Abstract
Background
Colorectal cancer (CRC) is a common but largely preventable cancer. Although fecal immunochemical tests (FITs) detect the majority of CRCs, they miss some of the cancers and most advanced adenomas (AAs). The potential of blood tests in complementing FITs for the detection of CRC or AA has not yet been systematically investigated.
Methods
We conducted a systematic review of performance of FIT combined with an additional blood test for CRC and AA detection versus FIT alone. PubMed and Web of Science were searched until June 9, 2017.
Results
Some markers substantially increased sensitivity for CRC when combined with FIT, albeit typically at a major loss of specificity. For AA, no relevant increase in sensitivity could be achieved.
Conclusion
Combining FIT and blood tests might be a promising approach to enhance sensitivity of CRC screening, but comprehensive evaluation of promising marker combinations in screening populations is needed.
Introduction
With approximately 1.4 million incident cases and 700,000 deaths annually, colorectal cancer (CRC) ranks third among men and second among women among the most common cancers globally. 1 Although the vast majority of all or distal CRCs and adenomas can be detected by colonoscopy or sigmoidoscopy, respectively, less-invasive screening tests are desirable. Inconvenience of bowel preparation and potential (albeit rare) complications 2 limit adherence to screening endoscopy. Furthermore, many countries lack the resources to offer colonoscopy as a primary screening test, 3 resulting in long waiting times.
Fecal immunochemical tests (FITs) have substantially higher sensitivity for CRC at similar levels of specificity compared to guaiac-based fecal occult blood tests (gFOBTs). 4 FITs on average detect nearly 80% of CRCs, although a wide range of sensitivities has been reported (25%–100%). 5 Sensitivities for advanced adenomas (AAs) are more limited, typically ranging from 20% to 30% and rarely exceeding 40%. 6 Because most CRCs develop slowly through the adenoma-carcinoma sequence 7 and are in principle preventable via removal of detected adenomas, improved AA sensitivity could reduce CRC incidence.
Blood (serum, plasma) tests appear to be more acceptable for screening participants than colonoscopy. 8 Many studies have assessed their potential as diagnostic tests for CRC screening, 9 but for none of them diagnostic performance equivalent to or better than performance of FIT 5 could be validated. However, combining the most promising tests with FIT might enhance diagnostic performance of FIT. To assess whether combining FIT and blood markers yields better sensitivity and/or specificity than individual testing with FIT, we systematically reviewed diagnostic studies reporting on diagnostic accuracy of FIT alone and combined with a blood marker test.
Methods
Data sources and search strategy
MEDLINE and Web of Science were searched for English-language articles from inception to June 9, 2017. The search terms (see Supplement) covered expressions for FIT, advanced colorectal neoplasms and diagnostic accuracy measures. Reference lists of relevant articles were also screened. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 10
Study selection methods
Eligible studies had to provide quantitative information on sensitivity and specificity or sufficient information to calculate them, and/or the area under the ROC curve (AUC/c statistic) for detection of CRC, AA or any advanced neoplasm (AN), and examine these outcomes for FIT alone and for the combination of FIT and a blood (serum/plasma) test. Non-AAs and “any adenomas” (advanced and non-advanced) were not investigated, because only a small minority of non-AAs are expected to develop into CRC. 11 To rule out verification bias, 12 we required conduction of colonoscopy as the gold standard for detecting colorectal neoplasms in all included participants.
Data extraction
Two authors (TN and KW) independently extracted data on study characteristics and outcomes from relevant articles.13–20 Eventual discrepancies were resolved by consensus. Sensitivities were extracted separately for CRC and AA. Sensitivities were calculated as the share of CRCs or AAs detected by FIT, the blood test or their combination. Specificities were defined as the share of participants with a negative result of FIT, the blood test or both combined among all individuals without AN.
Risk of bias in individual studies
Two authors (TN and KW) independently evaluated the risk of bias in the included studies with elements of the QUADAS-2 instrument 21 and the Cochrane Collaboration’s tool 22 that we deemed most important for our study question.
Data analysis
The combinations of two tests can be interpreted as positive if one out of two tests is positive, whereas the other one may be negative (“pn”), or if both tests are positive (“pp”). If 2 × 2 tables with true and false positives for both tests and their combinations were provided or reproducible, sensitivities and specificities for both interpretations were calculated.15,17 For one study, 17 “ROC-like plots” were created using R 23 and the “mada” package 24 to visualize diagnostic performance of the numerous reported test combinations.
Results
Study selection
Figure 1 shows the study selection process. Of 2054 unique articles, 26 were given full text assessment and eight of them were included in our systematic review.13–20 The 18 articles excluded after full-text review did not report sensitivities and specificities for FIT combined with a blood test. Among the suitable articles, one
16
was entirely based on an asymptomatic CRC screening population. Two reports13,14 comprised participants from a prospective screening study and additionally recruited cases from a case-control study. Two articles19,20 are apparently based on the same asymptomatic CRC screening population comprising first-degree relatives of CRC patients with the same FIT but different blood markers examined. Two studies used a case-control design.15,18 One report
17
was based on a prospective study among symptomatic participants.
Flow diagram of study selection.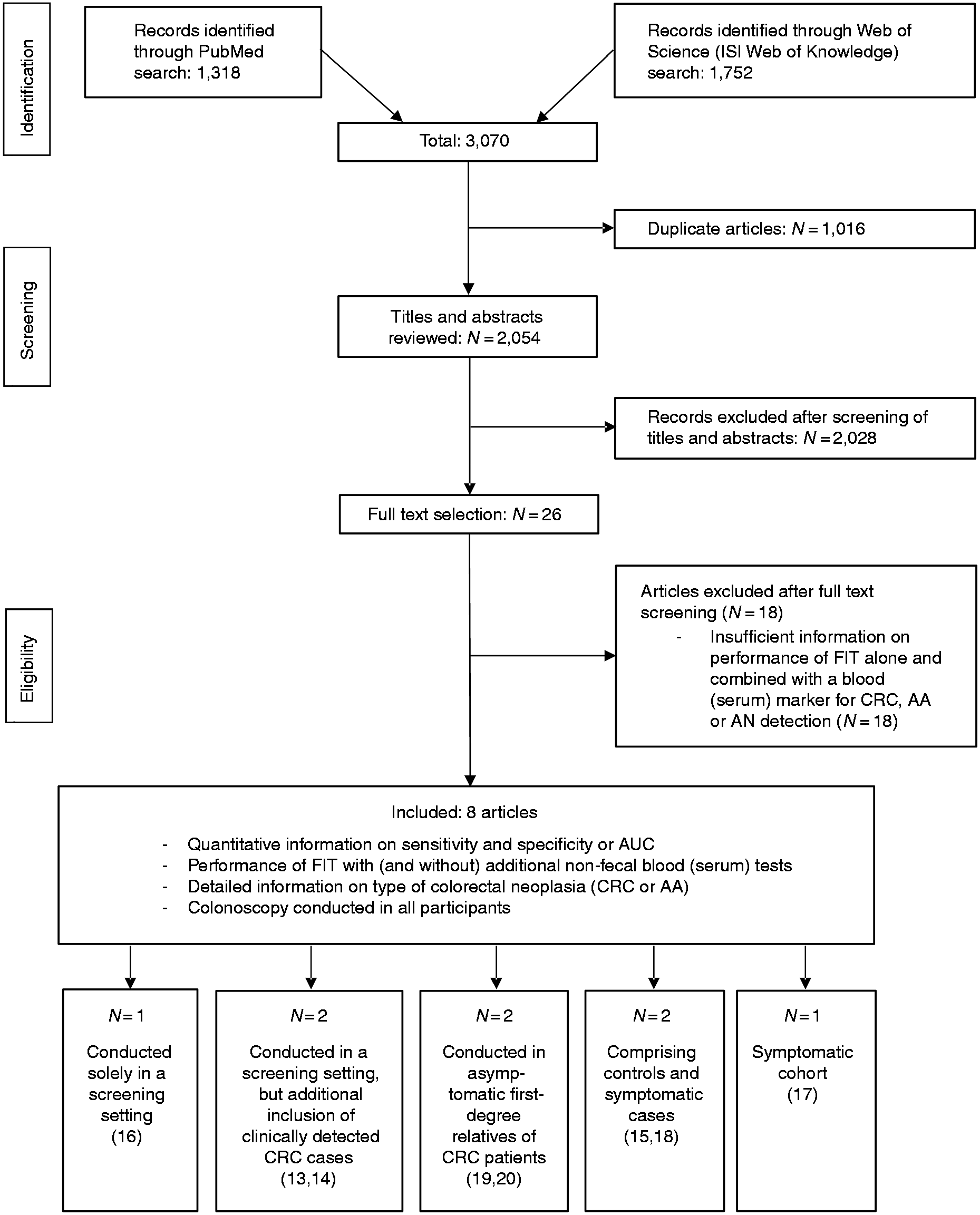
Blood markers added to FIT
Among the eight studies examining the performance of FIT alone in comparison to FIT combined with at least one blood marker, three14,15,18 examined methylation at the Septin9 gene and one study 13 examined a panel consisting of the inflammatory markers C-reactive protein (CRP), serum CD26 (sCD26), complement C3a anaphylatoxin, and tissue inhibitor of metalloproteinases 1 (TIMP-1). One study 16 examined a panel comprising carcinoembryonic antigen (CEA), ferritin, seprase, osteopontin and anti-p53 antibody. One study 17 investigated BCAT1 and IKZF1 DNA methylation. The studies conducted in first-degree relatives of CRC patients19,20 examined FIT plus sCD2619 and nucleoside diphosphate kinase A (NDKA). 20
Study characteristics
Characteristics of included studies conducted in average risk participants.
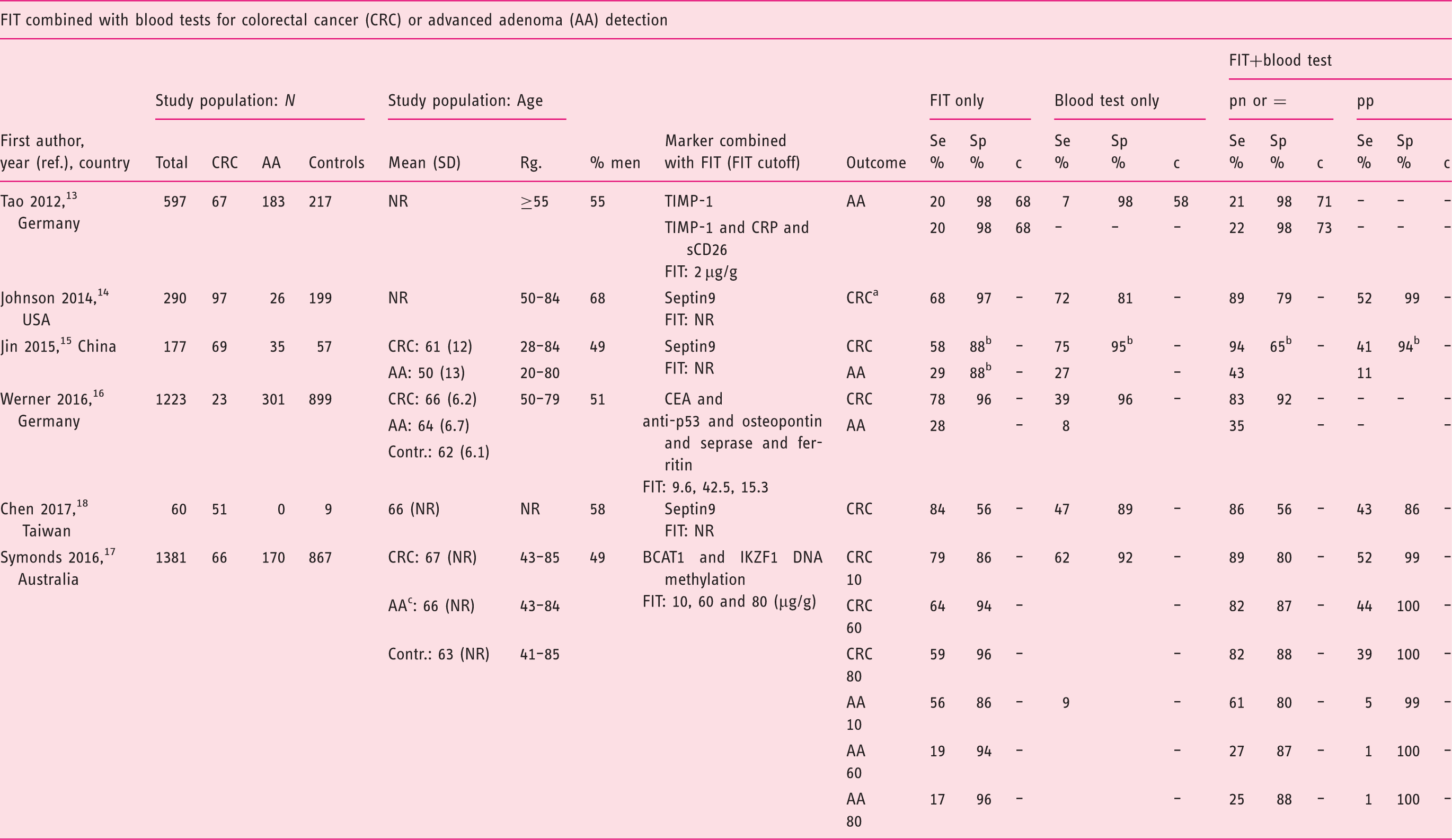
Negatives defined as non-CRC cases, including individuals with AA, small polyps, and no evidence of disease.
Hyperplastic polyps and non-advanced adenomas were included in the denominator of specificity.
Including high-grade dysplasia, >2 tubular adenomas and stage-0 CRCs.
AA: advanced adenoma; BCAT1: branched chain amino-acid transaminase 1; cytosolic; CEA: carcinoembryonic antigen; Clin.: clinical; Contr.: controls; CRC: colorectal cancer; CRP: C-reactive protein; c: c statistic (≙AUC: area under the receiver operating characteristics (ROC) curve); FIT: fecal immunochemical test; Hb: hemoglobin; IKZF1:IKAROS family zinc finger 1; NR: not reported; ref.: reference; TIMP-1: tissue inhibitor of metalloproteinases 1; Rg.: range; SD: standard deviation; Se: sensitivity; Sp: specificity; USA: United States of America. Definitions of a positive combined test: “pn,” at least one test positive; “pp,” both tests positive; “=”, cutoffs adjusted to obtain specificities equal to FIT alone.
Characteristics of studies focusing on first-degree relatives of CRC patients.
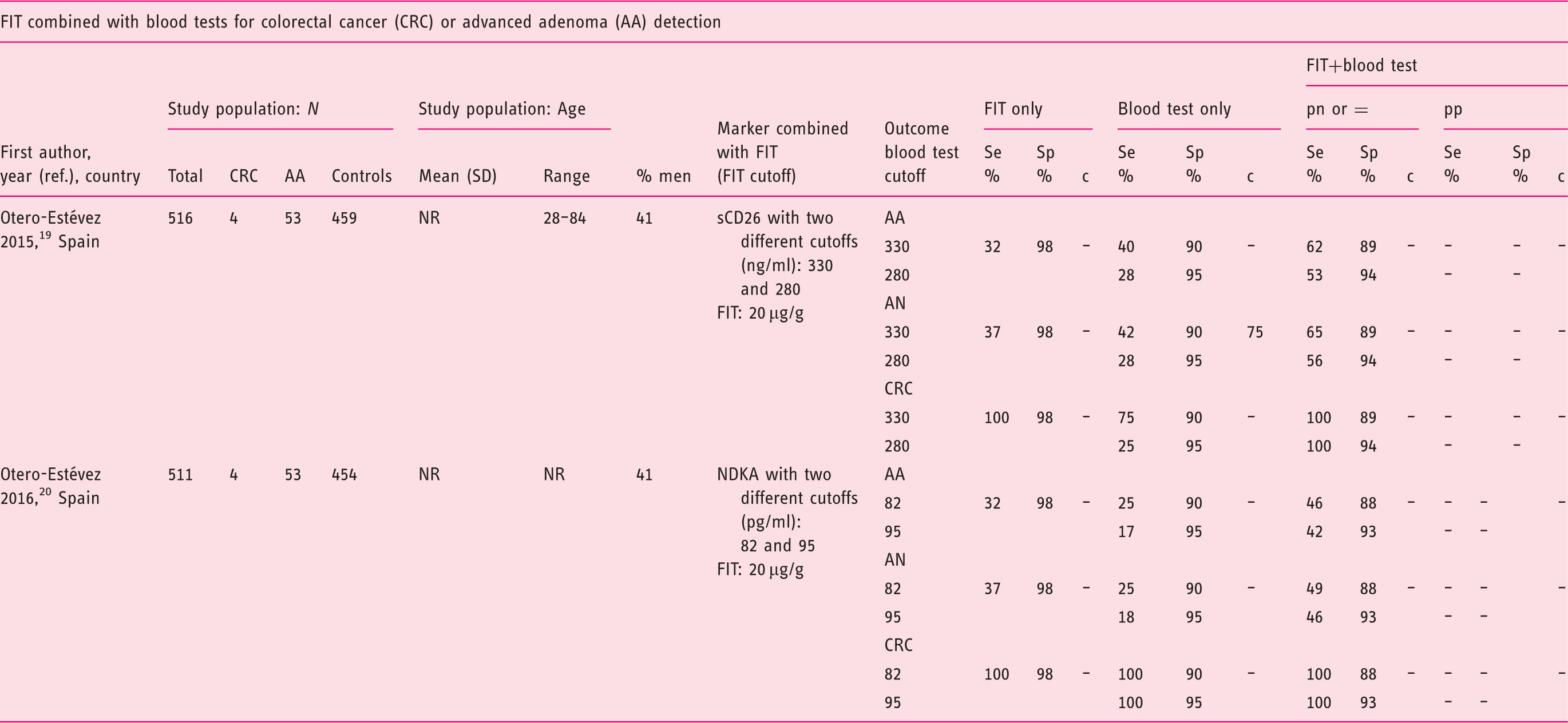
AA: advanced adenoma; AN: advanced neoplasia (CRC or AA); CRC: colorectal cancer; c: c statistic (≙AUC, area under the receiver operating characteristics (ROC) curve); FIT: fecal immunochemical tests; NDKA: nucleoside diphosphate kinase A; NR: not reported; ref.: reference; sCD26: soluble cluster of differentiation 26; Scr.: screening. Definitions of a positive combined test: “pn,” at least one test positive; “pp,” both tests positive; “=”, cutoffs adjusted to obtain specificities equal to FIT alone.
Results of the study quality assessment
Risk of bias assessment.
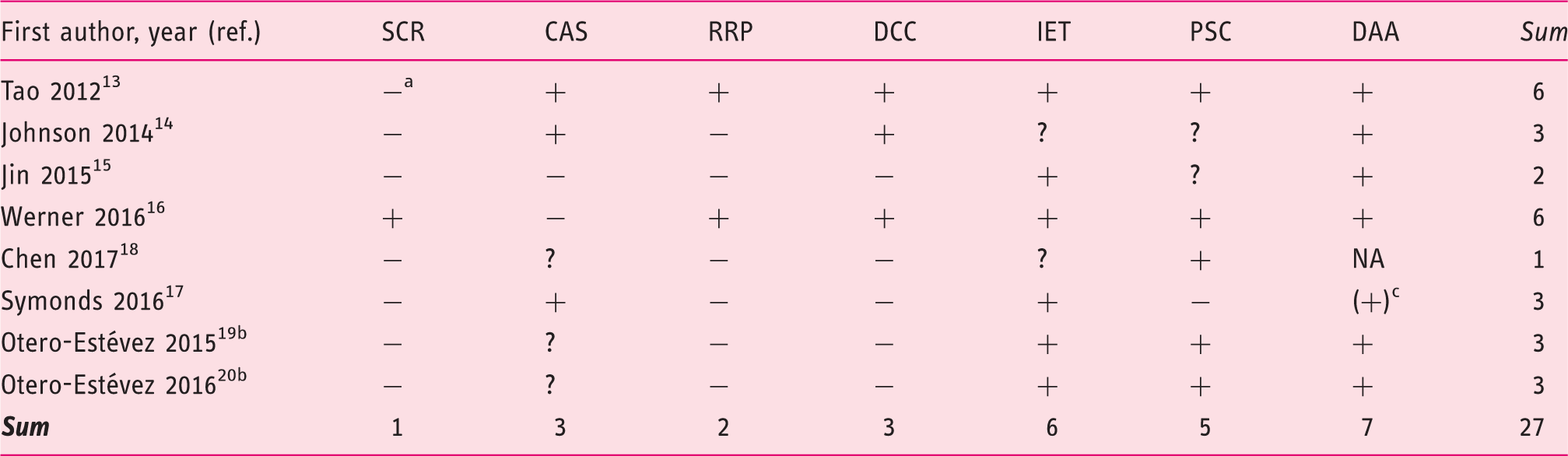
SCR: study conducted solely in a screening setting (asymptomatic individuals undergoing colonoscopy); CAS: comparable distribution of both, age and sex among cases and controls (<10% difference in average proportions of sexes, up to 10 years’ difference between cases and controls; “+” if both age and sex distribution are comparable, otherwise “–”); RRP: clear reporting of the study recruitment process with numbers and reasons of exclusions, ideally in a flow-chart; DCC: clear description of data collection and conduction of FIT; IET: independent examination of the index tests and the gold standard test (laboratory staff members who evaluated FIT and/or the blood test(s) were blinded to colonoscopic findings or endoscopists were blinded to the results of FIT and/or the blood test(s), whichever test was conducted first); PSC: marker cutoffs pre-specified in quantitative FIT (any qualitative FIT was rated as “prespecified” because no overoptimistic results can occur, in contrast to studies that optimize the AUC without splitting the data in a training and a test set. Adjusting cutoffs to a level yielding a fixed specificity was also rated as “prespecified”); DAA: study provided a common definition of advanced adenomas ((tubulo-) villous adenomas and/or adenomas >1 cm and/or high-grade dysplasia, with or without stage-0 CRCs); AA: advanced adenoma; CRC: colorectal cancer; FIT: fecal immunochemical test; “+”: fulfilled; “–”: not fulfilled; “?”: not reported; “NA”: not applicable; ref.: reference.
Comprising participants from a screening study (
Asymptomatic first-degree relatives of CRC patients.
Individuals with >2 tubular adenomas were also classified as AA.
Data synthesis
A meta-analysis was not conducted, because the studies investigating the same marker combination were heterogeneous regarding study population characteristics and other potentially influential factors. Consistently, studies investigating sensitivities at fixed specificities found minor sensitivity gains. In a “pn” approach, sensitivity gains were seen only at the cost of specificity. The opposite was observable in studies reporting a “pp” combination.
Septin9
Of the three studies examining Septin9,14,15,18 two15,18 were conducted in a clinical setting and one 14 used screening-identified cases and controls. Septin9 partly showed sensitivities and specificities similar to FIT across all studies, with large variation (Table 1). Sensitivity of Septin9 for CRC ranged from 47% to 75%. Although some studies14,15 reported large sensitivity gains by combining FIT and Septin9 (>30 percentage points, reaching 89% 14 and 94% 15 sensitivity for CRC), increased sensitivity was achieved only at decreases in specificity (in “pn” combination) and vice versa (in “pp” combination). No study indicated a sensitivity gain larger than the loss in specificity or a specificity gain at only a modest loss in sensitivity. Evidence of the complementary value in AA detection compared to FIT alone was limited to one study 15 reporting an increase from 29% to 43% sensitivity, while specificity decreased from 88% to 65%.
sCD26 and NDKA
sCD26 as a single marker added to FIT was examined in a study among first-degree relatives of CRC patients. 19 Sensitivity for advanced neoplasia with FIT achieved by combining it with sCD26 in a “pn” approach could be increased from 37% to 56% and 65%, depending on the sCD26 cutoff. Specificity decreased from 98% to 94% and 89%, respectively (Table 2). The positive likelihood ratios diminished compared to FIT alone, indicating no overall improvement. Adding NDKA to FIT 20 yielded higher sensitivities than FIT alone, but gains were smaller compared to adding sCD26. Sensitivity increases by the “pn” approach were large in proximal AAs (from 7% to 29%), but smaller in distal AAs (from 41% to 51%). In contrast to the study on combined performance of FIT and sCD26, 19 that marker had 0% sensitivity for CRC at 97.7% specificity in a multi-marker panel. 13
Multi-marker panels
One study
17
combined the DNA methylation markers BCAT1 and IKZF1 in a “pn” approach and the resulting combination with FIT in both a “pn” and “pp” approach for CRC and AA, each at three different FIT cutoffs. Graphical presentation of sensitivity versus 1-specificity in “ROC-like plots” (Figure 2) demonstrates small overall gains with the addition of the blood test. For CRC, “pn” combinations with higher FIT cutoffs seemed to yield a marginally higher sensitivity at no loss in specificity, compared to FIT alone. However, the minor differences might be due to chance. No strong stage-dependence was seen for the blood test.
Sensitivity versus 1-specificity of FIT alone and combined with BCAT1 and IKZF1, DNA methylation markers for CRC and AA detection in the study by Symonds et al.
17
FIT: fecal immunochemical test; CRC: colorectal cancer; AA: advanced adenoma.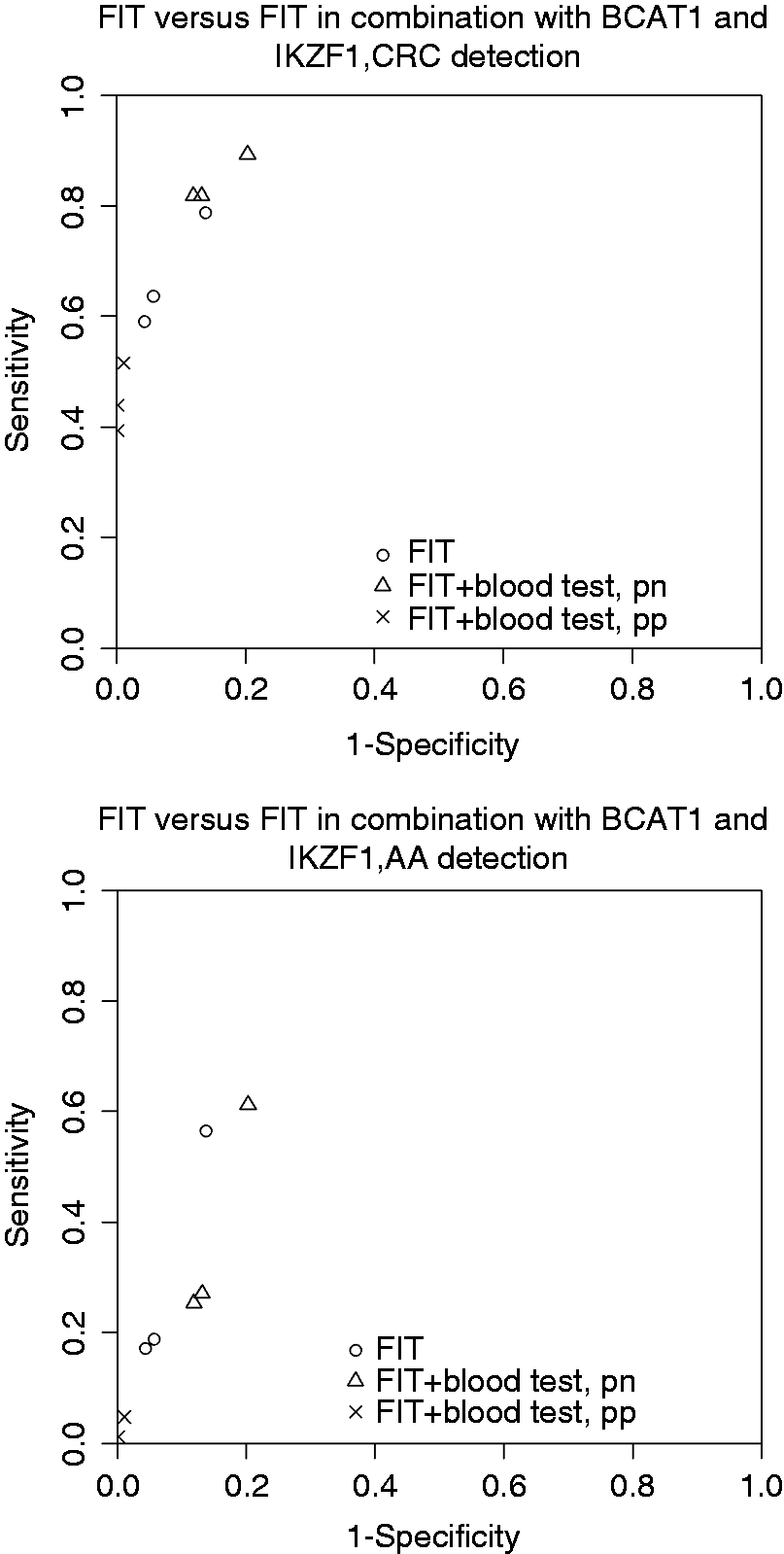
In a study examining FIT combined with five blood markers,
16
sensitivity for CRC and AA rose from 78% to 83% and from 28% to 35%, respectively, although CIs widely overlapped (Table 1). Specificity decreased from 96% for FIT alone to 92%. That study used a predefined algorithm trained on data of a previous study to avoid overfitting. Based on the same study, a previous report focusing on AA
13
found virtually no increases in sensitivity at constant 98% specificity when adding TIMP-1 alone or together with CRP and sCD26. The likelihood ratio tests nevertheless indicated a significant improvement in the AUC (
Discussion
We summarized the available evidence from studies evaluating sensitivity and specificity of FIT combined with blood markers for CRC or AA detection. Most studies did not find large improvements in FIT performance in such combinations, neither for CRC nor for AA. Sensitivity gains were achieved only at decreased specificity and vice versa. The only study comparing sensitivities for AA at fixed specificities 13 found virtually no gain in sensitivity. One study 17 demonstrated the methodological challenges for comparing combined diagnostic tests by providing a wide range of results for possible marker combinations.
Studies examining diagnostic performance of blood markers for CRC detection were systematically reviewed already.9,25,26 However, these studies did not examine their test performance when combined with FIT. Liu et al. 9 waived a conclusion on which biomarker to recommend, whereas Shah et al. 25 highlighted the promising results of DNA hypermethylation markers, specifically Septin9 expression and other DNA and microRNA (miRNA) panels. A meta-analysis 26 yielded 75% sensitivity and 89% specificity of Septin9 for CRC detection. Other markers had sensitivities ranging from 54% to 74% and specificities between 85% and 93%. However, these markers were not yet evaluated in an asymptomatic CRC screening population. One study 27 evaluated Septin9 (without FIT) prospectively in a CRC screening population with colonoscopy as the reference standard. With 91.5%, specificity was comparable to that of FIT. Sensitivity was only 48.2% for CRC, though, and 11.2% for AAs, which was similar to the false-positive rate (9.1%). By comparison, FIT was examined in screening studies and achieved 79% sensitivity for CRC and 94% specificity. 5
Our study has several strengths. This is the first systematic review investigating diagnostic performance of a combination of FIT and blood (serum, plasma) tests. Two authors extracted data independently. All studies verified test results with colonoscopy. We adhered to the PRISMA statement 10 and searched two databases and references of included articles. Information on potentially influential study characteristics was considered. Finally, most included studies were of moderate-to-high quality.
Our study also has limitations. We focused on English-language articles. The number of suitable articles was small and few studies investigated the same marker combination. Publication and reporting bias are conceivable. Blood tests in general are unable to detect AAs, 16 limiting their potential for CRC prevention. Predominant clinical settings in included studies and diagnostic or therapeutic measures for CRC patients might have influenced blood marker levels. Clinically detected CRC cases may present with more advanced, more easily detectable14,28 stages than in screening settings. 29 Thereby, potential “spectrum effects” might have led to some overestimation of sensitivities and specificities compared to a screening setting. 30
Only one study investigated diagnostic performance of a CRC blood marker combined with FIT in a screening population. 16 Future studies evaluating CRC blood markers in screening settings should additionally investigate the performance of a combination with FIT and of FIT alone. When combining FIT and blood markers, studies should examine sensitivity at equal specificities through cutoff adjustments. Ideally, performance over multiple cutoffs of FITs, blood tests or both tests using ROC approaches should be evaluated. Studies examining multi-marker panels might investigate if restricting the number of investigated markers would lead to comparable results, thereby facilitating application in clinical practice and improving cost-effectiveness. For example, one study 13 evaluated a combination of five different blood markers, one of which (sCD26) had 0% sensitivity for CRC at the observed specificity, probably making that marker expendable.
To be suitable for mass screening, serum/plasma markers should not only improve diagnostic accuracy compared to FIT alone. Further requirements are ease of conducting the test (e.g. requiring small amounts of blood), marker stability and reproducibility. For cost-effectiveness, a combined FIT and serum or plasma test should maintain high specificity. Microsimulation models might help to compare cost-effectiveness of the numerous possible screening methods comprising FIT and/or one or several blood tests. Such models should incorporate screening adherence rates, which may strongly affect cost-effectiveness. 31 Septin9, for instance, costs approximately 150 US$ per test 32 and studies indicate a significantly decreased specificity in a “pn” interpretation of a combination with FIT.
In conclusion, available evidence suggests very limited gain by combining currently available blood tests for CRC or AA detection with FIT compared to either test alone. The poor AA detection even when combining FIT and blood tests limits their ability to prevent CRC through early detection and removal of AAs. However, blood markers for CRC or AA detection are an emerging field and many markers available to date 9 have not yet been examined in combination with FIT.
Footnotes
Acknowledgments
Author contributions are as follows: HB designed the study. TN and KW extracted the data. TN conducted the literature search and drafted the manuscript. HB, MH and KW contributed to important intellectual content and critically revised the manuscript.
Declaration of conflicting interests
None declared.
Funding
Research on colorectal cancer screening conducted in the Division of Clinical Epidemiology and Aging Research at the German Cancer Institute has been or is currently being partly funded by industrial research grants from Eiken Chemicals, Epigenomics, Roche Diagnostics and Applied Proteomics to the German Cancer Research Center.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
