Abstract
Background
Thioguanine (TG) is efficacious in inflammatory bowel disease (IBD), but its toxicity, particularly nodular regenerative hyperplasia (NRH) of the liver, has limited its use. We assessed the long-term clinical outcomes and safety of TG in patients whom were intolerant or refractory to conventional immunomodulators.
Methods
This is a retrospective, single-centre study of IBD patients treated with TG from 2001–2013. Response was defined as clinical remission (Harvey–Bradshaw Index < 5 for Crohn’s disease (CD), Simple Clinical Colitis Activity Index < 4 for ulcerative colitis (UC)) without corticosteroids or, if receiving anti-tumour-necrosis-factor (anti-TNF) therapy, absence of dose escalation. We recorded TG failure, withdrawal and adverse events. Patients were monitored with biochemistry, liver biopsy and/or magnetic resonance imaging (MRI).
Results
54 patients (47 CD and 7 UC) whom received TG (mean dose: 27 mg/d (range: 20–40 mg/d)) as monotherapy (n = 36) or concomitantly with anti-TNF (n = 18) for a median inter-quartile range of 16 (5–37) months (126 patient-years of follow-up). 32 (59%) patients responded to TG at 6 months and 23 (43%) at 12 months. Pancreatitis did not recur amongst the 19 patients with prior thiopurine-induced pancreatitis. 16 (30%) patients ceased TG due to intolerance or toxicity (four serious); NRH was not observed. 6-thioguanine nucleotide concentrations did not correlate with efficacy nor with toxicity.
Conclusions
TG was efficacious and well tolerated in one out of two patients who had previously failed conventional immunomodulators. NRH did not occur.
Introduction
Immunomodulation remains the first-line therapy in inflammatory bowel disease (IBD). The conventional thiopurines, azathioprine (AZA) and 6-mercaptopurine (MP), are efficacious in maintaining steroid-free remission in IBD. 1 A substantial proportion of patients with Crohn’s disease (CD) require treatment with a thiopurine; however, approximately 20–30% of these patients discontinue due to intolerance. 2 A further 30–40% withdraw treatment due to non-response, in part because an effective therapeutic dose measured by 6-thioguanine nucleotides (TGNs) cannot be achieved. 3 Pharmacogenetic differences in thiopurine metabolism contribute to intolerance and non-response. 4
After ingestion, AZA is converted to MP, which then undergoes metabolism via the purine salvage pathway, to pharmacologically-active TGN. Concurrently, competitive metabolism by reduction to thiouric acid (via xanthine oxidase) and methylation to methyl-mercaptopurine (MMP) via thiopurine-S-methyltransferase (TPMT) determines the ultimate level of TGN. Despite targeted weight-adjusted dosing regimens, wide individual variation in the production of TGN and MMP has been observed, reflecting in part individual differences in the relative activities of the enzymes involved. 5 For many, intolerance or non-response to AZA/MP can be circumvented for a favourable clinical outcome. 4 However, in a proportion, intolerance is unavoidable and an alternative agent is needed. In particular, this applies to thiopurine-induced pancreatitis, where recurrence on re-challenge is likely. 6 Furthermore, certain patients, despite switching to MP or low-dose thiopurine and allopurinol, remain intolerant, due to nausea or flu-like symptoms. 4
Thioguanine, (TG) a purine analogue of the nucleobase guanine, is a less frequently utilised non-conventional thiopurine. It is converted directly to TGN by hypoxanthine phosphoribosyltransferase (HPRT), circumventing numerous intermediate metabolites involved in the conventional thiopurine pathway. 7 Despite being a substrate for TPMT with similar enzyme kinetics to MP, the putative advantages of TG include avoiding potential toxicity and adverse effects, which likely occur from intermediate metabolites generated with treatment by conventional thiopurines. 7
TG has been demonstrated in small, uncontrolled studies to be effective at inducing and maintaining clinical remission in CD and ulcerative colitis (UC).8,9 Initial interest in TG was tempered by high rates of hepatotoxicity, particularly nodular regenerative hyperplasia (NRH), which was reported in 18–76% of recipients.10,11 It has been suggested that TG-induced NRH may be dose-dependent, 12 with few cases arising on low-dose TG. 13 Splitting the dose of TG may also reduce the rate of NRH. 14
Overall, the literature on TG in IBD remains sparse, and, despite the drug being a logical alternative in cases of thiopurine toxicity or refractoriness, the use of TG is limited by uncertainties over the risk of toxicity. TG has been in regular use at our institution for 15 years. Here we report our long-term efficacy and safety data regarding TG in IBD.
Methods
Patient population
We performed a single-centre retrospective study of all IBD patients treated with TG between 2001–2013 at Guy’s and St. Thomas Hospitals, in London, UK. We previously reported the short-term outcomes of the first 30 patients. 15 Patients were identified by pharmacy dispensing records. Their diagnosis of IBD was based on standard criteria1,16 and confirmed after review of the patients’ medical records. Patients were included if they took a single dose of TG. Those with incomplete medical records were excluded.
Indications for TG were classified as active disease (induction of remission), as steroid-sparing in steroid dependency (>6 months corticosteroids or relapse on corticosteroid withdrawal), in order to maintain remission with episodic infliximab (IFX) or concomitant immunomodulation with scheduled maintenance IFX or adalimumab (ADA). Active disease was defined as a Harvey–Bradshaw index (HBI) 17 ≥5 (CD) or a Simple Clinical Colitis Activity Index (SCCAI) ≥ 4 (UC) 18 and was documented in the medical record at each clinical visit, as is our standard practice. Prior immunomodulator treatment and intolerance was documented. Where TG therapy was interrupted, only the first period of TG was reported.
Clinical response and failure
Clinical response was assessed at 6 and 12 months after commencing TG. Response was defined clinically (HBI < 5, SCCAI < 4). For steroid sparing, the response was only met if steroids were withdrawn within 6 months and maintained 12 months after starting TG. We performed a sub-group analysis of the patients treated with TG and a biologic, and their response was defined as not failing biologic therapy. In those patients treated episodically with a biologic, a response was defined as not requiring further doses. Failure was defined as TG withdrawal due to adverse effects, new corticosteroids, unplanned IBD-related surgery or biologic dose intensification or switching. Adherence was determined by pharmacy-initiated tablet counting and at clinical appointments. To represent real-world clinical practice, patients could have met criteria for failing TG, yet continued the drug, hence data was also collected pertaining to the timing and reason for subsequent TG cessation.
Monitoring for toxicity
After recognising the association between NRH and TG, 10 we advised patients (between 2001–2004) to undergo percutaneous liver biopsy after treatment for ≥12 months, performed under radiologic guidance using an 18G Trucut needle. Biopsies were stained with haematoxylin and eosin, reticulin silver, connective tissue trichrome stain and chromotrope aniline blue, and the slides were reviewed by an experienced histopathologist. After 2004, screening for NRH was recommended every 24 months with liver magnetic resonance imaging (MRI).
Upon starting TG, a full blood count and liver function tests (LFTs) were performed each fortnight for the first six weeks, and then every three months, unless otherwise indicated. Toxicity was defined as LFTs 2 × upper limit of normal (ULN), thrombocytopenia (<150 × 109) or leucopenia (white cell count (WCC) < 3.5 × 109). Red blood cell (RBC) TGNs were collected every six months and analysed by ultra high-performance liquid chromatography (HPLC), based on a method described elsewhere. 19
Statistics
Continuous variables were reported as the mean (SD) or the median inter-quartile range (IQR), and for categorical variables as the number (percent). We used Pearson’s correlation for relationships between parameters. Comparisons between clinical outcomes and TGNs were performed using the Mann–Whitney U-test. The cumulative probability of failing or withdrawing TG was calculated using the Kaplan-Meier method. The significance threshold was 0.05. Statistics were performed using Prism 6.0 (San Diego, USA).
Ethical considerations
According to the guidelines of the UK Health Research Authority, as the data were collected as part of routine clinical care and were evaluated retrospectively, the study was considered a review of clinical practice and ethical approval was not required. 20 This study was conducted in accordance with the Declaration of Helsinki. 21
Results
Study population
Patient demographics.
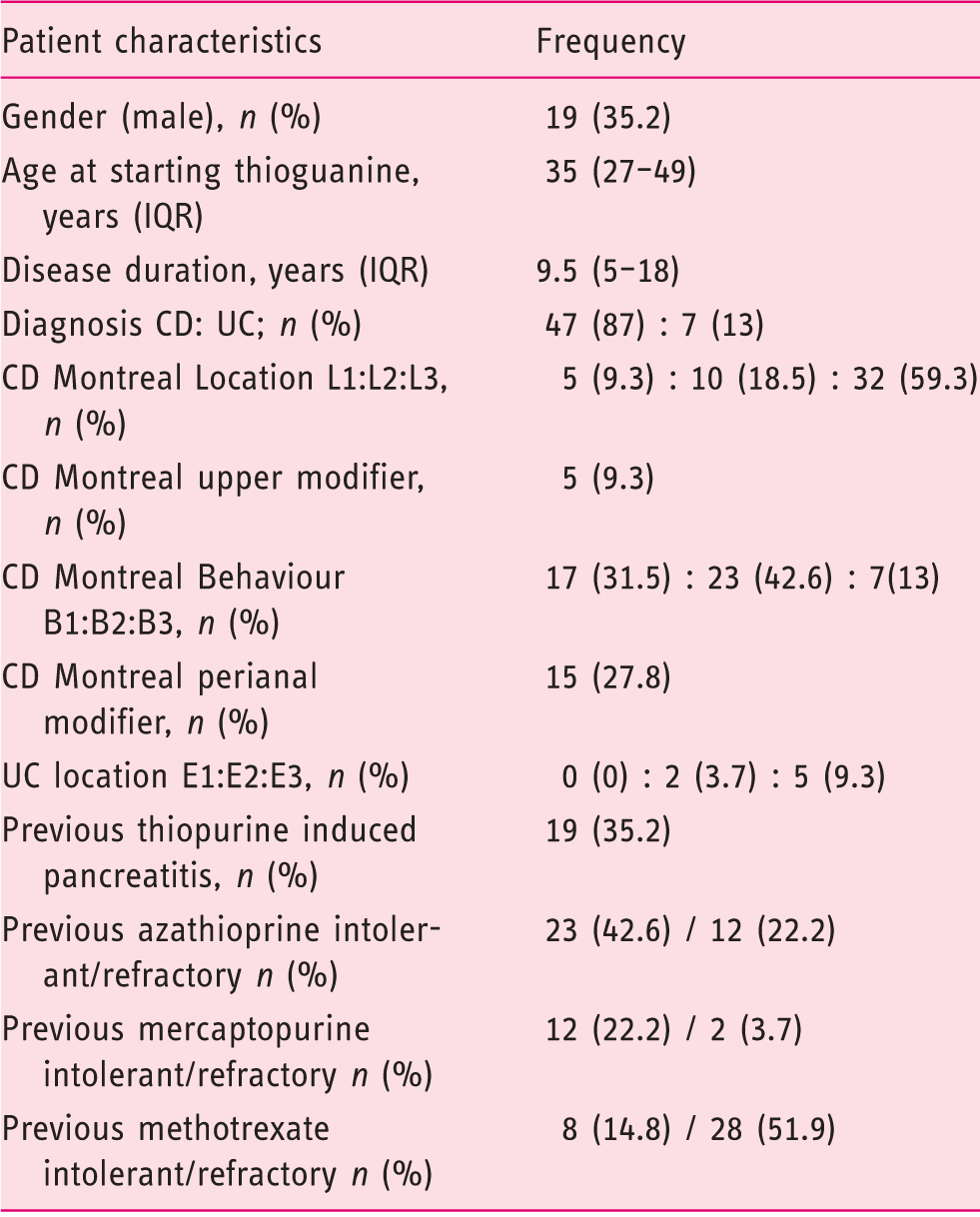
CD: Crohn’s disease; IQR: inter-quartile range; UC: ulcerative colitis.
Thioguanine dose
TG was started at 20 mg in 36 out of 54 patients and 40 mg in 18 out of 54 patients, and was continued as a single daily dose. The mean daily dose of TG was 27 mg (SD 8.4, range 20–40), equating to 0.44 mg/kg body weight (SD 0.19, range 0.2–0.9). The median cumulative dose of TG was 13 g (IQR 4.1–26). Considering the entire cohort, the median duration of treatment was 16 months (IQR 5–37). Of 34 out of 54 (63%) patients continuing TG for ≥12 months, the median duration was 26 months (range: 12–132).
Median TGNs during TG were 740 pmol per 8 × 108 RBC (IQR 445–1078). There was a trend towards a correlation between the dose and TGN (r = 0.28 and p = 0.06), but not between the normalised dose/body weight and TGN (r = 0.026, p = 0.87).
Response
Response rate by indication.
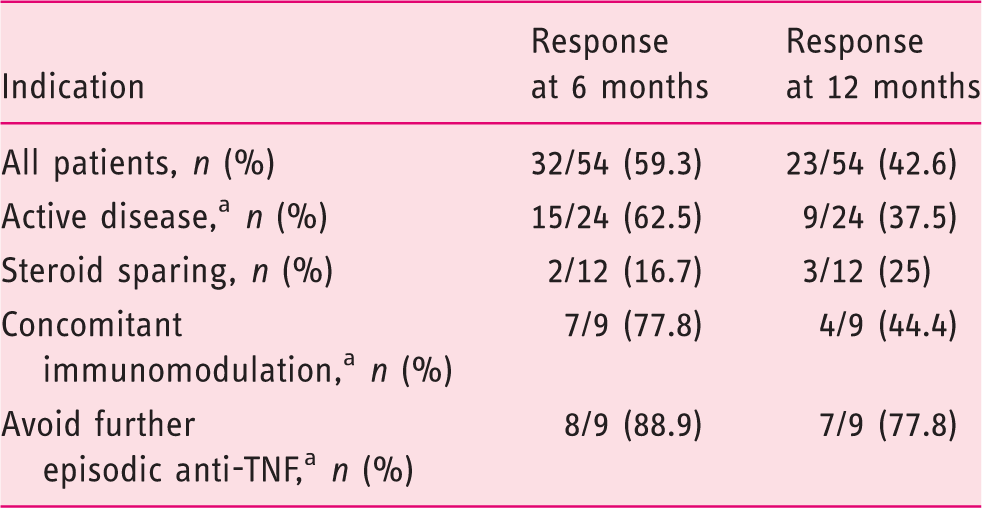
One patient with stoma for each indication.
TNF: Tumor necrosis factor.
Failure and withdrawal
Causes of thioguanine failure.

IBD: inflammatory bowel disease.
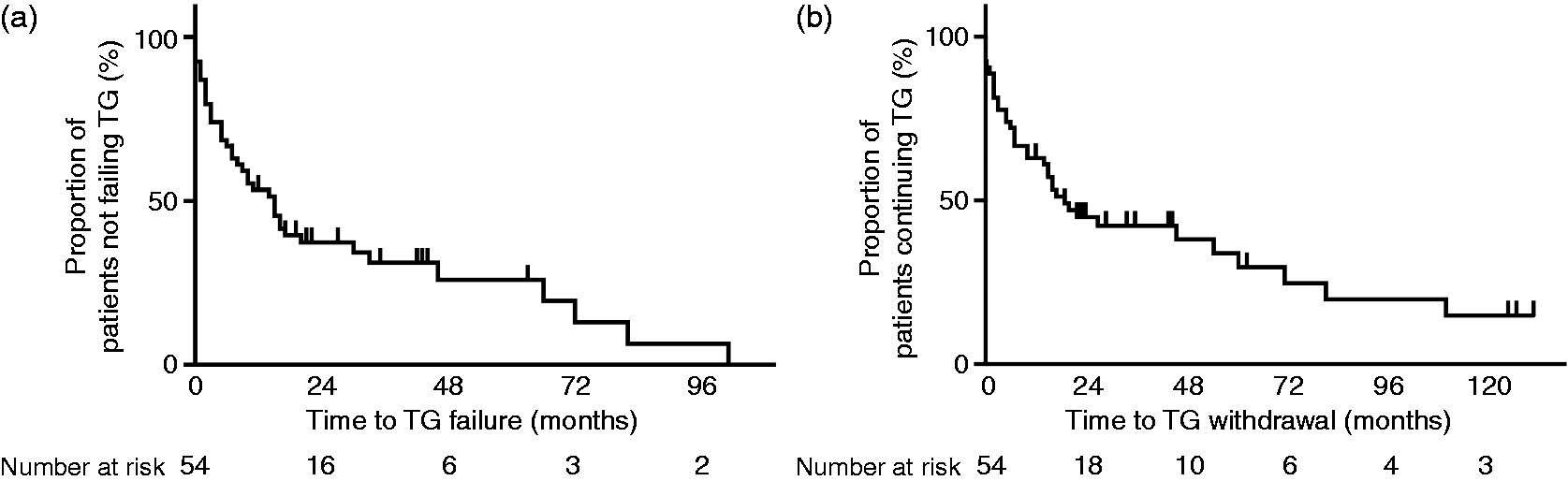
(a) Kaplan–Meier survival plot of 54 patients, showing the time to TG failure in months. The median time to TG failure was 15 months. Vertical lines represent the censored cases. (b) Kaplan–Meier survival plot of 54 patients, showing the time to TG withdrawal in months. Median time to TG withdrawal was 19 months. Vertical lines represent the censored cases.
Causes of thioguanine withdrawal.

Biochemical abnormalities
In two patients treated for two and five months, biochemistry was unavailable (performed externally). 18 out of 52 patients (34.6%) returned abnormal blood tests at least once during treatment. These were: deranged LFTs (n = 9), pancytopenia (n = 2), lymphopenia (n = 7) and neutropenia (n = 1). Abnormal biochemistry led to TG withdrawal in two patients and a dose reduction in one out of 18 patients. In the remaining 15 patients abnormal biochemistry was transient and resolved without dose modification.
Screening for hepatic complications
34 out of 54 patients treated with TG for ≥ 12 months were recommended screening for hepatotoxicity: 24/34 (70.6%) underwent MRI liver at a median of 20 months (IQR 12.3–29 months) after starting TG. 19/24 (79.2%) were normal and 4/24 (16.7%) demonstrated fatty liver disease, of which two were seen on ultrasound prior to TG. A single patient with portal hypertensive syndrome demonstrated splenomegaly, but no other abnormal findings. There were 11 out of 34 patients (32.4%) who underwent liver biopsy: four were normal, two had steatohepatitis and mild fibrosis, three had mild steatosis, one had solitary granuloma and one had a single focus of inflammation. There were no confirmed cases of NRH.
Safety
Four serious events occurred amongst 126 patient-years of treatment. Two patients developed malignancy: a 54-year-old male with gastric cancer, who died 15 months after starting TG (he had been intolerant of thiopurines and was biologic naïve), and a 61-year-old female, who developed metastatic breast cancer 10 months after co-treatment with TG and ADA. Both agents were withdrawn and she remains in remission after chemotherapy. A 57-year-old female previously on AZA and episodic IFX, with no history of liver disease, developed spider naevi, jaundice, ascites and cholestatic LFTs 13 months after starting TG. The cumulative TG dose was 16.8 g, the mean daily dose 40 mg and median TGN 1071 pmol per 8 × 108 RBC. MRI revealed splenomegaly with normal portal/hepatic venous flow and liver parenchyma. She declined a liver biopsy and recovered after ceasing TG. A patient treated with ADA and TG developed neutropenic sepsis and pneumonia, and recovered after interruption of immunosuppression.
Discussion
AZA and MP remain first-line immunomodulators in IBD and are important as concomitant immunomodulation to limit loss of response to anti-TNF agents.2,22,23 However, a proportion of patients fail to tolerate conventional thiopurines2,22 and typically are considered for MTX and in some, immunomodulation is no longer possible. In this setting, TG circumvents most adverse effects encountered with conventional thiopurines, but has been largely ignored. This likely relates to concerns regarding the risk of NRH10,11 and, in some, resigns the patient to anti-TNF monotherapy, despite an anticipated higher rate of loss of response. 23
In this study, we describe the largest series of IBD patients, with the longest period of follow-up after treatment with TG, whom were refractory or intolerant to conventional immunosuppression. Overall, clinical response to TG was seen in 59.3% and 42.6% of patients at six and 12 months, with a median exposure of 16 months. This compares favourably to response rates seen with conventional thiopurines, despite all having previously failed at least one immunomodulator. It is significant that within this difficult-to-treat cohort, 50% of patients continued TG for 19 months. Adverse events with conventional immunosuppression did not recur in the majority. At inclusion, 77.8% were intolerant of AZA, yet only 29.6% discontinued TG because of an adverse event or biochemical abnormality, demonstrating improved tolerability of TG. Myelotoxicity and hepatotoxicity were generally transient and resolved spontaneously, with TG withdrawn in only two patients.
Serious events occurred in four patients over the 126 patient-years of follow-up. One developed a portal hypertensive syndrome, resolving on TG withdrawal. Two developed solid-organ malignancy (both previously exposed to AZA and one also with anti-TNF): one gastric and one breast cancer. TG has historically been used as treatment for breast cancer, 24 and the causes of solid-organ malignancy are multi-factorial; hence, the aetiology of malignancy with thiopurines remains in debate. Aside from the recognised risk of lymphoma 25 and non-melanoma skin cancer, 26 thiopurines are probably associated with a modest increased risk of solid organ malignancy. 27 Of note, in our cohort, we observed no cases of lymphoma nor non-melanoma skin cancer, although given the small patient numbers, our study was underpowered to detect such a relationship.
Approximately 3% of patients treated with thiopurines developed pancreatitis. 6 Hypersensitivity to a component of AZA/MP is likely, supported by the association of thiopurine-induced pancreatitis with the HLA-DQA1*02:01–HLA-DRB1*07:01 haplotype. 28 The mechanism remains unclear; however, importantly pancreatitis did not recur with TG, as was demonstrated in our series where 35% of our patients had prior pancreatitis secondary to AZA. This implicates a metabolite of AZA/MP earlier in the purine salvage pathway, or the parent molecules themselves, as the cause of pancreatitis. Thiopurine-induced pancreatitis is therefore a key setting in which to consider TG.
The incidence of NRH with TG in IBD varies between 0–62%.9–11,13,29 Early studies report significant rates of NRH, confirmed by liver biopsy. This was not replicated in subsequent series using low-dose TG, and earlier concerns regarding TG and NRH may be overestimated. No study has performed baseline MRI or liver biopsy prior to TG. In the study reporting the highest frequency of NRH, 10 most patients had significant exposure to conventional thiopurines prior to TG, and TGNs on TG were significantly higher than in subsequent series, where NRH was not encountered.7,13,29 This implies a relationship between NRH and higher TG doses (>40 mg/day) and hence, higher TGN levels. Studies with TGNs < 1200 pmol per 8 × 108 RBC report no NRH.13,29 In our cohort, which is the longest follow-up reported to date, 33.3% of patients had a median exposure of 32 months, and 20 patients >35 months, without NRH.
Furthermore, NRH is associated with conventional thiopurines, 30 IBD itself, 31 and with other chronic inflammatory conditions. 32 A study utilising intra-operative liver biopsies from thiopurine-naïve IBD patients detected NRH in 6% of CD and 33% of UC patients. 31 Autopsy studies indicate rates of up to 2.6%. 33 Therefore, particularly with lower doses of TG, NRH appears unlikely to be more frequent than the inherent background IBD risk.
The optimal surveillance strategy for NRH is debated. At our institution prior to 2004, patients treated with TG for >12 months were recommended liver biopsy. Subsequently, screening with three monthly LFTs and platelet monitoring, in conjunction with MRI every 24 months, is offered. Screening by biochemistry alone is insufficient; three of nine patients biopsied in the Dubinsky series associating NRH with TG had normal biochemistry. 10 The sensitivity and specificity of liver MRI for NRH is 77% and 72% respectively 11 and is our preferred screening tool, with biopsy being recommended if MRI or biochemistry raise the possibility of NRH.
No correlation between TGNs and response, or between TGNs and side-effects, was seen. We aim for target TGNs of 600–1000 pmol per 8 × 108 RBC, on the grounds that NRH has not been reported with <1200 pmol/8 × 108 RBC. TGNs on TG are higher than those found with AZA/MP, most likely due to rapid uptake by circulating mature RBCs, rather than TGN formation by erythroid precursors in bone marrow. In contrast to the relationship between high TGNs and myelotoxicity seen with conventional thiopurines, high TGNs with TG do not typically cause bone marrow suppression, likely due to differences in thiopurine metabolism in leucocytes between therapies. 34 This is reflected in our study, where significant myelotoxicity was seen in only one patient. Therefore, TGNs on TG cannot be interpreted in the same way as AZA/MP, and future studies involving TG should involve measurement of TGNs in the target cells, namely leukocytes.
We acknowledge several limitations in this study. Clinical efficacy was assessed retrospectively using HBI/SCCAI, and, despite our practice to document this prospectively at every consultation, the response data must be interpreted with caution. Clinical indices (including Crohn’s disease activity index) correlate poorly with mucosal inflammation. 35 The indication for using TG was as concomitant immunomodulation, to maintain response to scheduled or episodic biologic use in a significant proportion of patients (33%). The response rates within this cohort were 83% and 61% at six and 12 months, respectively, which was higher than those seen when TG was prescribed as monotherapy for active disease or steroid sparing (47.2% and 33.3% at six and 12 months, respectively). Whilst this definition of ‘response’ is questionable, it does represent real-world clinical practice. As many patients did not have C-reactive protein measured routinely, we could not assess the biochemical response to TG. NRH was not formally excluded in all; hence, the true rate of NRH may be underestimated. One patient developed a portal hypertensive syndrome, but did not undergo liver biopsy; this could have represented NRH. Where TG was interrupted, we did not report efficacy nor the safety data of subsequent TG therapy, although the numbers were small; hence, the true duration of drug tolerance may have been longer than that reported.
Conclusions
In summary, we report our long-term experience of TG in a cohort of IBD patients’ refractory to, or intolerant of, conventional immunomodulators. The efficacy and side-effects were comparable to conventional immunomodulators and the safety data for TG is reassuring. We acknowledge that the latter, rather than the clinical effectiveness of TG, is the key message that clinicians may find most useful when translating our findings to everyday clinical practice. Thiopurine-induced pancreatitis did not recur with TG and the majority of patients who previously discontinued conventional thiopurines tolerated TG.
In contrast to earlier studies with TG, we did not observe NRH in this cohort after long-term follow-up (the largest reported in the literature). NRH may relate partly to dose and elevated TGNs; hence, TGN measurement and vigilance in monitoring for NRH are key issues when using TG. We recommend doses ≤40 mg/day, with dose adjustments based on TGN levels.
Head to head studies of TG versus conventional thiopurines would help establish the safety and efficacy of TG in IBD, but are unlikely to be undertaken. Nonetheless, TG remains an acceptable alternative in patients with IBD who have not tolerated or have failed conventional immunomodulators.
Footnotes
Acknowledgements
MW contributed to study design, data acquisition, analysis, and wrote and revised the manuscript. KP, VK, RG, BW, PB and TE contributed to data acquisition, manuscript writing and revision. PI, AM and JS contributed to study design, manuscript revision and intellectual content. All authors approved the final version of the manuscript.
Declaration of conflicting interests
There is no conflict of interest for any author to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial or non-for-profit sectors.
