Abstract
Background
Despite many publications regarding the role of faecal calprotectin (FC) in inflammatory bowel disease (IBD), clear recommendations for its use in clinical practice are currently lacking in the literature.
Aim
The aim of this article is to provide practical guidance for clinicians for the use of FC in the detection and management of patients with IBD.
Methods
All relevant publications were analysed and practical statements were proposed based on a Delphi consensus approach.
Results
Different commercial assays have been developed but international standardisation is lacking. FC can help in the diagnosis process of IBD. In IBD, FC can predict response to therapy, detect subclinical inflammation and help to drive treatment decisions to achieve better endoscopic and clinical outcomes. After Crohn’s surgery FC can identify patients with early endoscopic recurrence.
Conclusion
Although major therapeutic changes should not be based on FC alone, FC is a valuable tool to optimise the care for IBD patients.
Keywords
Introduction
Overall the sensitivity and specificity of classical biochemical parameters of inflammation is low in inflammatory bowel disease (IBD). Emerging evidence shows that faecal markers can be used to detect and monitor intestinal inflammation selectively. Despite this growing body of literature, recommendations for the use of faecal calprotectin (FC) are currently lacking. Current guidelines on diagnosis and treatment of IBD mention FC as useful makers but do not include values or practical recommendations in the statements.1,2 The Belgian IBD Research and Development (BIRD) group performed a literature review on the potential use of FC in several situations in IBD. This manuscript can serve as practical guidance for clinicians but it is not a comprehensive or systematic review of the literature.
Methodology
We searched for relevant publications in PubMed/MEDLINE, EMBASE and the Cochrane Central Register of Controlled Trials from their inception until October 2017. Relevant articles (in English) were critically reviewed and discussed among the authors of this manuscript. Priority was given to randomised controlled trials and meta-analyses published in the last five years. Relevant abstracts from major meetings were also considered.
The evidence-based recommendations for the use of FC in clinical practice were drawn up through an electronic Delphi process, which is a group facilitation technique including an iterative multistage process to transform opinion into group consensus. 3
A first draft of recommendations and statements was submitted to an expert panel of gastroenterologists and gastro-paediatricians within the BIRD. Thirty-two gastroenterologists (66%) answered the survey (47% from academic centres, 69% with ≥ 50% of medical activity in IBD, 91% involved in clinical or basic IBD research). No paediatrician participated. Final decisions on the statements were made if 80% agreement was achieved among the experts to come up with practical recommendations. A second round was necessary to validate two statements. The concordance rate within the expert panel is mentioned in brackets for each statement.
Results
Faecal sampling in practice
Stool sampling and dosage in clinical practice
– Use the morning sample or the first sample of the day. (91%) – Avoid highly liquid or too solid stools. (84%) – The samples can be stored up to 72 hours at room temperature. (94%) – Owing to variability among tests, it is recommended to use the same test in the follow-up of a single patient. (97%)
It has been recommended to collect the first stool sample of the day as this is the most concentrated. 4 The samples can be stored for 72 hours at room temperature and up to seven days at a temperature of 2℃–8℃. 4 Approximately 50 mg to 100 mg is required. After extraction, samples can be kept permanently when frozen.
Technical and methodological aspects of FC measurement
Faecal calprotectin assay characteristics.
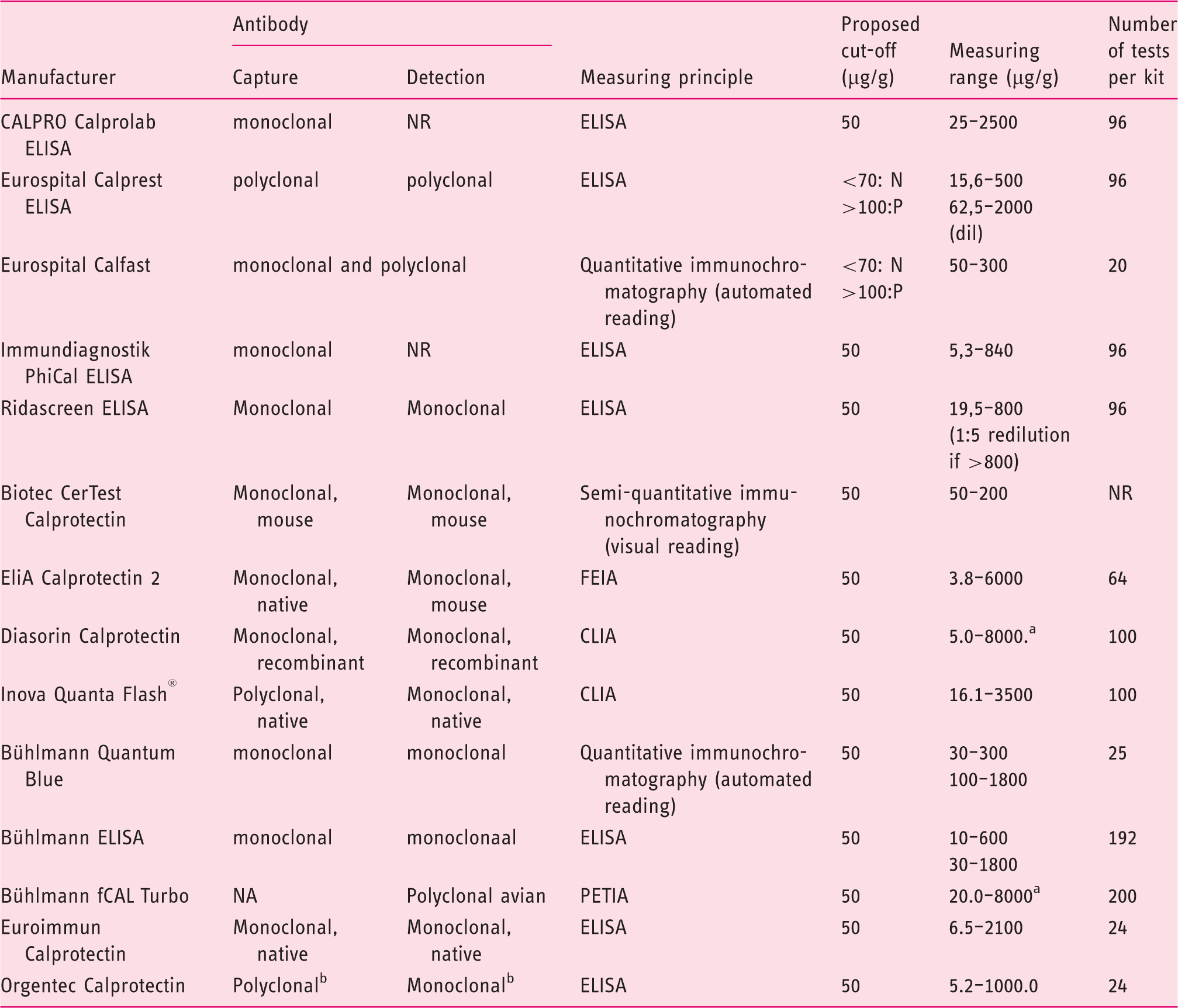
after 1:10 (Diasorin) and 1:4 (Bühlmann) dilution, concentrations up to 8000.0 µg/g faecal calprotectin can be obtained
Origin not reported
Abbreviations: FEIA: Fluoro Enzyme Immuno Assay; CLIA: Chemiluminescence Immunoassay, PETIA: Particle Enhanced Turbidimetric Immunoassay; ELISA: Enzyme Linked Immunosorbent assay; NA: Not Applicable; NR: Not Reported.
Use of FC in the detection of IBD
Use of FC as a diagnostic tool in clinical practice
– FC > 250 µg/g identifies patients who are most likely to have intestinal inflammation and justifies further endoscopic examination. (91%) – FC between 100 and 250 µg/g could require a second measurement within three months. (97%) – FC < 100 µg/g has a very high negative predictive value for IBD, justifying its use as a screening test to reduce the number of endoscopies and thereby the costs of health care management. This strategy delays the diagnosis in only a small proportion of patients. (97%)
The value of FC as a diagnostic marker for IBD has been extensively studied. Several meta-analyses and systematic reviews have summarised the results of individual studies and report an overall sensitivity of 80%–98% and a specificity of 68%–96% for cut-offs ranging from 30 to 100 µg/g.11,12 The specificity of the test tends to be lower in children.11–13 A cut-off of 50 µg/g is recommended by most test suppliers.11,12 Values below 100 µg/g have a very high negative predictive value for IBD, and this can be particularly helpful to guide the need for further investigations in patients with nonspecific gastrointestinal symptoms and a low pre-test probability of IBD. The use of FC can reduce the number of negative colonoscopies by about two-thirds in this setting and results in a cost savings of $417 per patient.
12
This strategy delayed diagnosis in only 7% of patients with actual IBD.
14
It is not recommended to use FC in the acute setting of diarrhoea or in symptomatic patients with a high pre-test probability of IBD because of the small but still existent risk of false-negative test results. In high-risk patients (age above 40 years old and with symptoms suggestive of malignancies), an immediate endoscopic work-up remains the best and most cost-effective approach. A positive FC test should always be interpreted in the global clinical context as FC may be elevated in many other gastrointestinal conditions (infections, malignancies, nonsteroidal anti-inflammatory drugs (NSAIDs)-related mucosal injury).
12
An algorithm illustrating the use of FC for the diagnosis of IBD is proposed in Figure 1.
Algorithm for the use of FC in the detection of IBD.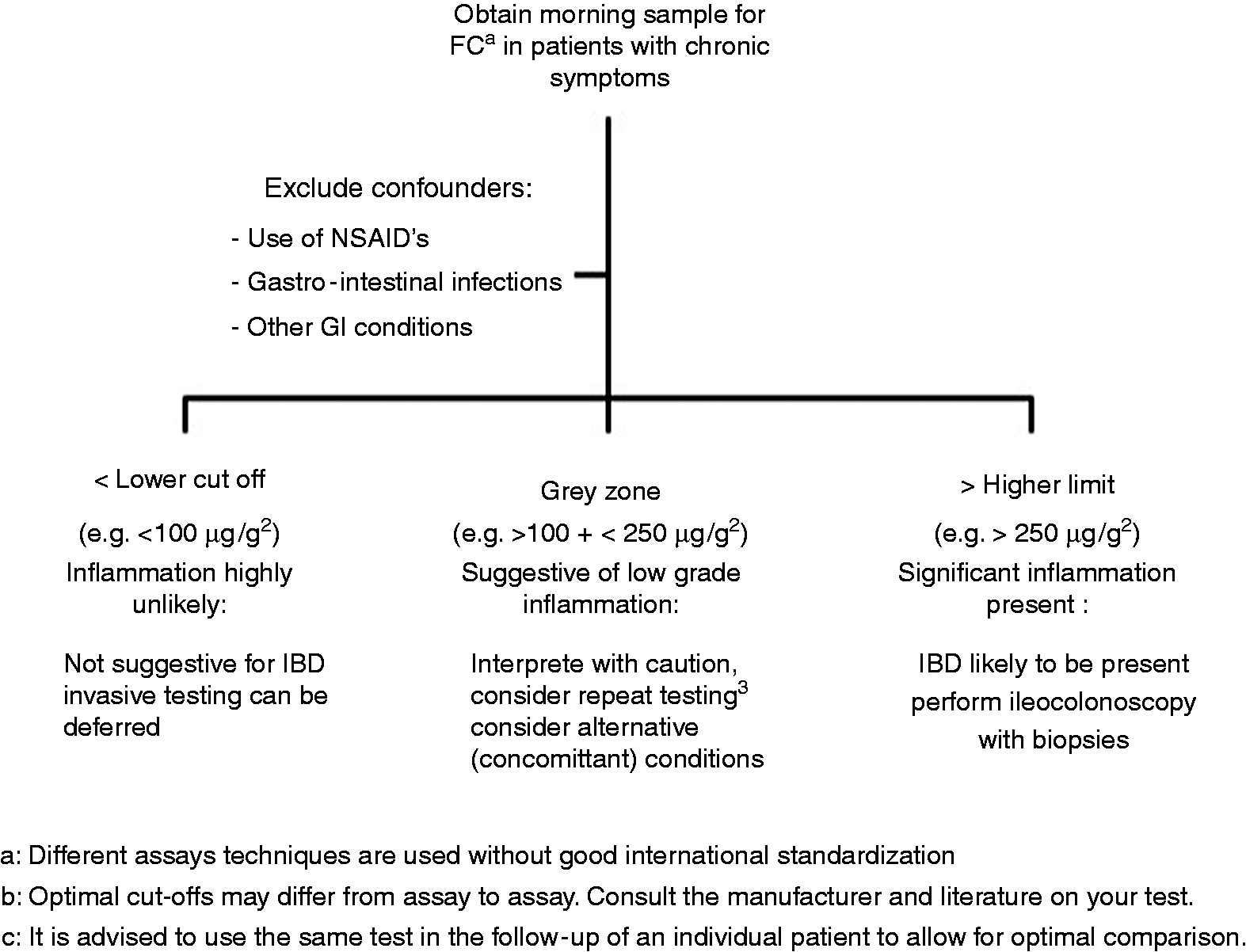
Use of FC for the clinical follow-up of symptomatic IBD patients
Clinical use of FC in symptomatic IBD patients
– FC should be measured at diagnosis of IBD or prior to major treatment changes for future comparison. (97%) – In symptomatic IBD patients FC > 250 µg/g can discriminate an IBD flare from noninflammatory complications or underlying associated irritable bowel syndrome. (91%) – FC decrease can predict clinical and endoscopic response to treatment. (94%) – FC levels before initiating IBD therapy and serial FC measurements three and six months following treatment initiation are recommended to evaluate response to therapy and to predict long-term remission. (84%) – In symptomatic IBD patients, endoscopy remains the gold standard to assess disease activity and major therapeutic changes are not recommended based on FC alone. (94%)
Objective assessment of the presence and degree of intestinal inflammation in symptomatic IBD patients is an essential part of disease management. Endoscopy remains the gold standard to detect and quantify mucosal inflammation in IBD patients, especially since the optimal therapeutic target to modify disease course should also be mucosal healing, which is associated with long-term improved outcomes.14,15 However, endoscopy is an expensive, invasive and time-consuming procedure that is, therefore, not ideal for repeated regular assessment of disease activity.
In current practice, FC can be used as an objective reliable marker of inflammation since it has been demonstrated to strongly correlate with endoscopic disease activity with high sensitivity and specificity (respectively 88% and 73% in Crohn’s disease (CD) and ulcerative colitis (UC)).14–17 FC correlates better with endoscopic activity than clinical activity
17
and better than C-reactive protein (CRP) with endoscopic activity.17–20 However, FC appears to reflect disease activity better in UC compared to CD and ileocolonic/colonic CD is associated with significantly higher FC compared with isolated ileal CD.
19
A practical algorithm is suggested in Figure 2. Optimal FC cut-off values for the detection of endoscopic active disease vary from 50 to 250 µg/g depending on the study and the test used as well as the type and location of the disease and intra-individual patient variability.4,21 To improve the reliability of FC testing, a baseline FC level during a period of known active inflammation should be obtained. In addition determining an FC value at the time of endoscopy allows the correlation of a patient’s individual value with endoscopic activity. Recent studies have shown that even in the absence of endoscopic signs of disease activity, levels of FC are predictive of long-term outcomes.
22
Algorithm for the use of FC in symptomatic IBD patients.
In the follow-up of symptomatic patients, FC also plays an important role in the assessment of response to IBD treatment that is generally based on symptoms, which may not accurately reflect the underlying inflammatory process, while endoscopic evaluations are not frequently performed. Mucosal healing is increasingly advocated as a therapeutic target in IBD and can be noninvasively identified by normalisation of FC. 23 A fast and significant fall in FC concentrations occurs in CD and UC patients treated with either high-dose corticosteroids, 24 infliximab, 25 adalimumab 26 or vedolizumab 27 and a normalisation or decrease in FC concentrations predicts clinical response and sustained remission at one year. 28 An 80% decrease at week 2 of FC levels as compared to pre-treatment levels predicts endoscopic remission at week 10 after infliximab induction with a specificity of 67% and sensitivity of 54%. 29 Looking at these data, a systematic endoscopy to assess mucosal healing after the initiation of new medications could be postponed when a normalisation or a decrease of 80% in FC concentrations is observed. Failure to reduce FC sufficiently may be a marker of nonresponse. In CALM, 28 a recently published multicentre randomised controlled study, higher rates of mucosal healing (46% vs 30%, p = 0.01), steroid-free remission (60 vs 39%, p < 0.001) and biologic remission (30% vs 16%, p = 0.006) at one year were achieved when the escalation of the treatment strategy with adalimumab was based on symptoms and biomarkers (CRP and FC) compared to symptoms alone. A persistent raised FC (>250 µg/g) was the main driver of escalating treatment in the first group. FC can therefore be measured before initiating or changing IBD therapy, and serial measurements could be recommended in the weeks following treatment initiation to evaluate response to therapy.
Role of FC in asymptomatic IBD patients (see Figure 3)
Clinical use of FC in asymptomatic IBD patients
– FC can identify patients in clinical remission with subclinical inflammation and high risk of short-term clinical relapse. (97%) – FC measurements can be performed according to the risk profile of the patient every three to six months. (94%) – In case of elevated FC: (97%) – Exclude confounders (C. difficile infections, use of NSAIDs). – Confirm disease activity by imaging (magnetic resonance imaging, endoscopy). Interpretation of the different cut-off levels of FC in asymptomatic IBD patients.
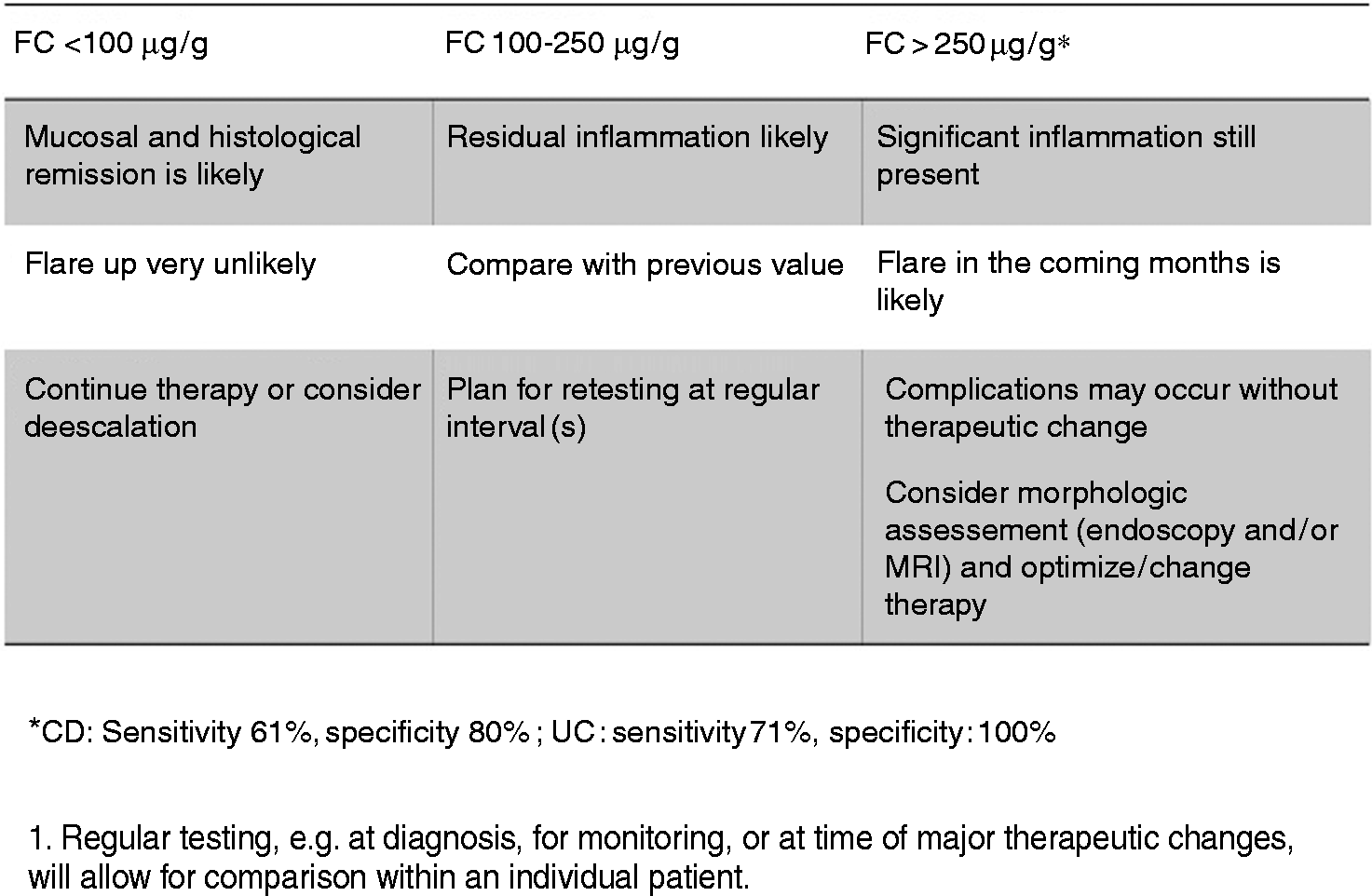
Faecal biomarkers have been identified as surrogate makers of endoscopic and histological healing in IBD patients. An FC cut-off value of less than 250 µg/g is associated with the absence of large ulcers in CD, according to the Simple Endoscopic Score for Crohn’s Disease, with a specificity and a sensitivity of 61% and 80%, respectively, and can discriminate a Mayo endoscopic sub-score of zero vs ≥1 in UC with a sensitivity and a specificity of 71% and 100%, respectively.19–21,30 Prospective studies have demonstrated that FC can discriminate patients with a higher risk of future relapse even if they are in clinical remission. 31 In CD with either ileal or colonic involvement, the patients having an FC value ≤200 µg/g had a four-fold lower risk of clinical relapse within the following year compared to patients with an FC > 200 µg/g. In UC and colonic CD, a lower cut-off was able to identify the patients with a low risk of clinical relapse: FC ≤120 µg/g was associated with a six-fold lower risk of clinical relapse within the following year. 32 In UC it was demonstrated that patients in deep remission on infliximab experienced an increase of FC three to four months before clinical relapse. 27 In CD, FC progressively increased two to four months before clinical relapse in patients whose anti-tumour necrosis factor was withdrawn while in clinical remission. FC > 200 to 300 µg/g predicted the relapse with 83% specificity and 50% sensitivity.33,34 The implication in clinical practice is considerable because the patient might be offered an escalated treatment strategy at earlier stages of inflammation.
FC in the postoperative setting in IBD
Clinical use of FC in postoperative IBD patients
– Endoscopy six to 12 months after surgery remains the gold standard to assess postoperative recurrence in CD. (100%) – FC <100 µg/g six to 12 months after surgery has a 90% negative predictive value for endoscopic recurrence (i2 or more) and is well correlated to the Rutgeerts score. (81%) – FC cannot replace an endoscopy six months after surgery. FC measurements three months after surgery can identify patients with early endoscopic recurrence and can select the patients who need an early postoperative endoscopy. (85%) – In UC pouchoscopy remains the gold standard to assess inflammatory activity after pouch surgery, but FC can be a useful marker to diagnose pouchitis noninvasively. (81%)
Despite an expanded therapeutic armamentarium, about one-half of patients with CD still require surgery in the first 10 years of the disease course. 35 Post-surgical recurrence is inevitable in a vast majority of patients although aggressive postoperative approaches have been introduced, mainly in patients with a high-risk profile. 36 Endoscopic evaluation six to 12 months after surgery is still the gold standard to detect the recurrence of early endoscopic lesions and to introduce an effective treatment strategy before the occurrence of severe lesions and tissue damage. 37 A single FC measurement six to 12 months after surgery is predictive for clinical recurrence over time. 38 A cut-off of 100 µg/g has a negative predictive value of 90% to identify patients without endoscopic recurrence and is well correlated to the Rutgeerts score.22,38,39 In a post-hoc analysis of the Post-Operative Crohn’s Disease Endoscopic Recurrence trial, the combined FC levels at six and 18 months postoperatively correlated with endoscopic recurrence by using a cut-off of 100 µg/g. 39 Another approach is the serial measurement of FC in the postoperative setting to guide the clinician for the best timing to perform the endoscopy. Continued low FC levels indicate continuous remission. 40
In patients with UC who underwent a proctocolectomy with ileo-anal pouch, increased FC has been correlated with pouchitis (using a cut-off of 56 µg/g) and increases two months before the occurrence of clinical symptoms and of endoscopic inflammation of the pouch. 41
Conclusion
FC is a sensitive biomarker to detect histological inflammation in the gastrointestinal tract. Although nonspecific, the presence of high titres of FC in chronic symptomatic patients is indicative for (active) IBD and urges further examination including endoscopy with biopsies.
In known IBD patients, FC is increasingly used to predict response to therapy, and for monitoring disease activity and postoperative recurrence, thereby avoiding repeated endoscopic investigations. However, FC should be used not only as a marker of treatment efficacy, but it could also play an important role in the assessment of disease activity to alert the clinician of the need for further endoscopic or radiographic evaluation.
Footnotes
Acknowledgements
We acknowledge all BIRD members who kindly answered the survey: Dr T Billiet (UZ Leuven), Dr JL. Coenegrachts (Hasselt), Dr A. Colard (Cliniques Saint Joseph Liège), Dr Delen (Ziekenhuis Maas en Kempen, Maaseik), Dr N. de Suray (G.H.d.C. Gilly), Dr P. Dewint (AZ Maria Middelares), Dr O. Dewit (Cliniques Universitaires Saint-Luc), Dr Dutre (XX), Dr M. Etienne (CHC Hermalle), Dr M. Ferrante (UZ Leuven), Dr G. Lambrechts (AZ Damiaan Oostende), Dr C. Liefferinckx (ULB Erasme), Dr E. Louis (CHU Liège), Dr E. Macken (UZ Antwerpen), Dr F. Mana (UZ Brussels), Dr F. Mokkadem (Clinique Libramont), Dr T. Moreels (Cliniques Universitaires Saint-Luc), Dr Ouziel (CHU de Charleroi), Dr H. Peeters (AZ Sint Lucas Gent), Dr Ph. Potvin (Sint-Jozefkliniek Bornem), Dr Ph. Van Hootegem (AZ Sint Lucas Brugge), Dr Van De Mierop (GZA Ziekenhuizen, Wilrijk), Dr H. Vafa (ULB Erasme), Dr C. Vankemseke (CHU Liège), Dr S. Vermeire (UZ Leuven), Dr B. Verstockt (UZ Leuven) and Dr B. Willandt (AZ Sint-Jan Brugge).
The authors thank Saskia Appelmans, chief operating officer of the BIRD, for her excellent support in driving this project.
Declaration of conflicting interests
Catherine Reenaers has received speakers and consultancy fees from Abbvie, Cellgen, Ferring, Janssens, MSD, Mundipharma, Pfizer, and Takeda. Peter Bossuyt is part of the advisory board for Mundipharma, Dr Falk Benelux, MSD, Hospira, Takeda, Pfizer, Janssens, and Roche; has received lecture fees from Abbvie, Takeda, and Vifor Pharma; and has received an educational grant from Abbvie. Pieter Hindryckx has received consulting fees from Abbvie and Takeda; and speakers fees from Ferring, Falk Pharma, Vifor Pharma, Tillotts Pharma, Chiesi, Takeda and Abbvie. Hilde Vanpoucke has nothing to declare. Anneline Cremer has received a grant from the Fonds Erasme for medical research. Filip Baert’s department has received research grants from Abbvie, Chiesi, Ipsen, MSD, and Roche. He has received speakers and consultancy fees from Abbvie, Falk, Ferring, Janssen, Mundipharma, MSD, Pfizer, Takeda, and Vifor.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The study was performed according the the recommendations of the ethical committee.
