Abstract
Introduction
Approximately 30% of healthy volunteers (HVs) show dilated intercellular spaces in the esophageal epithelium suggesting a functionally reduced epithelial integrity. We aimed to evaluate the presence of an altered epithelial integrity in HVs and whether physiological acid could explain such a difference.
Methods
Biopsies for Ussing chamber experiments were taken between 3 cm and 5 cm proximal to the gastroesophageal junction. Twenty-four-hour impedance-pH (MII-pH) monitoring was performed in the same 15 HVs. MII-pH tracings from 24 HVs before and after treatment with esomeprazole (40 mg b.i.d., two weeks), a proton pump inhibitor, were analyzed. Reflux parameters and impedance baseline (IB) at different levels of the esophagus were calculated.
Results
Epithelial integrity in the distal esophagus presents a large variability in vivo and in vitro (transepithelial electrical resistance 196.9 ± 16.27Ω. cm2; IB measurements 2022 ± 143.5Ω). Esomeprazole highly suppressed the total acid exposure time (AET) (1.9 (0.8–3.1) vs 0 (0–0)%, p < 0.0001). After splitting our participants into “high” and “low” IB, based on the median value, we observed only in the distal esophagus a higher total AET before (2.8 (1.6–4.8) vs 1.0 (0.5–2.2), p = 0.04) and increased IB values after esomeprazole (1620 (1347–1898) vs 2192 (1784-2503)Ω, p = 0.002) in the “low” IB group.
Conclusion
A subgroup of HVs presents a low epithelial integrity in the distal esophagus probably due to the increased presence of physiological acid reflux. Whether these individuals have a higher chance to develop gastroesophageal reflux disease is unknown. The role of epithelial integrity in symptom perception needs to be further explored.
Keywords
Current knowledge
The presence of a reduced epithelial integrity by means of dilated intercellular
spaces is found in 20% to 40% of healthy volunteers. Impedance baseline (IB) values are lower in patients with gastroesophageal reflux
disease (GERD). IB values in GERD increase after treatment with proton pump inhibitors.
New findings
Epithelial integrity in the distal esophagus of healthy volunteers shows a large
variability both in vitro as well as in vivo. There is a subgroup of healthy volunteers showing a low epithelial integrity and
higher physiological acid exposure time in the distal esophagus. After treatment with esomeprazole (b.i.d.) for two weeks, epithelial integrity
markedly improves in the low epithelial integrity group. Our study shows that physiologic acid reflux can impair epithelial integrity in
healthy individuals.
Introduction
Gastroesophageal reflux disease (GERD) may present with typical manifestations (heartburn/regurgitation) and atypical symptoms (chronic cough/wheezing/hoarseness) that can coexist in the same individual. 1 GERD affects about 20% of the adult Western population. 2 Although the majority of patients with typical reflux symptoms (±60%) have no evidence of erosive esophagitis (EE), quality of life is adversely affected by GERD. 3 The Vevey consensus therefore defined nonerosive reflux disease (NERD) as “a subcategory of GERD characterized by troublesome reflux-related symptoms in the absence of esophageal mucosal erosions/breaks at conventional endoscopy and without recent acid-suppressive therapy.” 4 Since acid was long believed to be the main pathological factor in provoking symptoms, treatment with proton pump inhibitors (PPIs) is the first-line approach in GERD. PPIs have a very high healing rate for EE; however, a large proportion of patients (∼40%), mostly NERD patients, remain symptomatic while using a standard dose of PPIs.5–8
Not only are PPIs very successful in suppressing gastric acid secretion, they also exhibit an antioxidant effect, both in vitro as well as in vivo, and they have an additional anti-inflammatory effect.9,10
Although NERD patients are characterized by the absence of esophageal mucosal damage on routine endoscopy, previous studies have provided evidence for impaired mucosal integrity in these patients. The presence of dilated intercellular spaces (DIS) in the esophageal mucosa, suggesting reflux-induced damage, was demonstrated by electron microscopy and histology.11–13 Nevertheless, the presence of DIS was also found in 20% to 40% of healthy volunteers (HVs).14,15 The Ussing chamber technique is the gold standard in the analysis of epithelial integrity, but is used only in the research environment since it requires expertise and is time-consuming. Epithelial integrity by means of esophageal transepithelial electrical resistance (TEER) has been studied in small cohorts of HVs as a marker of mucosal integrity, but many of these studies have suffered from methodological issues.16–24
Several recent studies support the use of intraluminal impedance baseline (IB) measurements to evaluate esophageal epithelial integrity. Intraluminal impedance monitoring was developed to detect reflux of all types of acidity and enables us to classify esophageal content as gaseous, liquid or mixed. 25 In between meal and reflux episodes, impedance values return to baseline, depending on the characteristics of the esophageal wall. We previously showed that exposure of rabbit esophageal epithelium to acid decreases TEER and reduces IB values, with a correlation between these in vivo and in vitro measurements. 26 Moreover, similar correlations were reproduced in HVs. 27 Furthermore, IB values are lower in GERD patients compared to HVs and IB values in the distal esophagus correlate with the degree of total esophageal acid exposure time (AET). 28 Additionally, IB values in GERD patients increase after PPI treatment. 29 Based on these data, IB measurements can be considered a marker for esophageal epithelial integrity, sensitive to change.
Based on this observation and the knowledge that up to 30% of HVs have DIS, we hypothesize the existence of two subpopulations of HVs: one with a low and one with a high epithelial integrity. 15 We postulate that variation in physiological reflux parameters may be responsible for such differences. Although PPI treatment effectively reduces AET and improves epithelial integrity in GERD patients, the effect of PPI treatment on the epithelial integrity in HVs is unknown. 29 Hence, the aim of our study was to evaluate in HVs first, the presence of an altered epithelial integrity both in vitro and in vivo and second, whether physiological esophageal acid exposure could explain such variability.
Materials and methods
Study 1: In vitro and in vivo evaluation of esophageal epithelial integrity
HVs
Fifteen HVs were recruited in Leuven, Belgium. Exclusion criteria were the presence of typical (heartburn/regurgitation) and atypical (chronic cough/asthma/wheezing/hoarseness/chest pain) reflux symptoms, history of thoracic or digestive surgery and the use of medication altering intragastric acidity or esophageal motility. The study was approved by the Ethical Committee of the University Hospital Gasthuisberg (S52721) and informed consent was obtained from all HVs.
In vitro evaluation of esophageal epithelial integrity
During upper endoscopy, three biopsy samples were taken with a biopsy forceps (Radial Jaw3, Boston Scientific, MA, USA) from the distal esophagus 3 cm to 5 cm above the esophagogastric junction (EGJ) by two experienced endoscopists (JT/TV), and put in ice-cold Hanks’ Balanced Salt Solution buffer for Ussing chamber experiments.
Biopsy specimens were mounted in modified 3 ml Ussing chambers (Mussler Scientific Instruments, Aachen, Germany) under the magnification of a binocular as described previously. 30 Both mucosal and serosal compartments of the Ussing chambers were filled with 3 ml 10 mM glucose in Krebs buffer. Solutions were kept at 37℃ and continuously carbogenated with O2/CO2 (95/5%). Experiments were performed in open-circuit conditions. Fluorescein (Fluorescein; molecular mass 400 Da, 20 mg/200 µl; Sigma-Aldrich, MO, USA), was added to the mucosal side. Serosal samples were collected every 30 minutes over two hours.
In vivo evaluation of esophageal epithelial integrity
Approximately one hour after the upper endoscopy, we started 24-hour esophageal impedance-pH (MII-pH) recordings using a bifurcated MII-pH catheter (Sandhill Scientific Inc, Highlands Ranch, CO, USA), placed with one pH-sensor located 5 cm above the lower esophageal sphincter (LES) and one in the stomach. Impedance channels were located at 3 cm, 5 cm, 7 cm, 9 cm, 15 cm and 17 cm above the LES. The catheter was calibrated in pH 4.0 and 7.0 buffer solutions before the start of the study. Participants were asked to remain upright during daytime and lie down at their usual bedtime. Meal times and postural changes were recorded as event markers by the data logger.
Data analysis
In vitro evaluation of esophageal epithelial integrity
TEER was calculated from the voltage deflections induced by bipolar constant-current pulses of 16 µA every 60 seconds with a duration of 200 ms and was recorded every 30 minutes over two hours. The average of all time points of the biopsy samples was taken and results are presented as Ω.cm2. The fluorescein level was measured using a fluorescence reader (FLUOstar Omega; BMG Labtech, Ortenberg, Germany). The average of time points 60, 90 and 120 minutes was taken and passage of fluorescein is presented as pmol.
In vivo evaluation of esophageal epithelial integrity
Analyses of the MII-pH tracings were performed using criteria described in a consensus report on detection and definitions of acid, nonacid and gas reflux. 31 Esophageal impedance identifies retrograde bolus movements as reflux and enables the detection of the nature (liquid, mixed and gas reflux) and the proximal extent of reflux.
A reflux episode is defined as a progressing drop in impedance to less than 50%, starting at the LES and propagating to at least the next two more proximal impedance channels. Reflux was considered acid when pH <4 for at least four seconds or if pH was already below 4, as a decrease of at least one pH unit sustained for more than four seconds. Weakly acidic reflux was defined as a reflux event with the basal pH remaining between 4 and 7. 31 The number of reflux episodes, AET and bolus exposure were considered increased if values were >95th percentile of data obtained in the HVs. 32 Reflux events were considered proximal if the impedance drop reached the impedance site located 15 cm above the LES.
Study 2: Effect of PPI on in vivo evaluation of esophageal epithelial integrity
HVs
Twenty-four HVs, recruited in Bordeaux and Lyon, France, were studied retrospectively. Exclusion criteria were the presence of typical (heartburn/regurgitation) and atypical (chronic cough/asthma/wheezing/hoarseness/chest pain) reflux symptoms, history of thoracic or digestive surgery, alcohol consumption >40 g/day, smoking >10 cigarettes/day, pregnant or nursing women, diabetes, neurological disorders, allergy to esomeprazole or benzimidazole derivatives, and the use of medication altering intragastric acidity or esophageal motility. The study was approved by the Comité de Protection des Personnes Sud-Ouest Outre-Mer 3 and all HVs gave written informed consent before inclusion.
In vivo evaluation of esophageal epithelial integrity
All HVs were studied after an overnight fast. Twenty-four-hour esophageal MII-pH recordings were performed using an MII-pH catheter (Sandhill Scientific Inc, Highlands Ranch, CO, USA), placed with the pH-sensor 5 cm above the LES and impedance channels located at 3 cm and 5 cm above the LES. A second MII-pH catheter was placed in the proximal esophagus, with impedance channels located 2 cm and 4 cm under the upper esophageal sphincter (UES). Both catheters were calibrated in pH 4.0 and 7.0 buffer solutions before the start of the study. An external reference electrode was attached to the anterior chest wall. Participants were asked to remain upright during daytime and lie down at their usual bedtime. Meal times and posture changes were recorded as event markers by the data logger. A first recording was performed after which all HVs received esomeprazole 40 mg twice daily (b.i.d.) for two weeks. A second measurement was performed while taking PPIs, using the same protocol. Volunteers were asked to reproduce their usual physical activity and meal periods as far as possible.
Data analysis
Analysis of MII-pH tracings (for reflux parameters) was performed as described above.
We measured esophageal IB in the upright period since we previously showed that this better discriminates HVs from GERD patients when compared with the gold-standard technique, measurements in Ussing chambers.33,34 IB at all three segments was assessed in two steady periods of 30 seconds at the beginning and the end of consecutive time windows of 30 minutes, 60 minutes after the placement of the catheter, avoiding values taken close to any period of swallowing or reflux. We assessed the association between different reflux parameters and epithelial integrity, as measured by IB values. 34
Statistical analysis
Data are presented as median (25th–75th percentile) unless stated otherwise. Deviations from Gaussian distribution were tested using the Shapiro-Wilk test (PRISM5.0, GraphPad5).
All correlations were made using Pearson’s or Spearman’s test, as appropriate. Comparison of IB values before and after PPI treatment was performed using a paired or Wilcoxon signed rank test, as appropriate. Comparison between high and low IB values were performed using an unpaired t-test or Mann-Whitney test, as appropriate. Statistical significance was considered when p < 0.05.
Results
Study 1: In vitro and in vivo evaluation of esophageal epithelial integrity
We studied 15 HVs with a median age of 22 years (range 20–38) (seven male/eight female).
None of the 15 HVs showed any signs of erosive esophagitis at upper endoscopy. Epithelial
integrity assessed in vitro and in vivo showed a large variability (Figure 1). TEER and passage of fluorescein were
196.9 ± 16.27Ω.cm2 and 29.81 ± 5.54 pmol, respectively. IB values at 3 cm,
5 cm and 15 cm were 2022 ± 143.5Ω, 1715 ± 110.6Ω and 2174 ± 196Ω, respectively. Variability of (a) transepithelial electrical resistance (TEER) and (b) impedance
baseline values at 3 cm above the esophagogastric junction in healthy
volunteers.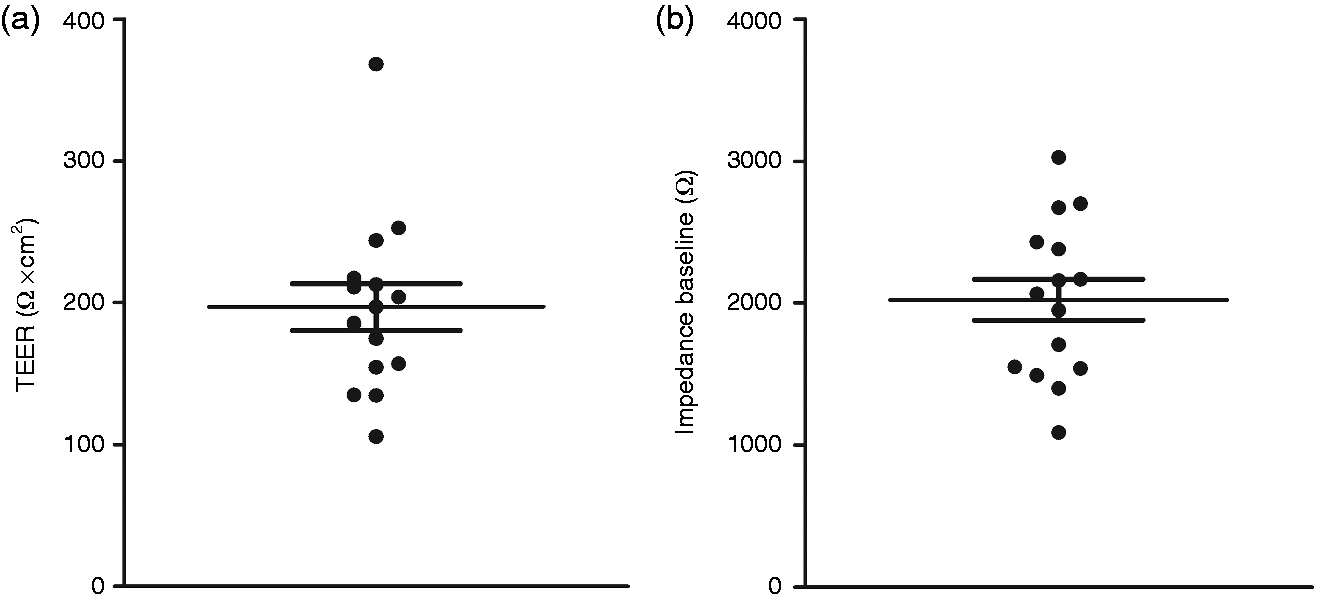
Study 2: Effect of PPI on in vivo evaluation of esophageal epithelial integrity
We studied 24 HVs with a median age of 54 years (range 24–78) (14 male/10 female). As expected, PPI treatment significantly suppressed total AET (1.9 (0.8–3.1) vs 0 (0–0)%, p < 0.0001). We confirmed a shift from acid to nonacidic reflux events with a significant decrease in the number of acid reflux events (26 (12–35) vs 0 (0–2), p < 0.0001) and a significant increase in the number of nonacidic reflux events (11 (3–16) vs 30 (16–37), p = 0.0003). We did find a small but significant reduction in the total number of reflux events after two weeks of PPI therapy (37 (25–51) vs 33 (17–39), p = 0.02).
Impedance baseline values at different levels off and on PPI therapy.
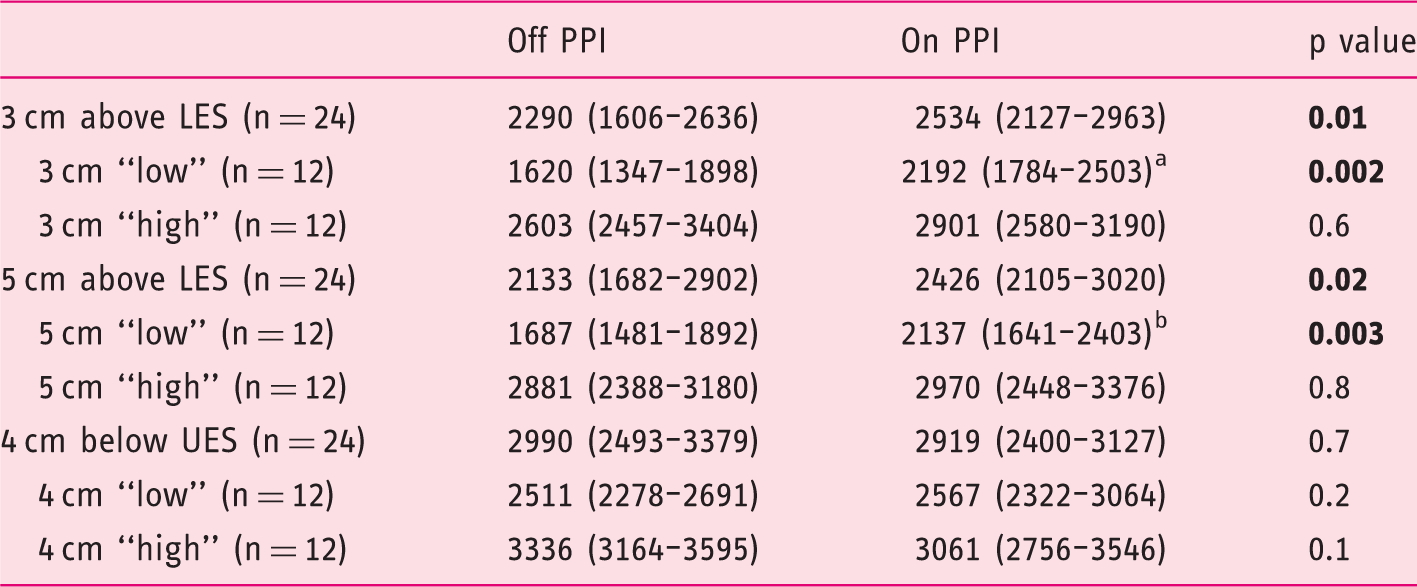
Significantly different with 3 cm “high” on PPI (p = 0.0019).
Significantly different with 5 cm “high” on PPI (p = 0.0038).
PPI: proton pump inhibitors; LES: lower esophageal sphincter; UES: upper esophageal sphincter.
We then split our HVs into a “high” (n = 12) and “low” (n = 12) IB group, based on the median value at different levels in the off-PPI protocol. Both at 3 cm and 5 cm above the LES, IB values did not change after PPI therapy in the “high” IB group (Table 1). Nevertheless, there was a significant increase in IB levels in the “low” IB group after two weeks of PPI use (p = 0.002 and p = 0.003, respectively) (Table 1). No differences were found at the level of 4 cm below the UES in the “high” or in the “low” IB group (Table 1).
Although IB values increased after two weeks of PPI treatment in the “low” IB group in
the distal esophagus, values of IB did not fully recover to values of the “high” IB group,
both at 3 cm and at 5 cm above the LES (Table 1/Figure 2). Impedance baseline (IB) values of healthy volunteers before and after two weeks of
proton pump inhibitor (PPI) treatment, at 3 cm and 5 cm above the lower esophageal
sphincter (LES) and at 4 cm below the upper esophageal sphincter (UES).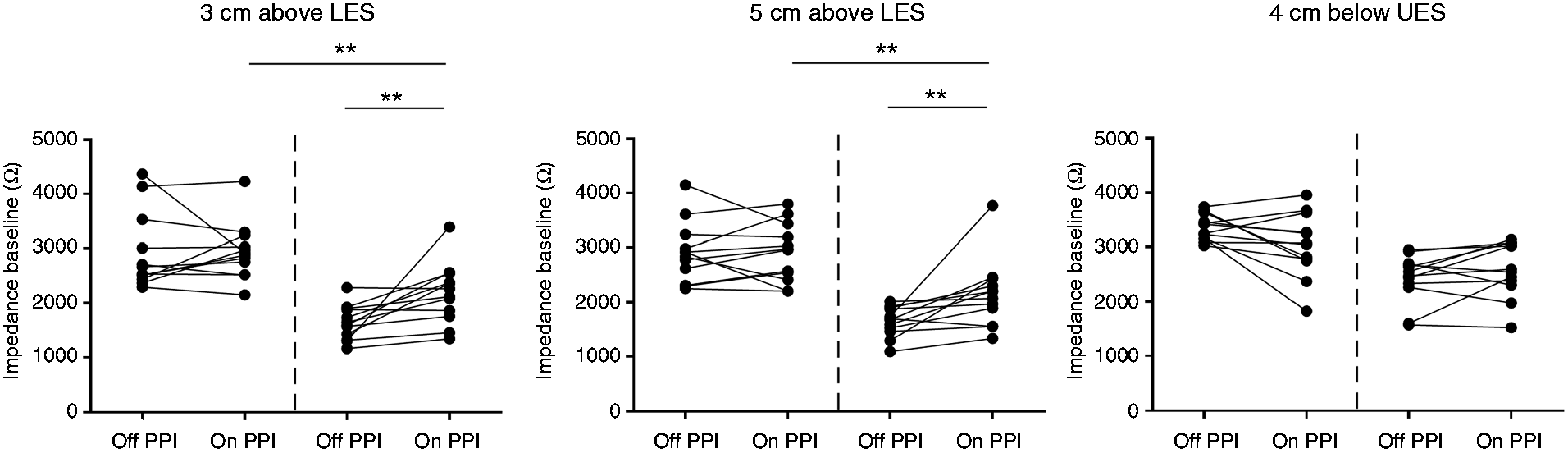
Demographics and reflux parameters in HVs with “low” and “high” epithelial integrity at 3 cm above the LES.
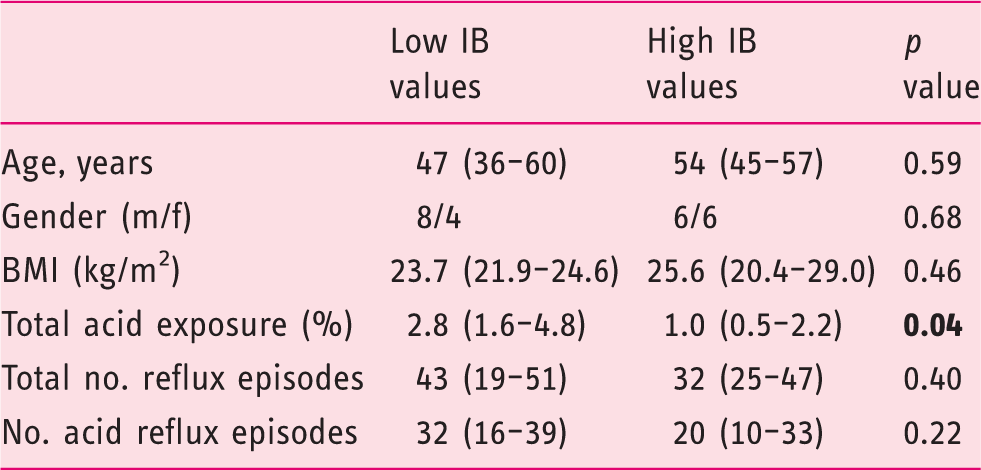
HVs: healthy volunteers; LES: lower esophageal sphincter; m: male; f: female; IB: impedance baseline; BMI: body mass index.
Discussion
Reduced epithelial integrity has been demonstrated not only in GERD patients, but also in NERD patients and even in some asymptomatic HVs.11–15,35 Several reports have established that IB values, derived from MII-pH measurements, can be used as a marker for esophageal epithelial integrity in GERD. IB values are significantly lower in GERD and NERD patients; however, we found (unpublished data) a subgroup of HVs with similar low IB values compared to GERD patients.28,35,36 While it has been shown that IB values of GERD patients increase after PPI treatment, 29 it is unclear whether the lower IB in HVs is attributable to GER, and whether it is affected by PPIs. The goal of our study therefore was, firstly, to evaluate the variability of epithelial integrity measurements in vitro and in vivo and the effect of PPIs in a group of HVs, and secondly, to know whether physiologic reflux parameters could explain at least in part such variability.
In the first part, we found a large variability both in TEER values and passage of fluorescein in HVs. Values for TEER in the distal esophagus range from 106 to 368 Ω.cm2. Previous studies from Orlando’s group showed similar values, with average TEER in the distal esophagus ranging from 224 to 344 Ω.cm2, but studies were performed in a small cohort and often limited by using one biopsy per individual.17–19 Other studies showed much lower values: In a Dutch study, median values for distal TEER in 11 HVs was 116 Ω.cm2 and in two studies in London (n = 14 and n = 22), average values of TEER in the distal esophagus were 151 and 107 Ω.cm2.20–24 The reason for these differences is unclear, but this could be related to technical issues. 16
PPIs are very effective in healing esophagitis in GERD patients. 6 In our cohort of asymptomatic HVs, we found a significant reduction in total AET and confirmed a shift from acid to weakly acidic reflux. 37 However, while the total number of reflux episodes remained similar after PPI treatment in previous studies, we demonstrated a small decrease in the total number of reflux episodes. This might be due to the decrease in gastric secretion volume after PPIs, which has been previously shown. 38
Overall, IB values in the distal esophagus of a group of HVs increased significantly after two weeks of PPI treatment, while there were no differences in the proximal esophagus. This increase was driven by a subgroup of HVs that had “low” IB values and that presented with slightly higher esophageal AET. Since PPIs reduce gastric and hence esophageal acid, this might explain the increase in epithelial integrity in the “low” IB group. However, PPIs not only increase the pH of the gastric contents, they exhibit an additional anti-inflammatory and antioxidant effect.9,10 It is conceivable that this reduction in pro-inflammatory cytokines also contributes to an increase in mucosal integrity, but this would require additional studies for confirmation. 39
Although IB values in the distal esophagus in the “low” IB group increased after two weeks of PPI treatment, they did not fully recover. Since acid is well suppressed with PPI treatment, we believe that other nonacidic components, such as bile acids, pepsin and trypsin, might play an important role. We recently showed that exposure of rabbit esophageal mucosa to solutions containing bile acids is able to impair epithelial integrity. 27 A second explanation could be that it requires more than two weeks of PPI treatment for a full recovery of the esophageal epithelium.
The present study shows the existence of two subgroups of HVs: one with “high” and a second with “low” epithelial integrity. It is only in the “low” integrity subgroup that values increase after PPI use. Both groups, however, were not different in terms of demographics and none of the volunteers had any reflux symptoms. In the “low” subgroup, IB values were substantially lower compared to those studied by de Bortoli and colleagues. 36 It is not clear whether HVs with low IB were excluded in the latter study, or whether none of the HVs presented with low IB values. Despite the fact that we didn’t perform gastroscopy in these HVs, since acid exposure was not pathologically high, we assume endoscopy would be negative in most individuals. There is a wide range in the epithelial integrity measurements in our cohort of HVs, and when comparing our data to a previous published study in GERD patients, almost half of our HVs show IB values comparable to those of GERD patients.29,36 The reason for this lower integrity probably can be found in the AET, which was higher in this subgroup. However, it is still unclear why these HVs do not present with symptoms despite the presence of an abnormal integrity. One hypothesis is that these HVs are primed to develop GERD symptoms over time; however, there are currently no longitudinal data available to support this hypothesis. Alternatively, findings could indicate that an impaired integrity alone, although important, is not sufficient to cause symptoms.
In conclusion, we demonstrated in HVs a high variability of epithelial integrity assessed with in vitro and in vivo techniques. Secondly, we described the presence of two subgroups of HVs: one with “high” and one with “low” epithelial integrity probably as a consequence of physiological esophageal acid exposure. Nevertheless, other components besides acid might be involved in this impaired esophageal epithelial integrity.
Footnotes
Acknowledgment
Author contributions were as follows.
A. Pauwels: analysis and interpretation of data; drafting of the manuscript
C. Broers: acquisition of data; critical revision of the manuscript for important intellectual content
T. Vanuytsel: critical revision of the manuscript for important intellectual content
N. Pardon: acquisition of data
S. Cocca: analysis and interpretation of data
S. Roman: acquisition of data; critical revision of the manuscript for important intellectual content
F. Zerbib: acquisition of data; critical revision of the manuscript for important intellectual content
J. Tack: critical revision of the manuscript for important intellectual content
R. Farré: study concept and design; critical revision of the manuscript for important intellectual content
Declaration of conflicting interests
None declared.
Ethics approval
This study was approved by the Ethical Committee of the University Hospital Gasthuisberg (S52721) and the Comité de Protection des Personnes Sud-Ouest Outre-Mer 3.
Funding
A. Pauwels is funded by a personal Research Foundation Grant (FWO)–Flanders (grant number 1278018N).
Informed consent
Informed consent was obtained from all healthy volunteers.
