Abstract
Background
Inflammatory bowel diseases are consequence of an intestinal homeostasis breakdown in which innate immune dysregulation is implicated. Toll-like receptor (TLR)2 and TLR4 are immune recognition receptors expressed in the intestinal epithelium, the first physical-physiological barrier for microorganisms, to inform the host of the presence of Gram-positive and Gram-negative organisms. Interleukin (IL)-10 is an essential anti-inflammatory cytokine that contributes to maintenance of intestinal homeostasis.
Aim
Our main aim was to investigate intestinal IL-10 synthesis and release, and whether TLR2 and TLR4 are determinants of IL-10 expression in the intestinal tract.
Methods
We used Caco-2 cell line as an enterocyte-like cell model, and also ileum and colon from mice deficient in TLR2, TLR4 or TLR2/4 to test the involvement of TLR signaling.
Results
Intestinal epithelial cells are able to synthesize and release IL-10 and their expression is increased after TLR2 or TLR4 activation. IL-10 regulation seems to be tissue specific, with IL-10 expression in the ileum regulated by a compensation between TLR2 and TLR4 expression, whereas in the colon, TLR2 and TLR4 affect IL-10 expression independently.
Conclusions
Intestinal epithelial cells could release IL-10 in response to TLR activation, playing an intestinal tissue-dependent and critical intestinal immune role.
Key summary
Summarize the established knowledge on this subject. Intestinal Toll-like receptors (TLRs) are immune receptors responsible for the recognition of luminal microorganisms, causing a release of cytokines. Interleukin (IL)-10 is a critical anti-inflammatory cytokine, essential for intestinal homeostasis. However, little is known about the relation between intestinal IL-10 and TLRs. What are the significant and/or new findings of this study? TLR2 and 4 can regulate intestinal IL-10 expression in a tissue-specific way. Here we describe how IL-10 expression is mediated by TLR2 and 4 supporting the idea that intestinal epithelial cells could play a role in innate immune response by IL-10 release and how microbiota could interfere in intestinal homeostasis controlling IL-10 levels.
Introduction
Intestinal immunological activity is determinant in homeostasis since it contributes to recognition and tolerance of the intestinal microbiota. Inappropriate mucosal immune responses, due to dysregulation of tolerance to microbiota, contribute to the development and maintenance of inflammatory bowel disease (IBD). 1
Intestinal epithelium is the first physical-physiological barrier for microorganisms to interact with the host. Intestinal epithelial cells have been well documented to express Toll-like receptors (TLRs), which are immune receptors responsible for the recognition both of microbiota and pathogens.
2
TLRs develop an important immunological activity for precise maintenance of intestinal homeostasis,3,4 inducing the synthesis and release of pro- and anti-inflammatory cytokines.
2
In recent years, several studies have demonstrated that TLR2 and TLR4 may play an important role in intestinal homeostasis as well as in inflammatory processes. TLR2 recognizes Gram-positive bacteria, and requires co-expression with TLR1 and TLR6 for their activation,
Interleukin-10 (IL-10) is an anti-inflammatory cytokine synthesized mainly by immune cells, whose main function is the inhibition both of immune activation and cell death. 7 IL-10 has a critical role in the tolerance to self- and mucosal antigens;8,9 in fact, IL-10-deficient mice spontaneously develop intestinal inflammation, in part due to defective hyporesponsiveness to microbiota. 10 In addition, IL-10 modulates enteroendocrine activity. 11 These effects reveal the essential contribution of IL-10 to intestinal homeostasis.
The intestinal epithelium is a rich source of pro-inflammatory cytokines12,13 where their imbalance is related to several gastrointestinal pathologies such as IBD. 14 Despite the critical role of IL-10 and TLRs in maintaining intestinal homeostasis, little is known about IL-10 production in the intestinal epithelium and its possible interaction with TLR activity. Therefore, our aim was, initially, to analyze IL-10 synthesis and release in human intestinal epithelial cells (Caco-2 cells), and secondly to assess the role of TLR2 and TLR4 activation in the regulation of epithelial IL-10 expression, by studying the dynamics of IL-10 in the intestinal tract of mice deficient in TLR2, TLR4 or both.
Methods
Cell culture
Culture conditions for the human enterocyte-like cell line Caco-2/TC7 has been described elsewhere.
15
The experiments were carried out 14 days after seeding (nine days after confluence), since previous results have shown that Caco-2/TC7 cell differentiation and expression levels are maximal at this time. Cells were treated for 24 hours with culture medium free of fetal bovine serum (FBS) containing either 5 µg ml−1 Pam3CSK4, 50 ng ml−1 Pam2CSK4 (InvivoGen, San Diego, CA, USA); or 30 µg ml−1 LPS (
Animals
The experiments were approved by the Ethics Committee for Animal Experiments from Zaragoza University (PI36/12). The care and use of animals were performed in accordance with the Spanish Policy for Animal Protection RD53/2013 and the European Union Directive 2010/63/EU on the protection of animals used for scientific purposes. Inbred C57BL/10 and mouse strains deficient for TLR2 (
RNA extraction, reverse transcription (RT), polymerase chain reaction (PCR) and real-time PCR
Primers designed from human (h) or mice (m) sequences of the different genes analyzed by polymerase chain reaction (PCR) and real-time-PCR.

Western blotting analysis
Preparation of homogenates from Caco-2/TC7 cells and mouse intestinal samples has been described previously. 3 Proteins were electrophoresed in 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels, and transferred to polyvinylidene fluoride (PVDF) membranes by electroblotting. The membranes were probed with rabbit polyclonal antibody anti-human IL-10 (1:5000) (ABF13, Millipore, Billerica, MA, USA), rabbit monoclonal anti-mouse TLR2 (1:1000) (ab108998) or rabbit polyclonal anti-mouse TLR4 (1:1000) (ab13556) (Abcam, Cambridge, UK). Signal was detected using a secondary antibody coupled to horseradish peroxidase (Santa Cruz Biotechnology, Santa Cruz, CA, USA) and ECL Plus detection kit (GE Healthcare, Buckinghamshire, UK) and visualized with VersaDoc™ (Imaging System, Bio-Rad, Hercules, CA, USA). The blots were reprobed with goat polyclonal anti-β-actin, and protein/β-actin ratio calculated in densitometric units with Quantity One Analysis Software (Bio-Rad).
Enzyme-linked immunosorbent assay (ELISA)
Caco-2/TC7 cells and mouse intestinal explants (30 mg) were maintained in growth medium (Dulbecco Modified Eagle Medium (DMEM) containing 10% FBS, 10 mM glutamine, 10 mM HEPES, 100 U ml−1 penicillin and 100 µg ml−1 streptomycin) for 24 hours at 37℃ in an atmosphere of 5% CO2. After 24 hours of incubation, the medium was collected, centrifuged at 500
Statistics
All results are expressed as the mean ± standard deviation (SD), with
Results
IL-10 expression and release in Caco-2/TC7 cells: Role of TLR2 and TLR4 activation
First, IL-10 expression (mRNA and protein) in Caco-2/TC7 cells was analyzed. An amplification product of expected size (96 bp) was obtained for IL-10 expression upon RT-PCR, and IL-10 protein expression was immunodetected as a band-sized 20 kDa in the cell homogenate (Figure 1(a) and (b)).
IL-10 expression in Caco-2/TC7 cells: Role of TLR2 and TLR4 activation. (a) IL-10 mRNA was detected in two independent preparations. (b) IL-10 protein was analyzed by Western blotting (60 µg of protein), and a band with a size of 20 kDa was immunodetected. (c) IL-10 mRNA expression in cells treated for 24 hours with Pam3CSK4 5 µg ml−1 (TLR2/1), Pam2CSK4 50 ng ml−1 (TLR2/6) or LPS 30 µg ml−1 (TLR4). Results are expressed in arbitrary units and are the mean ± SD of four independent experiments (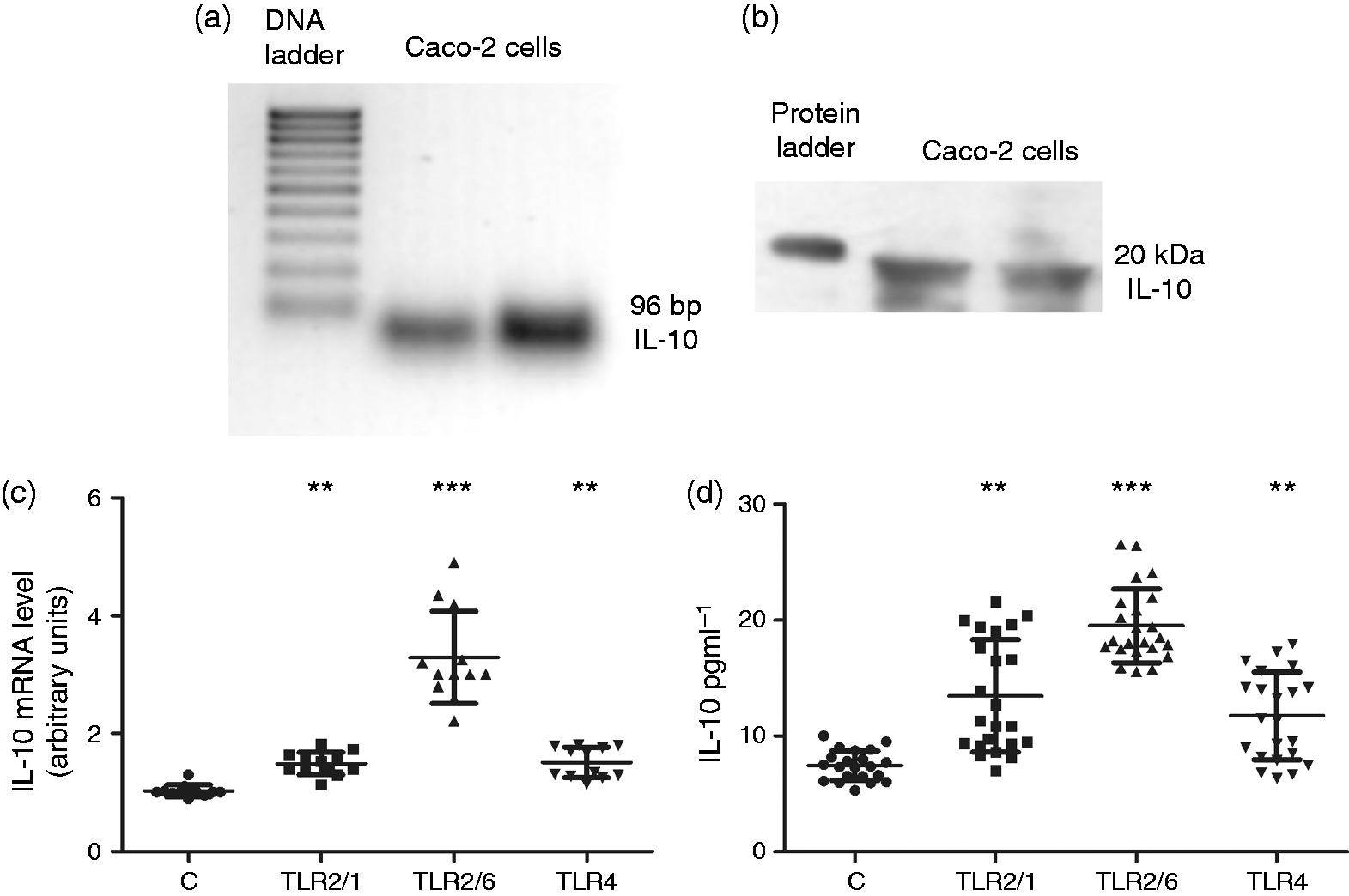
The roles of TLR2 and TLR4 on IL-10 expression and release in Caco-2/TC7 cells were analyzed in cells treated for 24 hours with Pam3CSK4 (5 µg ml−1) or Pam2CSK4 (50 ng ml−1) (TLR2/1 or TLR2/6 ligands, respectively) or with LPS (30 µg ml−1) (TLR4 ligand) concentrations that have been shown to affect the activity of epithelial cells.3,4,16 The results show that the activation of TLR2 or TLR4 increased significantly both IL-10 mRNA levels (Figure 1(c)) and IL-10 release (Figure 1(d)).
IL-10 expression in the intestinal tract of Tlr2–
/
–, Tlr4–
/
– and Tlr2/4–
/
– mice
To corroborate the role of TLR2 and TLR4 in IL-10 expression in the intestinal tract, we carried out the analysis of IL10 expression in the ileum and colon of
Our results show that the in ileum IL-10 mRNA expression (Figure 2(a)) and release (Figure 2(b)) were significantly increased both in Analysis of IL-10 expression and release in the ileum ((a), (b)) and colon ((c), (d)) from WT, 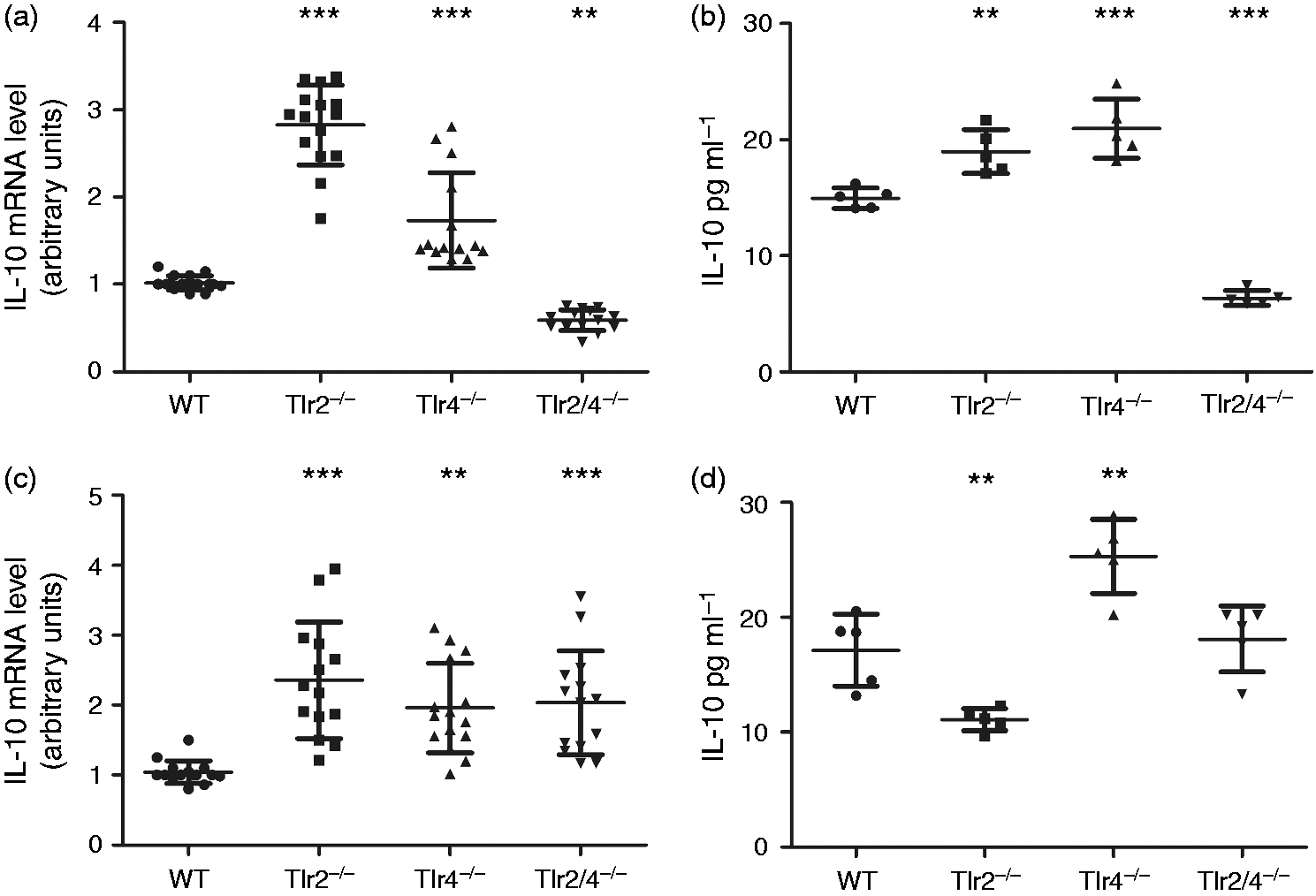
These findings are in line with previous studies that demonstrated TLR2 and TLR4 share potential synergy by crosstalk mechanisms to carry out cell responses;18,19 therefore, a cross-interaction between TLR2 and TLR4 might explain our results on IL-10 expression and release in the intestinal tract. To confirm this hypothesis, IL-10 expression was measured in the ileum and colon of
Cross-regulation between TLR2 and TLR4 expression in mouse intestinal tract
To clarify the previous results, the expression of TLR2 or TLR4 was analyzed in the ileum and colon of Modulation of TLR2 and TLR4 expression in the ileum ((a), (b)) and colon ((c), (d)) of deficient mice (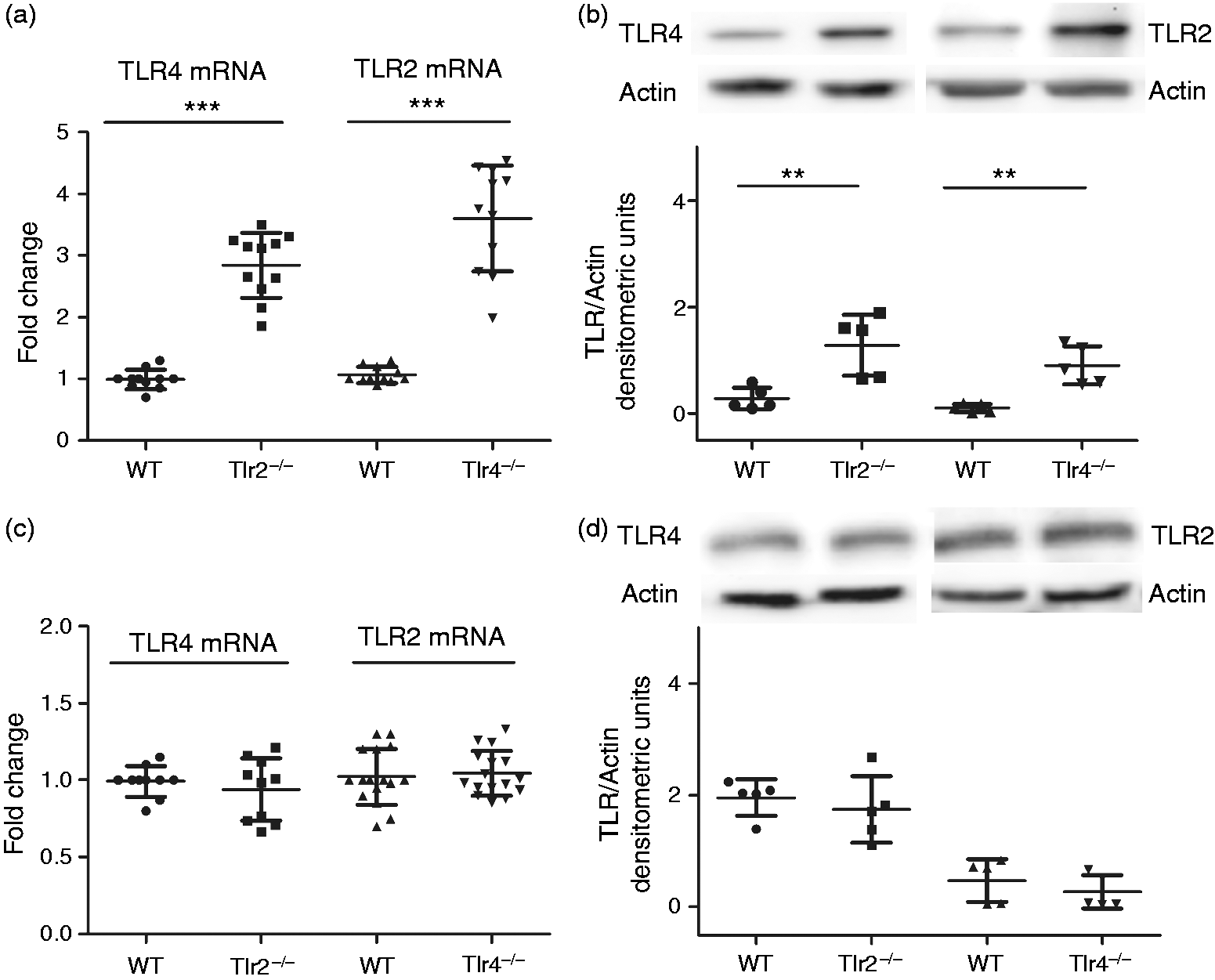
Discussion
The innate immune system plays an essential role in intestinal homeostasis, developing several crucial regulatory mechanisms and interactions. However, deregulation of these mechanisms may be implicated in intestinal pathologies such as IBD, in which a cytokine imbalance and altered microbiota are present.
A number of cell types can produce IL-10, including lymphocytes, monocytes, macrophages, mast cells, keratinocytes, endothelial cells, neurons of the enteric nervous system and intestinal epithelial cells. 20 Intestinal macrophages are robust producers of IL-10, but depend on the presence of commensal bacteria, and the most steady-state macrophages do not express TLRs. In contrast dendritic cells are not believed to be major producers of IL-10 in the lamina propia. 21 Interestingly, intestinal epithelial cells have been identified as the major cell type expressing IL-10 in the normal intestine,22,23 which appears to be physiologically indispensable for epithelial homeostasis. This suggests that in normal, non-inflammatory situations, the intestinal epithelial cells would be the major source of IL-10 production, due to being the major cell type expressing IL-10, but also due to the elevated number of these cells in the gut. IL-10 has been described as an essential cytokine in maintaining intestinal homeostasis 24 and has been shown to be modulated by different microorganism residing in the gut lumen. 25 However, the effect of TLR2 and TLR4, which recognize Gram-positive and Gram-negative bacteria, respectively, on IL-10 expression in the intestinal epithelium, remained unknown.
Our results suggest that enterocytes may act as an IL-10 source for intestinal epithelium that could be increased by TLR2 and TLR4 activation. The role of several TLRs in IL-10 expression has been previously described in dendritic, 26 T-regulatory cells 18 and B cells. 27 Moreover, a recent study has concluded that IL-10 expression in intestinal epithelial cells seemed to be mediated by a crosstalk with macrophages involving TLR4. 23 In this way, another study using Caco-2 and HT-29 cells has described that co-culture with pathogenic bacteria increases IL-10 mRNA expression, together with other anti- and pro-inflammatory cytokines, but co-culture with commensal bacterial induces a reduction of cytokines expression, 28 suggesting different ways of modulation as a function of the bacteria specie and the TLR activated.
The present study has also analyzed the role of TLR2 and TLR4 on IL-10 expression in the intestinal tract. Surprisingly, in the ileum of
The involvement of a cross-interaction between TLR2 and TLR4 in the regulation of IL-10 expression in the intestinal tract was confirmed by the significant reduction of IL-10 expression obtained in the ileum of
The analysis in the colon has indicated that TLR2 and TLR4 did not appear to modulate their expression mutually, and therefore, they did not seem to collaborate in the IL-10 regulation. In fact, the mice deficiency of either TLR2 or TLR4 increased IL-10 mRNA, and
TLR2 and TLR4 are widely known for their role in immune response activating pro-inflammatory cytokine production by nuclear factor (NF)-κB. However, some studies have described a TLR2 and TLR4 critical role in intestinal homeostasis and in the prevention of inflammation.33,34 The results of the present work confirm the role of TLR2 and TLR4 in intestinal inflammatory processes, by acting on IL-10 regulation from the intestinal epithelial cells. Studies in patients suffering IBD have described alterations in both TLR2 and TLR4 expression 35 and a significant decrease in IL-10 levels, 36 supporting our results. In addition, our study has also demonstrated a compensation between TLR2 and TLR4, which may contribute to an essential homeostatic role for preventing inflammation.
Overall, we have demonstrated that intestinal epithelial cells could produce and release IL-10, and actively take part in the immune response. This release appears to be mediated by a compensation between TLR2 and TLR4 expression in the ileum, to ensure the levels of IL-10 in the gut. This work opens the possibility of an immune role for intestinal epithelial cells and illustrates how bacteria in the gut may modulate intestinal activity through TLRs. In addition, the tissue-dependent modulation exerted by TLR2 and TLR4 on IL-10 might explain why certain IBDs are restricted to different regions of the digestive tract.
Footnotes
Acknowledgments
We would like to acknowledge the animal care staff of CITA for assistance in the animal work and maintenance of the mice colony. The authors also wish to acknowledge Dr Lorna W Harries for her critical reading of the manuscript. E. Layunta is a PhD student fellow from the Government of Aragón (B022/13).
Declaration of conflicting interests
None declared.
Funding
This work was funded by grants from the Spanish Ministry of Science and Innovation and the European Regional Development Fund (ERDF/FEDER) (BFU2010-18971), Zaragoza University (UZ2014-BIO-03), European Social Found (ESF) and the Government of Aragón (B61) and the Foundation for the Study of Inflammatory Bowel Diseases in Aragón (ARAINF 2012/0567).
Ethics approval
The experiments were approved by the Ethics Commitee for Animal Experiments from University of Zaragoza (PI36/12). The care and use of animals were perfomed in accordance with the Spanish Policy for Animal Protection RD53/2013 and the European Union Directive 2010/63/EU on the protection of animals used for scientific purposes.
Informed consent
This paper does not need this kind of consent as we do not use humans in our study..
