Abstract
Background
Refractory coeliac disease type II (RCDII) frequently transforms into an enteropathy-associated T-cell lymphoma (EATL) and therefore requires intensive treatment. Current evaluated treatment strategies for RCDII include cladribine (2-CdA) and autologous stem cell transplantation (auSCT).
Objective
The purpose of this study was to evaluate long-term survival and define clear prognostic criteria for EATL development comparing two treatment strategies.
Methods
A total of 45 patients were retrospectively analysed. All patients received 2-CdA, after which they were either closely monitored (monotherapy, n = 30) or a step-up approach was used including auSCT (step-up therapy, n = 15).
Results
Ten patients (22%) ultimately developed EATL; nine of these had received monotherapy. Absence of histological remission after monotherapy was associated with EATL development (p = 0.010). Overall, 20 patients (44%) died with a median survival of 84 months. Overall survival (OS) within the monotherapy group was significantly worse in those without histological remission compared to those with complete histological remission(p = 0.030). The monotherapy group who achieved complete histological remission showed comparable EATL occurrence and OS as compared to the step-up therapy group (p = 0.80 and p = 0.14 respectively).
Conclusion
Histological response is an accurate parameter to evaluate the effect of 2-CdA therapy and this parameter should be leading in the decisions whether or not to perform a step-up treatment approach in RCDII.
Keywords
Introduction
Coeliac disease (CD) is a chronic immune-mediated enteropathy. Once patients are diagnosed with CD, a life-long gluten-free diet (GFD) is the only accepted treatment. In the majority of patients compliant to a GFD, clinical remission is induced and histological abnormalities resolve. Nevertheless, in approximately 0.5–1% of patients histological abnormalities and clinical symptoms remain or reoccur despite a strict GFD. 1 After excluding other causes of villous atrophy these patients are diagnosed as refractory coeliac disease (RCD). 2
Based on phenotypical analysis of intra-epithelial lymphocytes (IELs) RCD patients are either diagnosed with RCD type I (RCDI) or RCD type II (RCDII) characterised by respectively the absence or the presence of aberrant IELs. 3 In contrast to RCDI, RCDII should be defined as low-grade intraepithelial lymphoma and frequently transforms into an aggressive enteropathy associated T-cell lymphoma (EATL) with dismal prognosis.4–6 Therefore, it is of utmost importance to evaluate treatment strategies for RCDII in order prevent or delay progression to an overt EATL.
Unfortunately, to date there is no standardised treatment approach for RCDII. RCDII is, at least in part, resistant to most evaluated therapies so far.7–9 Previous reports from our centre showed that cladribine (2-CdA) therapy is feasible and well tolerated for RCDII.10,11 Nevertheless, 2-CdA did not prevent EATL development in all treated patients. 11
In patients with RCDII refractory to conventional 2-CdA treatment, autologous haematopoietic stem cell transplantation (auSCT) is an increasingly accepted effective treatment option. AuSCT appears to be feasible and safe and might result in clinical remission in 2-CdA-failures.12,13 Nevertheless, little is known concerning sustainability of clinical and histological remission after long-term follow-up and the possible risk of developing EATL. Furthermore, no criteria for when to proceed to auSCT therapy are available. Therefore, we evaluated clinical response, EATL development and survival in RCDII patients treated with either 2-CdA monotherapy or 2-CdA-auSCT step-up therapy in order to evaluate which response parameters should be leading in a decision whether or not to proceed to auSCT and to determine factors associated with EATL development and survival.
Material and methods
Patients
The medical files of 58 RCDII patients were retrospectively studied. This analysis evaluates all RCDII patients diagnosed between 1998–2014 in the VU Medical Centre who received at least one cycle of 2-CdA and includes the RCDII patients of the open-label cohort studies performed by Tack et al.11,13 Patients with RCDI, uncharacterised CD, insufficient data, EATL at RCDII diagnosis or patients without intention to receive at least one cycle of 2-CdA were excluded. In both regimens patients from outside of The Netherlands were included.
Clinical evaluation and diagnosis of RCDII
In the case of suspicion of RCD (due to persisting or recurring CD symptoms) a standard diagnostic work-up was performed including clinical, laboratory, serological and endoscopic evaluation. Dietary compliance and nutritional status was evaluated by a specialised dietician (NJW). 14 Patients were classified into three groups based on baseline body mass index (BMI); less than 18.5 kg/m2 (underweight), 18.5–25.0 kg/m2 (normal weight) and more than 25 kg/m2 (overweight) according to the definition of the World Health Organization in 2000. 15 Nutritional support was started when indicated. When inadvertent gluten ingestion was reasonably excluded, the initial CD diagnosis was re-evaluated and confirmed (i.e. using patients' history and confirmation of human leukocyte antigen (HLA) DQ2 or DQ8). Laboratory evaluation included haematologic and liver function evaluation. Hypoalbuminaemia and anaemia were evaluated using the local laboratory reference values and were defined as <35 g/l and <7.5 mmol/l respectively. In all patients, fresh duodenal biopsies for histological analysis and flow cytometric immunophenotyping of IELs were taken during upper endoscopy. When duodenal biopsies revealed persisting villous atrophy, histological and clinical evaluation focussed on identifying other causes of villous atrophy. 16 When these were excluded, the patient was diagnosed with RCD. The clinically validated cut-off value of 20% aberrant IELs (lacking surface CD3 and generally CD8 but expressing intracellular CD3) detected by flow cytometric analysis was used to distinguish RCDI (<20%) and RCDII (>20%). 17
Diagnosis of EATL
At RCDII diagnosis, EATL was excluded by several imaging techniques including computed tomography (CT), whole-body positron emission tomography (PET-CT), magnetic resonance imaging enteroclysis (MRE), 18 upper gastrointestinal endoscopy, video capsule endoscopy (VCE) 19 and/or double-balloon enteroscopy (DBE). 20 When RCDII patients showed clinical deterioration or symptoms suggestive of lymphoma development during follow-up, patients were evaluated on having an EATL using the fore-mentioned imaging techniques. In those patients who developed or died of EATL, the diagnosis was established according to the World Health Organization Classification of tumours of hematopoietic and lymphoid tissues. 21
Treatment protocol
Patients were hospitalised, and 2-CdA was given (0.1 mg/kg) intravenously for two hours daily for five days as previously described. 11 When indicated, patients had supplemental folic acid, vitamin B12, and/or iron. Patients aged <70 years and diagnosed with RCDII who showed no clinical, histological or immunological response (i.e. persisting diarrhoea and weight loss, persisting high percentages of aberrant IELs and/or the absence of histological remission) after 2-CdA were intended to receive auSCT. This step-up approach was based on expert opinion since no criteria for 2-CdA response exist, depending on the judgement of the treating physicians. The auSCT regimen consisted of mobilisation using Granulocyte-colony stimulating factor (G-CSF), a conditioning regimen with fludrabine and intermediate melphalan, the exclusion of EATL after leukapheresis, followed by auSCT (as previously described by Tack et al.). 13 The purpose of this conditioning regimen was both intensive T-cell depletion and myeloablation. Contraindications for auSCT were age of, or above, 70 years, the presence of comorbidity and/or unsuccessful leukapheresis.
Follow-up and criteria of response
A clinical assessment was carried out at three-month intervals during the first year, thereafter at six-month intervals. Clinical evaluation included evaluation of signs and symptoms, BMI and possible complications of treatment. Clinical remission is defined as complete disappearance of all clinical symptoms at the end of follow-up. For example, the complete disappearance of diarrhoea. When a patient failed to achieve clinical remission but showed no increase of previous symptoms and no new symptoms, response was defined as stable disease. Progressive disease was defined by any new symptoms or increase of the previous symptoms. Patients with progressive symptoms including persisting diarrhoea and progressive weight loss without the evidence of EATL were diagnosed as suffering from progressive refractory disease. When a patient was asymptomatic at diagnosis, clinical remission during follow-up was not scored. BMI was classified according to the fore-mentioned categories. Laboratory and immunological evaluation was performed and classified as described above. Immunological response was evaluated using the absolute decline or rise in percentages of aberrant IELs. Histological response was either defined as complete or no histological remission. Complete histological remission is defined as a normalisation of the architecture of the small intestinal mucosa, classified as Marsh 0 or I lesion. No histological remission as Marsh II and III according to the modified Marsh criteria.22,23 Response was defined as response at the last follow-up closest to may 2014, or as response at the moment patients died or were lost to follow-up. All response parameters were calculated in comparison with baseline values (T = 0). In addition, OS and EATL occurrence were evaluated during follow-up. OS was defined as the time from diagnosis to death. Surviving patients were censored at the end of follow-up.
Statistical analysis
In general continuous variables were summarised with standard descriptive statistics including means, standard deviations (SDs), medians and ranges. The flow cytometric data were analysed using the Cellquest Pro software program (Becton Dickinson). Continuous variables were compared using the independent samples and paired t-tests or the nonparametric Mann-Whitney and Wilcoxon signed-rank test. Categorical variables were compared using Fisher’s exact test or Chi-square test. Survival was analysed using Kaplan-Meier curves and significance compared using the log-rank test. Hazard ratios (HRs) for time to development of EATL were computed with a Cox proportional hazard model. Values of p less than 0.05 were considered statistically significant. All analyses were performed in SPSS 20 (IBM Corp., Armonk, New York State, USA).
Results
Patients
Baseline characteristics at refractory coeliac disease type II (RCDII) diagnosis.
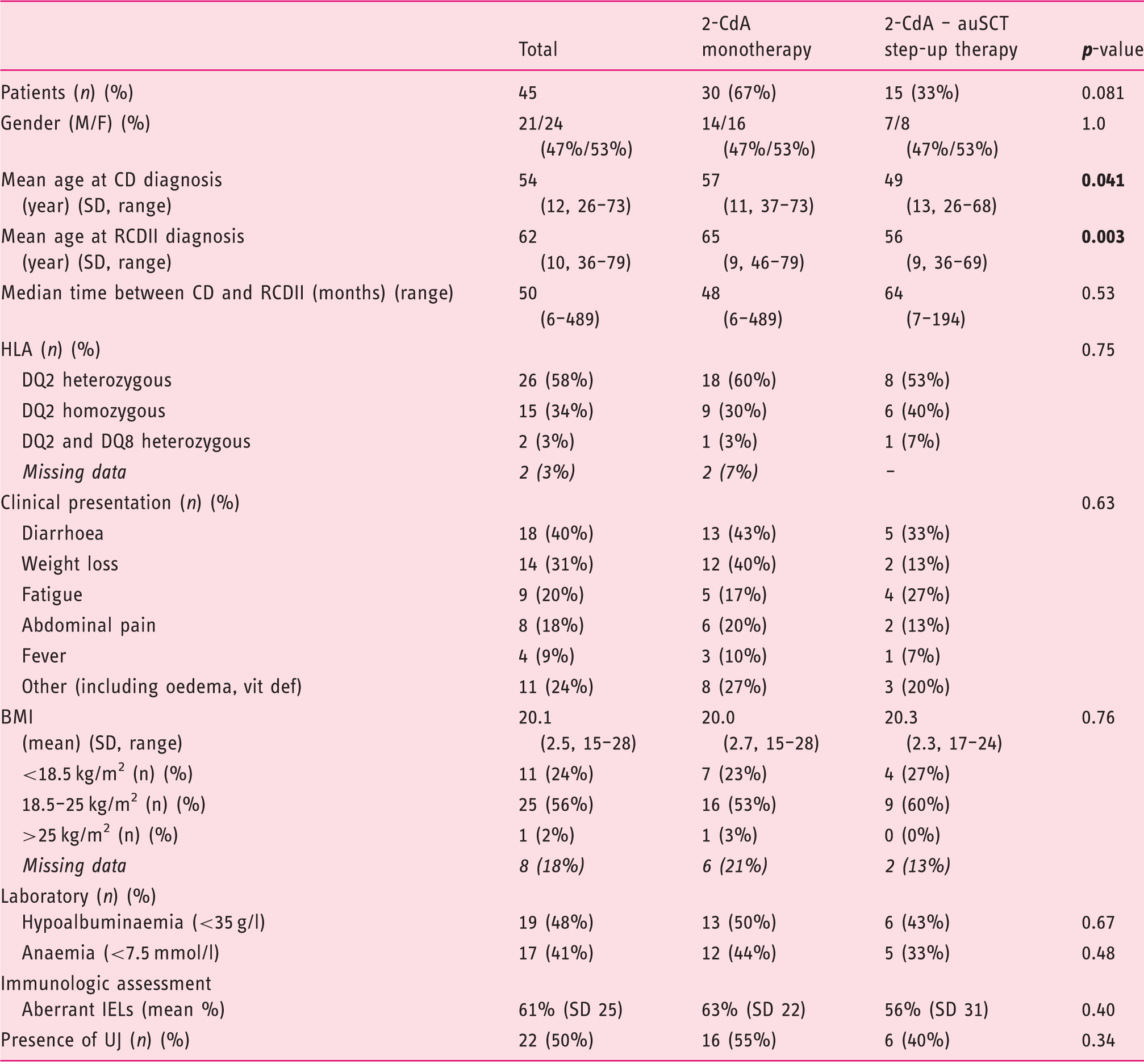
2-CdA: cladribine; auSCT: autologous stem cell transplantation; BMI: body mass index; CD: celiac disease; IEL: intra-epithelial lymphocyte; RCDII: refractory CD type II; SD: standard deviation; UJ: ulcerative jejunitis; HLA: human leukocyte antigen; vit def: vitamin deficiency.
Percentages were calculated considering all patients who underwent clinical evaluation for the variable. Missing data was excluded.
Clinical characteristics
Diarrhoea and weight loss are the most common presenting symptoms in RCDII patients (40% and 31% respectively). Others presented with non-specific symptoms including fatigue (20%), abdominal pain (18%) or other malabsorption and enteropathy suggestive symptoms like oedema (7%) and vitamin deficiencies as presenting ‘symptom’ (5%). Almost half of the patients (48%) presented with hypoalbuminaemia and 41% with anaemia. Five patients were diagnosed based on the presence of anaemia or vitamin deficiency and had no physical complaints at presentation. Endoscopic signs of ulcerative jejunitis (UJ) were present in half of the patients (n = 22). Sixty-one per cent of patients were HLA-DQ2.5 heterozygous and 35% were HLA- DQ2.5 homozygous, which is remarkably higher than in the background population (2.1%; p<0.001). 24 Two patients (5%) were compound heterozygous for HLA-DQ2.5 and DQ8.
Treatment
Figure 1 gives an overview of the treatment strategies. All patients received primary 2-CdA therapy (n=45). 2-CdA was started after a median of two months following diagnosis RCD (with a range from three days to more than four years). The majority of patients received one course of 2-CdA (n = 26, 58%). The other patients received two courses (n = 14), three courses (n = 3) and four or seven courses (both n = 1) based on the judgement of the treating physicians. Treatment was well tolerated. Side effects were seen in one patient who developed (toxic) hepatitis and pancytopenia three weeks after initial therapy which resolved without additional therapy. One patient died two weeks after 2-CdA therapy due to an opportunistic infection. Twenty-seven patients (60%) were intended to receive additional auSCT. Reasons for auSCT were the absence of histological remission (n = 9), a persisting high percentage of aberrant cells (arbitrarily defined as>50%, n = 4), progressive clinical symptoms (n = 1) or a combination of these (n = 13). The step-up therapy consisting of auSCT was eventually not performed in 12 of the patients (44%) due to comorbidity (n = 5), age>70 (n = 4), or unsuccessful leukapheresis (n = 3). Ultimately, 15 patients (33%) underwent auSCT and in total 30 patients (66%) had 2-CdA monotherapy and were closely monitored. AuSCT was usually well tolerated, and no major transplant-related events occurred. One patient died of chronic encephalitis nine months after transplantation, a complication associated with CD rather than transplantation. AuSCT-related toxicity was relatively mild. Two patients had transient diarrhoea and two had a fever of undetermined origin, which was treated with intravenous antibiotics.
Overview of treatment strategies.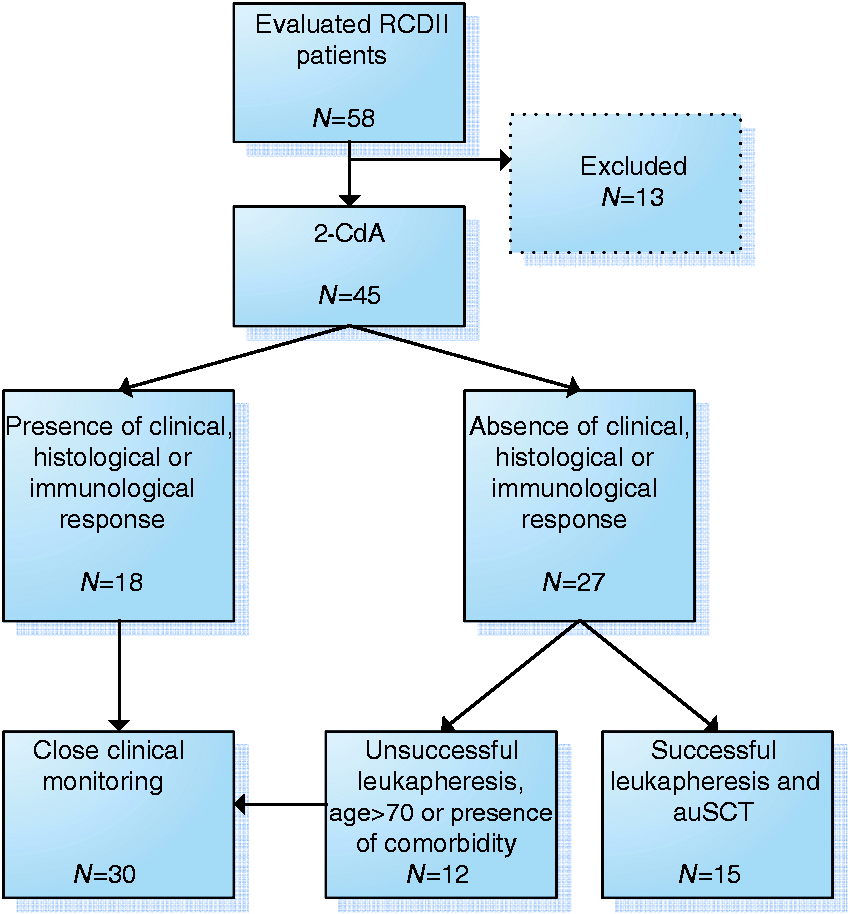
Treatment response
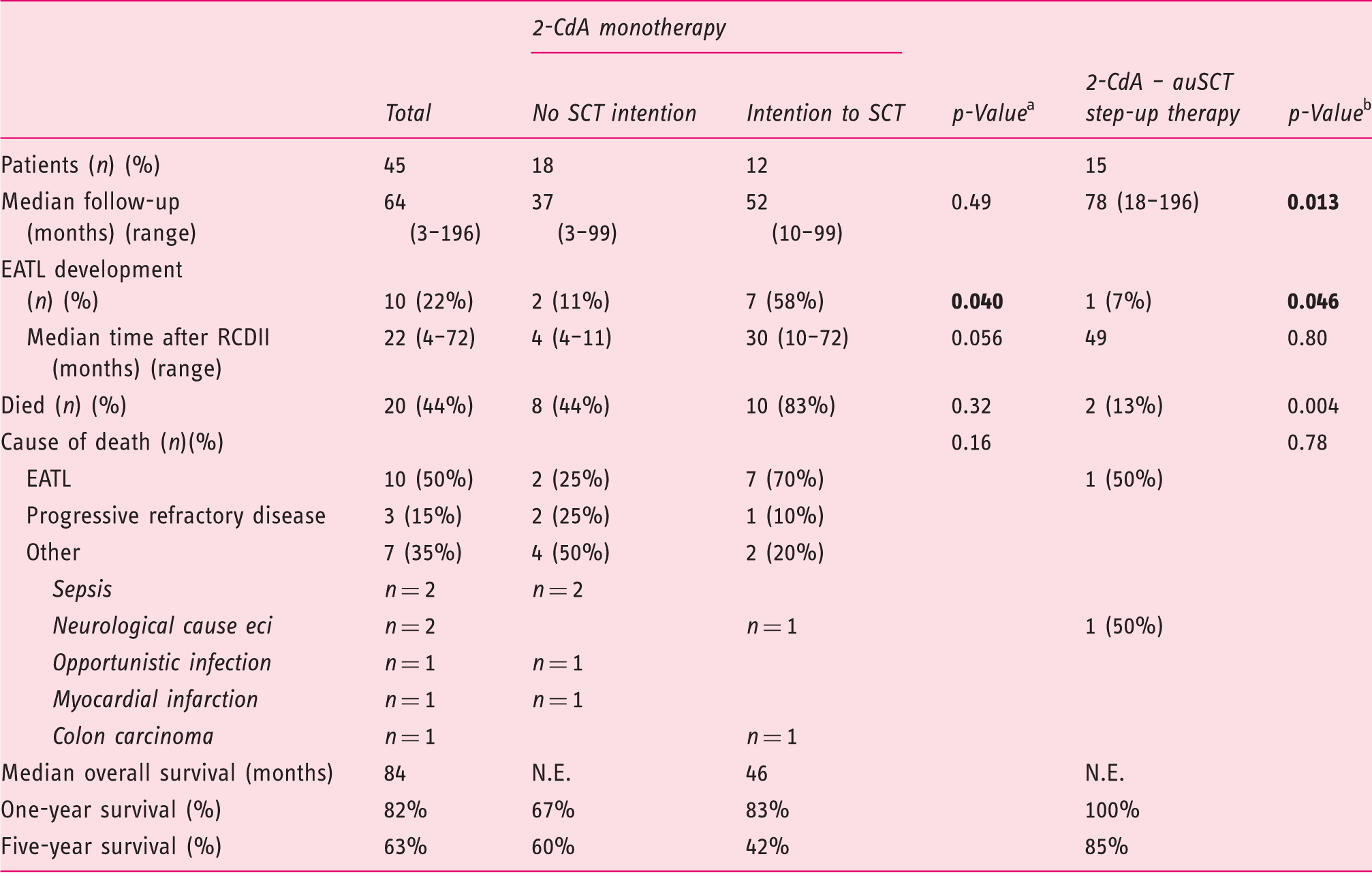
Outcome after cladribine (2-CdA) monotherapy and 2-CdA-autologous stem cell transplantation (auSCT) step-up therapy
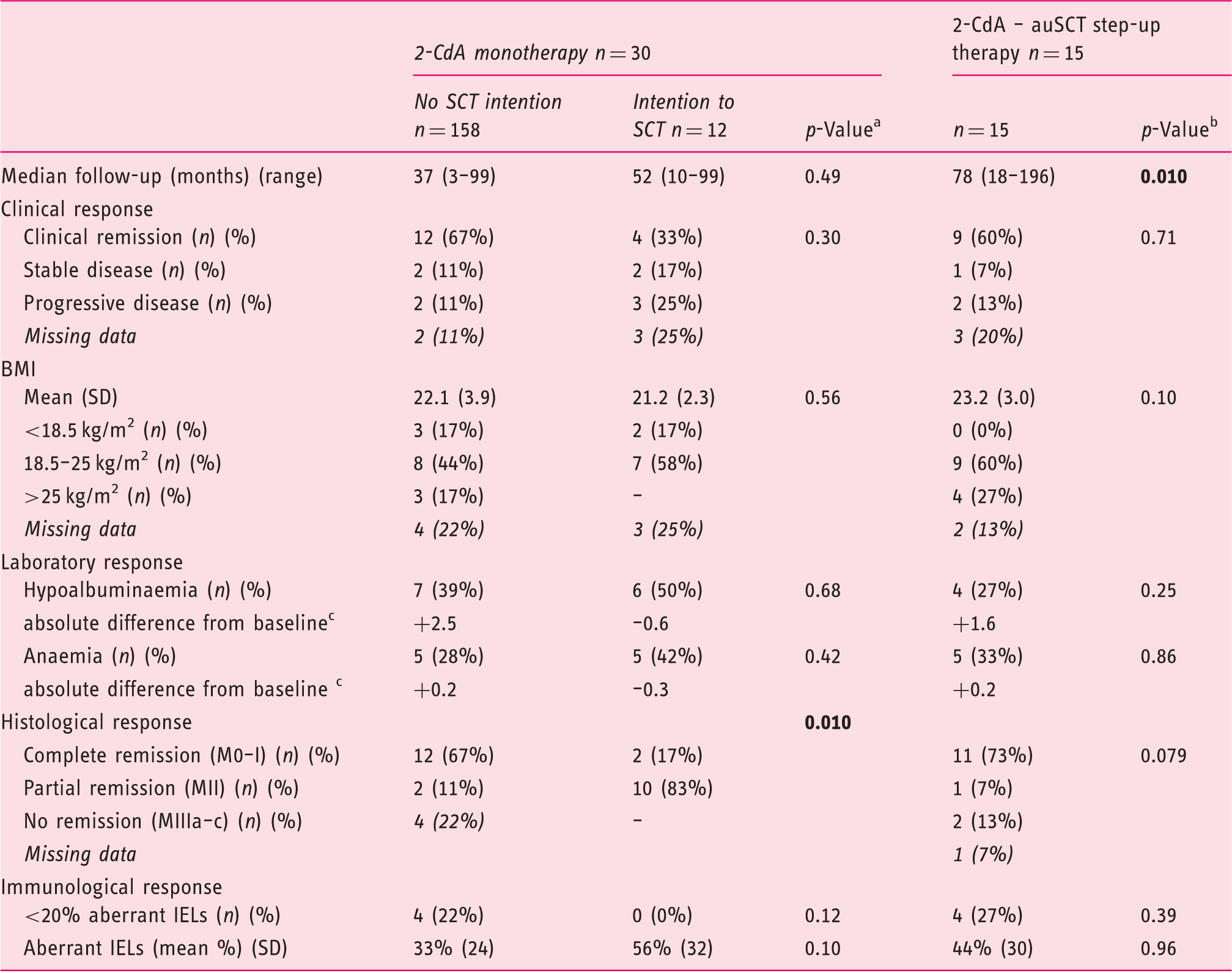
2-CdA: cladribine; auSCT: autologous stem cell transplantation; BMI: body mass index; CD: coeliac disease; IEL: intra-epithelial lymphocyte; RCDII: refractory CD type II; SD: standard deviation; UJ: ulcerative jejunitis
Comparing the outcome of monotherapy without the intention to transplant with monotherapy with the intention to transplant after therapy at maximal follow-up.
Comparing the outcome of the complete monotherapy group with the outcome of step-up therapy group at maximal follow-up.
Calculated as the mean from the absolute difference compared with T = 0.
When comparing patients who underwent 2-CdA monotherapy with those who underwent step-up therapy, there was a trend for better histological response (p = 0.079) and a higher mean BMI (p = 0.10) in the patients who underwent auSCT. Notably, when comparing patients without the intention to transplant with those who underwent auSCT, at maximal follow-up no significant differences in clinical parameters were observed, which underlines the conflicting outcomes within the monotherapy group. Evaluating the immunological response compared to baseline reveals a significant decrease in aberrant IELs in the monotherapy group without the intention to transplant from a mean of 63% (SD 22) to a mean of 33% (SD 24)(p = 0.003). The decrease of aberrant IELs in the monotherapy group with an intention for step-up therapy and the decrease in those who underwent auSCT was non-significant (mean of 63% decreased to a mean of 56% (SD 32, p = 0.68) and mean of 56% (SD 31) decreased to a mean of 44% (SD 30, p = 0.14) respectively).
EATL development
During a median follow-up of 64 months (range 3–196), 10 out of 45 patients developed an EATL. This occurred almost exclusively in the monotherapy group (n = 9, with a median time after RCDII diagnosis of 49 months). In the step-up therapy group one patient developed an EATL 78 months after RCDII diagnosis. Within the monotherapy group, EATL development was significantly higher in the group with the intention to transplant (n = 7) compared to the group without the intention (n = 2) (p = 0.04). In the latter group, EATL development was comparable to those treated with the step-up approach(p = 0.54) (Figure 2(a)).
Kaplan-Meyer curve (a) for time to enteropathy-associated T-cell lymphoma (EATL) development stratified by treatment (p = 0.046) and (b) by histological remission within the monotherapy group (p = 0.010).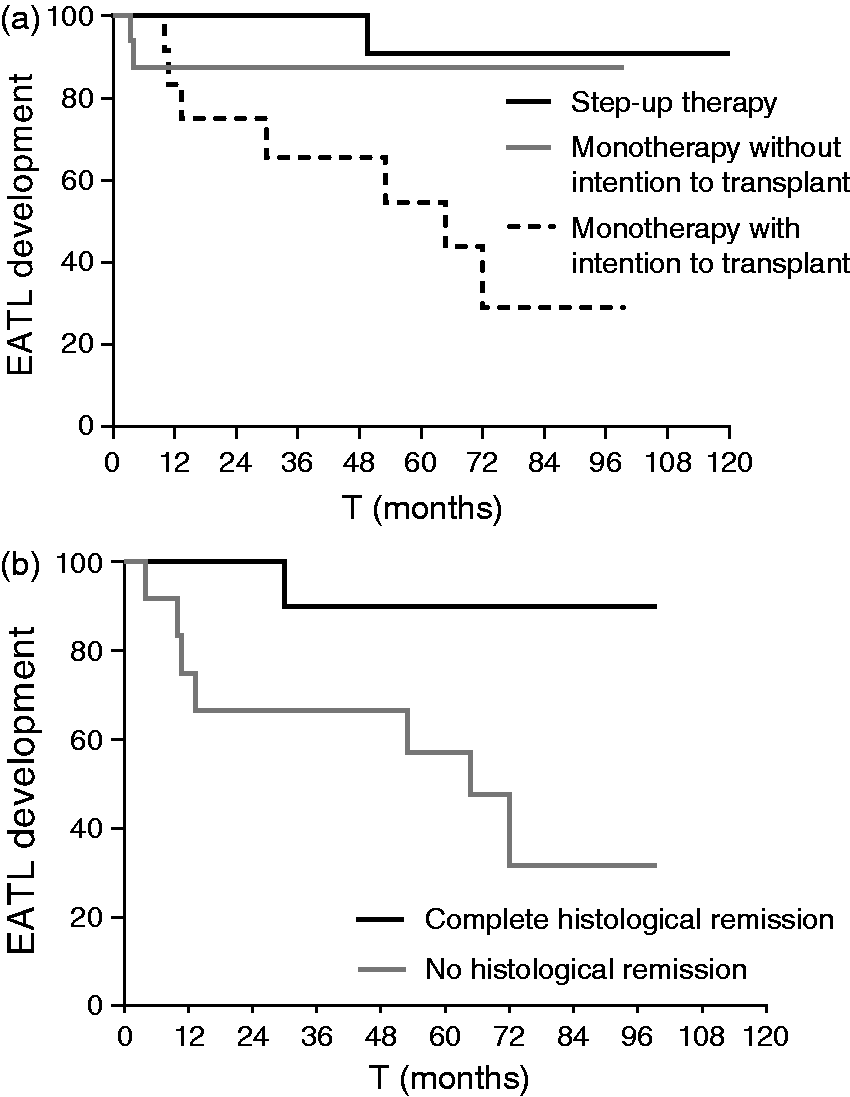
Univariate analysis evaluating prognostic factors for time to enteropathy-associated T-cell lymphoma (EATL) development within the monotherapy group
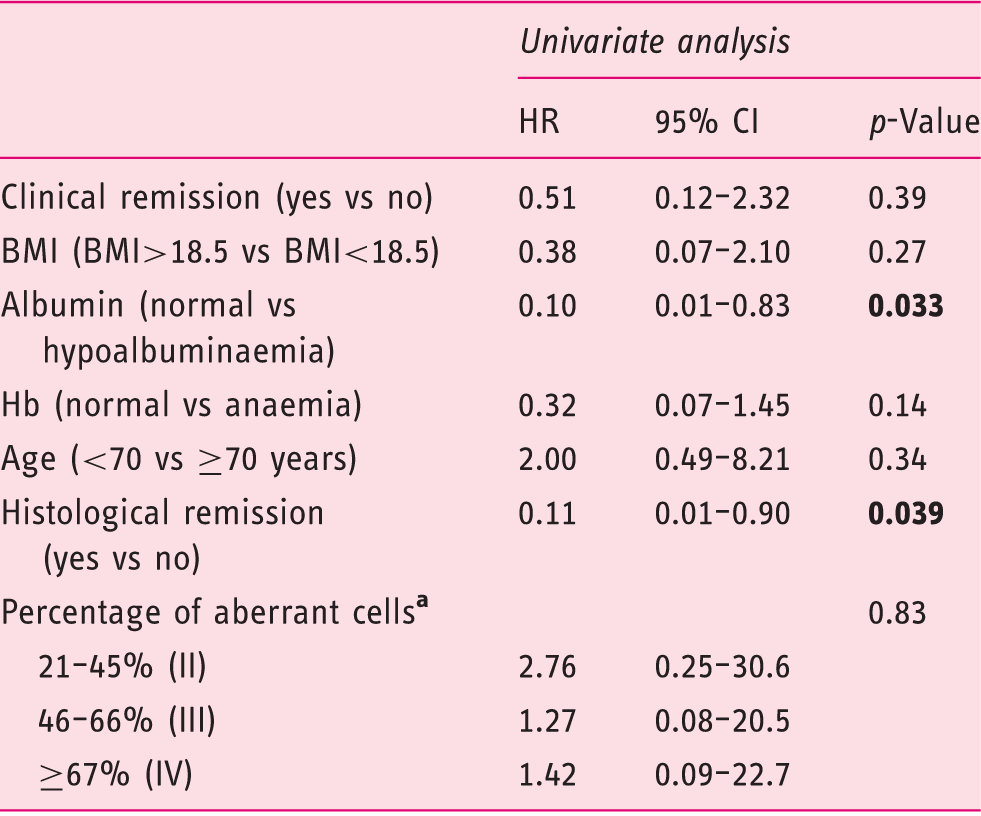
BMI: body mass index; CI: confidence interval; Hb: haemoglobin: HR; hazard ratio.
As compared to 0–20% (I).
Within the monotherapy group, there was no association between the occurrence of EATL and the presence of a high percentage of aberrant IELs after 2-CdA therapy (p = 0.82) (Figure 3) and aberrant percentages could not be identified as prognostic factor for EATL development (p = 0.60) (Table 3). Moreover, somewhat surprising and counter-intuitively, histological remission was not associated with the percentage of aberrant cells in all groups; patients with complete histological remission showed no significant difference in the percentage of aberrant cells compared to patients without histological remission at maximal follow-up (p = 0.34).
Boxplot showing the percentage of aberrant intra-epithelial lymphocytes (IELs) in patients with and without enteropathy-associated T-cell lymphoma (EATL) development within the monotherapy group (p = 0.82).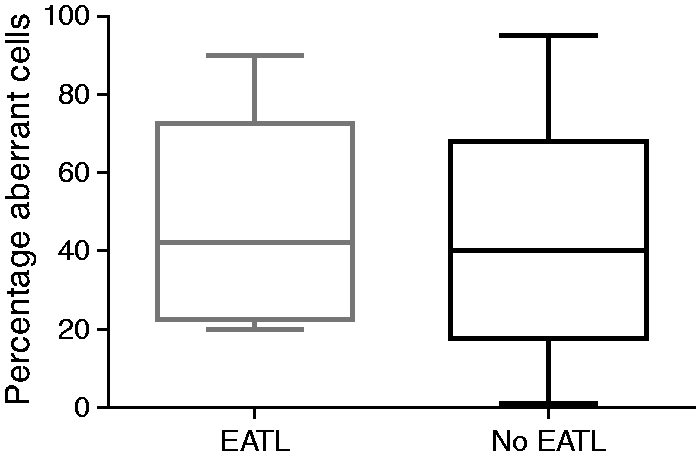
Survival outcome
Overall outcome and outcome in different therapy groups
2-CdA: cladribine; auSCT: autologous stem cell transplantation; EATL: enteropathy-associated T-cell lymphoma; N.E.: could not be estimated; RCDII: refractory coeliac disease type II; SCT: stem cell transplantation.
Comparing monotherapy without the intention to transplant with monotherapy with the intention to transplant.
Comparing the monotherapy group with the outcome of step-up therapy group.
Comparing both treatment regimens, patients in the 2-CdA-auSCT step-up therapy group showed a significantly better survival rate compared to the the complete monotherapy group (p = 0.004). Patients receiving step-up auSCT had one- and five-year survival rates of 100% and 85% respectively and a median follow-up in the survivors of 79 months (range 27–196). With a median follow-up of 49 months within the monotherapy group, eight patients (44%) died in the group without the intention to transplant, compared to 10 patients (83%) in the group with a transplantation intention (p = 0.32). Five year survival was 60% and 42% respectively (Table 4) (Figure 4(a)).
Kaplan-Meyer curve (a) for overall survival stratified for therapy (p = 0.004) and (b) for histological remission within the monotherapy group (p = 0.030).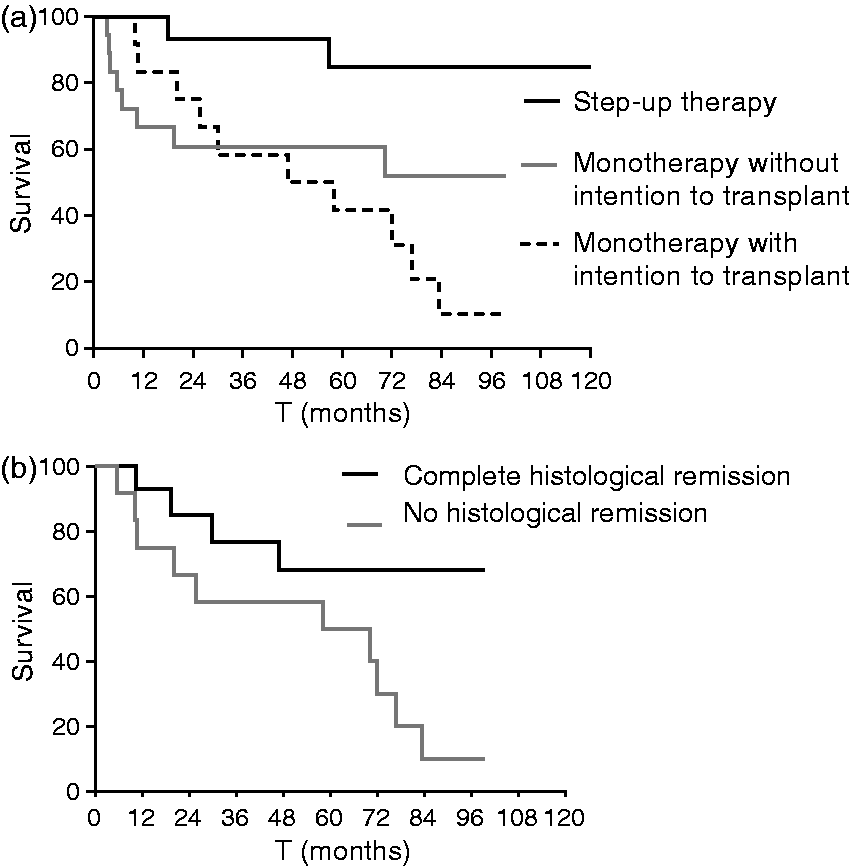
Survival within the monotherapy group was significantly worse in the patients without histological remission (f = 0.030) (Figure 4(b)). One- and five-year survival rates in the group without histological remission were 75% and 50% respectively and median OS was 58 months. From the patients in complete histological remission, ultimately four out of 14 (29%) died with a median follow-up of 58 months (range 10–99). Causes of mortality in this group were EATL (n = 1), sepsis (n = 1), opportunistic infection following 2-CdA (n = 1) and colon carcinoma (n = 1). One- and five-year survival rates in this group were 93% and 68% respectively. Comparing patients from the monotherapy group who achieved complete histological remission with the 2-CdA-auSCT step-up therapy group revealed no significant difference in OS (p = 0.14).
Discussion
With an overall five-year survival of 63% and EATL development in 22% despite (intensive) therapy the present results support the notion that RCDII is a pre-malignant condition. Although 2-CdA has been proven feasible and safe, it does not prevent EATL development in all treated patients. Here we showed that histological response is a good clinical parameter to evaluate the effect of 2-CDA therapy. When histological remission is achieved after 2-CdA therapy, the frequency of EATL development is low and OS is comparable with that in the 2-CdA-auSCT step-up therapy group.
To the best of our knowledge, this is the first study to describe response parameters on the effectiveness of a step-up strategy used in RCDII. Hence, this study has some important limitations. Due to the low incidence of RCDII, this study included a limited number of patients in the different treatment groups which makes statistical comparison difficult and prognostic analysis via multivariable analysis impossible. Selection of patients non-responsive for 2-CdA and eligible for auSCT was non-standardised.
Immunological response was not associated with time to EATL development and OS. Although all patients treated with 2-CdA showed a significant decrease in aberrant IELs as compared to baseline, the monotherapy group showed a significant higher frequency of EATL occurrence. Moreover, persisting high percentages of aberrant IELs were not associated with EATL development. This is in line with a study by Rubio-Tapia et al. describing the same observation. 9 It can be hypothesised that a small percentage, of phenotypically different heterogenous aberrant IELs 25 might be 2-CdA-resistant and these result in EATL development over time, although there are no data yet to substantiate this thesis. As a consequence of this observation, follow-up after 2-CDA therapy can be diminished to only clinical and histological follow-up, which results in lower costs and the possibility of performing the follow-up after 2-CDA without the necessity of performing phenotypical flow-cytometric analysis.
Although ultimately 22% of patients developed an EATL, the monotherapy and the step-up therapy described in this study are both more effective in the delay and prevention of EATL as compared to alternative RCDII treatment strategies previously described.8,9,26 Whether the benefits from the addition of auSCT outweigh the costs and the risks of this aggressive treatment strategy needs to be substantiated in a larger cohort with longer follow-up. Unfortunately, auSCT can only be performed in a selected population and is restricted to age in most countries. Considering this, alternative treatment strategies and/or the possibility of performing auSCT at a higher age are urgently needed, especially for 2-CdA failures.
In conclusion, 2-CDA-auSCT step-up therapy and 2-CdA monotherapy show a good and comparable effect on EATL prevention and OS in those patients who achieve histological remission. Patients who fail to respond histologically on 2-CDA monotherapy are at risk for EATL development and should be considered for an additional step-up strategy consisting of auSCT and/or newly developed treatment regimens. Therefore, regular histological and clinical evaluation is essential in the follow-up of RCDII patients and early step-up treatment strategies could prevent EATL development.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
