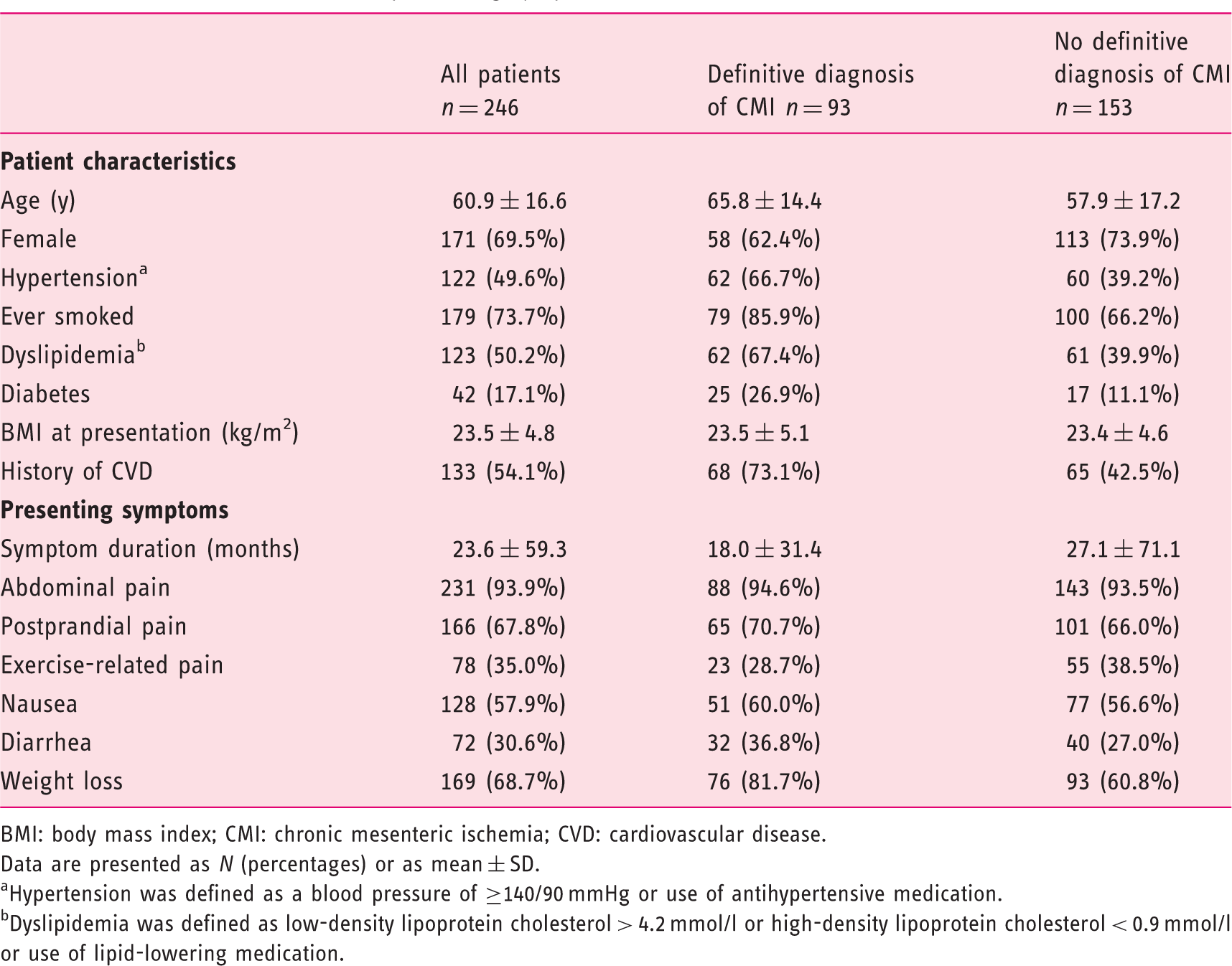
BMI: body mass index; CMI: chronic mesenteric ischemia; CVD: cardiovascular disease.
Data are presented as N (percentages) or as mean ± SD.
Hypertension was defined as a blood pressure of ≥140/90 mmHg or use of antihypertensive medication.
Dyslipidemia was defined as low-density lipoprotein cholesterol > 4.2 mmol/l or high-density lipoprotein cholesterol < 0.9 mmol/l or use of lipid-lowering medication.
Diagnosis, vascular lesion, and cause of the vascular lesion specified for each risk group according original score chart of the validation cohort.
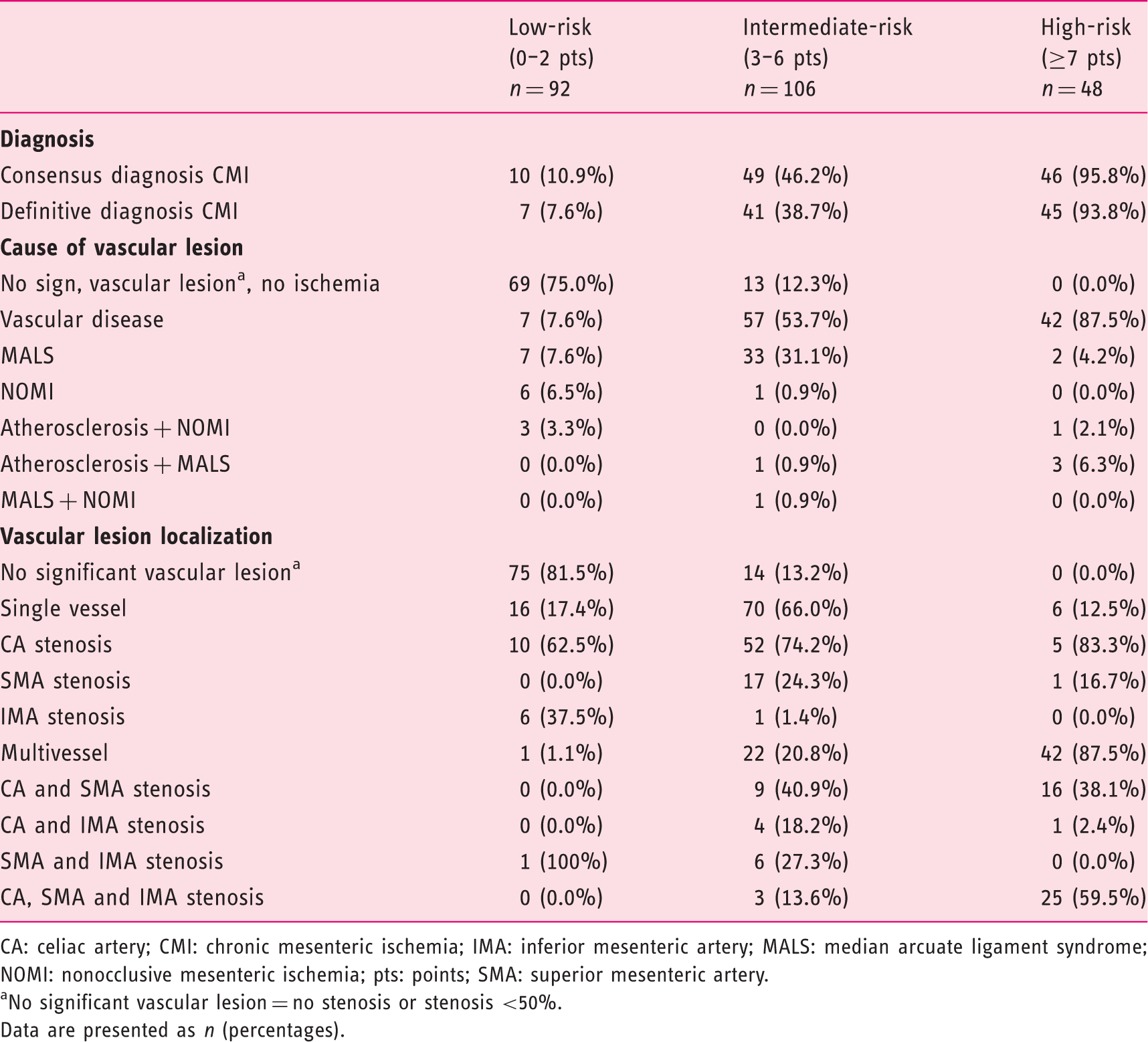
CA: celiac artery; CMI: chronic mesenteric ischemia; IMA: inferior mesenteric artery; MALS: median arcuate ligament syndrome; NOMI: nonocclusive mesenteric ischemia; pts: points; SMA: superior mesenteric artery.
No significant vascular lesion = no stenosis or stenosis <50%.
Data are presented as n (percentages).
The follow-up period was 10.5 ± 6.6 months for the patients with a consensus diagnosis of CMI and 3.7 ± 6.6 months for the patients with a consensus diagnosis of no CMI.
Performance original score chart
The original score chart performed well in the validation cohort with an absolute CMI risk of 8% in the low-risk group (original cohort: 21%
13
), 39% in the intermediate-risk group (original cohort: 46%
13
) and 94% in the high-risk group (original cohort: 79%
13
). Discriminative ability was acceptable with a c-statistic of 0.79 (original cohort: 0.79). Calibration results of the original model in the combined cohort are shown in Figure 1(a). Perfect prediction of CMI would show when all points are at 45 degrees (dashed line) because of a slope of 1 and calibration intercept of 0.
Performance of the (a) original score chart and (b) the updated score chart based on the combined cohort (n = 666).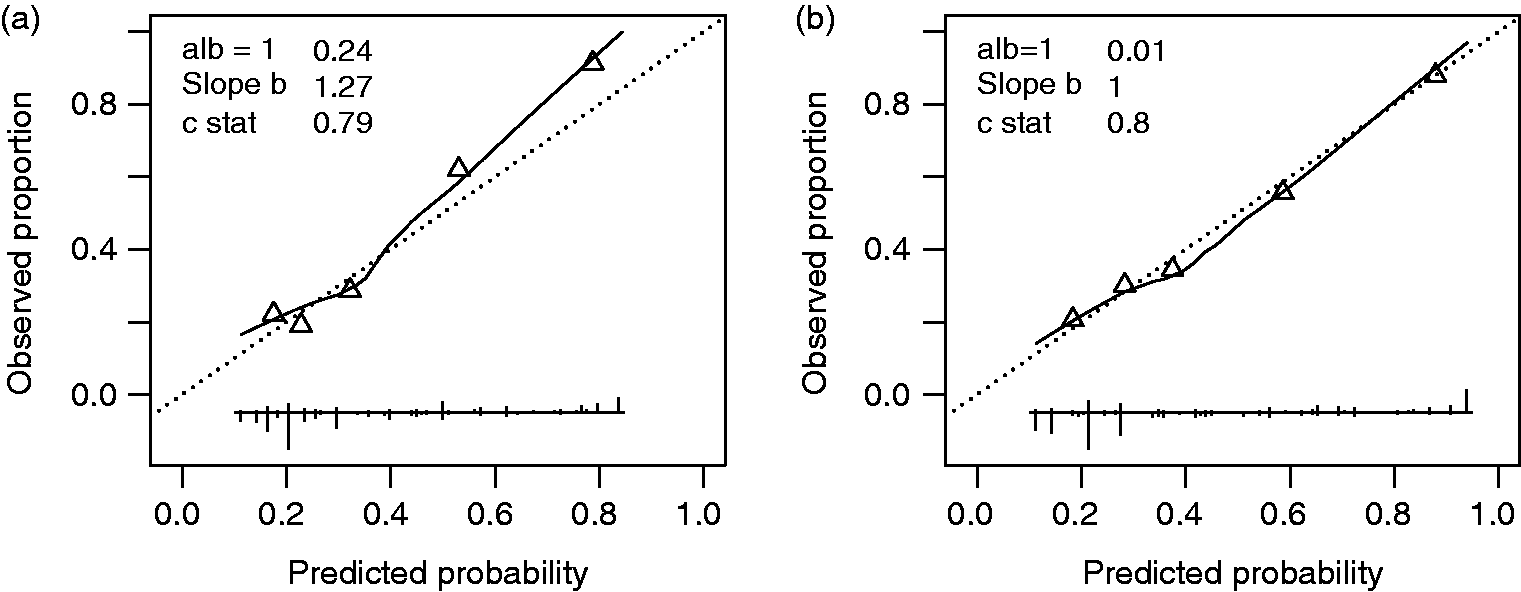
Combined cohort
Observed frequencies of chronic mesenteric ischemia in original, validation and combined cohorts' hospital specified.

Results of multivariable logistic regression analyses in the combined cohort; odds ratio (95% confidence interval).
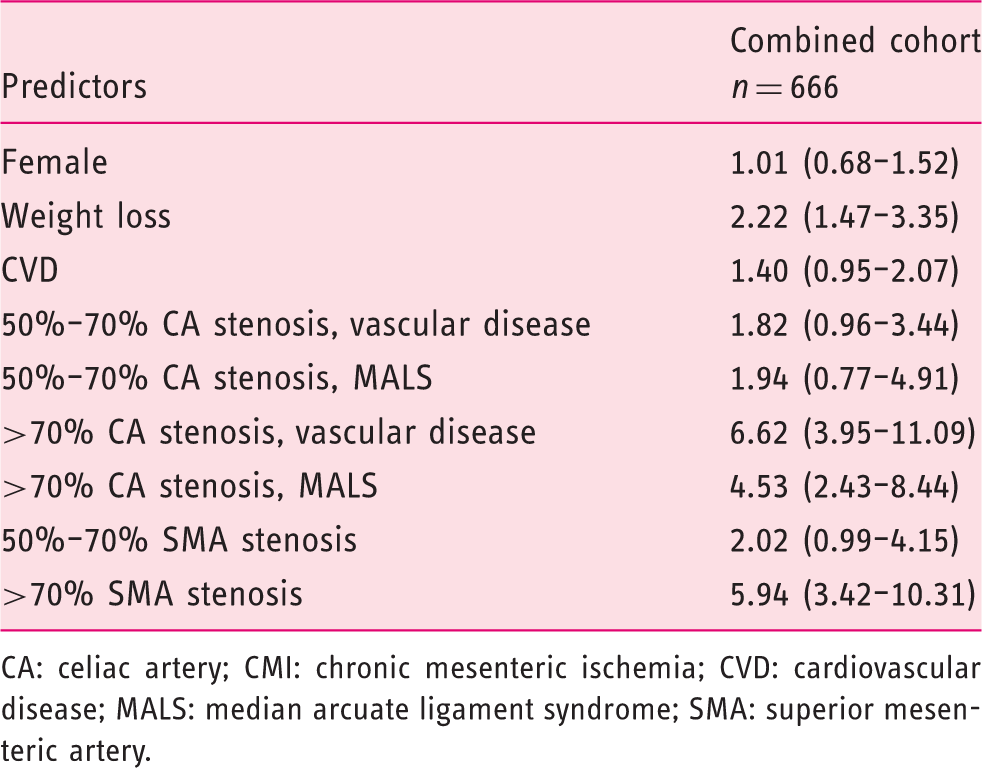
CA: celiac artery; CMI: chronic mesenteric ischemia; CVD: cardiovascular disease; MALS: median arcuate ligament syndrome; SMA: superior mesenteric artery.
Updated score chart
Updated score chart for prediction of CMI.
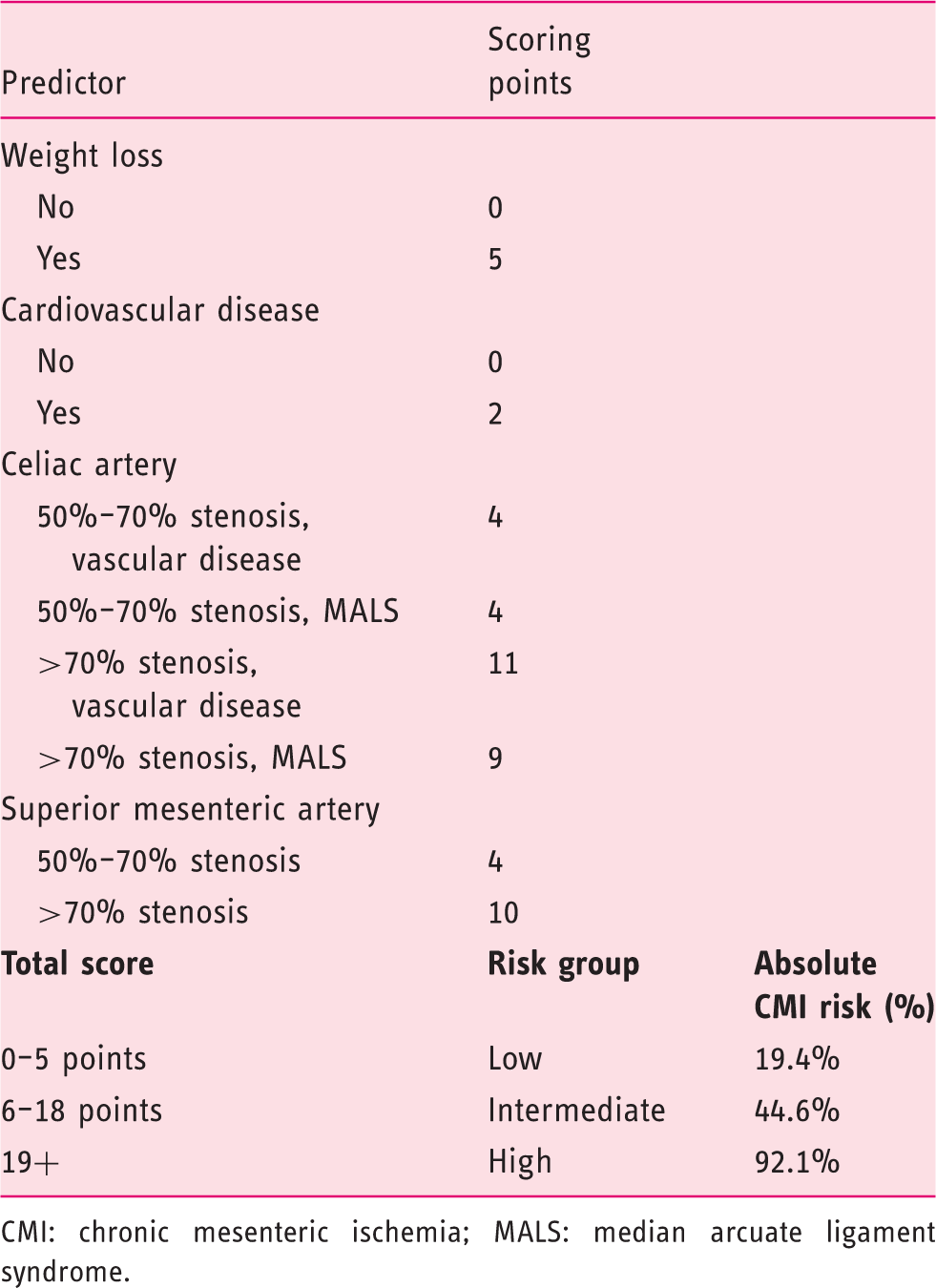
CMI: chronic mesenteric ischemia; MALS: median arcuate ligament syndrome.
Figure 1(b) shows the calibration of the updated score chart. The updated model is based on the data of the combined cohort and resulted in a calibration intercept close to 0 and calibration slope b (slope b) close to 1. The discriminative ability of the updated model is excellent with a c-statistic of 0.80.
Discussion
In this multicenter study, we performed an external validation of the recently developed score chart to predict the risk of CMI. Next, we developed an updated version of the score chart based on the performance of the score chart in the combined cohort and with inclusion of the CA stenosis cause. CMI predictors of the updated score chart are presence of weight loss, presence of CVD, the degree of CA stenosis combined with the cause of CA stenosis and the degree of SMA stenosis. The updated score chart is an easy-to-use and reliable tool to discriminate the risk of CMI (c-statistic 0.80).
The findings of our cohort analysis in 666 CMI suspected patients, the largest cohort described, correspond with the recently published clinical practice guidelines Management of the diseases of mesenteric arteries and veins 1 by the European Society of Vascular Surgery. Weight loss is an important symptom for the diagnosis of CMI according to the guidelines, and the advice is given to perform additional analyses for an alternative diagnosis in patients suspected of having CMI without substantial weight loss. Our data confirm that weight loss is the only predictive clinical symptom in the prediction model. In accordance with the guidelines stating that patients with CMI have atherosclerotic involvement in other locations,26,27 our score chart incorporated the presence of CVD as a predictor of CMI. Finally, the guidelines recommend considering the diagnosis of CMI in patients with otherwise unexplained abdominal symptoms and occlusive disease of two or three mesenteric arteries. This corresponds with our findings that multivessel disease is present in 88% of high-risk patients as opposed to only 1% multivessel disease in low-risk patients.
In contrast with the original score chart, female sex is not a predictor in the updated score chart. Female preponderance for CMI is reported in the literature,26,27 and we show that the majority (70%) of patients with definitive CMI are female in the combined cohort. However, the majority of patients without CMI are also female (65%).
We recommend the updated model for clinical practice since it is based on a more heterogeneous data set from a multicenter cohort, which supports its generalizability. Its applicability is also boosted by inclusion of the cause of CA stenosis. Experts expressed reservations regarding the original score chart because it lacked the cause of CA stenosis, whereas clinical presentation and patient characteristics differ between those with atherosclerotic CMI and CMI based on MALS. The updated score chart shows a higher predictive value for vascular disease in case of greater than 70% CA stenosis.
Both the original and the validation cohort include patients with chronic NOMI. Patients with NOMI present with typical symptoms of CMI. Chronic NOMI patients, however, will not be readily identified as high-risk using the score chart because of a maximum score of seven points in the absence of mesenteric artery stenoses. Subsequently, chronic NOMI patients are classified as low risk or intermediate risk. Based on symptom development, they will undergo immediate or later additional testing to establish or negate the diagnosis of CMI.
We suggest a wait-and-see policy for patients classified as low risk by the updated score chart since the symptoms in this patient group are minor and immediate treatment is not required. According to the score chart, 19% of the patients in the low-risk group will have CMI and their CMI is caused by NOMI or single-vessel disease. The delay in CMI diagnosis for the 19% patients suffering from CMI in the low-risk group can be defended since these patients are under the control of a physician during the wait-and-see policy who may intervene when the clinical situation during follow-up worsens. With this wait-and-see strategy, on the other hand, patients without CMI in the low-risk group (81%) are spared unnecessary diagnostic procedures.
This study has several limitations. An inevitable limitation is the absence of a gold-standard clinical test for the diagnosis of CMI. A working diagnosis is established by multidisciplinary consensus opinion and a definitive diagnosis is established when treatment results in durable symptom relief. 3 Symptom relief is considered the most important and relevant patient-related outcome. This response was noted as a dichotomous variable to limit interpretation bias. However, the patients reported symptom relief to the same team that participated in the multidisciplinary meeting reaching the consensus diagnosis of CMI, for which reason reporting bias cannot be excluded. Patients with a consensus diagnosis of no CMI were not selected for therapy and discharged without further follow-up. The results of our prediction model can be extrapolated only to patients suspected of having CMI. Therefore, this model should not be used for patients with gastrointestinal symptoms in general, but only for those with a clear suspicion of CMI after other causes have been excluded. Incorporation bias may have possibly led to overestimation of the diagnostic accuracy of the score chart for CMI. 28 Finally, the original cohort described by Harki et al. consisted of 436 patients. 13 The data set used for the current combined cohort analysis consisted of 420 patients from the original cohort because of missing data. In view of the sizable number of 666 patients in the current analysis, we assume that the effect of the missing data of these 16 patients is negligible.
In conclusion, we externally validated a previously published score chart to predict the risk of CMI. We also updated the original score chart and included the cause of CA stenosis based on expert view. The updated score chart shows good performance and an excellent discriminative ability. This updated score chart is a useful tool to be used in clinical practice to stratify CMI-suspected patients in three groups: 1. wait-and-see policy justified, 2. additional functional testing indicated and 3. immediate vascular intervention justified.
Supplemental Material
Supplemental material for Validation of a score chart to predict the risk of chronic mesenteric ischemia and development of an updated score chart
Supplemental Material for Validation of a score chart to predict the risk of chronic mesenteric ischemia and development of an updated score chart by Louisa JD van Dijk, Desirée van Noord, Robert H Geelkerken, Jihan Harki, Sophie A Berendsen, Annemarie C de Vries, Adriaan Moelker, Yvonne Vergouwe, Hence JM Verhagen, Jeroen J Kolkman, Marco J Bruno and on behalf of the Dutch Mesenteric Ischemia Study group in United European Gastroenterology Journal
Footnotes
Acknowledgments
Author contributions include the following: Louisa J.D. van Dijk: collecting, interpreting and analyzing data, drafting the manuscript. Desirée van Noord: planning and conducting the study, interpreting data and critical revision of the manuscript. Robert H. Geelkerken: interpreting data and critical revision of the manuscript. Jihan Harki: collecting data and critical revision of the manuscript. Sophie A. Berendsen: collecting data and critical revision of the manuscript. Annemarie C. de Vries: critical revision of the manuscript. Adriaan Moelker: critical revision of the manuscript. Yvonne Vergouwe: statistics, interpreting and analyzing data and critical revision of the manuscript. Hence J.M. Verhagen: interpreting data and critical revision of the manuscript. Jeroen J. Kolkman: interpreting data and critical revision of the manuscript. Marco J. Bruno: planning and conducting the study, interpreting data and critical revision of the manuscript. All authors approved the final draft submitted.
The Dutch Mesenteric Ischemia Study group (DMIS) consists of the following individuals: Ron Balm, Academic Medical Centre, Amsterdam; Gert Jan de Borst, University Medical Centre Utrecht, Utrecht; Juliette T. Blauw, Medisch Spectrum Twente, Enschede; Marco J. Bruno, Erasmus MC University Medical Center, Rotterdam; Olaf J. Bakker, St. Antonius Hospital, Nieuwegein; Louisa J.D. van Dijk, Erasmus MC University Medical Center, Rotterdam; Hessel C.J.L. Buscher, Gelre Hospitals, Apeldoorn; Bram Fioole, Maasstad Hospital, Rotterdam; Robert H. Geelkerken, Medisch Spectrum Twente, Enschede; Jaap F. Hamming, Leiden University Medical Center, Leiden; Jihan Harki, Erasmus MC University Medical Center, Rotterdam; Daniel A.F. van den Heuvel, St. Antonius Hospital, Nieuwegein; Eline S. van Hattum, University Medical Centre Utrecht, Utrecht; Jan Willem Hinnen, Jeroen Bosch Hospital, ’s‐Hertogenbosch; Jeroen J. Kolkman, Medisch Spectrum Twente, Enschede; Maarten J. van der Laan, University Medical Center Groningen, Groningen; Kaatje Lenaerts, Maastricht University Medical Center, Maastricht; Adriaan Moelker, Erasmus MC University Medical Center, Rotterdam; Desirée van Noord, Franciscus Gasthuis & Vlietland, Rotterdam; Maikel P. Peppelenbosch, Erasmus MC University Medical Center, Rotterdam; André S. van Petersen, Bernhoven Hospital, Uden; Pepijn Rijnja, Medisch Spectrum Twente, Enschede’ Peter J. van der Schaar, St. Antonius Hospital, Nieuwegein; Luke G. Terlouw, Erasmus MC University Medical Center, Rotterdam; Hence J.M. Verhagen, Erasmus MC University Medical Center, Rotterdam; Jean Paul P.M. de Vries, University Medical Center Groningen, Groningen; and Dammis Vroegindeweij, Maasstad Hospital, Rotterdam.
Declaration of conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not‐for‐profit sectors.
Ethics approval
This study complies with the Declaration of Helsinki on research ethics.
Informed consent
The medical research ethics committee of Erasmus MC University Medical Center approved that the Medical Research Involving Human Subjects Act does not apply to this study and that no informed consent was required according to local directives (MEC‐2013‐317).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
