Abstract
The European Society of Gastrointestinal Endoscopy (ESGE) together with the United European Gastroenterology (UEG) recently developed a short list of performance measures for small-bowel endoscopy (i.e. small-bowel capsule endoscopy and device-assisted enteroscopy) with the final goal of providing endoscopy services across Europe with a tool for quality improvement. Six key performance measures both for small-bowel capsule endoscopy and for device-assisted enteroscopy were selected for inclusion, with the intention being that practice at both a service and endoscopist level should be evaluated against them. Other performance measures were considered to be less relevant, based on an assessment of their overall importance, scientific acceptability, and feasibility. Unlike lower and upper gastrointestinal endoscopy, for which performance measures had already been identified, this is the first time small-bowel endoscopy quality measures have been proposed.
Introduction
The European Society of Gastrointestinal Endoscopy (ESGE) and United European Gastroenterology (UEG) have identified the need to benchmark the quality of endoscopic procedures as a high priority. The rationale for this was recently described in a manuscript that also addressed the methodology of the current quality initiative process. 1
The identification of small-bowel endoscopy performance measures presents several challenges, in contrast to the situation with upper and lower gastrointestinal (GI) endoscopy, for which several performance measures have been identified over recent years. To date, a specific set of quality measures for small-bowel endoscopy that could serve as a basis for quality assessment and improvement has not been produced. Moreover, in contrast to upper and lower GI endoscopy, small-bowel endoscopy encompasses two distinct modalities as the small bowel can be approached by means of a noninvasive, diagnostic modality (i.e. small-bowel capsule endoscopy (SBCE)) and/or a more-invasive, diagnostic, and therapeutic modality (i.e. device-assisted enteroscopy (DAE)). Although SBCE and DAE target the same organ, they differ greatly in terms of technique, procedure, process, and outcome.
The aim of the ESGE Small-Bowel Working Group was to identify a short list of performance measures for small-bowel endoscopy that were widely applicable to endoscopy services across Europe. Performance measures refer to specific issues identified for comparison and potential improvement, and they represent the minimally acceptable standard of care. This list would ideally consist of performance measures with the following characteristics: proven impact on significant clinical outcomes or quality of life; a well-defined, reliable, and simple method for measurement; opportunity for improvement; and application to all levels of endoscopy services.
Bearing in mind the lack of a previous comprehensive peer-reviewed paper describing quality measures, the ESGE Small-Bowel Working Group evaluated the available evidence and/or absence of evidence to identify research priorities. This manuscript reports the agreed-on list of performance measures for small-bowel endoscopy and describes the methodological process applied in the development of these measures. Performance measures are divided into key performance measures and minor performance measures. The list of performance measures and the methodological process applied in the development of these measures are described separately for SBCE and DAE.
Methodology
The multistep methodological process used to develop performance measures has previously been described. 1 Briefly, a modified Delphi consensus process was used to develop quality measures in the following domains: pre-procedure, completeness of procedure, identification of pathology, management of pathology, complications, procedure numbers, patient experience, and postprocedure. One or two key performance measures were selected for each quality domain. Minor performance measures, if any, were also described for each domain.
For each of the identified domains, every participant in the ESGE Small-Bowel Working Group was invited to identify performance measures. All the possible performance measures suggested were discussed through email correspondence and teleconferences. All possible performance measures identified by this process were then structured using the PICO framework (where P stands for Population/Patient, I for Intervention/Indicator, C for Comparator/Control, and O for Outcome) to inform searches for any available evidence to support these performance measures. Detailed literature searches were performed by an expert team of methodologists. Working group members also identified additional articles relevant to the performance measures in question.
The PICOs and the clinical statements derived from these were adapted or omitted during iterative rounds of comments and suggestions from the working group members during the Delphi process. In total, working group members participated in a maximum of three rounds of voting to agree on performance measures in the predefined domains and their respective thresholds, as discussed below. A statement was accepted if at least 80% agreement was reached after a minimum of two voting rounds. Statements not reaching agreement were modified according to the comments made in the voting rounds. Statements were discarded if agreement was not reached over three voting rounds. The agreement given for the different statements refers to the last voting round in the Delphi process (see online-only Supporting Information).
The key performance measures were distinguished from the minor performance measures based on the Importance, Scientific acceptability, Feasibility, Usability, and comparison with competing measures (ISFU) criteria, expressed by mean voting scores.
The performance measures are displayed in boxes under the relevant quality domain. Each box describes the performance measure, the level of agreement reached during the modified Delphi process, the grading of available evidence (the evidence was graded according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system), how the performance measure should be measured, and recommendations supporting its adoption. The boxes further list the measurement of agreement (scores), the desired threshold, and suggestions on how to address underperformance. The minimum number needed to assess whether the threshold for a certain performance measure is reached can be calculated by estimating the 95% confidence intervals (CIs) around the predefined threshold for different sample sizes. For reasons of practicality and to simplify implementation and auditing, the working group suggested that at least 100 consecutive procedures (or all, if < 100 were performed) should be measured to assess a given performance measure.
The assessment of performance measures should be applied at an individual level; however, when this is not feasible, assessment of performance measures should at least be applied at a service level. To facilitate service improvement, there should be at least an annual audit of a sufficient number of procedures with appropriate actions taken when suboptimal performance is identified. If the recommended threshold is not reached at a service level, further evaluation at an individual level is required to identify quality constraints and possible action(s) required for improvement.
SBCE
To identify performance measures, first a list of all possible performance measures for SBCE was created through email correspondence, teleconference, and face-to-face meetings that took place between May 2015 and October 2015. This process resulted in 29 possible performance measures and 50 PICOs. The performance measures and PICOs were consolidated for consistency, and any overlap was limited through email correspondence and teleconferences. After a discussion within the working group, a total of 23 PICOs were selected and retained as the basis of the literature searches, performed by a team of expert methodologists.
The evidence derived from the literature searches, along with input from the working group members, generated 38 clinical statements addressing 17 potential performance measures, grouped into eight quality domains. The PICOs and the clinical statements were adapted and/or excluded during the iterative rounds of voting and comments from the working group during the Delphi process. Over the course of three rounds of voting, consensus agreement was reached for 18 statements relating to 10 performance measures. The remaining clinical statements and performance measures did not reach agreement over the course of the three rounds of voting; it was also impossible to define performance measures for three domains (i.e. number of procedures, patient experience, and postprocedure). Therefore, a final total of 10 performance measures (18 statements) attributed to five quality domains were accepted for these guidelines (Figure 1).
Key performance measures for small-bowel capsule endoscopy (SBCE). DAE: device-assisted enteroscopy; GI: gastrointestinal; NA: not applicable.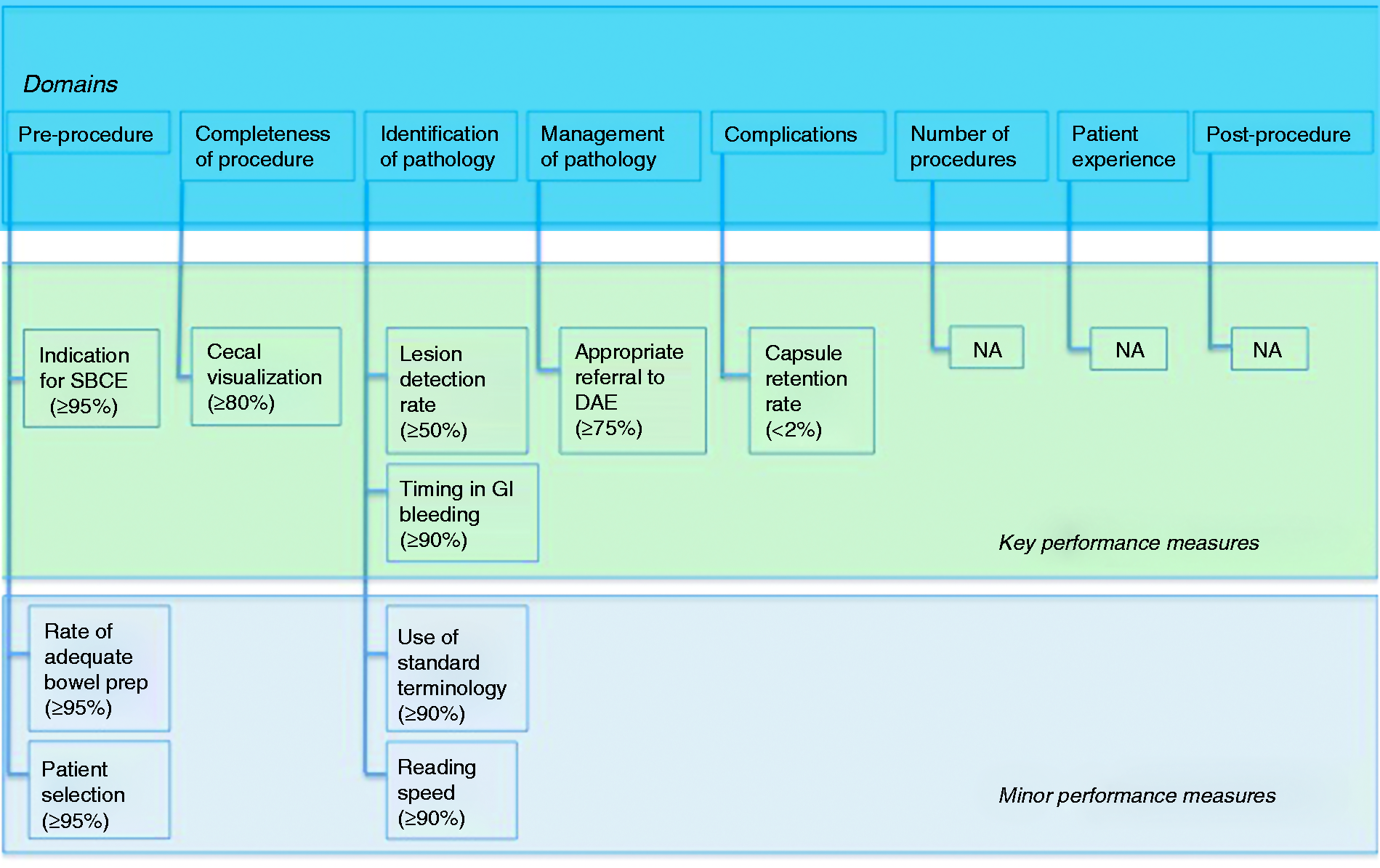
DAE
To identify performance measures, first a list of all possible performance measures for DAE was created through email correspondence, teleconference, and face-to-face meetings that took place between March 2016 and February 2017. This process resulted in 34 possible performance measures and 65 PICOs. The performance measures and PICOs were consolidated for consistency, and any overlap was limited through email correspondence and teleconferences. After a discussion within the working group, 25 PICOs were selected and retained as the basis of the literature searches. A detailed literature search was performed by a team of expert methodologists.
The evidence derived from the literature searches and input from the working group members were used to formulate 23 clinical statements addressing 16 potential performance measures, grouped into eight quality domains. The PICOs and clinical statements were then adapted and/or excluded during the iterative rounds of voting and comments from the working group during the Delphi process. Over the course of three rounds of voting, consensus agreement was reached for 19 statements relating to 10 performance measures. The remaining clinical statements and performance measures did not reach agreement over the course of three rounds of voting; it was also impossible to define performance measures for two domains (i.e. number of procedures and postprocedure). Therefore, a final total of 10 performance measures (19 statements) attributed to six quality domains were accepted for these guidelines (Figure 2).
Key performance measures for small-bowel device-assisted enteroscopy (DAE). NA: not applicable.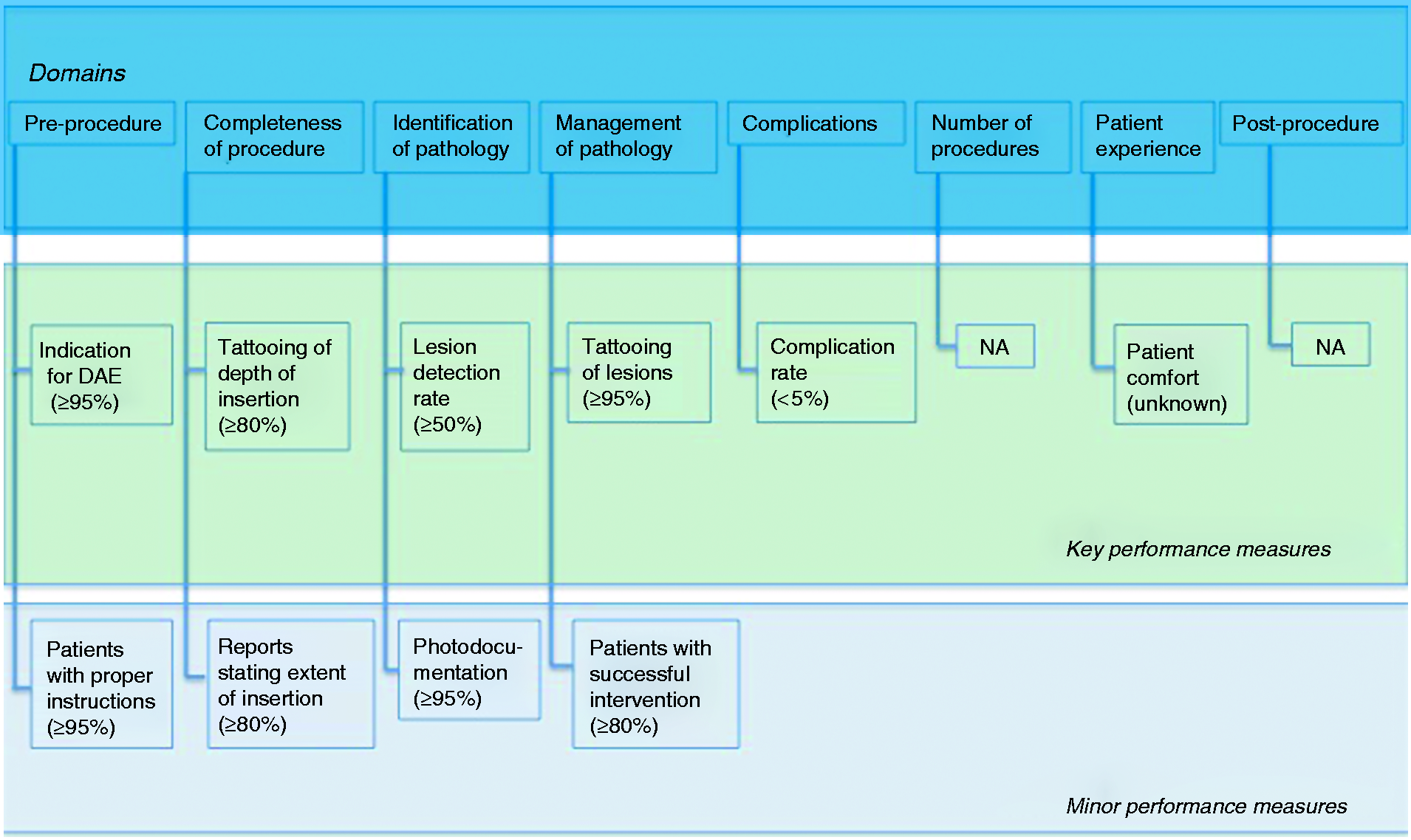
Key and minor performance measures
Although the overall evidence quality (as assessed using the GRADE criteria) for most of the performance measures for SBCE and DAE is low, this does not suggest that these performance measures are irrelevant. We used the highest mean voting scores to identify at least one key performance measure for each of the quality domains for each modality. When this process was not possible because performance measures had similar voting scores, the Quality Improvement Committee chair selected the key performance measure, and this was subsequently agreed on by the whole working group.
The remaining performance measures were considered to be minor performance measures, but nevertheless all performance measures (identified by the rigorous process described above) were deemed to be valuable by the working group members. It was however agreed that, from a practical standpoint, it may be more acceptable to concentrate on the implementation of the key performance measures in the first instance. Once a culture of quality measurement (with the aim of optimizing practice, outcomes, and patient experience) is accepted and supporting software is available, the minor performance measures may then further aid the monitoring of quality in small-bowel endoscopy. The use of appropriate endoscopy-reporting systems is key to facilitate data retrieval on identified performance measures.
Performance measures for SBCE
1 Domain: Pre-procedure

The acceptance of this performance measure is based on the strength of agreement with the following statements:
The indications for SBCE should be guided by published recommendations (e.g. ESGE guidelines). (Statement number 1.1) Agreement: 81.8% The percentage of SBCE procedures performed by indication should be audited. (Statement number 1.1) Agreement: 81.8% Studies performed for indications not included in a published standard list of appropriate indications approved by an internationally recognized endoscopy professional society should be documented and reviewed. (Statement number 1.1) Agreement: 81.8%
Adherence to appropriate indications for SBCE may help to optimize the use of limited resources (considering the high costs relating to SBCE) and to protect patients from the potential harms of unnecessary procedures. SBCE performed for an appropriate indication is associated with significantly higher diagnostic yields for clinically relevant lesions, when compared with SBCE performed without an appropriate indication.2–8
The ESGE and American Society for Gastrointestinal Endoscopy (ASGE) both have produced guidelines with similar recommendations for the appropriate use of SBCE: obscure GI bleeding, iron deficiency anemia, Crohn disease (known or suspected), small-bowel tumors, inherited polyposis syndromes, abnormal radiological imaging, and subgroups of patients with celiac disease (i.e. complicated and/or refractory celiac disease). Other indications such as abdominal pain, diarrhea, and malabsorption are associated with a low diagnostic yield and are therefore not considered to be appropriate indications for SBCE.9–11
Studies performed for indications not approved by an internationally recognized endoscopy professional society should be documented, audited, and reviewed regularly.

The acceptance of this performance measure is based on the strength of agreement with the following statements:
Visualization is higher in patients who received purgative agents. (Statement number 17.2) Agreement: 81.8% The mucosal visualization obtained for SBCE should be adequate or good in greater than 95% of cases using accepted bowel preparation methods. (Statement number 17.1) Agreement: 90.9%
Optimal bowel preparation for SBCE is controversial. As recommended by the ESGE clinical guideline, prior to SBCE, patients are requested to follow a modified diet and to ingest a purgative for better visualization. Suboptimal bowel preparation results in further costs and inconvenience because patients need to undergo either a repeat SBCE or an alternative investigation. Five systematic reviews13–17 and 16 randomized controlled trials (RCTs)18–32 were included for the analysis of the role of bowel preparation in patients undergoing SBCE and were considered potentially relevant. Four main interventions were examined: (a) purgatives, (b) antifoaming agents, (c) a combination of purgative and antifoaming agent, and (d) prokinetics.
Laxatives do not significantly improve diagnostic yield or completion rate in SBCE but do improve quality of small-bowel mucosal visualization. The use of laxatives may therefore be beneficial in patients likely to have subtle findings, although there is recent evidence to the contrary. 33 Bowel preparation is less well tolerated by patients and the addition of laxatives remains controversial.
RCTs demonstrate that antifoaming agents improve the quality of mucosal visualization, 10,14 and meta-analyses have concluded that simethicone has a positive impact on the quality of preparation as it significantly decreases the presence of luminal bubbles/foam.14,17,18,24,26,34–55 The combination of purgatives and antifoaming agents does not increase diagnostic yield; however, it may play a role in improving the quality of small-bowel mucosal visualization.
Prokinetics do not improve completion rate nor the diagnostic yield of SBCE, and their routine administration is not recommended. It is to be noted, however, that there is considerable heterogeneity in methodology and definitions used in different studies.14,15
A regimen of preparation as well as the most appropriate timing for small-bowel preparation has not been standardized. Unlike colonoscopy, there is no standardized, validated scale available for the evaluation of cleansing achieved for SBCE and such a tool should be developed for standardized scoring and recording of SBCE findings. Small-bowel luminal contents, such as food residue, blood, bile, and bubbles/foam, which hinder adequate small-bowel mucosal visualization, are usually subjectively evaluated. Subjectivity is also currently applied to the overall assessment of small-bowel preparation quality.
Several cleansing scales for capsule endoscopy have been proposed, some of which have already been validated. 56 Among the others, the Brotz and Park scales have been validated and are relatively commonly adopted.57,58
In a prospective, randomized, single-center validation study, Brotz et al. 58 assessed the adequacy of small-bowel cleansing for capsule endoscopy according to three scales: quantitative index, qualitative evaluation, and overall adequacy assessment. The authors showed a strong and highly significant association between the quantitative index, qualitative evaluation, and overall adequacy assessment. The authors also concluded that the adequacy of small-bowel cleansing should be incorporated into the standard capsule endoscopy report as an important quality measure.
Similarly, Park and colleagues 57 developed a small-bowel cleansing score assessing two visual parameters: proportion of visualized mucosa and degree of obscuration. The first parameter was the proportion of visualized mucosa. This was scored using a four-step scale ranging from 0 to 3: score 3, > 75%; score 2, 50%–75%; score 1, 25%–50%; score 0, < 25%. The second parameter was the degree of obscuration by bubbles, debris, bile, etc. This was scored using a four-step scale ranging from 0 to 3: score 3, no (<5%) obscuration; score 2, mild (5%–25%) obscuration; score 1, moderate (25%–50%) obscuration; score 0, severe (>50%) obscuration. For the evaluation, representative frames from small-bowel images were serially selected and scored at five-minute intervals. The reliability of the grading system was evaluated by assessing the interobserver, intrapatient, and intraobserver agreement, which resulted in excellent agreement.

The acceptance of this performance measure is based on the strength of agreement with the following statements:
Certain groups of patients undergoing SBCE have a greater risk of capsule retention. (Statement number 7.1) Agreement: 90.9% The use of a patency capsule can reduce the incidence of capsule retention in patients at higher risk. (Statement number 11.1) Agreement: 90.9%
It is well established that certain underlying conditions predispose to capsule retention. A recently published meta-analysis showed the capsule retention rate was 2.1% for patients with suspected small-bowel bleeding (95% CI 1.5%–2.8%) and 3.6% (95% CI 1.7%–8.6%) for suspected inflammatory bowel disease (IBD). For established IBD, the capsule retention rate reached 8.2% (95% CI 6.0%–11.0%). 61
The overall capsule retention rate is low, and it is related to clinical indication. Symptoms such as abdominal pain, abdominal distension, and nausea/vomiting are associated with a significantly higher rate of capsule retention. Furthermore, previous small-bowel resection, abdominal/pelvic radiation therapy, and chronic use of high-dose nonsteroidal anti-inflammatory drugs have all been shown to increase the risk of capsule retention.2,61 Therefore, a careful assessment of past medical history is mandatory to select patients who require a specific work-up aimed at preventing capsule retention.
The use of a patency capsule can reduce the incidence of capsule retention in high-risk patients. The use of a patency capsule has been shown to accurately identify the presence of stenosis as well as or better than standard radiological techniques and is at least comparable to dedicated cross-sectional imaging modalities. Although unable to provide direct visual information regarding the presence and location of strictures, masses, or other causes of luminal narrowing of the small bowel, a successful patency capsule examination minimizes the risk of retention in high-risk patients and allows for a safer SBCE procedure.
This performance measure can and should be implemented both at a service and individual endoscopist level. Variations from the expected capsule retention rates suggest suboptimal patient selection and procedure quality. There are insufficient data to set the minimum and target standards reliably, but the proposed values for proper selection of patients of ≥95%, respectively, were deemed appropriate to ensure safer SBCE.
2 Domain: Completeness of procedure

The acceptance of this performance measure is based on the strength of agreement with the following statements:
The incomplete study rate (failure to reach the colon or stoma bag) should be less than 20%. (Statement number 4.1) Agreement: 100% In all cases of an incomplete study, the patient should be asked to confirm excretion. If excretion is not confirmed after 15 days, an abdominal radiograph should be obtained. (Statement number 5.1) Agreement: 100%
Visualization of the colon or a stoma is a prerequisite for confirming complete visualization of the small bowel. Incomplete SBCE results in further costs due to the repetition of SBCE and/or an alternative investigation. Patients undergoing SBCE should be instructed to check for excretion of the capsule. In cases where the capsule did not reach the colon or the stoma within the duration of the recording and the patient does not confirm excretion within two weeks of ingestion, an abdominal radiograph should be obtained to rule out capsule retention (unless contraindicated). A completion rate < 80% may be associated with a higher risk of missing significant pathology; nevertheless, the true magnitude of this risk is unclear.
For grading this performance measure, 39 studies2,4,62–98 with 18,035 procedures were analyzed. All were retrospective or prospective analyses of registries of single- or multiple-center experiences. The percentage of complete examinations was reported in 23 studies. The results were heterogeneous, ranging from 64% to 96%, with a median of 80% complete SBCEs.
3 Domain: Identification of pathology

The acceptance of this performance measure is based on the strength of agreement with the following statements:
The overall diagnostic yield of SBCE depends on the referral population, and adherence to ESGE guidelines. (Statement number 2.1) Agreement: 100% Currently available data do not support a single optimal diagnostic yield per indication and as such regular audit is required to ensure adherence with ESGE guidelines on the indications for SBCE and >95% compliance achieved. (Statement number 2.1) Agreement: 100%
Available studies of SBCE diagnostic yield are mainly reports of clinical experience without clearly defined indications for the procedure. These indications include: suspected small-bowel bleeding (overt and/or occult); suspected or established Crohn disease, malabsorption, diarrhea, abdominal pain, polyp surveillance, suspected tumors, and abnormal radiological imaging. Therefore, the patient populations studied and diagnostic yields showed wide variation. Diagnostic yield for mixed indications varied between 27% and 77.3%2,63,64,66,72,74,76,79–82,86,88–92,95,97,99,100; for suspected GI bleeding, between 31% and 68%4,62,65,67,70,71,77,85,87,93,94,98,101; for suspected Crohn disease, between 6% and 38% (although definition of Crohn disease also varied)68,73,82,93; with a 39% yield of active disease in patients with established Crohn disease. 82
The working group agreed that a diagnostic yield of at least 50% in all patients having SBCE (for any indication) was a reasonable aim. It was acknowledged that there may be situations in which there was uncertainty in distinguishing pathology from normal variants and bowel content, and in terms of whether the lesions identified were relevant to the indication of the procedure and were a “true diagnosis.” Therefore, it was agreed there should be a minimum requirement for a written description and photodocumentation of any lesion considered significant.
For patients with suspected GI bleeding, the Saurin classification is advisable for the evaluation of the relevance of the lesions. 102 According to the Saurin classification, lesions detected at capsule endoscopy are classified as P0, P1, and P2. P0 lesions are those having no potential for bleeding, including visible submucosal veins, diverticula without the presence of blood, or nodules without mucosal break. P1 lesions are those regarded as having uncertain hemorrhagic potential, such as red spots on the intestinal mucosa, or small or isolated erosions. P2 lesions are those considered to have a high potential for bleeding, such as typical angiomas, large ulcerations, tumors, or varices. For indications other than GI bleeding, a universally accepted classification is lacking; however, the presence on SBCE of ulcerations, erosions, and/or strictures in the context of a patient with suspected/established Crohn disease, along with the findings of small-bowel tumors and small-bowel polyps, are considered significant findings.

The acceptance of this performance measure is based on the strength of agreement with the following statement:
Earlier timing of SBCE achieves a higher diagnostic yield in patients with overt small-bowel bleeding. (Statement number 16.1) Agreement: 100%
In the context of overt small-bowel bleeding, performing SBCE closer to an episode of bleeding appears to correlate with an increased diagnostic yield.3–8 Although the optimal timing for the performance of SBCE in this setting is unknown, in line with ESGE guidance 9 the working group adopted the cutoff point of 14 days from the episode of bleeding as a measure of quality for audit.

The acceptance of this performance measure is based on the agreement with the following statement:
Structured and standardized reporting improves the consistency of image interpretation, the description of findings, and patient management. It also facilitates audit and collation of study databases, but does not improve diagnostic yield. (Statement number 15.1) Agreement: 100%
No studies of the effect of using a standardized terminology on image interpretation or diagnostic yield were identified. However, the minimal standard terminology for documentation in flexible endoscopy was devised in recognition of the value this brings to the retrieval of information from databases for the purpose of audit, research, and the facilitation of education and training. 82 A similar process was followed in the creation of the capsule endoscopy structured terminology (CEST), in which the standard considers a report in two components: structure and content. 103
The CEST standardizes the documentation of: patient demographics, details of capsule used, indication, examination characteristics, findings, recommendations, and complications. It includes a detailed breakdown of descriptive methodology in terms of lumen, content, mucosal appearances, and any lesions identified. Validation was defined as the CEST inclusion for ≥90% of all descriptors used in any one section of a historical cohort of reports. 104

The acceptance of this performance measure is based on the strength of agreement with the following statements:
For all indications and in all cases, reading speed should be in accordance with the ESGE technical review. (Statement number 14.6) Agreement: 80% Reading speed should be appropriate such that lesion detection is not compromised, and sufficient diagnostic yields are achieved on regular audit, when patient selection and indication are in line with ESGE guidance. (Statement number 14.6) Agreement: 80% Reading time is significantly shorter with software that eliminates repeated identical images than conventional viewing. (Statement number 14.1) Agreement: 81.8%
In line with the ESGE technical review, 10 the Small-Bowel Working Group recommends recordings be read at a maximum speed of 10 frames per second (single view); if double-/multiple-view modes are used, a maximum reading speed of 20 frames per second is advised. Also, in line with the ESGE technical review, the working group supports the recommendation that particular vigilance must be paid (and the reading frame rate further slowed) within the proximal small bowel, where the risk of missing lesions appears to be higher.10,107
The Small-Bowel Working Group also supports the ESGE technical recommendations 10 relating to automated software algorithms designed to shorten reading time, in that these may be used to scan the small bowel for diffuse lesions (associated with Crohn disease, for example), but should not be relied on to detect an isolated lesion. Moreover, in accordance with the ESGE technical review, the use of virtual chromoendoscopy and “blue mode” imaging is not recommended for routine use because this has not been shown to improve diagnostic yield or enhance detection or characterization of small-bowel mucosal pathology. 10
4 Domain: Management of pathology

The acceptance of this performance measure is based on the strength of agreement with the following statement:
The use of SBCE prior to DAE improves the diagnostic yield. Prior SBCE is associated with an increased diagnostic and therapeutic yield during DAE. (Statement number 10.1) Agreement: 81.8%
Pathological findings at SBCE may warrant further investigation (and possible endotherapy) by DAE and, in this context, SBCE-reported findings should serve as a guide. Clear description of any lesion identified and its location (as described in the ESGE technical review 10 ) will help the enteroscopist to select the most appropriate route of approach (i.e. antegrade vs retrograde) and any potential endotherapy that may be applied.
5 Domain: Complications

The acceptance of this performance measure is based on the strength of agreement with the following statement:
Retention rates should be audited in all cases against known rates. Variations from expected rates suggest suboptimal patient selection and procedure quality. (Statement number 7.2) Agreement: 80%
SBCE is a generally safe, noninvasive modality to visualize the small-bowel mucosa and is now the recommended first-line small-bowel investigation for several indications. However, adverse events may occur in up to 2% of cases overall; these include: capsule retention, aspiration, and capsule-induced bleeding or perforation. 113 Knowledge of SBCE-related complications improves patient care and safety by ensuring that only patients for whom the procedure is appropriate are selected. This knowledge, and awareness of it, is critical for appropriate informed consent prior to SBCE and facilitates early identification of adverse events and their appropriate management.
Currently there is insufficient evidence to reach a consensus on an acceptable overall complication rate for SBCE or to define individual target standards for any complication, other than for capsule retention. In light of this, regular audit of SBCE-related complications is advised to enhance the SBCE user knowledge base and to identify individual as well as unit variance over time.
Capsule retention is defined as a capsule remaining in the GI tract for 15 days (or less if medical, endoscopic, or surgical intervention was deemed necessary). Although uncommon, capsule retention is a serious potential complication of SBCE and warrants assessment as a key performance measure for all centers and individual endoscopists. Appropriate patient selection and the use of a patency capsule, where indicated, all play an important role in avoiding retention and can influence retention rates, which vary considerably by indication. The evidence base suggests a target standard of 2% for overall capsule retention, irrespective of indication, in any given population is reasonable, with reported overall retention rates of 0.3%–3%,84,88,113–115 and this should be audited yearly.
In all cases of capsule retention within the small bowel, a management plan to promote natural excretion or to retrieve the capsule should be agreed on with the patient to avoid subsequent complications, including perforation, obstruction, and bleeding. Most cases of retention do not require surgical intervention. In asymptomatic patients, a “watch and wait” policy, with or without the addition of laxatives, prokinetics, or disease-specific medical therapy, may be a reasonable approach as spontaneous passage of the capsule has been reported in up to 50% of reported cases. 88 Conversely, symptomatic patients and those with significant small-bowel pathology or with tight stenosis on cross-sectional imaging may benefit from early endoscopic or surgical intervention, 115 especially where the pathology is suspected to be malignant.
6 Domain: Number of procedures
As a safe, noninvasive procedure with high patient acceptability and proven clinical use, the role and demand for SBCE continues to increase; delivering this requires the provision of well-trained, competent endoscopists. As with all endoscopy, competency involves a broad knowledge of the procedure, including indications and contraindications, lesion identification and interpretation, accurate reporting, and follow-up, as well as technical and practical skills.
It is well accepted and established that there is a need to combine both formal training courses and supervised practical training for most endoscopy procedures and to perform formal structured assessment before a trainee is considered competent. The Small-Bowel Working Group unanimously agreed that completion of a formal training course, compliant with core curriculum recommendations for SBCE, and a minimum number of practical procedural experiences are both needed to become proficient in SBCE. Although there are several studies that show enhanced accuracy with reading experience and confirm the value of formal training courses, the overall evidence base in support of this recommendation is scant. In addition, there is some evidence to show that trainees with prior endoscopy experience are at an advantage in gaining competence and dual training should be encouraged. 116
It is unclear whether there is a need to regularly undertake a minimum number of procedures to maintain competency. While this may appear prudent and would be in keeping with recommendations for other flexible endoscopy procedures, there are insufficient data currently to make a recommendation. During the discussions and the Delphi process, the working group agreed on the suggestion that a minimum of 30–50 SBCEs be required to acquire proficiency and that 30–50 procedures per year be needed to maintain competence. However, ESGE is currently developing a curriculum for training in small-bowel endoscopy that will address this matter in more detail.117–119
Performance measures for DAE
7 Domain: Pre-procedure

The acceptance of this performance measure is based on the agreement with the following statement:
DAE examinations should be performed for recognized indications as published in international guidelines. (Statement number 19) Agreement: 100%

The acceptance of this performance measure is based on the strength of agreement with the following statements:
All patients (100%) undergoing DAE should receive adequate pre-procedure preparation, including fasting for antegrade DAE and approved bowel preparation for retrograde DAE. (Statement number 18) Agreement: 100% All patients (100%) referred for antegrade DAE should be fasting for solids for at least six hours prior to the procedure. (Statement number 18.1) Agreement: 88.9% All patients (100%) referred for antegrade DAE are allowed to drink water until two hours prior to the procedure. (Statement number 18.2) Agreement: 88.9% All patients (100%) referred for retrograde DAE should follow the same regimen of preparation as recommended by ESGE guidelines for colonoscopy. (Statement number 18.3) Agreement: 88.9%
The Small-Bowel Working Group, in line with ESGE guidance (technical and clinical),9,10 strongly recommends adherence to appropriate preparation instructions for DAE. As for other endoscopic procedures, 121 good-quality preparation is essential for adequate detection of small-bowel mucosal pathology at DAE. Additionally, particularly in the case of retrograde DAE procedures, the presence of intraluminal debris is detrimental not only to lesion identification but also to the technical success of procedures as this may lead to excessive friction between the enteroscope and overtube, causing hindrance to the progress of the procedure. Although there are no comparative studies on preparation for antegrade DAE,122–125 a prolonged fast of at least six hours is usually sufficient. Retrograde DAE procedures require optimal, purgative-based preparation as per the local protocol for colonoscopy.126,127
The presence or suspicion of a stenosis may potentially increase the risk of residual intraluminal debris and, in such cases, more prolonged fasting (and potentially additional preparation) may be required. 10
8 Domain: Completeness of procedure

The acceptance of this performance measure is based on the strength of agreement with the following statement:
Depth of insertion should be marked with a submucosal tattoo of sterile carbon particles. (Statement number 24.1) Agreement: 100%

The acceptance of this performance measure is based on the strength of agreement with the following statement:
In all cases, small-bowel depth of insertion should be estimated and recorded. (Statement number 24) Agreement: 90.9%
This statement corresponds to a very low level of evidence because of the low reliability of measurement of small-bowel insertion depth. The only scientific approach is that described by May et al., 124 who validated an estimated 40-cm progression for each insertion step; however, clinical experience suggests this measure is still rather uncertain, given the episodic “slippage” (in which previous advancement is “lost” by enteroscope and overtube “fallback”) and episodic failure of advancement, which are encountered at some point during all DAE procedures. Despite animal studies showing a low deviation (<10%) from the actual and calculated insertion depth,124,130 in actual clinical practice, there is a large variation in the techniques employed, in enteroscopist ability, and procedure efficacy, with human studies failing to confirm the earlier animal studies. Therefore, the impact of this measure on diagnostic yield at DAE is unknown.
9 Domain: Identification of pathology

The acceptance of this performance measure is based on the agreement with the following statements:
Cases for DAE should be carefully selected to maximize diagnostic yield. (Statement number 21) Agreement: 88.9% Current literature is insufficient to set a minimal diagnostic yield for DAE by indication or per enteroscopist. DAE use and diagnostic yield should be audited regularly. (Statement number 22) Agreement: 100% Overall pathology detection rates for SBCE and DAE vary according to indication. Indications both for SBCE and DAE procedures should be regularly audited for adherence to international guidelines, and reasons for variations should be examined. (Statement number 23) Agreement: 100%
Appropriate detection and management of pathology is one of the cornerstones of quality in endoscopy. Reducing lesion “miss rates” results in improved patient outcomes and warrants consideration as a specific quality measure.
DAE is most often performed following a less-invasive small-bowel investigation (SBCE and/or dedicated cross-sectional imaging) and is therefore undertaken following identification of pathology and with a specific therapeutic intent. This approach is favored, where possible, and can assist with planning the appropriate approach route (antegrade or retrograde), as well as the appropriate intervention. In such cases, correlation between the expected and achieved lesion detection rates, and the proportion of successful interventions, are likely to prove useful performance measures, reflecting appropriate patient selection and operator competency.
In general, unlike adenoma detection rate in screening colonoscopy, there is inadequate evidence available in the literature to support a recommendation of specific overall diagnostic yield for DAE, with significant variation in reported diagnostic yields from 47% to 75%.2,4,63,64,95,97
As with other endoscopic modalities, published evidence confirms lesion detection and diagnostic yield vary according to indication, further affecting the ability to set specific quality targets for DAE. In addition, the route of DAE affects identification of pathology, with lower rates in general reported for retrograde procedures.81,93
As such, in the absence of specific targets for overall and per-indication lesion detection rates, to maintain quality DAE should be performed only for approved clinical indications. ESGE guidelines recommend DAE: for therapy in patients with positive findings at capsule endoscopy; in patients with obscure GI bleeding when SBCE is not available or is contraindicated; in selected patients with ongoing overt obscure GI bleeding; in patients with ongoing obscure GI bleeding and an unremarkable capsule endoscopy; for biopsy in patients with noncontributory ileocolonoscopy and a suspicion of Crohn disease on radiologic imaging tests or capsule endoscopy; in patients with Crohn disease when endotherapy is indicated; for biopsy and/or tattooing when an imaging test is suspicious of a small-bowel tumor, for biopsy and/or tattooing in patients for whom there is an uncertain diagnosis of small-bowel tumor at capsule endoscopy; when a submucosal mass is detected by capsule endoscopy; in patients with inherited polyposis syndromes when polypectomy is indicated; and in patients with nonresponsive or refractory celiac disease for biopsy.

The acceptance of this performance measure is based on the agreement with the following statement:
It is recommended to use photodocumentation as a record of findings in all cases. (Statement number 24.3) Agreement: 90.9%
Endoscopic image documentation has become a routine part of clinical care and has gained an important role in quality control, with many international societies advocating documentation of specific landmarks and pathological lesions. Despite the lack of specific data in support of this approach, there was almost unanimous agreement among experts and in quality recommendations that photodocumentation reflects current best practice, is clinically useful, and can be considered a standard performance measure.
A formal set of photographic images to confirm cecal intubation on reports is the best example of photodocumentation as a quality measure, regarded by many as an essential tool to assess individual and institutional colonoscopy quality. Documentation of other endoscopic landmarks, both in upper and lower GI disease, is also advocated and is becoming established practice.
While there are few small-bowel anatomical landmarks and none likely to be viable as DAE quality indicators, taking a photographic record of individual lesions, as done with other endoscopic procedures, is strongly advised. Photodocumentation of lesions facilitates accurate reporting and interpretation, assists with onward referral, and enables direct comparison if subsequent follow-up procedures are required. As such, regular audit and a minimum compliance of 95% is recommended for lesion photodocumentation as a quality performance measure for DAE.
10 Domain: Management of pathology

The acceptance of this performance measure is based on agreement with the following statement:
It is recommended practice to mark a lesion that may later be a target for therapeutic intervention. (Statement number 24.2) Agreement: 100%
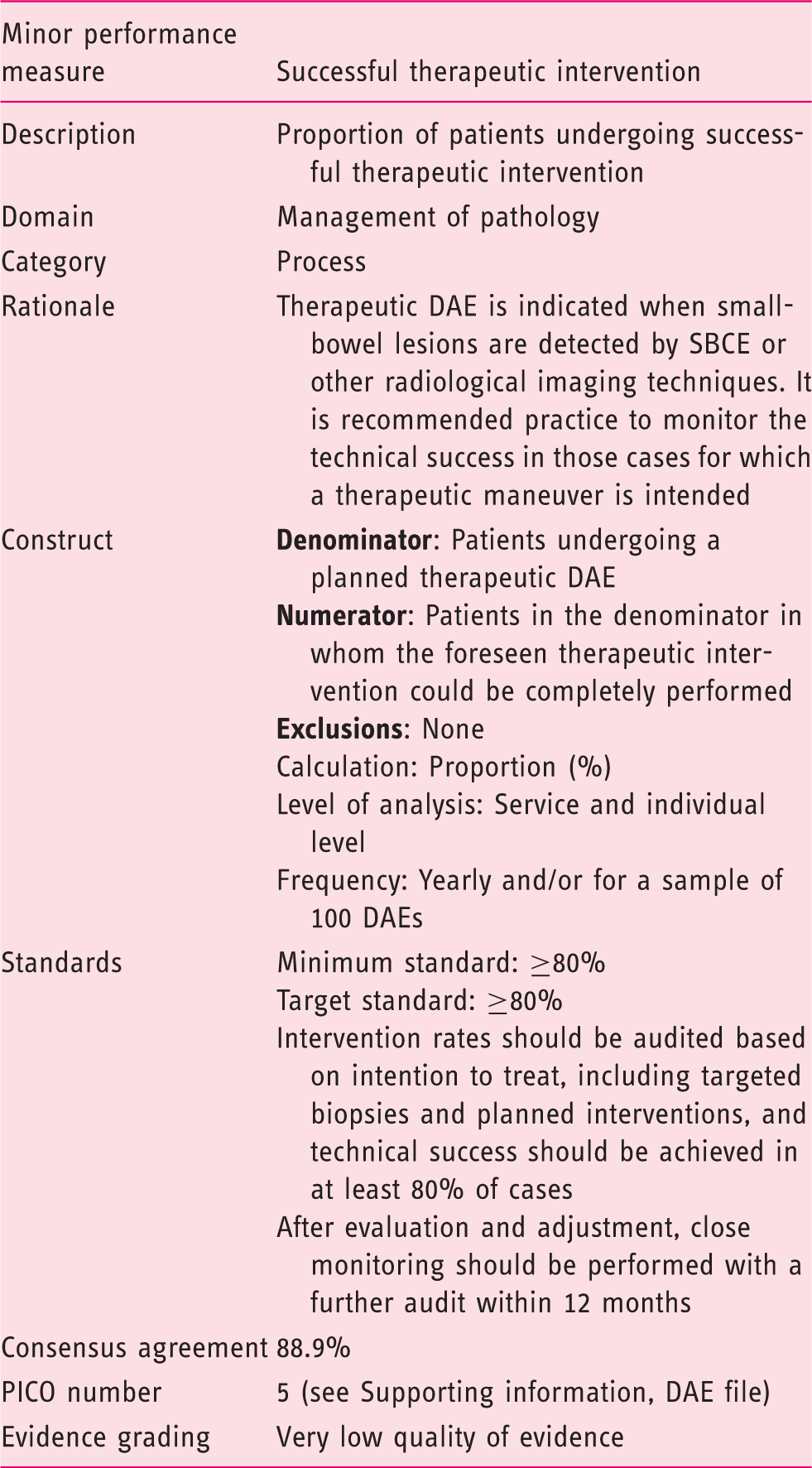
The acceptance of this performance measure is based on agreement with the following statement:
Intervention rates should be audited based on an intent-to-treat basis, and successful completion of the planned intervention or outcomes should be achieved in at least 80% of cases. (Statement number 21) Agreement: 88.9%
Although data on intended vs actual intervention rates for DAE are lacking, based on the available evidence and expert opinion, a target rate of 80% was considered appropriate by the working group. If the minimum standard is not achieved on a case-by-case basis, audit should be performed including review of the operator's DAE technique, as well as a critical review of the initial diagnostic tests, SBCE, or radiology that prompted the DAE procedure. It should also be borne in mind that insertion depth is frequently only a rough estimate, so an apparent lack of success may be due to the lesion not being reached. In addition, once identified, as previously stressed, a tattoo-based marking of the lesion location is required to facilitate further intervention.
11 Domain: Complications

The acceptance of this performance measure is based on the strength of agreement with the following statements:
The rate of severe complications (overall, including perforation, bleeding, and pancreatitis) resulting from diagnostic DAE should not exceed 1% in an unselected population. (Statement number 26) Agreement: 100% The rate of severe adverse events (overall, including perforation, bleeding, and pancreatitis) resulting from therapeutic DAE should not exceed 5% in an unselected population. (Statement number 26.1) Agreement: 100% The overall rate of pancreatitis in DAE should not exceed 0.3%. (Statement number 26.2) Agreement: 88.9% Adverse event rates by operator and indication should be audited for all DAE procedures against known rates of adverse events. Reasons for variations from these rates should be examined. (Statement number 27) Agreement: 100%
DAE is a safe procedure: The complication rates for DAE from published pooled series indicate the complication rate for diagnostic procedures is less than 1% and for therapeutic procedures is less than 5%. There are limited data to suggest that the complication rate is greater in patients who have had previous abdominal surgery resulting in altered bowel anatomy, especially with the retrograde route, and caution is advised when performing the procedure in this group of patients. A meta-analysis by Lipka and colleagues
130
showed complication rates of SBE and DBE were similar (overall adverse events: relative risk 1.41, 95% CI 0.32–6.3;
12 Domain: Number of procedures
In the absence of any evidence regarding the number of procedures required for training for individual certification of DAE competence, we were not able to set any minimum standard. Any recommendation in terms of the minimum annual number of procedures per endoscopist that are required to maintain adequate levels of quality, as well as which kind of training should be provided to beginners and/or to poor performers, would need to be based on an established strong association of poor quality with a minimum threshold number of procedures performed per year. Such data are currently unavailable. Nevertheless, after an extensive discussion the working group agreed on the following suggestion: (i) training should be provided only by experienced enteroscopists in units with a sufficient volume of work (50–100/year) to ensure an appropriate case mix, and trainee proficiency should be assessed by direct observation of procedures prior to being signed off on by their supervisor; (ii) combined training in capsule endoscopy and DAE may enhance lesion recognition and detection, and is encouraged in those intending to perform DAE.
Definitive data regarding the need for training and its potential benefits on diagnostic accuracy are lacking. In fact, there are no formal guidelines that state the minimum training requirements before performing DAE. According to an unpublished expert consensus on DBE, only advanced trainees should train in DAE. Advanced endoscopy skills should mean having enough experience both in diagnostic and therapeutic endoscopy, including hemostasis (endoclipping, argon plasma coagulation, and injection endotherapy), polypectomy and lesion resection, endoscopic balloon dilation, stenting, and/or ERCP. This approach is also supported by a recent trial conducted in high-volume centers in Japan that demonstrated that DBE can be safely carried out by advanced trainees under the supervision of an expert after a dedicated training program. 131
Based on available early publications, although performance will vary according to the individual's endoscopic skillset, at least 10–15 cases performed under expert supervision are necessary to achieve appreciable small-bowel insertion depth for antegrade DAE procedures. The retrograde insertion route is usually found to be more challenging, and at least 30–35 cases are needed to achieve an appreciable, effective insertion depth.132,133 However, these numbers are likely to be an underestimate of the experience required to achieve actual, effective clinical competence and the lack of a solid evidence base should be noted in this regard. The impact of SBCE knowledge and experience in DAE training remains unknown and warrants further study; albeit, it may reduce the training period as lesion appearance is similar for both modalities.
13 Domain: Patient experience

The acceptance of this performance measure is based on the strength of agreement with the following statements:
Patient comfort should be audited for all DAE procedures. (Statement number 28) Agreement: 90.9% Inadequate comfort levels should be audited against route of insertion, sedation, insufflation method, and endoscopist experience. (Statement number 29) Agreement: 90.9%
Patient comfort is a surrogate marker for procedure quality. Monitoring patient experience is feasible, yet it is not universal, and no standardized approach exists. The Global Rating Scale134,135 is the most validated questionnaire for assessing patient experience. Comfort of patients undergoing DAE is related to enteroscopist competence, available equipment, sedation protocol, and overall setup.
Three meta-analyses136–138 have demonstrated that the use of CO2 insufflation, when compared with air, is associated with reduced abdominal discomfort as measured on a visual analog scale for pain assessment at one hour (
Future research
The potential areas needing future research in SBCE and DAE are broad, encompassing pre-procedure, procedure, and postprocedure domains, and all research designed to address quality issues is to be encouraged and welcomed. However, through the process of developing these recommendations, important areas pertinent to procedure quality with an insufficient evidence base were highlighted that in our opinion warrant particular mention. Future research is required to address these issues, to strengthen the evidence base, and thereby support the refinement of some of the quality measures and the development of new ones going forward.
In relation to capsule endoscopy, the timing, selection, and use of different bowel preparation combinations remain issues. Despite numerous publications and several meta-analyses, the optimal approach to enhance not only visualization but also procedure completion and lesion detection in all patients and in those at risk of a poor-quality procedure remains debatable.
In addition to preparation, an acceptable diagnostic yield remains inconclusive for capsule endoscopy overall and for any given indication. As there is no true gold-standard comparator for capsule endoscopy, it is difficult to define its accuracy. Although correlation with subsequent DAE or surgery and long-term longitudinal follow-up studies offers some form of accuracy assessment, a robust approach to defining acceptable diagnostic tolerances for capsule endoscopy is required.
Similarly, diagnostic accuracy in capsule endoscopy is heavily reliant on individual reader and reading mode characteristics, but is without a simple measure to assess performance. As such, studies on reading and reader-related parameters (reading speeds, enhanced reading technologies, reader experience, prior training, reading/caseload volume, as well as novel parameters) should be a focus of future research.
With regard to DAE, further studies to help define accuracy are also needed. As with capsule endoscopy, currently there are no gold standards with which to compare DAE. There is no clear definition of a complete DAE, and no true understanding of the value of a negative diagnostic DAE or the likelihood of missed lesions overall or for any given indication. Although comparisons with capsule endoscopy and radiology can be helpful, they are not without potential flaws, and a more robust approach to accuracy assessment is required.
As with other endoscopy procedures, DAE is operator dependent and a clearer idea of the optimal training, competency assessment tools, including key performance indices, and the impact of experience and caseload are needed.
Finally, within the DAE arena there is a need to understand the advantages and disadvantages of different devices and their approaches, and to develop appropriate recommendations, if warranted, for their selection in a given clinical scenario, along with the development of specific quality measures where necessary.
Footnotes
In brief
The ESGE and United European Gastroenterology present a short list of performance measures for SBCE and DAE. For each procedure, six key measures and four minor metrics were proposed. A systematic and scientifically based methodology was applied to substantiate the proposed measures using the available evidence where possible. The adoption of these performance measures in all endoscopy services across Europe is recommended.
Supporting Information
Acknowledgments
The authors gratefully acknowledge the contributions of Dr Stuart Gittens, ECD Solutions, in the development and running of the web platform; all at Hamilton Services for project administrative support; the Scottish Intercollegiate Guidelines Network for hosting the critical appraisal module; and the Research Foundation-Flanders (FWO) for providing funding for Prof Raf Bisschops. UEG supplied co-funding and additional project governance to this endeavor.
Declaration of conflicting interests
C. Spada receives consultancy fees from Medtronic (2016 to present), and speakers and teaching fees and travel support from Olympus (2018–2019). D. McNamara was a member of the Colon Expert Group at the Medtronic session in Tarquinia. E.J. Despott has received honoraria from Fujifilm and Olympus (2017–2019) and academic and research funding from Fujifilm, Olympus, and Pentax Medical (2017–2019). S. Adler gives teaching seminars in capsule endoscopy sponsored by Medtronic (2015 to present). I. Fernández-Urién receives consultancy fees from Given Imaging, Covidien, and Medtronic (2010–2018). M. Keuchel has received speakers fees from Medtronic and Olympus (2002–2019), Given Imaging study support (2010–2013), and DGVS – guideline quality in endoscopy (2012–2015). M. McAlindon receives consultancy fees from Medtronic (2016–2019) and research support from Ankon Ltd and Intromedic Ltd (2017–2019). J.C. Saurin has provided consultancy for Intromedic Capsovision Medtronic (2014–2018). S. Panter has received support for advisory boards from Medtronic (from 2010), for a capsule course from Diagmed (from 2005), and for training guideline development from the Joint Advisory Group on GI Endoscopy training working group (from 2010). He was also a member of the Colon Expert Group at the Medtronic session in Tarquinia. C. Senore’s department received PillCAM2 Colon devices from Medtronic to conduct a comparative study (2014–2017); along with C. Belissario and S. Minozzi, he also received consultancy fees from ESGE to provide methodological expertise (PICOs evaluation, literature searches, and evidence summaries) for the present project (2014–2017). C. Bennett owns and works for Systematic Research Ltd, and received a consultancy fee from ESGE to provide scientific, technical, and methodological expertise for the present project (2014–2018). M. Bretthauer receives fees for being a member of the Norwegian government colorectal cancer screening advisory group (2012 to present) and receives funds from the American College of Physicians for editorial work for
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
