Abstract
Introduction
Endoscopic mucosal resection and endoscopic submucosal dissection are demanding procedures. This study aims to establish face, content and expert validity of the live porcine model in performing endoscopic mucosal resection, endoscopic submucosal dissection, complication management and to assess it as a training tool.
Material and methods
Tutors and trainees participating in live porcine model endoscopic mucosal resection and endoscopic submucosal dissection workshops filled out a questionnaire regarding the realism of the model compared to human setting and its role as a learning tool. A 10-point Likert scale was used.
Results
Ninety-one endoscopists (13 tutors; 78 trainees) were involved in four workshops. Median global classifications for the realism of the life porcine model ranged between 7.0–8.0 (interquartile range 5.0–9.0). Procedures resembled human cases with a median of 9.0 (8.0–9.0) for oesophageal multiband endoscopic mucosal resection; 8.5 (8.0–9.0) for oesophageal endoscopic submucosal dissection; 9.0 (8.0–10.0) for gastric endoscopic submucosal dissection; and 9.0 (8.5–9.75 and 8.0–9.69) for complication detection and management. The animal model as a learning tool had median scores of 9.0 (7.0–10.0) considering how procedures are performed; 9.0–9.5 (8.0–10.0) for usefulness for beginners; and 9.0–10.0 (5.0–10.0) regarding it a prerequisite.
Conclusions
Training in a live porcine model was considered very realistic compared to the human setting and was highly appreciated as a learning tool. This is the first study to establish face, content and expert validity of the live porcine model in performing multiband endoscopic mucosal resection, oesophageal and gastric endoscopic submucosal dissection. The validation of this model provides the rationale to incorporate it into formal teaching programmes.
Keywords
Key summary
1. Summarise the established knowledge on this subject
EMR and ESD are demanding procedures requiring extensive training. Numerous live animal courses are organised throughout the world and, until now, a scientific basis in the form of a formal validation of this model has been lacking.
2. What are the significant and/or new findings of this study?
This study provides face, content and expert validity of the live porcine model in performing EMR, ESD and complication management. This validation adds to the ethical justification of animal sacrifice for medical training purposes and provides the rationale to incorporate it into formal teaching programmes. This validation may have an impact on the justification to apply for funding and may form the basis for future studies.
Introduction
Complex advanced endoscopic procedures, such as endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) are considered treatment options for pre-malignant and early neoplastic lesions. These challenging procedures require practical skills obtained through extensive training, are time-consuming and have the potential for serious adverse events.1–3
Multiband mucosectomy (MBM) is one of the EMR techniques that uses a multiband ligation kit enabling sequential banding and snare resection without removing the endoscope, facilitating resections of extensive superficial lesions. It has been used mainly in the oesophagus for the treatment of Barrett’s early cancer.4,5
ESD was developed in Japan in the mid-1990s and has expanded gradually to other countries ever since. The major advantages of this technique are the ability to achieve en-bloc resections allowing for an accurate tumour histopathological staging and grading with precise evaluation of resection margins, R0 resections and lower rates of local recurrence.2,6,7
A learning curve in the performance of ESD has been demonstrated, and more experienced endoscopists have a higher rate of en-bloc resection, reduced procedure duration and fewer related adverse events.8,9
In the East, ESD teaching has followed the traditional master-apprentice model with hands-on human cases supervised by expert endoscopists.9,10 However in Western countries the number of highly qualified ESD experts is low and the prevalence of gastric superficial neoplasms is much lower, diminishing the opportunity to start performing ESD on the easiest and safest locations. Therefore, in Western countries, a pre-patient training programme, including simulation models, is recommended.2,11–13
Currently, existing training programmes have consistently included: acquiring basic knowledge, practicing on animal models (ex vivo animal organs and live porcine models), visiting centres with a high ESD volume, attendance of hands-on training ESD workshops and only then proceeding to clinical practice, ideally under supervision by an expert when available during the first cases.2,11–13
Workshops using animal models are being organised in many specialised training centres with the potential to aid in speeding up the learning process and achieving initial competence in ESD in a safe learning environment with direct one-on-one expert supervision.14–16
It seems logical to practise skills in advanced endoscopic resections in a live porcine model, which is considered to have the closest resemblance to the human anatomy.
However, for every technology, teaching tool or method, and particularly in the case of a live animal model, evidence to support its validity should first be established before its wide practice use, sacrifice of animals and financial resources employment.
The validation process should start from basic premises and, accordingly, it is necessary to prove the live porcine model provides a realistic comparison to the real-life human setting. Validity of the model can be established by studying face, content and expert validity.
Face validity refers to the degree to which a model appears to represent the intended purpose as assessed by its users. Content validity refers to the extent to which the content of the model incorporates all the relevant, appropriate, comprehensive and consistent domains of items that we want to assess. Expert validity is attained when this appreciation is recognised by experts in the field.17–19
This study aims to establish face, content and expert validity of the live porcine model in performing ESD and EMR and subsequent complication management, in order to assess its value as a training tool.
Material and methods
Study design
Animal model
Live pigs (Sus scrofus domesticus) weighing between 30–40 kg were used. The animals were given a liquid diet for three days and fasted for eight hours before the procedures. With the support of the veterinarian staff throughout the course, general anaesthesia with endotracheal intubation and mechanical ventilation was performed according to local protocol.
Participants
Faculty members (tutors) and trainees attending a course for EMR/ESD training using live porcine were invited to fill out a questionnaire at the end of the two-day workshop they were involved.
Validation questionnaire
The questionnaire contains 75 questions in three separate domains. This questionnaire is based on similar validation questionnaires from previous studies from our group.20,21 The first part is related to demographic data and includes level and range of endoscopic and simulators experience. The second part is centred on the realism of the animal model, namely in accomplishing multiband EMR, ESD and dealing with complications, compared to human setting. The final part is focused on the animal model as a learning tool. Appreciation is reported in a 10-point Likert scale. The complete questionnaire is available in Appendix 1.
Simulation
Each workshop had a duration of two days and took place in the training centres of the Erasmus School of Endoscopy at the Erasmus University Medical Center in Rotterdam, the Netherlands and the School of Health Sciences, University of Minho (ECS-UM) in Braga, Portugal.
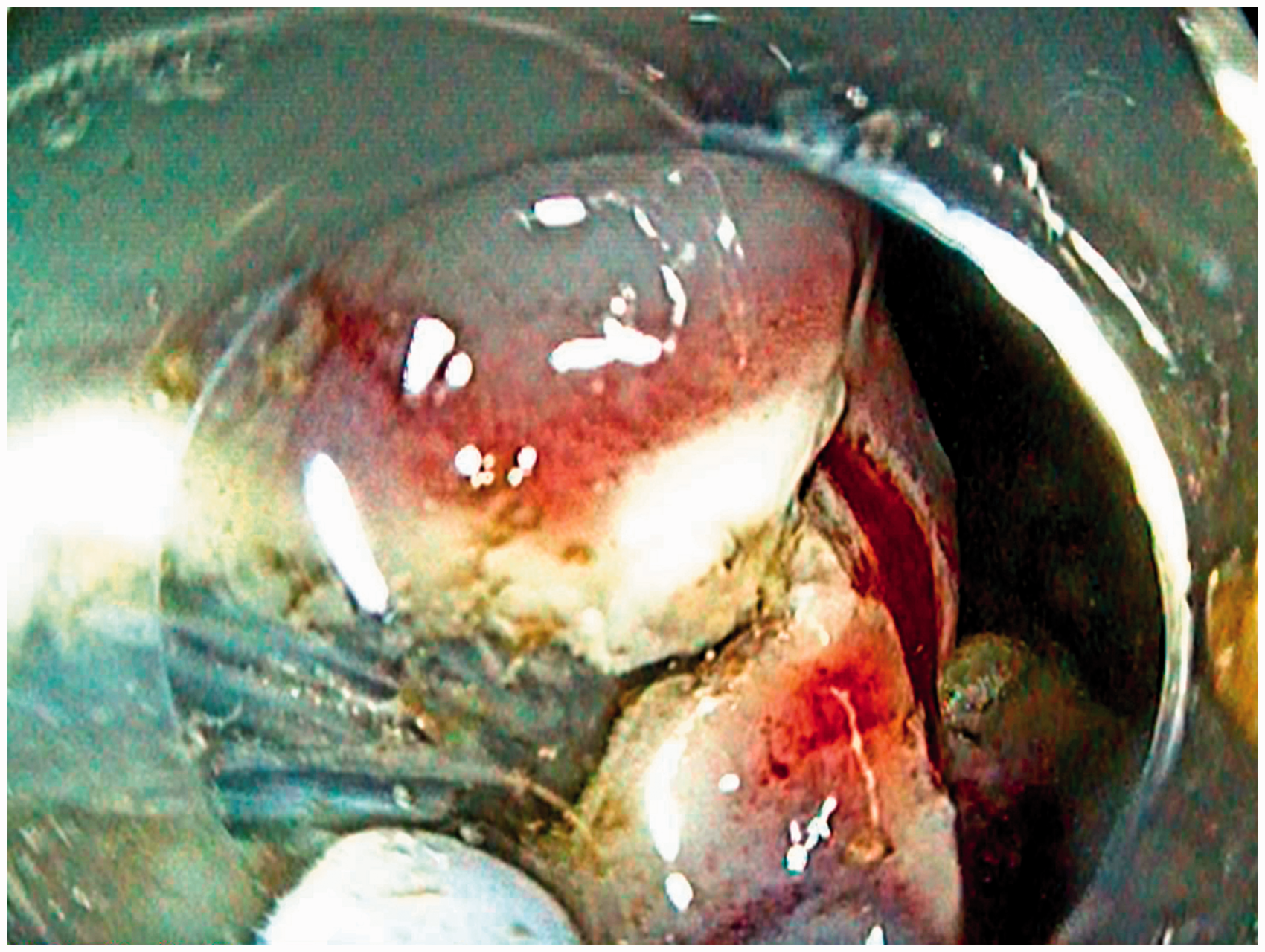
Six fully equipped interventional endoscopy workstations were used under the guidance of international faculty experts (Figure 1). Storz and Pentax flexible endoscopes were used. The electrosurgical units were the ERBE VIO 200s. The training modules consisted of EMR in the oesophagus using the Duette multiband mucosectomy kit (Cook Ireland Ltd, Limerick, Ireland) and ESD in oesophagus and stomach using ERBE Hybrid Knives (ERBE, Tübingen, Germany). During both EMR and ESD, trainees were instructed to mark the outer margins of a pseudo-lesion. EMR was carried out using multiband mucosectomy, where band ligation was applied followed by snare resection of the created pseudo-polyp and inspection of the ulcer. ESD was executed by performing submucosal injection and creating a circumferential incision, followed by submucosal dissection and re-lifting the pseudo-lesion when needed (Figure 2). Any complication such as bleeding or perforation was subsequently managed by the trainees as an integral part of the training course, using injection, coagulation with bipolar haemostatic forceps (HemoStat-Y; Pentax, Tokyo, Japan) and clip application (Instinct clip; Cook Medical, Winston-Salem, North Carolina, USA).
Live porcine model endoscopy workstation. Performance of endoscopic submucosal dissection on porcine model.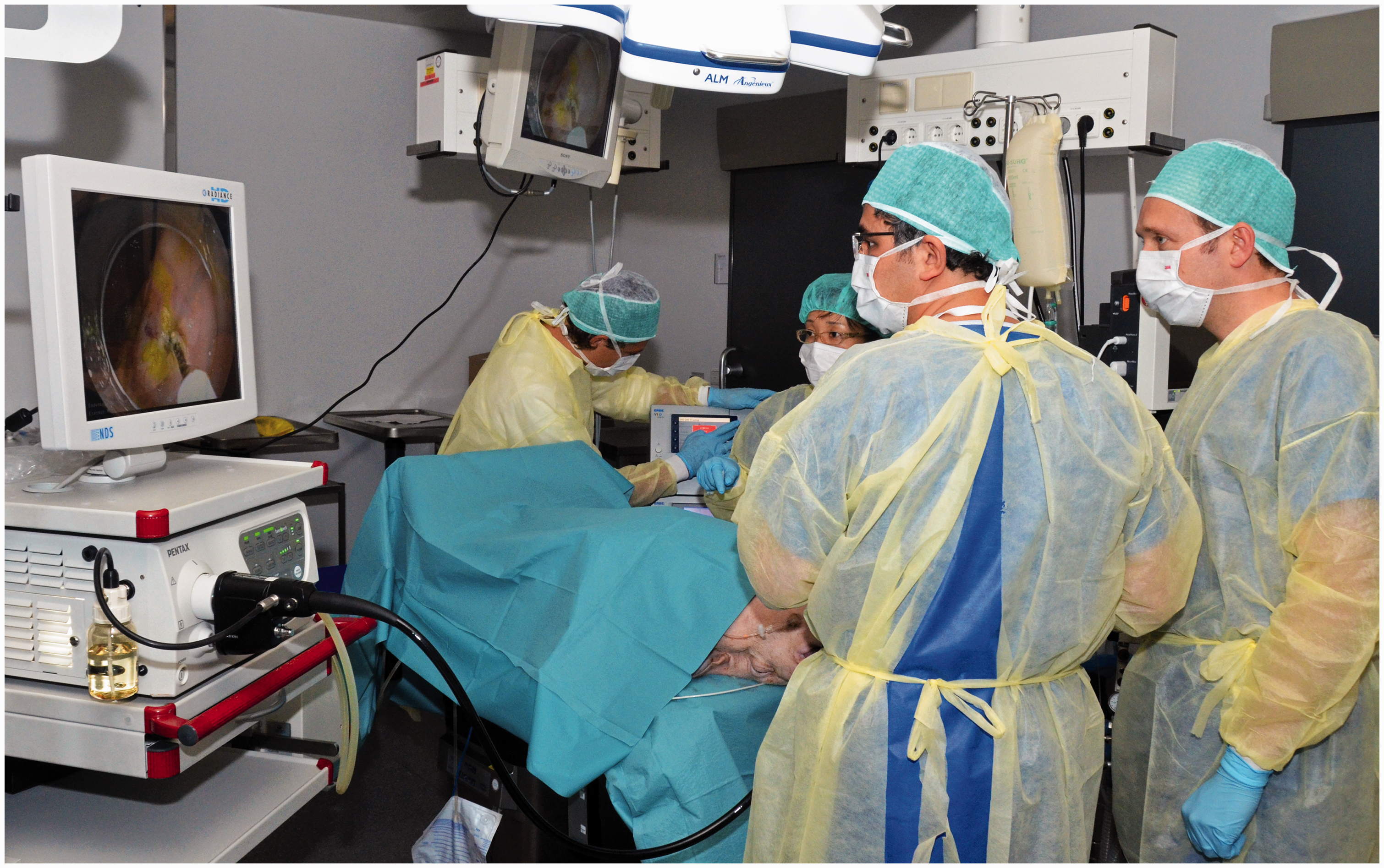
Statistical analysis
Descriptive statistics were determined for all measures according to type of variables. Proportions were reported for dichotomous and ordinal variables. For continuous variables the mean (as a more intuitive measure) and median (interquartile range (IQR) 25–75%) were described. Non-parametric tests were used to assess statistical differences.
The IBM Statistical Package for Social Sciences (SPSS Version 23.0.0; SPSS Inc., Chicago, Illinois, USA) was used to manage data.
Results
Participants
Background characteristics, endoscopic and courses experience of participants.
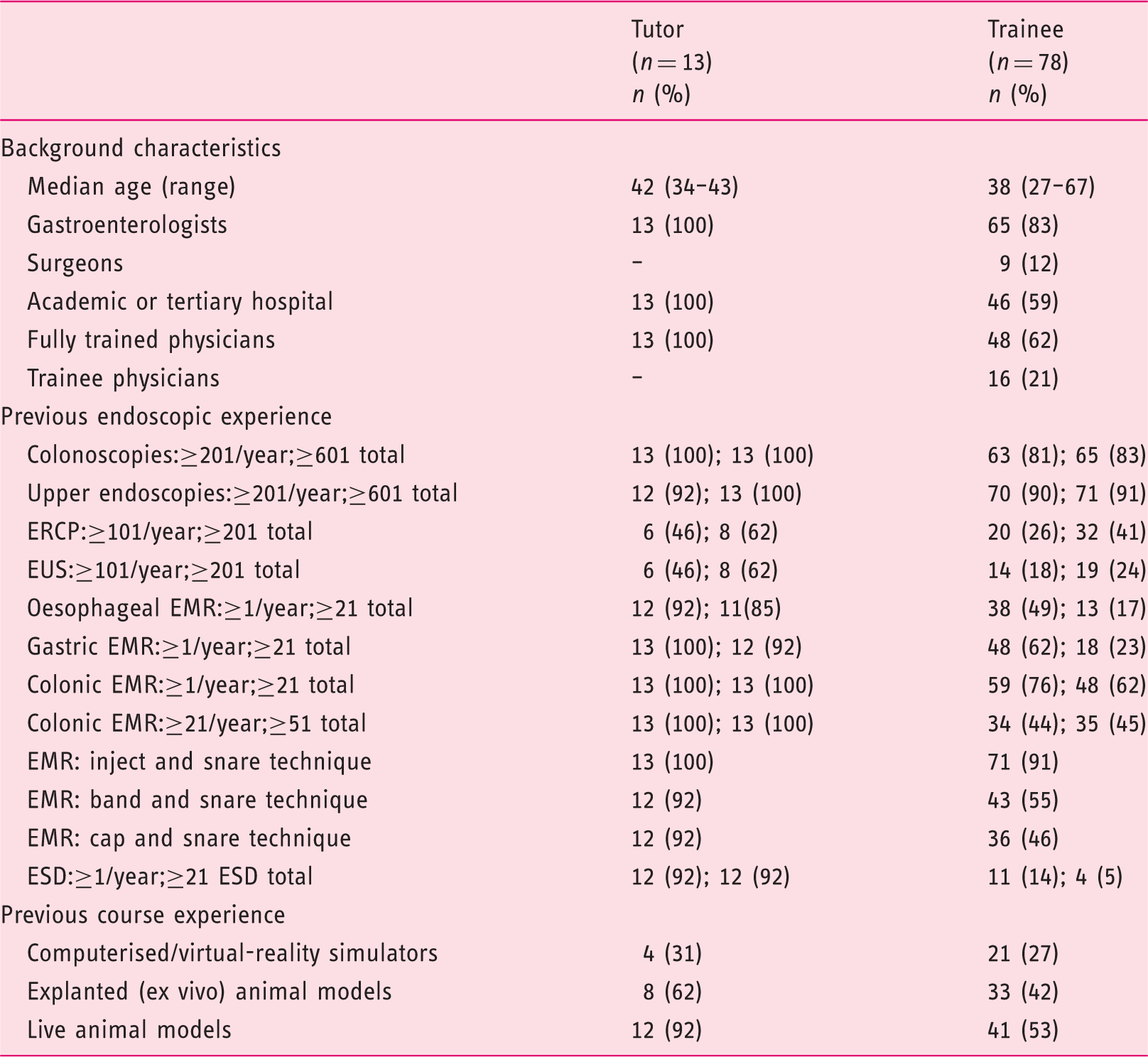
EMR: endoscopic mucosal resection; ERCP: endoscopic retrograde cholangiopancreatography; ESD: endoscopic submucosal dissection; EUS: endoscopic ultrasonography.
Realism of the live porcine model
Realism of the live porcine.
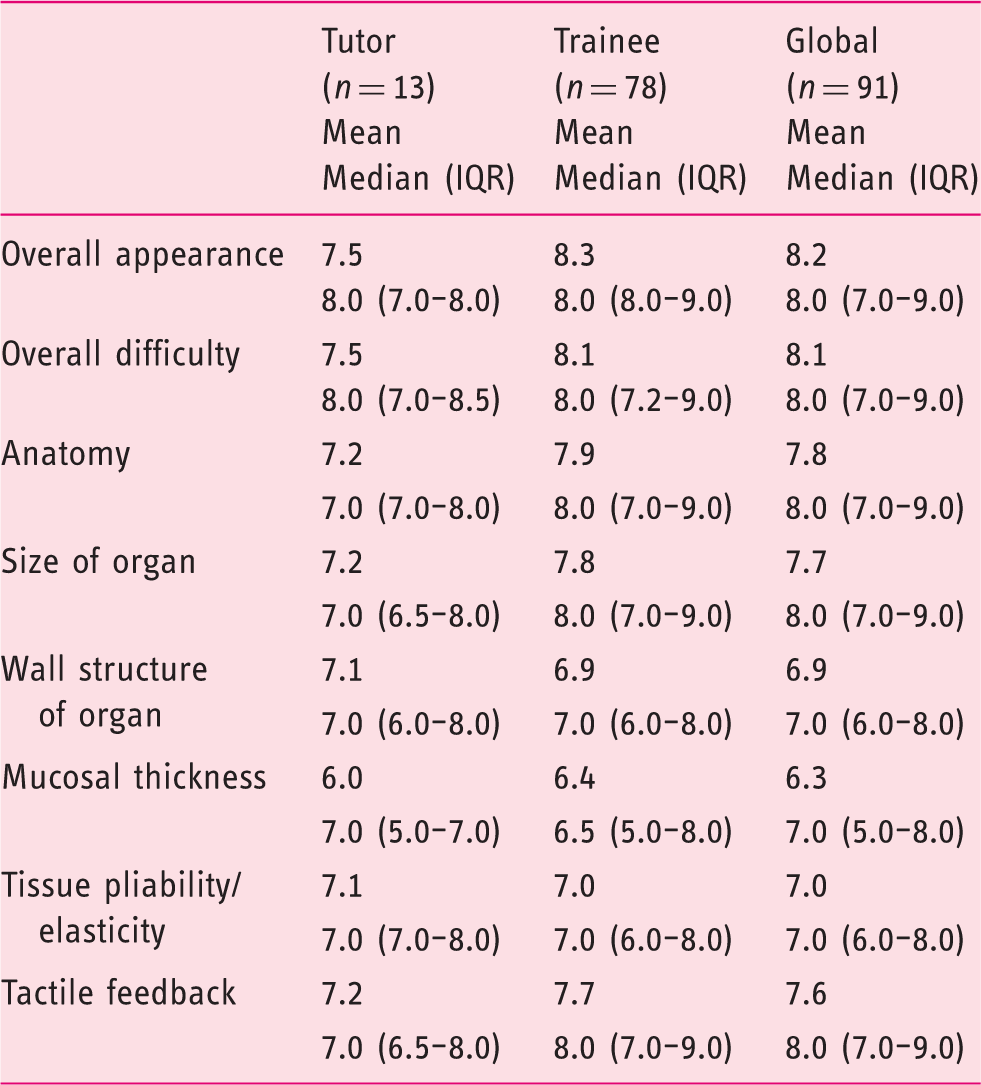
IQR: interquartile range.
Results are presented as mean and median (IQR 25–75%); Mann-Whitney test used as non-parametric test.
Realism of procedures
Procedures in the live porcine model.
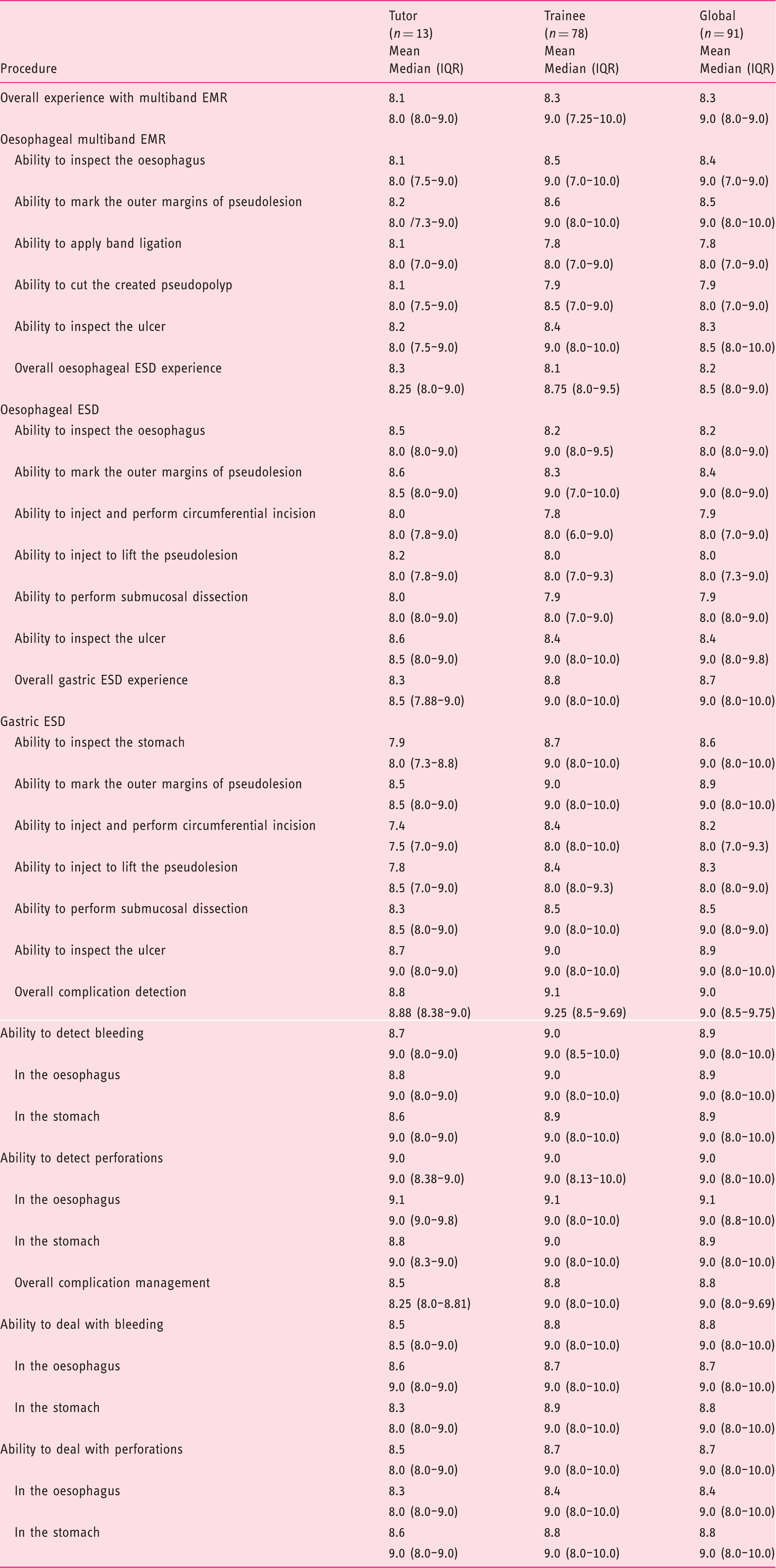
EMR: endoscopic mucosal resection; ESD: endoscopic submucosal dissection; IQR: interquartile range.
Results are presented as mean and median (IQR 25–75%); Mann-Whitney test used as non-parametric test.
The animal model as a learning tool
The animal model as a learning tool.
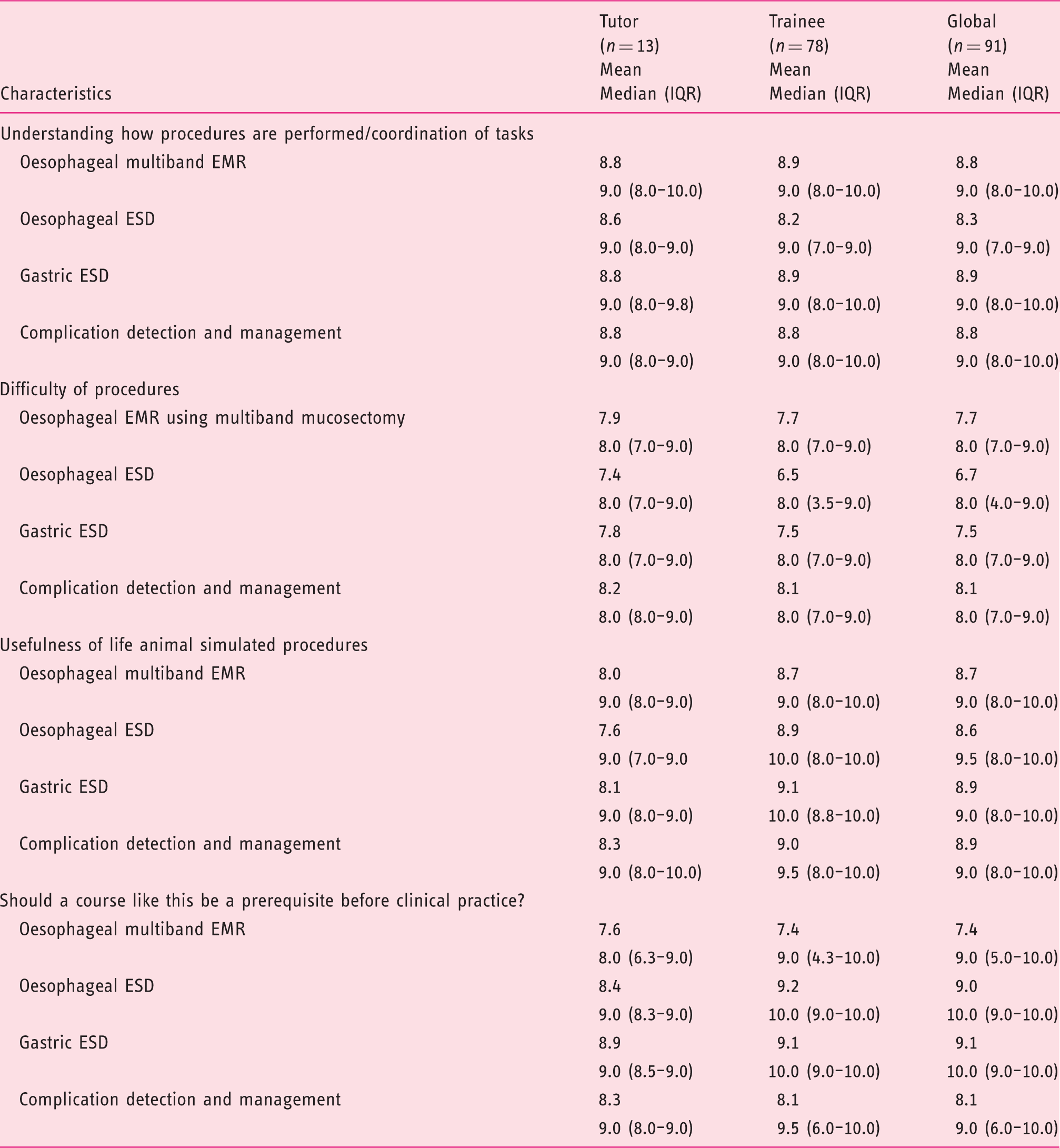
EMR: endoscopic mucosal resection; ESD: endoscopic submucosal dissection; IQR: interquartile range.
Results are presented as mean and median (IQR 25–75%); Mann-Whitney test used as non-parametric test.
Procedure difficulty had a median of 8.0 (IQR 4.0–9.0), whereas the lowest mean value was on oesophageal ESD (6.7) and the highest on complication detection and management (8.1).
Live animal simulated oesophageal EMR, oesophageal ESD and gastric ESD were considered highly useful for beginners with median global values ranging from 9.0 to 9.5 (8.0–10.0).
Participants considered that live animal courses should be a prerequisite before clinical practice (median global values of 9.0 (IQR 5.0–10.0) for oesophageal EMR and 10.0 (IQR 9.0–10,0) for oesophageal and gastric ESD).
Trainees considered the usefulness of life animal simulated procedures and the necessity of courses with live animal models to be a prerequisite before clinical practice with higher scores than tutors for oesophageal and gastric ESD.
Discussion
To our knowledge this is the first study to formally address the face, expert and content validity of the live porcine model in training for EMR and ESD. Our results demonstrate that the model is very realistic and that procedures accurately resemble human cases. Moreover, the simulation process is highly appreciated as a learning tool by both tutors and trainees alike. In our opinion the results of this study justify the application of the live porcine model to formally train endoscopists in performing EMR and ESD and complication management before proceeding to human cases.
Simulators allow a novice or expert to perform a new technique or use a new accessory thereby avoiding putting human patients to the potential risks associated with the initial, steepest part of the learning curve. 22 Pure mechanical simulators and virtual reality (VR) simulators are not ideal when addressing advanced endoscopic resection training, due to the inability to reproduce the elasticity, tissue properties and tactile feedback of human tissue. 23 This study demonstrates that the live porcine model received high ratings on these items, supporting its value and use for this training.
Trainees in our study were already experienced in performing endoscopic procedures such as upper endoscopy and colonoscopy (and to a lesser extent endoscopic retrograde cholangiopancreatography (ERCP) and EUS: endoscopic ultrasonography (EUS)). The majority had previous experience in EMR, mostly in the colon and used several EMR techniques. For that reason they seem well fitted to assess the realism of the porcine training model for EMR and ESD. The fact that appreciation by the trainees was similar to the ones given by the experts supports this view even more. It shows that both experts and trainees are highly capable of assessing the validity of this training model and strengthens the robustness of the data presented.
Regarding the live animal model, orientation of organs, thickness and stiffness of the mucosal layer have been considered distinct from the human scenario.24,25 Accordingly, in our study, wall structure of organ, mucosal thickness and tissue pliability scored the lowest mean values, although medians were comparable. This seems in line with the study by Horii et al. where mucosal layer of the porcine was found to be thinner in the proximal stomach and thicker in the distal area, particularly at the level of the greater curvature, where ESD speed is lower and difficulty is greater when compared to human procedures. It has been suggested that the greater curvature, particularly the distal part of the porcine stomach is the least suitable place for ESD training. 26
The ability to apply band ligation and to cut the created pseudopolyp, while performing oesophageal multiband mucosectomy, was rated slightly lower compared to the other steps in this technique. We feel that this is related to a subjective impression that, due to the difference in wall structure compared to human oesophagus, suction of the pseudolesion is different. Unlike in human practice, it is relatively easy to create a pseudopolyp containing a full thickness specimen resulting more often in a perforation. The aforementioned difference in the porcine oesophagus also explains why oesophageal ESD had lower overall scores.
In addition to the high scores on issues related to the realism of the live porcine model, the performance of the procedures itself was considered very similar to the human practice, as well. Moreover, the difficulty of procedures and usefulness of life animal simulated procedures had high ratings. We think that this is related to the fact that animal models, such as intubated, anaesthetised pigs, have breathing movements, heart beats, peristalsis, intraluminal secretions, tissue reaction to injection and electrocautery, and abdominal distension which makes the model approach the human setting. The same properties make it possible to deal with adverse events, such as bleeding and perforation.11,15,25,27
When teaching ESD, the process of submucosal dissection has been considered the most challenging one and its difficulty is related mainly to controlling haemorrhage. 28 We found that ratings addressing the porcine model on complication detection and management, namely controlling haemorrhage, were very high, emphasising the value of the model in training all the steps involved in ESD. Our study clearly demonstrates that the live porcine model is not only a valid model for training EMR and ESD procedures, but also for training of intraprocedural complication management.
It has been suggested that ex vivo models cannot substitute for in vivo training to acquire competence in ESD and some advocate only moving to the live animal model after acquiring basic ESD skills using explanted organs.16,27,29 Nonetheless, issues pertaining ethics, dedicated facilities, equipment, veterinarian support, availability of general anaesthesia and preparation of the animal, including intensive bowel cleansing (in the case of colorectal ESD), euthanasia or follow-up care (when performing survival studies), as well as financial considerations may limit its use.11,15,25,27,30 Animal models have been incorporated in ESD training programmes by different groups. Although previous papers have focused on feasibility of ESD, validation of the model itself has not been formally demonstrated. Considering the evidence gathered in this study, including the validation of the live porcine model in training EMR and ESD, we believe that live pig workshops should be formally incorporated in teaching programmes. This fact may have an impact on the justification to apply for funding. Similarly, the current scientific substantiation of a live pig model to train for EMR and ESD, adds to the ethical justification of animal sacrifice for such medical training purposes.
This article may serve as a framework for future studies on subjects such as learning curves and how to get the most out of animal training before proceeding to human cases.
With regard to the strengths of our study, we underline its relevance, the multicentre character of the hosting labs and the origin of tutors and trainees which, in turn, serves as an argument for the generalization of the achieved results.
Concerning limitations of our study, we stress that the porcine colon was not evaluated. Also, procedures were conducted on healthy porcine tissues without pathologic conditions, unlike the real human scenario where genuine mucosal/submucosal lesions are being treated and in which, for example, fibrosis may be an issue. Nevertheless this limitation is probably of reduced magnitude given that in a great number of patients these features will not alter the procedure and that novices in ESD, for whom this training is most valuable, should not start ESD in difficult, large and fibrotic lesions.
It is important to notice that in this work we have not addressed the effect of training in the animal model regarding outcomes in clinical practice. We believe that this objective should be a target for future studies.
In conclusion, EMR and ESD training in a live porcine model was considered very realistic compared to the human setting and was highly appreciated as a learning tool. This is the first study to establish face, content and expert validity of the live porcine model in performing multiband EMR, oesophageal and gastric ESD. The validation of this model provides the rationale to incorporate it into formal teaching programmes and provides a basis on which further studies can be conducted.
Footnotes
Acknowledgements
The authors would like to thank all of the participants in the workshops for their collaboration in filling out the questionnaires.
Declaration of conflicting interests
There are no conflicting interests to declare.
Ethics approval
The use of the live porcine model for training purposes in the workshops was approved by local Ethical Committees for the welfare of animals in medical training.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
This validity study was not a research conducted on humans, although it was based on questionnaires answered by humans. An oral informed consent was obtained, based on the fact that the questionnaires were freely answered only by the health professionals themselves that attended the workshops and that the act of answering the questionnaires assumed their implicit acceptance/consent. A prerequisite of this approach was the complete anonymity of all participants.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
