Abstract
Background
The use of an indwelling peritoneal catheter system in hospitalized patients with ascites could facilitate patient management by the prevention of repetitive abdominal paracentesis. Despite these possible benefits, the use of indwelling catheters is not widely established.
Objective
This retrospective study aimed to evaluate the feasibility, effectiveness and safety of the use of an indwelling catheter for ascites drainage in the clinical routine.
Methods
This retrospective study included all indwelling peritoneal catheter placements in our department in hospitalized patients with cirrhosis between 2014 and 2017.
Results
A total of 324 indwelling catheter placements for ascites in 192 hospitalized patients with cirrhosis were included. The catheter (7F, 8 cm) was placed ultrasound-assisted bed-side on the hospital ward. The technical success rate of the catheter placement was 99.7% (323/324). In 17.5% (64/324) the catheter was placed to optimize ascitic drainage prior to an abdominal intervention (e.g. transjugular intrahepatic portosystemic shunt). The median time of catheter retention was 48 hours (8–168 hours) and the median cumulative amount of drained ascites 8000 ml (550–28,000). The most common adverse event was acute kidney injury (49/324, 15.1%); the risk was particularly higher in patients with a Model for End-Stage Liver Disease (MELD) score ≥ 16 (p = 0.028; odds ratio 2.039). Ascitic fistula after catheter removal was observed in 9.6% (31/324). Catheter-related infections occurred in 4.3% (14/324), and bleeding was documented in three cases (0.8%) with one major bleeding (0.3%).
Conclusion
The placement of an indwelling catheter for repetitive ascitic drainage in hospitalized patients with cirrhosis can be established in the clinical routine, facilitating patient management. High-MELD patients especially have to be monitored for acute kidney injury.
Key summary
Established knowledge
Repetitive large-volume paracentesis is part of the clinical routine in the management of hospitalized patients with decompensated cirrhosis. The short-term use of indwelling peritoneal catheters could save repeated singular punctures and improve patient management, although data on effectiveness and safety are lacking.
Key findings of the study
Indwelling peritoneal catheters for repeated volume relief for hospitalized patients with cirrhosis and ascites can be established in the clinical routine. Repetitive ascitic drainage by the catheter is effective and can be used as a preparation for abdominal interventions like TIPS or liver biopsy. Acute kidney injury, especially in patients with high Model for End-Stage Liver Disease score, was the most frequent adverse event that warrants comprehensive clinical monitoring and further prospective evidence.
Introduction
First decompensation episodes occur in 5% to 7% of patients with cirrhosis per year and resemble a shift to a systemic disease with massive increased morbidity and mortality. The deteriorated prognosis is driven by cirrhosis-associated complications like gastrointestinal hemorrhage, hepatorenal syndrome or spontaneous bacterial peritonitis (SBP) in patients with ascites. 1 Ascites is a common clinically overt sign of decompensation that is associated with a mortality of 50% within two years. 1 Patients with ascites are not only prone to SBP but also experience a massive deterioration of quality of life due to the repetitive need for large-volume paracentesis (LVP) and hospitalization. 1 Moreover, the removal of large volumes can lead to a condition known as post-paracentesis circulatory dysfunction (PPCD), clinically mainly associated with renal impairment and hyponatremia. The risk for PPCD can be reduced effectively by the substitution of albumin, which is recommended by international guidelines.2,3 In clinical experience repetitive LVP, especially for hospitalized patients, becomes necessary. Furthermore, residual ascites after LVP represent an obstacle for several interventional procedures like biopsy of liver lesions or transjugular intrahepatic portosystemic shunt (TIPS) placement, impeding interventional success.4–6 Therefore, the use of indwelling peritoneal catheters that remain in the abdominal cavity for several days enables repeated volume relief without the need for repetitive punctures and minimizes the amount of residual ascites. These ascites catheters can be placed ultrasound-assisted bed-side on the hospital ward without additional effort compared with routine paracentesis. Although data on the use of indwelling catheters for several days with repetitive fluid drainage in hospitalized patients are lacking, this approach could be associated with several advantages: (a) saving repetitive punctures, (b) reducing the risk of PPCD by the gentle removal of large volumes over several days and (c) increasing the safety/success of interventional procedures by the complete removal of ascites via the catheter, for example, directly before the intervention. On the contrary any indwelling peritoneal plastic catheter could be associated with an increased number of adverse events in this serious ill patient cohort, that is, (i) catheter-associated infections, (ii) bleeding complications or (iii) ascitic fistulas.
Because indwelling peritoneal catheters with repetitive fluid removal for hospitalized patients with ascites are used as clinical routine in our department, data from all consecutive patients who received such a catheter between January 2014 and December 2017 were analyzed retrospectively and followed until hospital discharge. The aim of this study was to investigate feasibility, effectiveness and safety of the indwelling peritoneal catheter in hospitalized patients with ascites.
Materials and methods
In this retrospective study all consecutive cases with an indwelling peritoneal catheter placement for ascites drainage were included while hospitalized in the Frankfurt University Hospital, Department of Medicine 1, Germany, between January 2014 and December 2017. Inclusion criteria were (a) peritoneal catheter placement during hospitalization, or (b) grade 3 ascites due to liver cirrhosis. Figure 1 illustrates the eligibility criteria for either standard paracentesis or indwelling catheter placement in our department. Patients with ascites for other reasons (e.g. malignancy and loculated ascites) were not included. The indication for catheter placement and the daily amount of drainage volume were at the discretion of the treating physician. No general threshold of maximum drainage volume was defined. In general, patients with clinical signs of SBP or an episode of SBP shortly before the actual hospital stay were not considered eligible for catheter placement, although a history of SBP in general was not an exclusion for catheter placement. Albumin after LVP was substituted according to the European Association for the Study of the Liver (EASL) guidelines (8 g/l) when drainage volume exceeded 5 l or in case of hepatorenal syndrome. A total of 324 catheter cases matching inclusion criteria in 192 patients were included in the study. Cases were followed from the time of catheter placement until discharge from the hospital. Demographic data, lab values and follow-up data were collected from the electronic patient files. Complications of the peritoneal catheter placement were defined as follows: Ascitic fistula was defined as continuous discharge of ascites at the puncture site after removal of the catheter. Catheter-associated peritonitis was assumed when leukocytes in ascites increased above 500/µl or neutrophilic granulocytes above 250/µl within one week of catheter placement or removal. Acute kidney injury (AKI) was defined according to EASL guidelines as either an absolute increase in creatinine of more than or equal to 0.3 mg/dl in less than 48 hours, or by a percentage increase in creatinine of more or equal to 50% (1.5-fold from baseline) in less than seven days.2,7,8 Baseline creatine was measured the day of catheter placement. Bleeding was defined as clinically evident bleeding together with decrease of hemoglobin levels of more than 1 g/dl.
Flowchart of patient selection for either large-volume paracentesis (LVP) or indwelling peritoneal catheter. All consecutive patients between January 2014 and December 2017 were included in the study. Grade 3 ascites: Large or gross ascites that provokes marked abdominal distension. SBP: spontaneous bacterial peritonitis; TIPS: transjugular intrahepatic portosystemic shunt. *Patients with malignant ascites were not included in the study although this is not a general contraindication for an indwelling peritoneal catheter.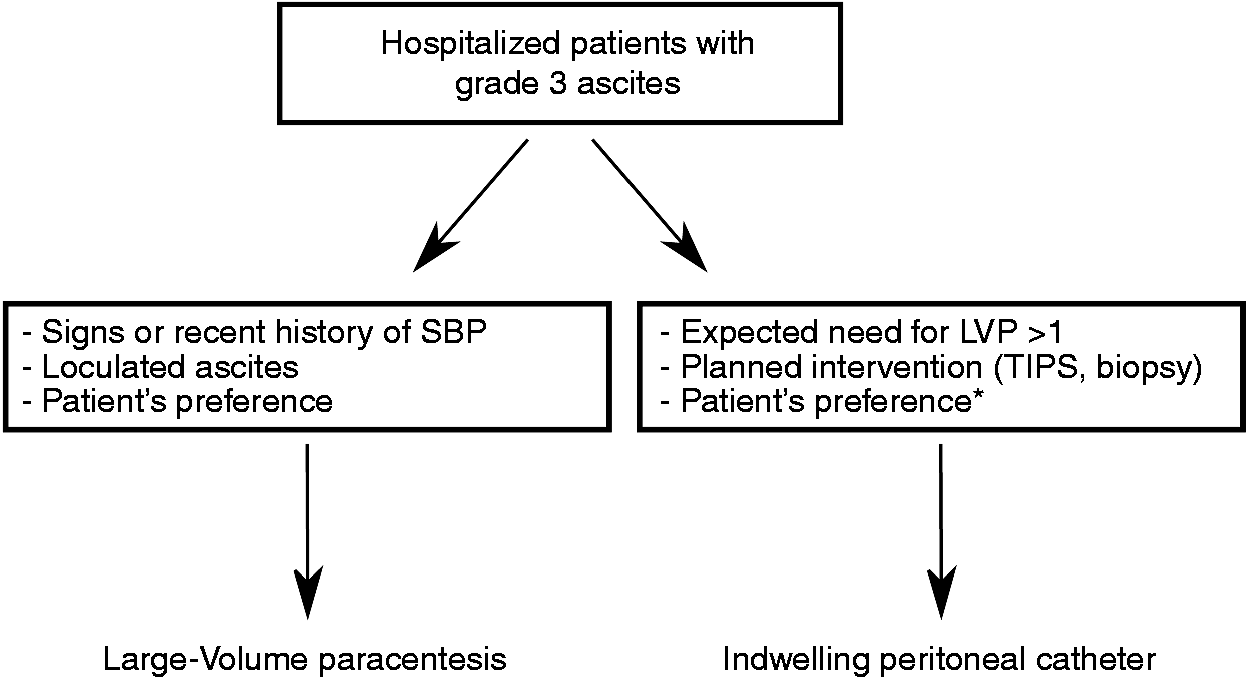
This study was approved by the institutional review board of the Frankfurt University Hospital (Ethikkommission der Goethe-Universität Frankfurt, date: August 21, 2018; decision 329/18). Written, informed consent for catheter placement was obtained from each patient prior to the procedure. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Catheter and procedure
In all cases the catheter was placed ultrasound-assisted bed-side on the hospital ward under sterile conditions after application of local anesthesia. A 7 F plastic straight catheter with a length of 8 cm was used (PerkuStay Drainage Catheter, Schlottman type, Pflugbeil, Germany) (Figure 2). The catheter was secured with a single suture and sterile bandage.
Preparation for peritoneal catheter placement. (a) 7 F peritoneal catheter with needle, (b) connection tube, (c) suture, (d) needle holder, (e) scalpel, (f) drainage bag.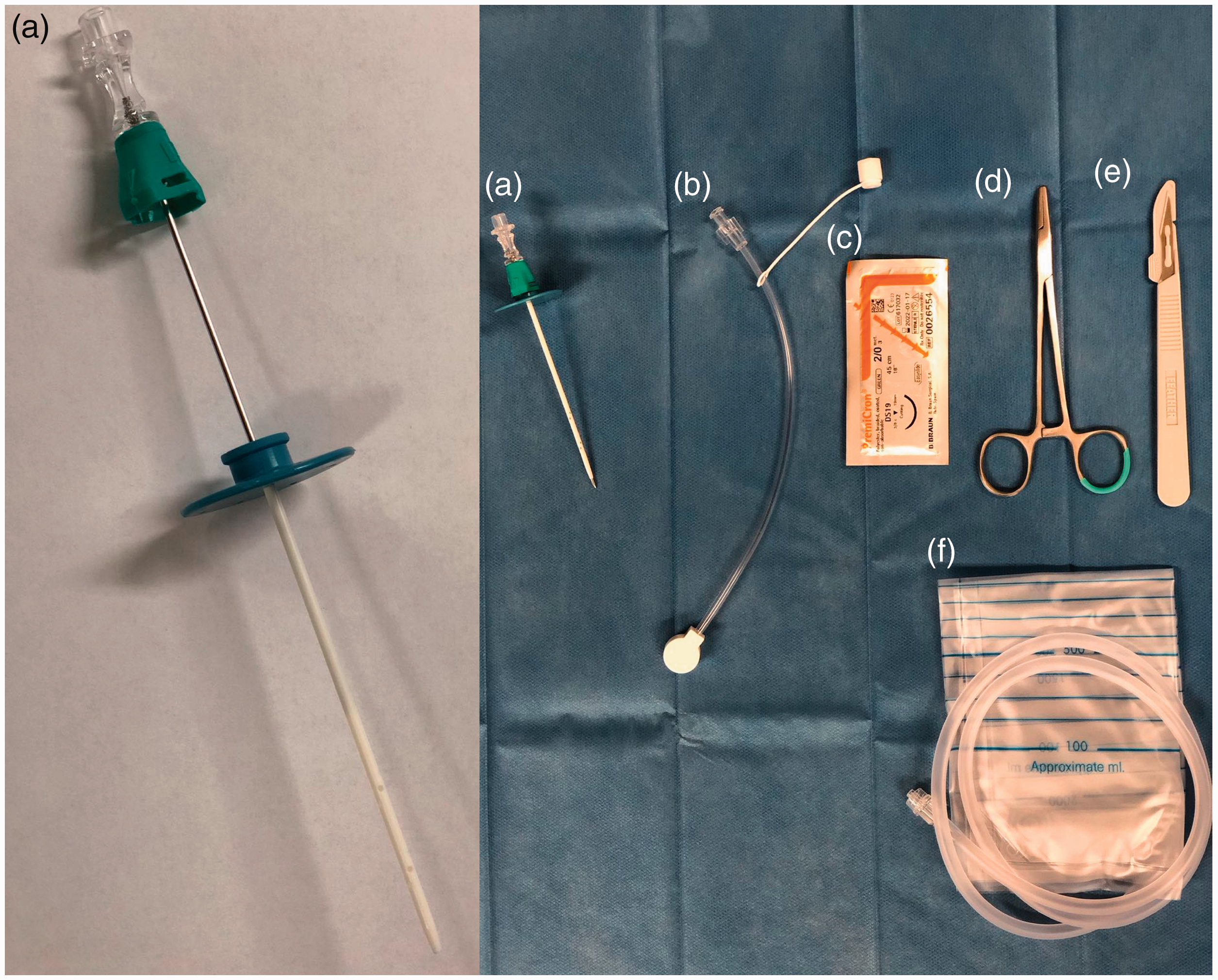
Statistical analysis
Data collection, data management and statistical analyses were performed with the SPSS software package, release 17.0 (SPSS Inc, Chicago, IL, USA). For the regression analysis to identify factors associated with complications of the catheter placement, a logistic regression model was used. For comparison of the rate of AKI in the two Model for End-Stage Liver Disease (MELD) groups, Fisher exact test was used. P values < 0.05 were considered statistically significant.
Results
Patient population
Demographics of study population by time of ascites catheter placement.
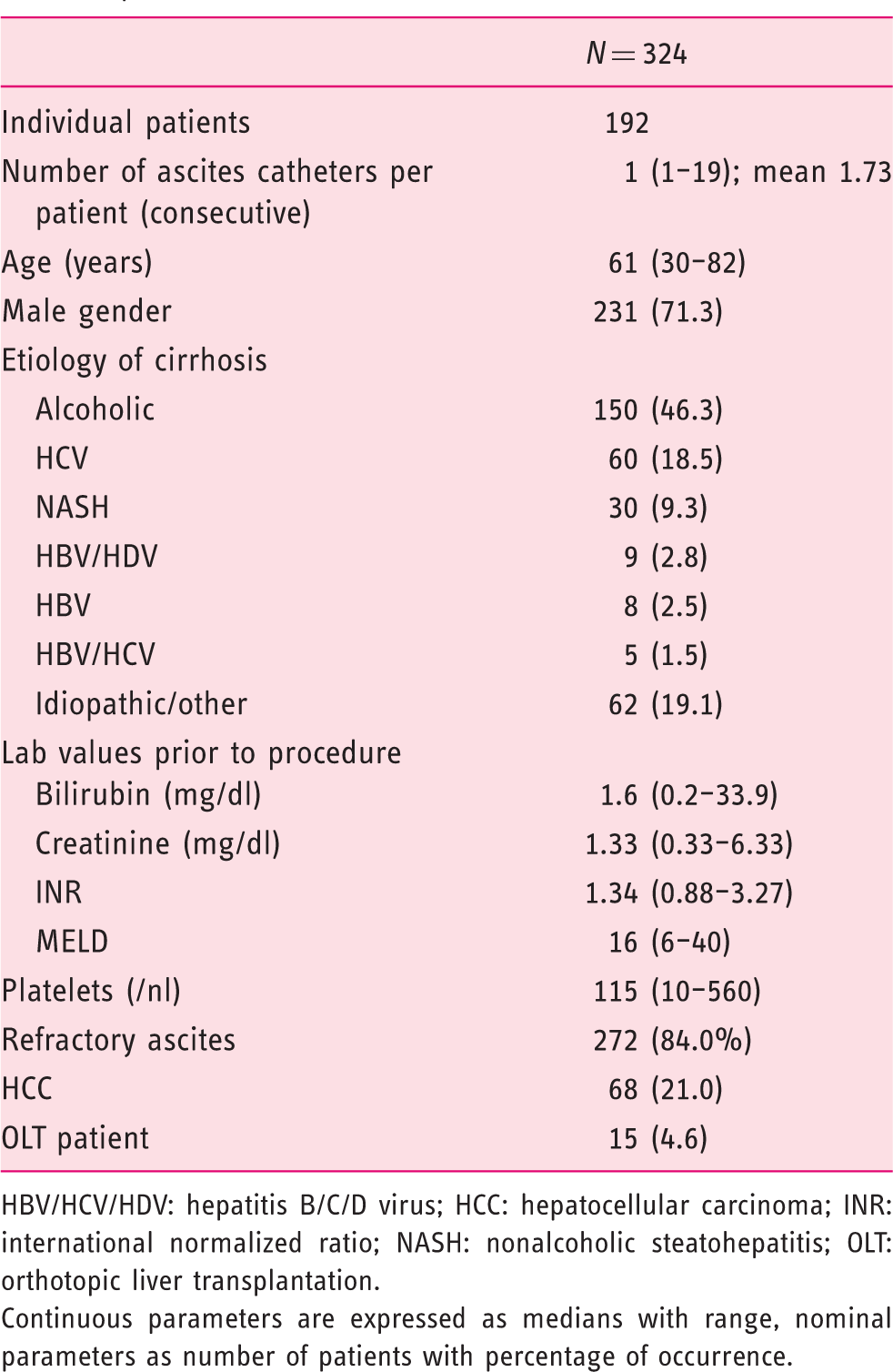
HBV/HCV/HDV: hepatitis B/C/D virus; HCC: hepatocellular carcinoma; INR: international normalized ratio; NASH: nonalcoholic steatohepatitis; OLT: orthotopic liver transplantation.
Continuous parameters are expressed as medians with range, nominal parameters as number of patients with percentage of occurrence.
Effectiveness
Efficacy of peritoneal catheter placement.
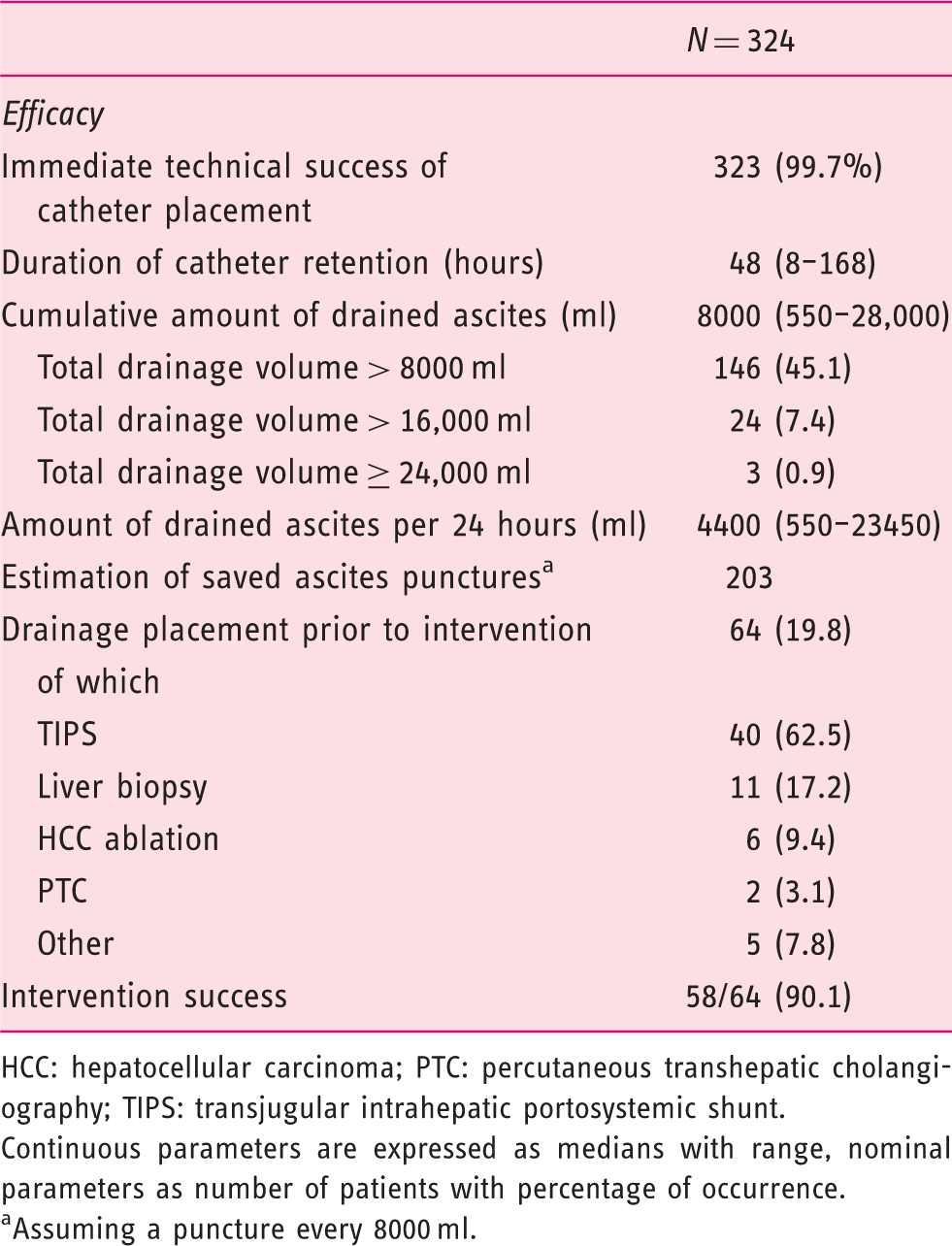
HCC: hepatocellular carcinoma; PTC: percutaneous transhepatic cholangiography; TIPS: transjugular intrahepatic portosystemic shunt.
Continuous parameters are expressed as medians with range, nominal parameters as number of patients with percentage of occurrence.
Assuming a puncture every 8000 ml.
The immediate technical success rate of the catheter placement was 99.7% (323/324). One patient suffered splenic injury during the catheter placement (see below). The median time of catheter retention was 48 hours with a maximum of 168 hours. A median of 8000 ml ascites were drained per catheter with a median drainage of 4400 ml per 24 hours. The ascites drainage volume exceeded 8000 ml in 146 cases (45.1%), 16,000 ml in 24 cases (7.4%) and 24,000 ml in three cases (0.9%). When assuming conservatively, the need for an additional paracentesis every 8000 ml by an estimated number of 202 additional ascites punctures were saved by the use of the peritoneal catheter.
In 64 (19.8%) cases the catheter was placed prior to an interventional procedure to improve intervention conditions by a preferably complete removal of ascites. This preparation was performed particularly before transjugular intrahepatic portosystemic shunt (TIPS) placement (40/64, 62.5%) and biopsy of liver lesions (suspected HCC) (11/64, 17.2%). The intervention was successful in 58 of 64 cases (90.1%).
Safety
Safety of peritoneal catheter placement.
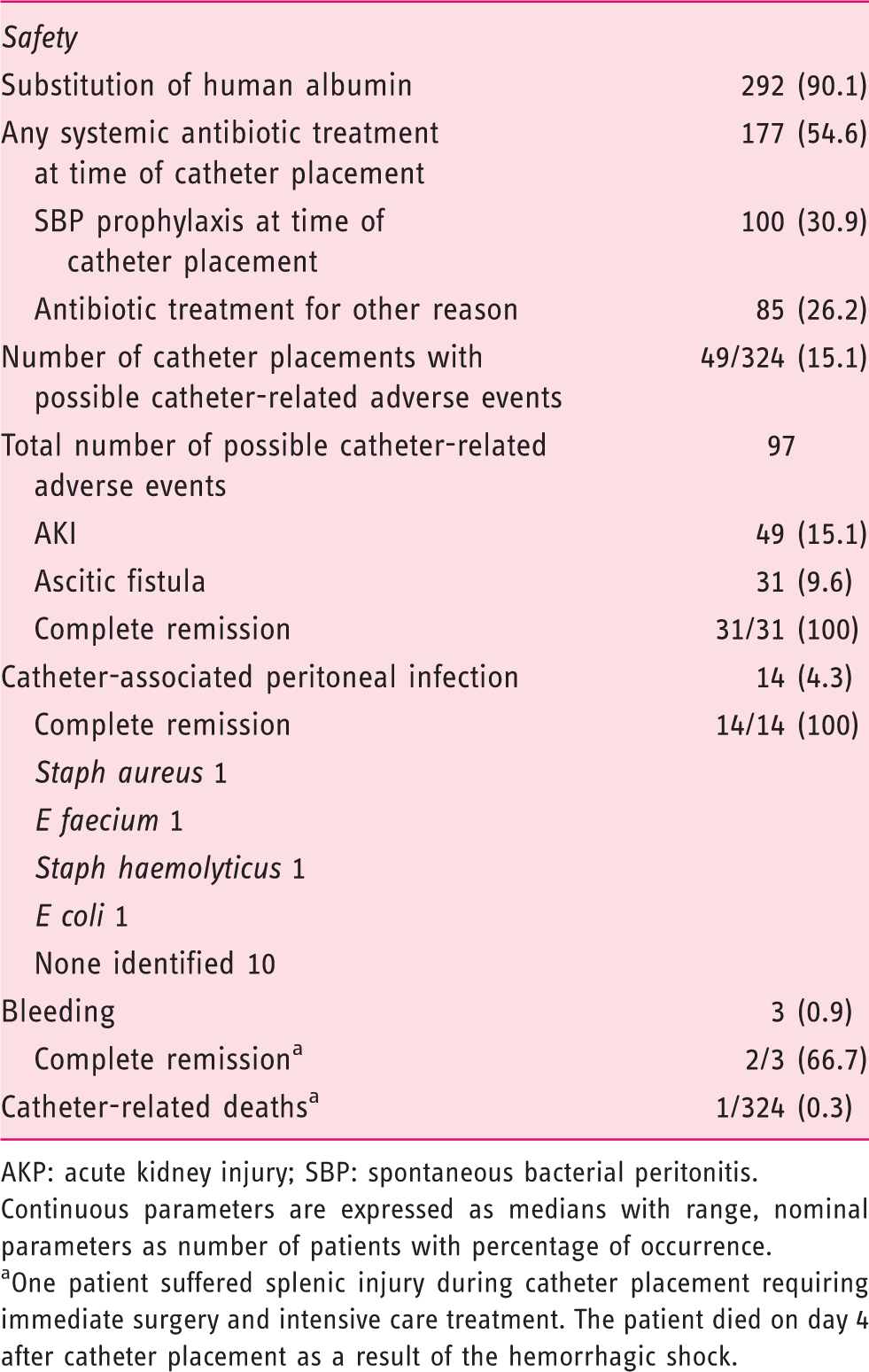
AKP: acute kidney injury; SBP: spontaneous bacterial peritonitis.
Continuous parameters are expressed as medians with range, nominal parameters as number of patients with percentage of occurrence.
One patient suffered splenic injury during catheter placement requiring immediate surgery and intensive care treatment. The patient died on day 4 after catheter placement as a result of the hemorrhagic shock.
Albumin substitution after large-volume ascites drainage was performed in the majority of cases (292/324, 90.1%). Patients not receiving albumin had drainage volumes less than 5 l.
More than half of the patients (177/324, 54.6%) received antibiotic treatment at the time of the catheter placement, of which 100 cases were permanent treatment for SBP prophylaxis (ciprofloxacin or norfloxacin). The remaining 77 cases received antibiotic treatment during the whole time of catheter retention for reasons other than SBP. The treatment consisted of 31 cases ceftriaxone, 22 imipenem, nine piperacillin/tazobactam, seven ciprofloxacin, three meropenem, two metronidazole, one cefotaxime, one cefuroxime and one levofloxacin.
Possible catheter-related adverse events were documented in 82 of 324 cases (25.3%) with a total of 97 possible catheter-related adverse events.
AKI (according to actual EASL definition) was the most common adverse event with 49 of 324 catheter placements (15.1%). An increased serum creatinine to a value greater than 1.5 mg/dl occurred in only five cases (1.5%).
Ascitic fistula after removal of the catheter was documented in 31 of 324 catheter placements (9.6%). Almost all cases resolved spontaneously after one to three days by application of a pressure dressing; one patient needed a single suture that successfully occluded the fistula.
Possible catheter-related infections were all peritoneal infections with an increase of leukocytes in the ascites. No localized tissue infections of the abdominal wall surrounding the catheter site were observed. A total of 14 (4.3%) possible catheter-related infections were documented. In four cases the causing bacterial pathogen was detected by ascites culture (Staph aureus, E faecium, Staph haemolyticus, E coli) while in the remaining 10 cases no pathogen was identified. The infection resolved in all 14 cases after antibiotic treatment.
Bleeding complications were documented in three cases. One venous bleeding from the punctual site after removal of the catheter stopped after application of a pressure dressing for 24 hours. In one case a larger bleeding into the abdominal wall occurred. Again the bleeding stopped after application of pressure. A large hematoma of the abdominal wall resolved slowly afterward.
One fatal bleeding occurred after the spleen was injured by the catheter needle during catheter placement. The patient had pronounced splenomegaly and coughed exactly at the time when the needle was inserted into the abdominal cavity causing the splenic injury. The bleeding was noticed immediately and emergency surgery was performed promptly after computed tomography scan of the abdomen. The patient died four days after the complication in the intensive care unit of multiorgan failure as a consequence of the hemorrhagic shock. The patient was a 76-year-old woman with NASH cirrhosis. Her MELD was 22 at the time of the catheter placement (international normalized ratio; 1.42, platelets 73/nl).
Association of patient and procedural factors with complications
Association of patient factors with adverse events.
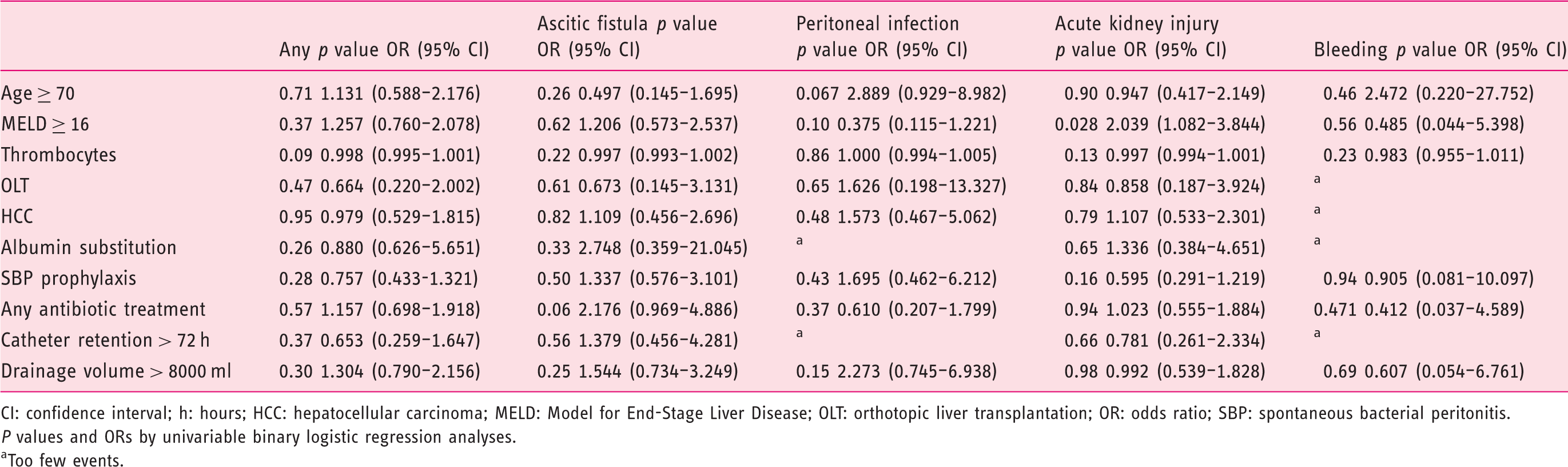
CI: confidence interval; h: hours; HCC: hepatocellular carcinoma; MELD: Model for End-Stage Liver Disease; OLT: orthotopic liver transplantation; OR: odds ratio; SBP: spontaneous bacterial peritonitis.
P values and ORs by univariable binary logistic regression analyses.
Too few events.
Overall no association of age, MELD score, orthotopic liver transplantation status, HCC, thrombocytes, albumin substitution, antibiotic treatment, time of catheter retention or drainage volume with catheter-related complications was observed. In the separate analysis of specific complications, only MELD score ≥ 16 was associated with the risk for AKI (p = 0.028, odds ratio (OR) 2.039, 95% confidence interval (CI) 1.082–3.844). In patients with a MELD score < 16 the rate of AKI was 10.6% compared with 19.5% in patients with a MELD score ≥ 16 (p = 0.03).
Antibiotic therapy was not associated with fewer infectious complications (p = 0.37, OR 0.610, 95% CI 0.207–1.799). Furthermore, large drainage volumes (>8000) ml were not associated with AKI (p = 0.98, OR 0.992, 95% CI 0.539–1.828).
Discussion
The present study is to our knowledge the first to analyze the feasibility, effectiveness and safety of the use of an indwelling peritoneal catheter in the clinical routine for hospitalized patients with ascites for repetitive ascites drainage. The main findings of the study are that catheters can be used reliably in the clinical routine, saving repeated paracentesis and allowing reliable ascites removal prior to abdominal interventions like TIPS or biopsy. Still, a possible increased number of catheter-related AKI, especially in patients with high MELD scores, have to be considered as well as the risk of serious bleeding complications.
The technical success rate of the catheter placement was high (99.7%), which is in line with previous studies on the use of pigtail catheters for singular paracentesis.9,10 The effectiveness seems to be superior to the reported rate of standard paracenteses by venous catheters with only slightly higher complication rates.9–11 Even more experience exists with the longer implementation of tunneled indwelling peritoneal catheters for outpatients with refractory ascites.12,13
A drainage volume of higher than 8000 ml was achieved in more than half of the patients, the mean cumulative drainage volume being 9028 ml. In a large, retrospective, multicenter study that analyzed more than 4000 paracenteses, the mean drainage volume was 4900 ml. 11 Accordingly, other studies report the drainage volume by singular paracentesis most commonly lying between 4 l and 6 l.14,15 Additionally, the risk for PPCD seems lower when the drainage volume is limited to 8000 ml. Based on these data and our clinical experience, we assume that repeated paracenteses can be saved by the use of indwelling catheters although the study design cannot prove this finding.
Other studies on catheters for only singular paracentesis documented an increase of drainage volume by only 250 ml compared with the standard use of indwelling venous catheters.9,10 Obviously, the advantage of higher drainage volumes by the catheter is achieved when the fluid removal is performed more than once over several days.
The preparation for abdominal interventions by the catheter was excellent, with an intervention success rate of greater than 90%. Because ascites can impede the success of TIPS placement4,5 and liver biopsy, 6 the reduction of residual ascites by an indwelling catheter that remains during the procedure could be an advantageous approach for these interventions.
Regarding the safety analysis, it has to be taken into account that all complications were assumed to be catheter related that fell into the time of catheter placement up to one week after removal. Furthermore, standard LVP is performed as an outpatient procedure in our clinic whereas catheter placement is performed only in hospitalized patients. This might have led to an over- rather than underestimation of catheter-related AKI and infection.
The safety profile of the procedure is clouded by a fatal splenic injury by the catheter needle during catheter placement. The patient died from the hemorrhagic shock after four days although emergency treatment and surgery were initiated immediately. Local bleeding complications were reported in large studies of routine paracentesis in 2.3% to 2.9% of cases11,13 while severe intraperitoneal hemorrhage was reported in 0.19% to 1.0% and death due to hemorrhage in 0.016% to 0.39% of cases.13,16 Although being in line with reported complication and mortality rates for regular paracentesis, a higher risk for bleeding or organ damage might be associated with the longer catheter needle, warranting a careful selection and thorough local anesthesia of the puncture site to optimize safety conditions. To our knowledge no case of fatal splenic injury by paracentesis has been reported in the literature so far, assuming it to be a very rare complication. No patient or procedural factors were associated with the risk for bleeding, which can be attributed to overall low rate of bleeding complications. The rate of AKI possibly associated with large-volume drainage was rather high with 15% of catheter placements. This high number is most likely driven by the strict EASL conform definition of AKI that was not used in previous studies though it is sensitive and reliable. Additionally, the reported AKI rate for any hospitalized patient with cirrhosis was reported to be 9.6%, which is not far away from our finding. 17 A less-rigorous threshold of > 1.5 mg/dl absolute creatinine identified only five cases, all of which resolved rapidly with conservative treatment. After LVP the rate of PPCD with renal impairment is reported to be between 0% and 5.9%,18,19 which is probably underestimated. The association of MELD with AKI in our cohort should warrant monitoring for AKI in high-MELD patients after drainage placement.
We report a prolonged ascites outflow from the puncture site (ascitic fistula) in 9.6% of patients, which is slightly higher than reported for standard paracentesis (3.2% to 5.0%).11,13 This finding could be caused by the larger diameter of the catheter and the longer catheter retention. Still, the outcome of this mild complication was benign with spontaneous occlusion of the fistula within one to three days in almost all cases (only one requiring suture).
Possible catheter-related infections occurred in 4.3% of patients, all of whom recovered completely after antibiotic treatment. For routine paracentesis, the rate of procedure-related infection is reported to be 0.6% to 4.3%.11,13,20 It has to be taken into account that more than half the patients were on antibiotic treatment during the time of catheter retention, in most cases for SBP prophylaxis, which probably contributes to that low number.
Patients with advanced liver disease are particularly prone to infections, thus drained ascites via the catheter should be tested for leukocyte and neutrophil counts repeatedly during the time of catheter retention to detect infectious complications early. Although the time of catheter retention was not associated with infectious complications, we recommend a maximum of 72 hours. A prophylactic antibiotic treatment cannot be generally recommended based on our data. In the outpatient setting tunneled catheters that adduce a preservation of infection are an already established device. 21
The strengths of the presented study lie in its innovative nature on a field with lacking data and the large number of cases. Thus, questions on feasibility, effectiveness and safety could be addressed adequately. Still, the study has several limitations that need to be addressed. Owing to the retrospective nature of the study, a direct comparison with standard paracentesis was not possible. This has to be addressed in prospective randomized trials. Furthermore, the data quality relies on the completeness of documentation in the patient file. Accordingly, complications might have been missed because of incomplete documentation or when they occurred after hospital discharge. Especially all aspects of PPCD, like hyponatremia or hemodynamic monitoring, could not be taken into account retrospectively because of lack of data. Of course, the estimated number of saved paracenteses by the use of the catheter is hypothetical, although, based on published data on mean amounts of fluid drainage by singular paracentesis and our clinical experience, the number of paracenteses can most likely be reduced efficiently. The analyses are based on individual catheter placements; patients were included several times whenever a new catheter was placed during a new hospital admission. This might be a source of bias although the actual clinical parameters of every new case were used for the analyses. Finally, patient opinion on the use of catheters could not be addressed because a patient survey was not performed.
In conclusion, repetitive ascites drainage by an indwelling peritoneal catheter proved to be feasible and effective in the clinical routine. The safety profile was satisfactory in many respects but AKI especially in patients with high MELD score and bleeding complications have to be taken into account and warrant further prospective evidence.
Footnotes
Declaration of conflicting interests
The Authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethics approval
This study was approved by the institutional review board of the Frankfurt University Hospital (Ethikkommission der Goethe-Universität Frankfurt, date: August 21, 2018; decision 329/18). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee.
Informed consent
Written, informed consent for catheter placement was obtained from each patient prior to the procedure.
