Abstract
Background
Terlipressin is used as pharmacological treatment for variceal bleeding. The drug’s physiological effect favours hyponatremia, and rapid changes in plasma sodium (PNa) may cause brain injury. Cirrhosis patients seem to be largely protected against this effect but patients without cirrhosis may not be so.
Objective
The objective of this study was to examine whether terlipressin treatment of patients without cirrhosis leads to more serious fluctuations in PNa than in cirrhosis.
Methods
In a retrospective cohort design, during a 39-month period, 11 patients with prehepatic portal hypertension and no cirrhosis and 134 patients with cirrhosis received a minimum cumulative terlipressin dose of 4 mg during at least 24 hours for variceal bleeding. The groups’ PNa changes were compared.
Results
During terlipressin, the non-cirrhotic patients developed a greater reduction in PNa [mean 8.3 (95% confidence interval (CI) 1.9–14.6) vs. 1.8 (1.0–2.7) mmol/l; p = 0.048], a lower nadir PNa [129 (123–135) vs. 133 (132–134) mmol/l; p = 0.06], and within 48 hours after terlipressin a greater increase in PNa [12.6 (3.4–21.7) vs. 2.3 (1.5–3.0) mmol/l; p = 0.03]. Severe (>10 mmol/l change) hyponatriemia or PNa rebound were seen in 27% of these patients but in only 4% of those with cirrhosis (p = 0.02). One non-cirrhotic patient developed permanent brain damage.
Conclusion
Terlipressin treatment of bleeding varices carries a high risk of potentially dangerous PNa fluctuations in patients with non-cirrhotic prehepatic portal hypertension.
Keywords
Key summary
Terlipressin’s physiological effect favours hyponatremia and rapid changes in plasma sodium (PNa) may cause brain injury. This study shows that terlipressin treatment of bleeding varices carries a particularly high risk of potentially dangerous PNa fluctuations in patients with non-cirrhotic prehepatic portal hypertension compared to patients with cirrhosis. Our results call for caution if terlipressin is used in patients without cirrhosis.
Introduction
Terlipressin is a vasopressin analogue that given in supra-physiological doses decreases the portal pressure.1–3 This is the background for the drug’s use in cirrhosis patients as acute pharmacological treatment for variceal bleeding4,5 which controls the bleeding in 80% and reduces the mortality by 40%.6,7 The drug’s physiological effect in increasing renal sodium excretion leading to hyponatremia 8 is generally not a problem in the usually habitually sodium-retaining cirrhosis patients and side effects are less important compared to the risk from bleeding. However, patients with portal hypertension and no cirrhosis may be at higher risk for terlipressin’s physiological effects. Accordingly, two cases of development of severe hyponatremia is reported.9,10 As a result of studies demonstrating terlipressin-induced hyponatraemia also in cirrhosis,11–16 the latest European Baveno recommendations for treatment of bleeding varices make a note on this risk. 4
Acute hyponatremia can induce neurological complications by cerebral oedema, and rapid correction of it may lead to permanent brain injury by osmotic demyelination. This risk is highest at plasma sodium (PNa) increases above 10 mmol/l per 24 hours.17,18 It is therefore relevant to examine systematically whether terlipressin treatment carries a higher risk for such PNa fluctuations in patients without cirrhosis.
The purpose of this study was to compare PNa fluctuations during and after terlipressin treatment for bleeding varices between patients with non-cirrhotic prehepatic portal hypertension and patients with cirrhosis. We did so in a consecutive retrospective patient cohort.
Materials and methods
Study population
From November 2011 to February 2015, a total of 284 patients were included based on their first episode of terlipressin treatment at the Department of Hepatology and Gastroenterology, Aarhus University Hospital, Denmark, identified from the electronic patient record system, which is complete also as regards medications. Of these, 177 (62%) were treated for variceal bleeding, 100 (35%) for hepatorenal syndrome, and 7 (3%) for other causes. Sixteen patients received an accumulated dose of terlipressin of less than 4 mg, which is considered irrelevant for the present purpose, and they were excluded. Further excluded were 16 patients without hepatic or perihepatic disease or with non-cirrhotic hepatic lesions. Thus, the final study cohort comprised 145 patients, of whom 11 had non-cirrhotic prehepatic portal hypertension and 134 had cirrhosis. The non-cirrhotic portal hypertension was due to portal vein thrombosis (n = 6), splenic vein thrombosis (n = 1), portal vein anomaly (n = 2), or compression (n = 2).
PNa measurements
Venous PNa was recorded at baseline (at commencement of terlipressin treatment) and thereafter every 24 hours. The terlipressin-induced decrease in PNa is given as the baseline PNa minus the nadir PNa during treatment. The post-treatment PNa increase is given as the maximum PNa within 48 hours after terlipressin minus the PNa on the day of treatment discontinuation. The PNa measurements were secured according to clinical routine. Thus, the time from commencement and discontinuation of terlipressin to the first following PNa measurement can be less than 24 hours. For this reason, the 48-hour post-discontinuation measurement was chosen to ensure that the effect of terlipressin discontinuation would not be missed in patients who received their final dose of terlipressin late the previous day. PNa data were 94% complete during terlipressin. PNa after terlipressin was available in 8 (73%) non-cirrhosis and 108 (81%) cirrhosis patients.
Statistical analysis
Data were analysed using Stata v.14.2 (StataCorp, College Station, TX, USA). Data are presented as mean if nothing else is stated. Skewed data that obtained normality when log-transformed are presented as median values. Estimates are given with their 95% confidence interval (CI). Student t test was used for comparison between the groups, but for data with few observations the Wilcoxon–Mann–Whitney test was used. Fisher exact test was used for ordinal data. Spearman rank correlation coefficient (ρ) was used to describe the relationship between two variables. A p value <5% was regarded as significant in a two-sided test.
The study protocol conformed to the Declaration of Helsinki and the Central Denmark Region Committees on Health Research Ethics. The study was approved by the Danish Data Protection Agency (journal no.: 1-16-02-545-15, date of approval: 21 October 2015) and surrogate patient consent was obtained from the Danish Patient Safety Authority (journal no.: 3-3013-1258/1, date of approval: 4 February 2016).
Results
Cohort characteristics
Patient clinical and biochemical characteristics upon terlipressin initiation.
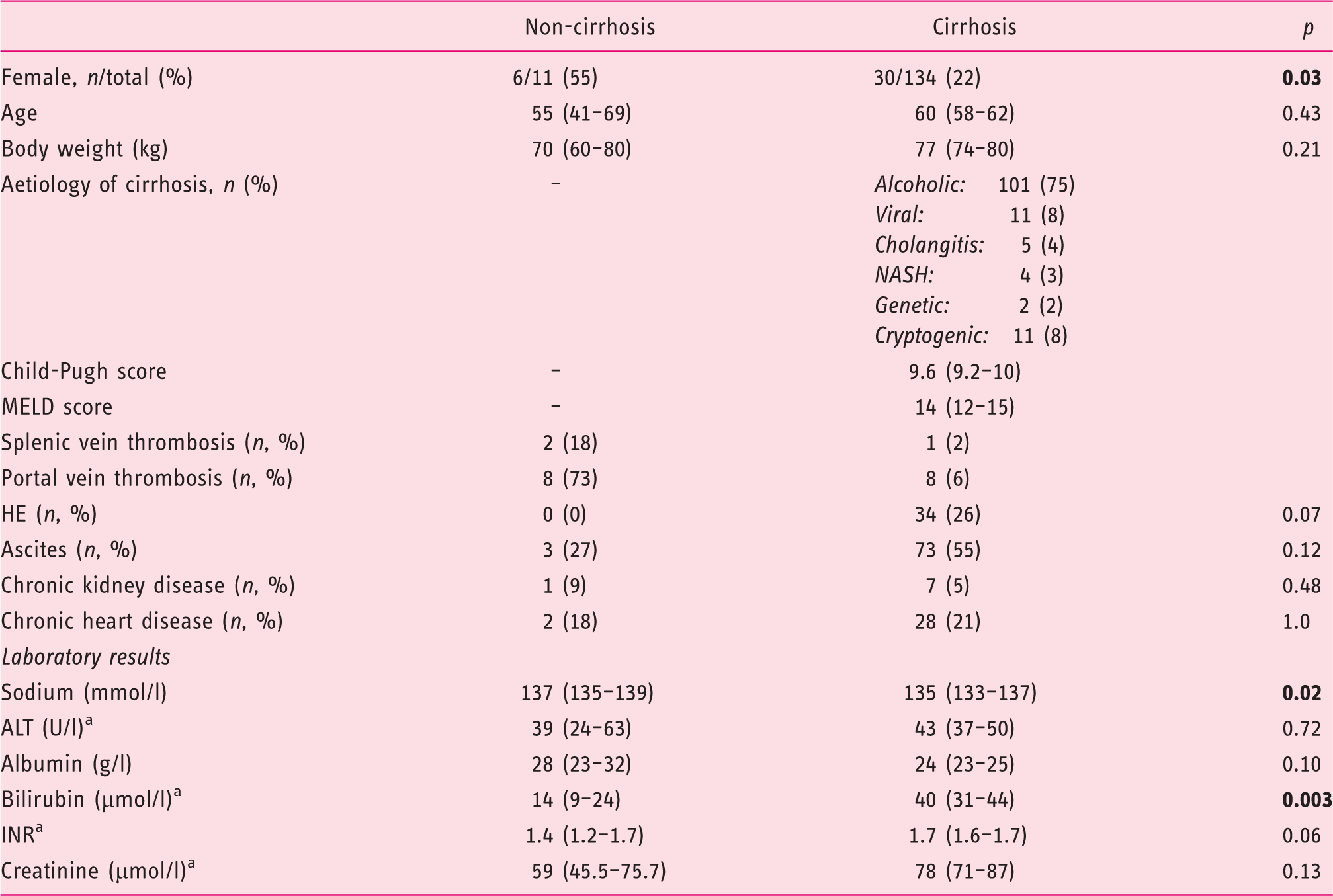
ALT: alanine aminotransferase; HE: hepatic encephalopathy; INR: international normalised ratio; MELD: Model for End-Stage Liver Disease; NASH: non-alcoholic steatohepatitis.
Estimates are given as mean (95% confidence interval) except fora, where median (95% confidence interval) is given. Bold values represent statistical significance (p < 0.05).
Bleeding severity and treatment protocol
Severity of bleeding and details regarding treatment.
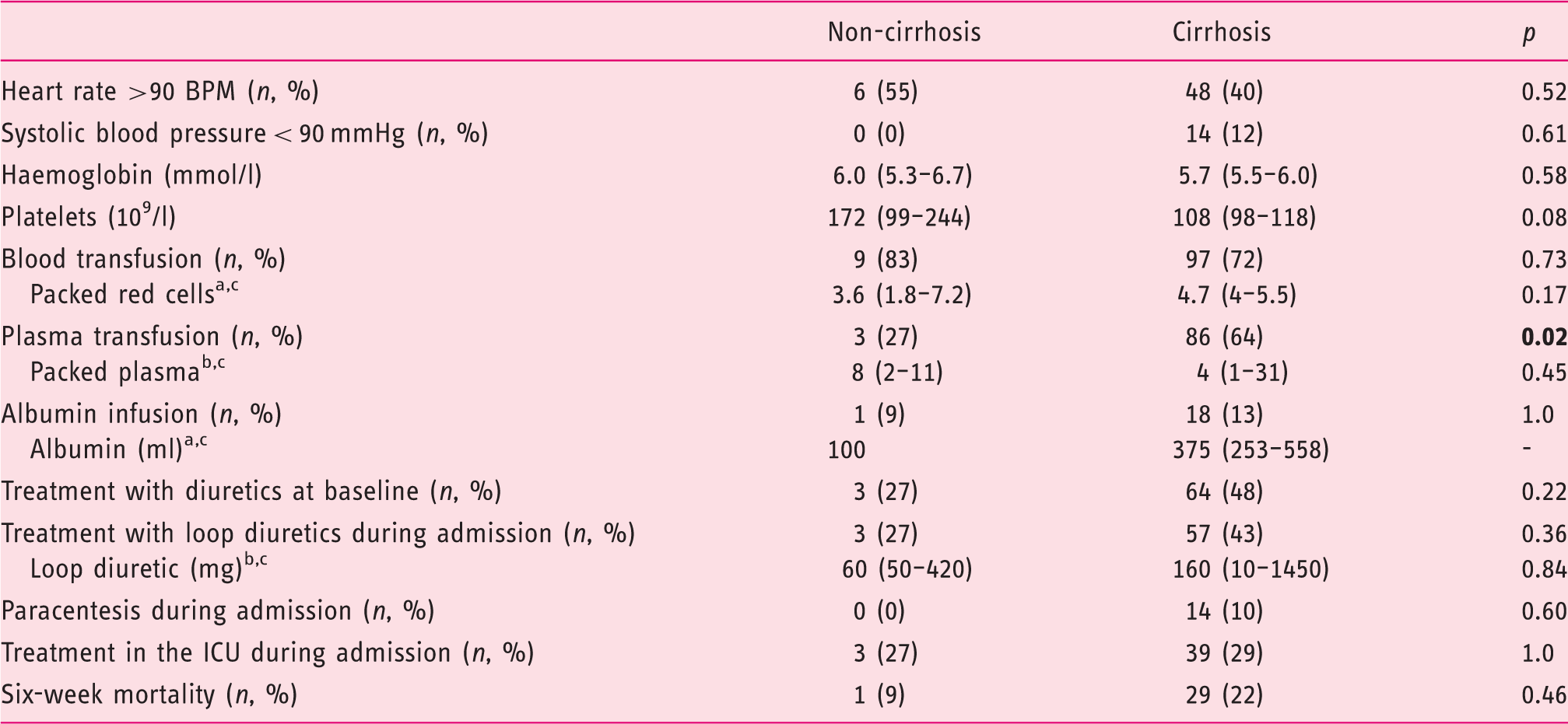
BPM: beats per minute; ICU: intensive care unit.
Estimates are given as mean (95% confidence interval) except fora, where median (95% confidence interval) andb, where median (range) are given.
Estimated for patients receiving at least one pack/dose. Bold values represent statistical significance (p < 0.05).
Terlipressin treatment was initiated at a dose of 1–2 mg/4 hours and tapered after bleeding cessation before discontinuation. The two groups were similar in regard to median cumulative dose of terlipressin [16.2 (9.0–29.2) vs. 13.2 (11.6–15.1) mg; p = 0.40] and median days of treatment [3.3 (2.0–5.6) vs. 2.6 (2.3–3.0); p = 0.32]. In 1 (9%) non-cirrhotic patient and in 18 (13%) of patients with cirrhosis, terlipressin was stopped because of side effects (signs of organ ischaemia or hypertension) or failure of bleeding control.
Changes in plasma sodium concentration
The baseline PNa was marginally higher in the non-cirrhotic patients [137 (135–139) vs. in cirrhosis 135 (133–137) mmol/l; p = 0.02] (Figure 1). Terlipressin induced a greater decrease in PNa in the non-cirrhotic patients [8.3 (1.9–14.6) vs. in cirrhosis 1.8 (1.0–2.7) mmol/l; p = 0.048]. The non-cirrhotic patients also tended to reach a deeper nadir PNa [129 mmol (123–135) vs. in cirrhosis 133 mmol/l (132–134); p = 0.06].
Average plasma sodium profiles during and after terlipressin treatment.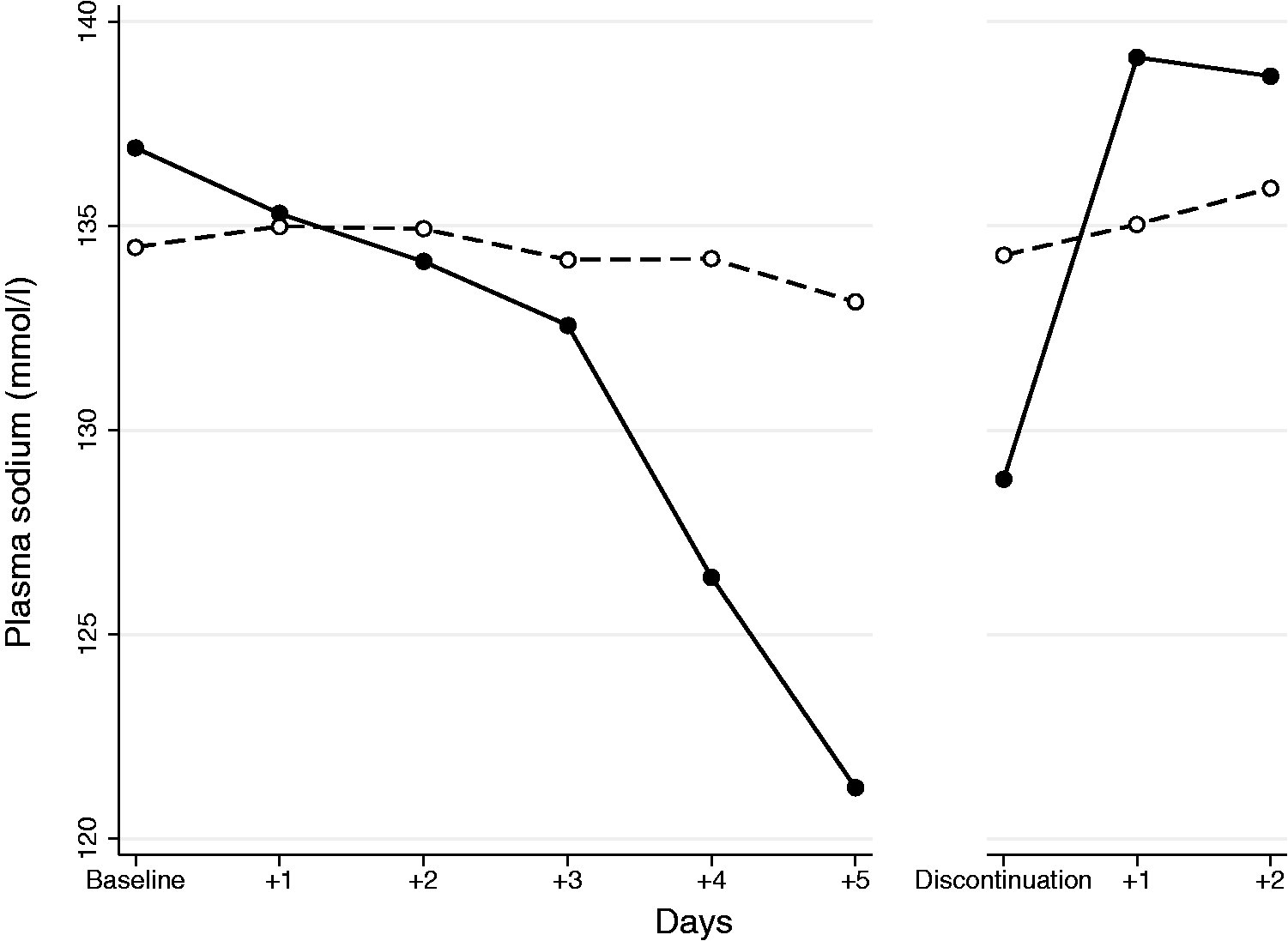
After terlipressin discontinuation, PNa increased in both groups but more so and more rapidly in the non-cirrhotic patients [12.6 (3.4–21.7) vs. 2.3 (1.5–3.0) mmol/l; p = 0.03], and the increase in these patients occurred within 24 hours (Figure 1 and Appendix S1). PNa within 48 hours of terlipressin discontinuation was similar in both groups [139 (135–144) mmol/l vs. in cirrhosis 136 (135–137) mmol/l; p = 0.16].
Three (27%) of the non-cirrhotic patients experienced a severe treatment-induced PNa decrease of >10 mmol/l and the same three patients a severe increase after terlipressin. This happened in only 4% of the cirrhosis patients (p = 0.02).
A higher cumulative terlipressin dose as well as more days of treatment were associated with a more marked PNa decrease in both non-cirrhosis (ρ = –0.84, p = 0.002 and ρ = –0.74, p = 0.009, respectively) and cirrhosis (ρ = −0.27, p = 0.002 and ρ = –0.21, p = 0.02, respectively) (Appendix S2 and S3). In addition, pre-treatment normonatremia was associated with larger PNa fluctuations than baseline hyponatremia (Appendix S4). There was no difference in PNa decrease between patients receiving and patients not receiving diuretics within the groups (p > 0.25).
Nine cirrhosis patients had portal vein thrombosis and their PNa profile was not different. Alcoholic vs. non-alcohol aetiology had no influence on the PNa profiles. There was no gender difference in the use of terlipressin or PNa fluctuations.
Neurological manifestations related to PNa fluctuations were reported in one patient with non-cirrhotic portal hypertension who developed permanent brain damage on the basis of myelinolysis.
Discussion
This is the first cohort study on PNa fluctuations during terlipressin treatment for variceal bleeding in patients with non-cirrhotic prehepatic portal hypertension. Our main finding was that these patients more often developed severe hyponatremia and more rapidly normalised their PNa after treatment discontinuation than did patients with cirrhosis. In one case this had disastrous consequences.
The present study is in line with the two case reports of non-cirrhotic prehepatic portal hypertension patients who experienced a reduction of serum sodium of ≥20 mmol/l during terlipressin treatment and a prompt serum sodium recovery after discontinuation.9,10 Less marked PNa lowering effect of terlipressin has otherwise been reported for patients with cirrhosis.11–16 In accordance, we observed slight PNa reduction in the majority of the cirrhosis patients but this was >10 mmol/l in only a few cirrhosis patients. We observed PNa decrease >5 mmol/l in 28% of cirrhosis patients, which is comparable to 35% reported by Kang et al. 15 but lower than the up to 67% reported by others.14,16 These studies, however, used more terlipressin for a longer time.
Dose and duration of treatment were equal in the two groups of our cohort and cannot explain the marked difference in PNa profile. One difference is that the non-cirrhotic patients were all normonatriemic before treatment, which all the cirrhosis patients were not. Normonatriemia was a risk factor for terlipressin-induced hyponatremia in cirrhosis patients (Appendix S4) as also reported by others.14–16
Our and others’ data suggest that the cessation of terlipressin treatment causes the sudden increase in PNa. Previous studies of cirrhosis patients report the recovery time of PNa after terlipressin to be three to six days,14–16 which is in accordance with the PNa profiles of our cirrhosis patients. In contrast, in our study, all but one non-cirrhotic patient recovered their baseline PNa already within 24 hours of the final terlipressin dose, even though terlipressin over the two preceding days was tapered to 1 mg on the day of discontinuation. So, dose reduction of terlipressin does not protect against the PNa rebound in non-cirrhotic patients.
Our study, despite comprising a limited number of patients in the non-cirrhotic group, still was able to demonstrate consistent and marked differences in PNa profiles between the two groups. The retrospective design is likely not a serious problem because the patients were well characterised and because we used a complete list of all patients treated with terlipressin over a defined period of time. In support of our findings, the two groups were comparable regarding potential confounding factors such as cumulative dose and duration of the terlipressin treatment, bleeding severity, baseline kidney function and supportive treatment during the bleeding episodes. Diuretic treatment was comparable between the groups and the PNa fluctuations were unrelated to the use of diuretics in both groups.
Our results are of clinical relevance because severe hyponatremia and its normalisation can induce immediate neurological disturbances by cerebral oedema17,18 and in worst case permanent brain injury due to osmotic demyelination, such as we unfortunately experienced in the one case that motivated this study. Our patients were not systematically observed for neurological symptoms that could be related to PNa fluctuations and, if present, could easily have been ascribed to hepatic encephalopathy.
The mechanism of the large PNa fluctuations remains unclear. Terlipressin stimulates the vasopressin V2 receptors in the renal collecting ducts and increase aquaporin-2 expression, decreasing free water clearance.20,21 This could favour dilutional hyponatremia. Patients with cirrhosis have a high level of endogenous vasopressin which may lead to tachyphylaxis against the physiological effects of terlipressin.22–24 If so, patients without cirrhosis may not be protected this way. This line of thought seems to be supported by primary normonatremia being a risk factor for serious hyponatremia in cirrhosis.
Our study gives no clear guidance as to how the hyponatraemia during terlipressin should be managed, neither during development nor during recovery. Our data do not allow for identification of risk factors related to the correctional sodium infusion regimens applied. However, the worst risk evidently is carried by the avid PNa normalisation or rebound occurring upon final discontinuation of terlipressin, even after tapered doses. This may imply that attempts of correction of the hyponatraemia should be performed cautiously and conservatively as regards the amount of sodium infused in relation to the estimated deficit. Our study does not allow for identification of a safe threshold for the cumulative terlipressin dose. The Baveno guidelines recommend terlipressin to non-cirrhotic patients with variceal bleeding, based on studies of patients with cirrhosis.4,19 The guidelines acknowledge this limitation and state a note of caution. However, on the basis of the large PNa fluctuations observed in this study, the benefits and risks of terlipressin for bleeding non-cirrhotic patients should be carefully gauged and alternative vasoactive regimens considered.
In conclusion, terlipressin treatment of bleeding varices carries a high risk of marked and rapid PNa fluctuations in non-cirrhotic patients with prehepatic portal hypertension. As illustrated by our disastrous case, this may be dangerous. Therefore, when terlipressin is administered to such patients, PNa should be particularly carefully monitored and cessation of the treatment considered if the PNa profile shows signs of a decrease in the magnitude of 10 mmol/l.
Supplemental Material
Supplemental material for Terlipressin for variceal bleeding induces large plasma sodium fluctuations in patients without cirrhosis
Supplemental material for Terlipressin for variceal bleeding induces large plasma sodium fluctuations in patients without cirrhosis by Peter Lykke Eriksen, Anne Luise Hartkopf-Mikkelsen, Peter Ott, Hendrik Vilstrup and Niels Kristian Aagaard in United European Gastroenterology Journal
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
Surrogate patient consent was obtained from the Danish Patient Safety Authority (journal no.: 3-3013-1258/1, date of approval: 4 February 2016).
Ethics approval
The study protocol conformed to the Declaration of Helsinki and the Central Denmark Region Committees on Health Research Ethics. The study was approved by the Danish Data Protection Agency (journal no.: 1-16-02-545-15, date of approval: 21 October 2015).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
