Abstract
Background
Endoscopic submucosal dissection (ESD) allows “en bloc” resection of superficial gastric lesions.
Objective
The aim of this study is to report “en bloc" resection rate, procedure results, post resection features, analysis of complications, and relapses at mid–long term follow-up on an Italian center case series of patients with superficial gastric neoplastic lesions treated with ESD.
Methods
This is a retrospective evaluation of a prospectively collected series of patients diagnosed with gastric superficial lesions who underwent ESD between November 2007 and May 2015. The analysis included demographic, clinical, endoscopic, and histological data. In detail, the following features were analyzed: (1) “en bloc” resection rate; (2) R0, R1, and RX rate; (3) complication rate; and (4) neoplastic recurrence during follow-up. A comparison was performed between lesions treated according to standard and to extended criteria.
Results
In the analysis, therefore, 70 superficial lesions and 70 endoscopic procedures were considered.
Overall, 53 (76%) procedures were performed according to the extended-ESD criteria. The remaining 17 (24%) cases were performed according to standard criteria. In 68/70 (97%) procedures “en bloc” resection was successful. Forty-four (65.6%) patients had a R0 resection, while 15 (22%) patients had a R1 and the remaining 8 (12.4%) patients were RX. Two patients (2.8%) experienced a major complication. Overall, 7 patients (10.4%) required surgery: in two cases because of local recurrence, in one case for an intraprocedural complication not amenable to endoscopic treatment, in three patients with R1 vertical margins, and in the last patient because of an aggressive pathology (i.e. signet ring cell adenocarcinoma). The mean follow-up of patients with neoplastic lesions was 36.6 ± 13.1 months.
Conclusion
ESD for early gastric cancer is a safe and effective technique when performed by experienced Endoscopists also in Western setting. When comparing the outcomes of the ESD in patients with standard and extended indication there is no significant difference in terms of complete resection, complications and local recurrence rates. Larger Western studies are needed to clearly define the role and the outcomes of ESD in regions at low incidence for gastric neoplasia.
Introduction
Gastric cancer is the third highest cancer-related cause of death worldwide.
1
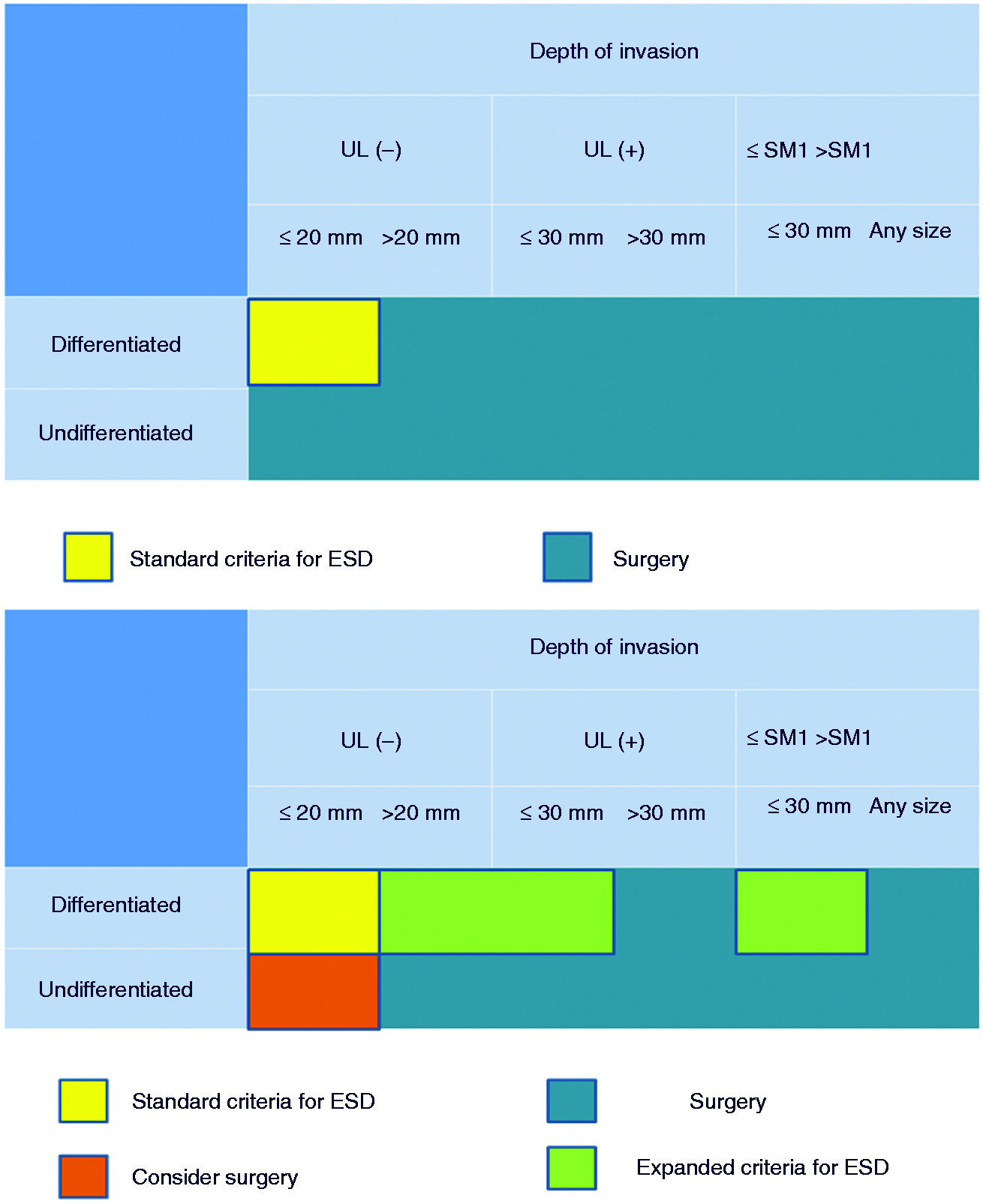
Early detection of pre-neoplastic lesions, early neoplastic lesions and their appropriate removal are of paramount relevance. Both endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) may allow a curative treatment for early gastric cancers (EGC) with low risk of lymph node metastasis. When lesions are larger than 20 mm in diameter, EMR often requires a “piecemeal” resection which impairs R0 resection rate. ESD allows “en bloc” resection of the lesions, allowing an adequate histopathological evaluation and a radical treatment with lower risk of relapse.2,3 The criteria for the endoscopic treatment of EGC proposed by the Japanese Gastric Cancer Association (JGCA) were successively extended by Gotoda et al. (Figure 1).4,5 Extended criteria are widely accepted, so that several EGC patients may be safely managed endoscopically avoiding a surgical resection. A recent meta-analysis reported that the ESD approach was superior to EMR in patients with extended criteria, in term of “en bloc” resection rate, histologically complete resection rate, and local recurrence, although associated with a higher perforation rate.
6
In Western countries, EGC is still largely treated with a surgical approach, being EMR/ESD procedures still marginal and performed in few, highly specialized endoscopic centers.7–13 This results in lack of data coming from Western countries.
Standard criteria for gastric ESD according guideline and extended criteria for gastric ESD (SM: submucosal, UL: ulceration). Modified by Gotoda et al.
5
The aim of this study is to report on a case series of patients with superficial gastric neoplastic lesions treated with ESD in a single Italian center. “En bloc” resection rate, procedure results, post resection features, analysis of complications, and relapses at mid–long term follow-up were evaluated.
Patients and methods
Patients
This is a retrospective evaluation of a prospectively collected series of patients diagnosed with gastric superficial lesions who underwent ESD between November 2007 and May 2015. The analysis included demographic, clinical, endoscopic, and histological data. In detail, the following features were analyzed: (1) “en bloc” resection rate; (2) R0, R1, and RX rate; (3) complication rate; and (4) neoplastic recurrence during follow-up. A comparison was performed between lesions treated according to standard and to extended criteria. Patients with advanced gastric cancer and/or with contraindications to endoscopic treatment or general anesthesia were excluded from the study.
All patients were informed about the endoscopic technique, including advantages and disadvantages, and possible complications. All patients signed an informed consent.
This study was conducted in accordance to the Declaration of Helsinki and was approved by the Ethical Committee of our Hospital (Prot n 0040189/16).
Endoscopic procedures
Initial diagnosis was performed by esophagogastroduodenoscopy (GIF H-180 or GIF-H180J, Olympus, Japan, Tokyo) with chromoendoscopy using indigo carmine and narrow band imaging (NBI). Endoscopic ultrasound (EUS) was not routinely performed. Lesions were classified according to the Paris classification. 7 All procedures were performed either under deep sedation with propofol or general anesthesia. All the ESD procedures were performed by two expert endoscopists (GC and LP) using an Olympus GIF H-180 or GIF-H180J video endoscope system (EVIS EXERA II; Olympus, Tokyo, Japan). In all cases the electrosurgical unit was the VIO 300 (ERBE Elektromedizin GmbH, Tübingen, Germany) set at Endocut I, Dry Cut effect 2, 30W, Swift Coag 30 W.
A soft, straight, and transparent distal attachment (D-201-11304; Olympus, Tokyo, Japan) to facilitate the evaluation of the submucosal (SM) layer was used during ESD. Different cutting tools as Dual-Knife, IT-Knife and IT-Knife2 (KD-611L; Olympus) were used to mark the lesion, to perform the mucosal incision, and then the submucosal dissection. A grasper forceps (Coagrasper; Olympus) was used for bleeding control and hemostasis (ERBE VIO 300 D, Soft Coag 60 W), whereas endoscopic clips (EZ Clip HX-610-135L; Olympus) were used for bleeding control and closure of perforations. Figure 2 shows the procedure steps. In the first phase the lesion was observed in white light (a) and subsequently, to define the side edges, traditional indigo carmine 0.5% chromoendoscopy and high definition virtual chromoendoscopy (NBI) was used (b). Border marking was performed at 2–5 mm from the side margins of the lesions using closed toe cutting device Dual-Knife (c). Submucosal injection was done with a 10% glycerol solution (Chugai Pharmaceutical Co, Tokyo, Japan) with indigo carmine 0.5% and adrenaline at different dilutions or hydroxypropyl methyl cellulose (HMPC) with addition of indigo carmine and adrenaline at different dilutions (d). Circumferential incision to access the submucosa was performed with Dual-Knife externally to the border markings. Finally, ESD was performed using different cutting tools (Dual-Knife, Knife-IT, IT-Knife 2). When necessary, monopolar coagulation was performed by hemostatic forceps (Coagrasper).
Endoscopic submucosal dissection of an angulus lesion (extended criteria): (a) white light lesion evaluation; (b) NBI lesion analysis; (c) border marking; (d) submucosal injection; (e) circumferential incision; (f) endoscopic submucosal dissection; (g) post “en bloc” resection eschar; (h) gastric lesion removed and staged oriented on cork.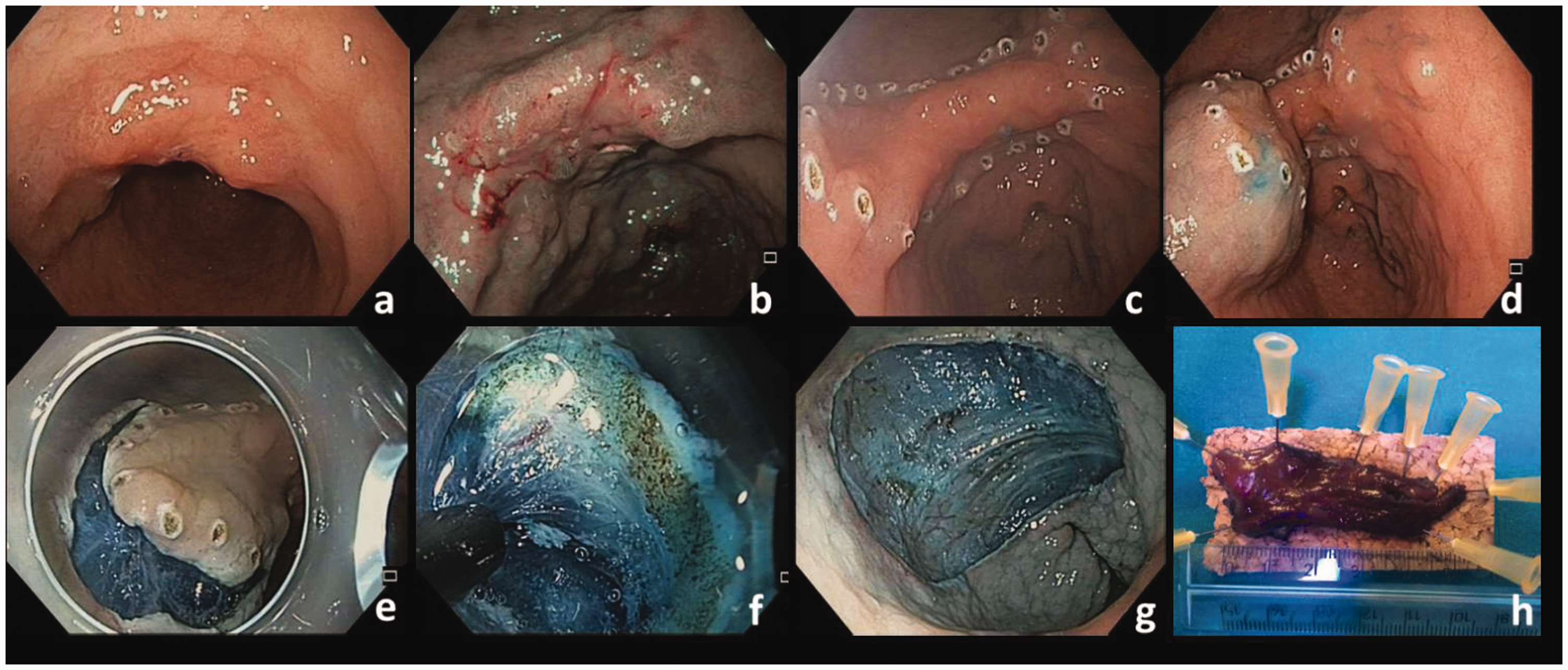
Histopathological assessment
All resected lesions were placed and oriented on cork immediately after the procedure. Complete resection was confirmed by the presence of the markings on the side edges of the lesion. Prior to cutting, specimen bases were marked with India ink; complete resection was confirmed by the absence of neoplastic tissue on the marked edges of the resected specimen. The “piecemeal” removed lesions were reconstructed trying to respect the topography of the lesion and fixed on a cork. Lesions sizes were recorded. Samples were fixed in formalin for histopathological evaluation. Two millimeters cuts were performed and lateral margins, depth of invasion and the presence of tumor budding and lymphovascular embolization were evaluated. 8 Lauren classification for histological diagnosis was used. 9
Endpoints
Feasibility was defined as the success in removing the entire endoscopic lesion. The ESD was defined complete if dissection was completed, while incomplete if the lesion removal was completed by using EMR snares (Oval plurifilament type, Olympus, or monofilament type, Riab Medical s.r.l., Cesena, Italy). The JGCA definitions were adopted to classify the completeness of dissection. In detail, “en bloc” resection (ER) was defined as resection in a single piece as opposed to piecemeal resection (in multiple segments). Complete en bloc resection (R0) was defined when en bloc resection was achieved, with lateral and deep tumor-negative margins. A free margin was defined as a 2 mm distance from the lesion to the margin in the resection specimen. Curative resection (CR) was defined when R0 was achieved, with absence of lymphovascular invasion and submucosal infiltration <500 µm. Unclear horizontal and/or vertical margins were analyzed as RX. 10 A resection with histopathological confirmation of residual lesion of deep and/or lateral margins was defined R1. Recurrence was defined as presence of neoplastic tissue (intraepithelial neoplasia or high and/or low-grade dysplasia) at the scar during follow-up endoscopy. Follow-up was performed by upper endoscopy and biopsies every 3 months for the first year, and thereafter once a year.
Adverse events
Major and minor adverse events were routinely recorded. Perforation was considered a major complication. Perforation was defined as the presence of mesenteric fat or intra-abdominal space visualization through the gastric wall during the procedure. Procedural bleeding was considered a major adverse event only when not manageable endoscopically or requiring red blood cells transfusion. A delayed bleeding was defined a major adverse event in case of hematemesis and melena appearing >24 h post-procedure and/or a reduction of more than 2 g/L of hemoglobin. Microperforations or bleeding episodes that occurred during the procedure and that could be successfully managed by endoscopy during the procedure were considered as minor complications.
Statistical analysis
Statistical Package for Social Sciences (SPSS 17.0) software was used for statistical analysis of the data. Comparisons between groups of extended criteria (Ext-G) and the standard criteria (St-G) was performed using the Fisher test. The difference was defined as statistically significant for p < 0.05. The comparison of the size and characteristics of the lesions was performed with the t-test. The difference was defined as statistically significant when p < 0.001.
Results
A total of 70 patients (34, 49% males, 36, 51% females, mean age 67.7 years, range 34–85 years) were eligible for ESD, for a total of 71 superficial gastric lesions. One patient presented with two lesions, while in one case the lifting sign was absent. In this latter patient the endoscopic resection was not attempted and he underwent surgical resection. In the analysis, therefore, 70 superficial lesions and 70 endoscopic procedures were considered.
Demography, classification of lesions, and pathology results.
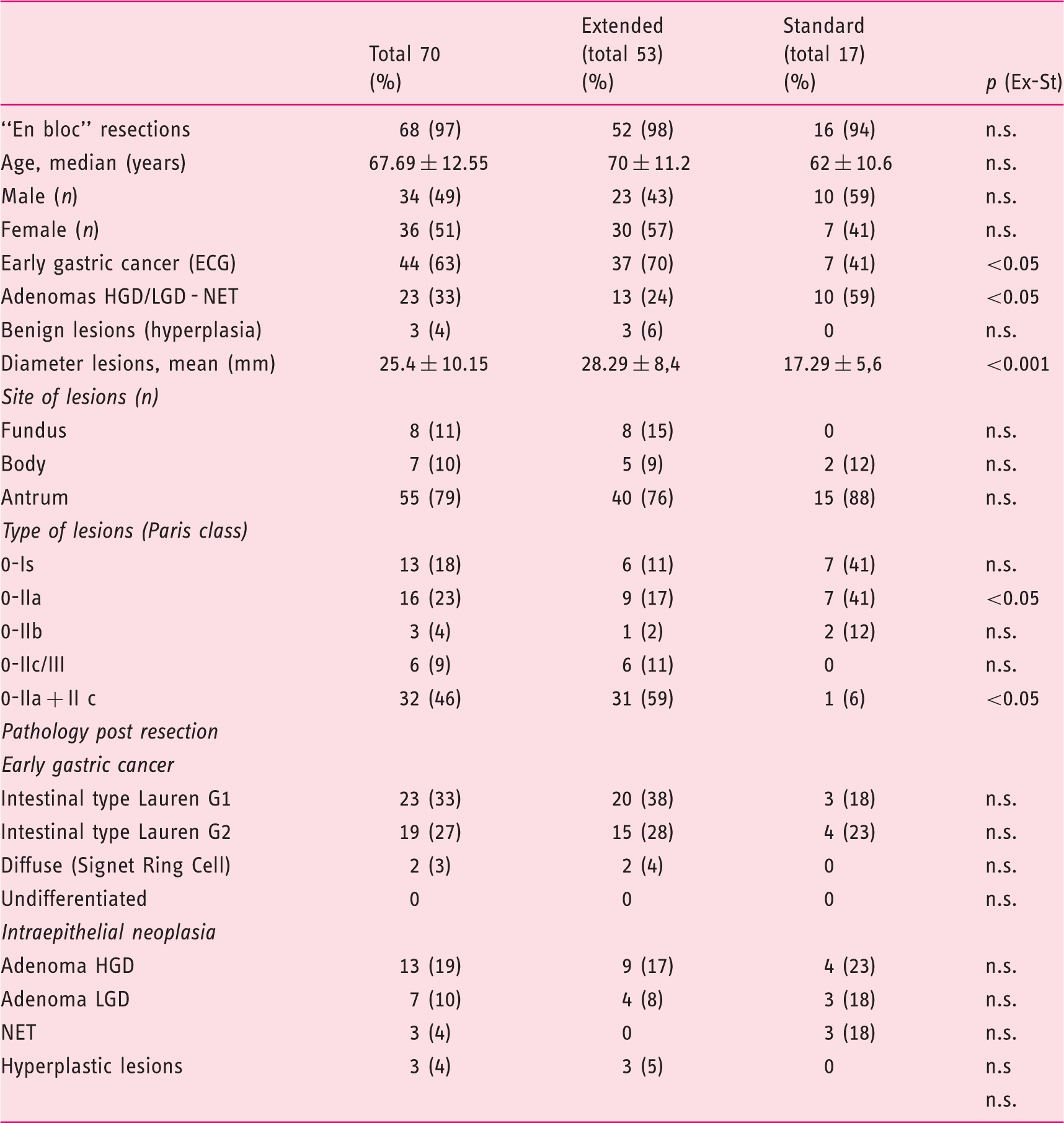
In 68/70 (97%) procedures, “en bloc” resection was successful; in one case a circumferential incision followed by lesion removal in two pieces by diathermy snare was performed (Hybrid-ESD), while in the remaining patient a partial ESD was done and completed with “en bloc” EMR. The average procedure time was 70 ± 42 min (Table 1).
The histology of the resected lesions is reported in Table 1. Sixty-seven out of the 70 resected lesions were neoplastic, while the remaining three were hyperplastic lesions.
Post resection features and follow-up.
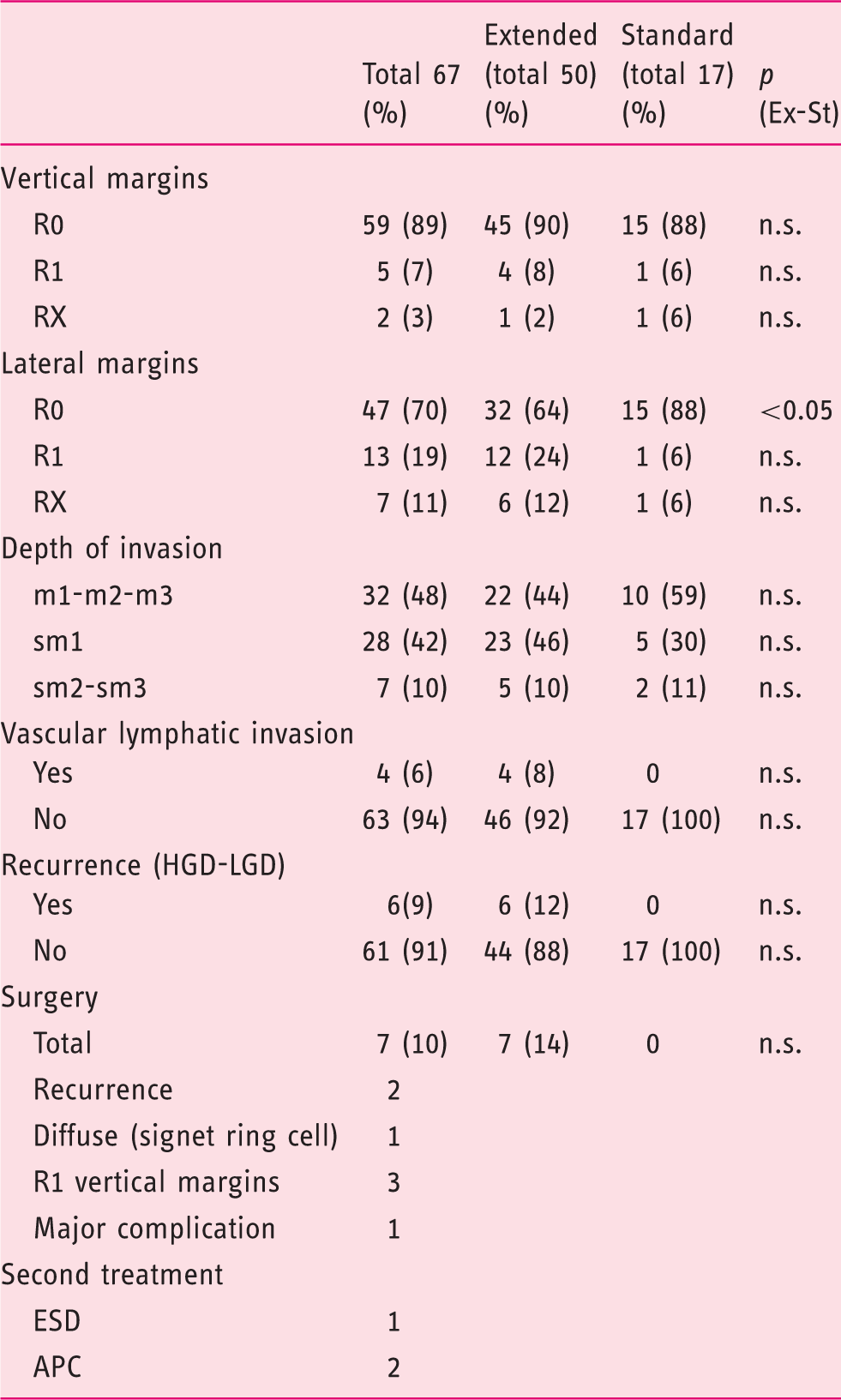
sm: submucosa; m: mucosa; APC: argon plasma coagulation; HGD: high-grade dysplasia; LGD: low-grade dysplasia.
Details of patients with R1 and/or RX.
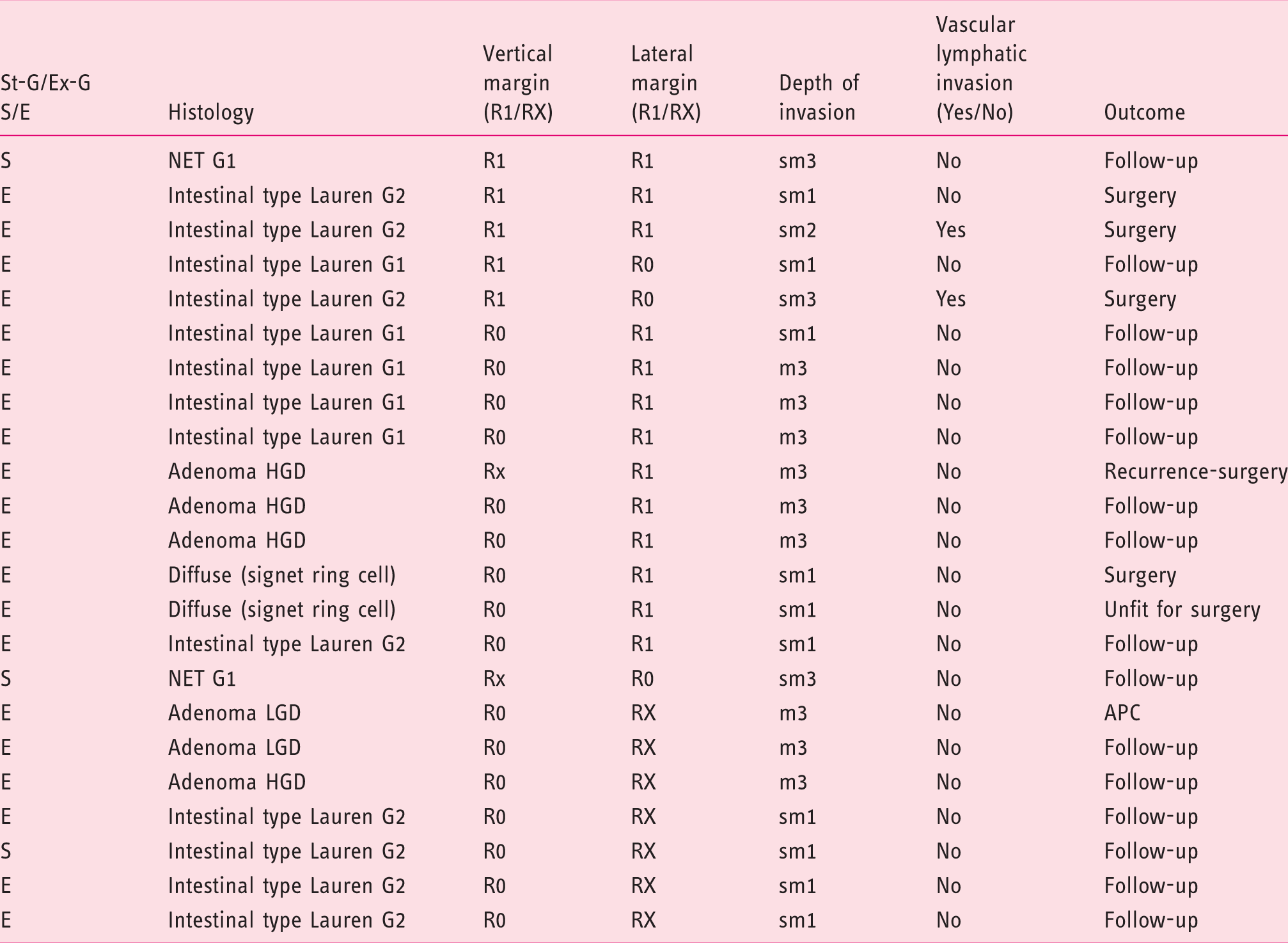
Two patients (2.8%) experienced a major complication. One patient presented a delayed (24 hours post procedure) bleeding with hematemesis and reduction of 4 g/L of hemoglobin. An urgent endoscopy was performed and a combined hemostasis (monopolar coagulation with Coagrasper and clipping (Type EZ LONG, Olympus) was successfully attempted. Another patient had an intraprocedural perforation that was unsuccessfully treated by clips (OTSC System, Ovesco Endoscopy AG, Tübingen, Germany). This patient was, therefore, referred for surgical resection. Two intraprocedural microperforations occurred. These patients were treated successfully by clipping, without sequelae.
Details of patients who presented recurrence.
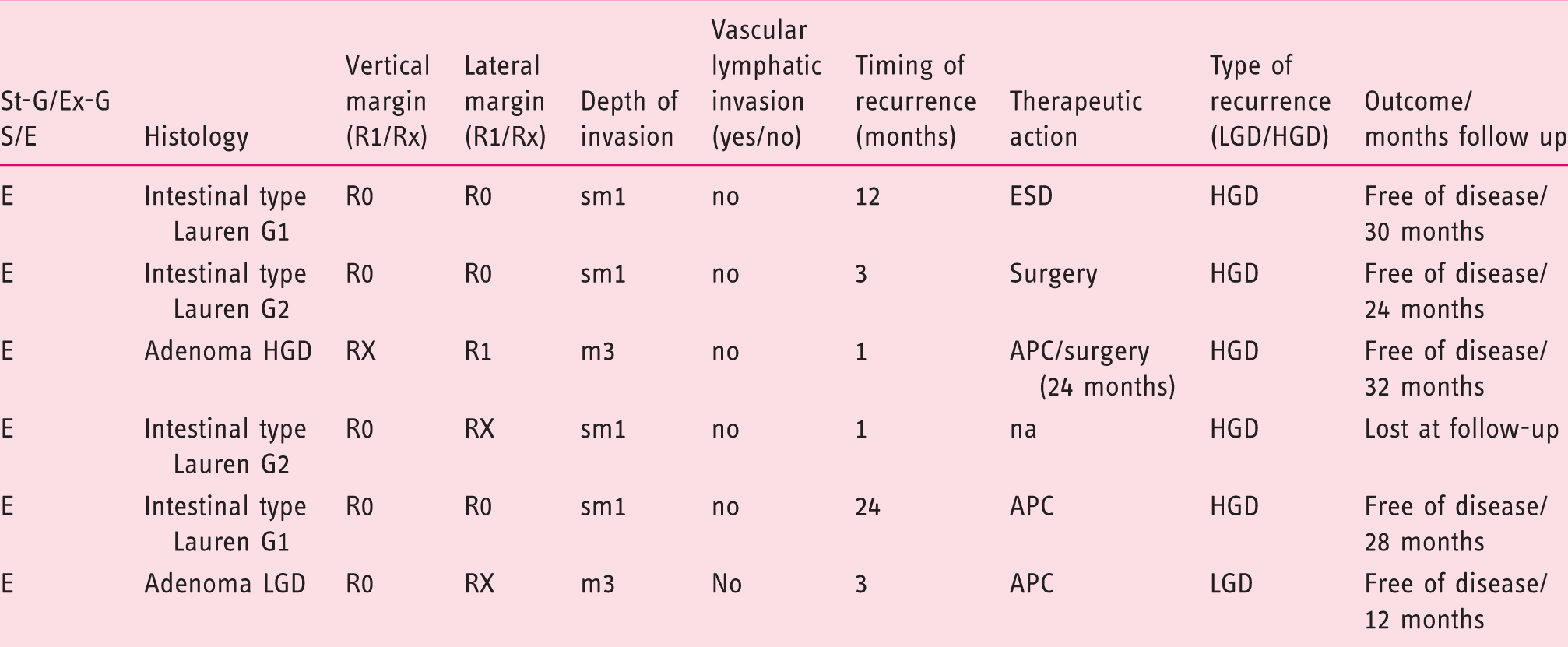
Tables 1 and 2 report the details of a comparison between the extended criteria group (Ex-G) and standard criteria group (St-G). To note, a statistical significant difference was observed when considering the histology, diameter, type (Paris classification) and completeness of resection when comparing lesions with extended and standard criteria. In details, lesions with extended criteria were more frequently EGC (70% vs. 41%), were bigger in diameter (28.3 mm vs. 17.3 mm) and they were more frequently complex lesions (i.e. 0-IIa + IIc) (59% vs 6%). Lesions with standard criteria had a statistically higher R0 resection for lateral margins when compared to lesions with extended criteria. No differences were observed between the two groups in terms of en-bloc resection, R0 resection for deep margin, lympho-vascular invasion, recurrence and adverse events.
Discussion
The results of the present study show that there was no difference between standard and extended criteria group in terms of complete resection rate (98% vs. 94%; p = ns). Complication rate (0% vs. 2.8%; p = ns) and local recurrence rate (0% vs. 12%; p = ns) were lower in the standard indication group, nevertheless the difference was not statistically significant. Endoscopic resection has become the standard treatment for pre-neoplastic or early malignant lesions in the stomach. Conventional EMR, although very useful for the removal of small lesions, has the limitation at not allowing a safe and potentially curative “en bloc” resection when the lesions are bigger than 20 mm in diameter. ESD has been developed in Japan to overcome this limitation. 11 The advantage of ESD is the ability to perform “en bloc” resections without any dimensional limitation. This advantage leads to an adequate histopathological evaluation of R0 resections and reduces the risk of local recurrence. 12 Large studies have reported “en bloc” resection rates with no cancer margins in more than 90% of EGC and large flat gastric adenomas. 12 Traditionally, only mucosal lesions smaller than 20 mm were considered suitable for endoscopic treatment. The study by Gotoda et al. on a large series of 5265 patients who underwent gastrectomy with lymph node resection for EGC proposed new criteria suggesting a low risk of lymph node metastasis even in large and complex lesions. 5 Based on the results of this trial an extended indication for endoscopic resections was proposed for EGC. In adjunction, the advance of instruments as well the development of new techniques allowed to perform safe endoscopic resections also of complex gastric lesions, avoiding unnecessary surgery. Therefore, the number of patients receiving ESD for EGC has continuously increased during the last years.
Several studies showed that ESD in extended indication has similar recurrence rate and disease-free survival rate when compared to standard indication. However, the vast majority of the published data comes from Japan or Eastern countries. This is due to a lower incidence of gastric cancer and especially of EGC in the Western world in comparison to Asian regions, 13 where there are widely diffused mass screening programs.
Comparison with previous major series.
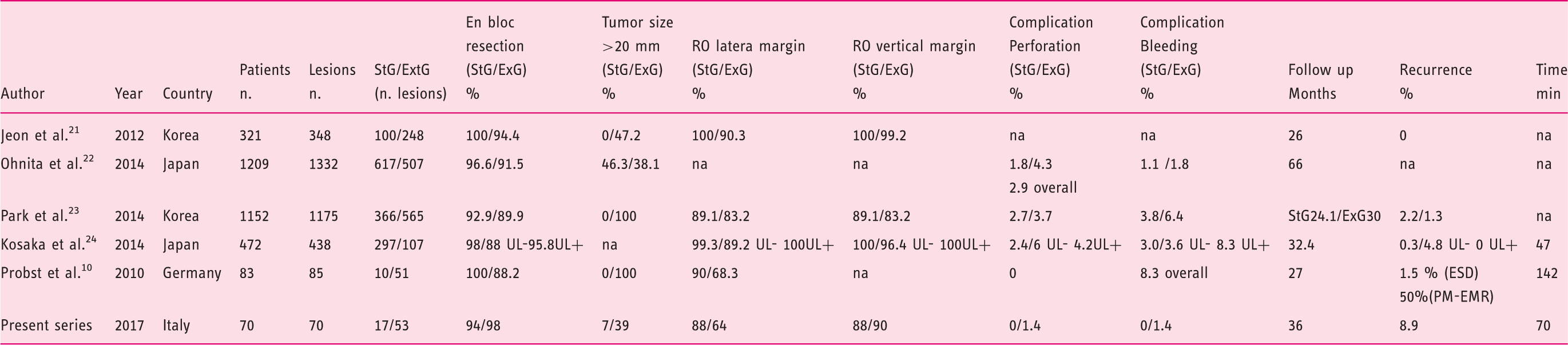
Both complications in the present series occurred in patients with extended indications. Although there is no statistically difference in terms of recurrence rate and adverse event occurrence between the two groups, the higher recurrence and adverse event rates observed in the extended group should be considered with caution suggesting the higher complexity of ESD in patients with bigger and/or more advanced lesions. Sixth, EUS was not routinely performed in the present series. Several studies have compared the accuracy of invasion depth measurement between conventional endoscopy and EUS. Two recent meta-analyses concluded that EUS shows low accuracy for staging the depth of EGC invasion limiting the role of EUS in the staging algorithm and for the selection of patients with early gastric neoplasia suitable to ESD treatment.19,20
Finally, to note, the 70 patients included in the present study were collected during a 8 years period of enrollment while, for example, Kosaka et al. collected 472 cases of EGC during a 5 years period of enrollment. 17 The low incidence of gastric lesions with an indication for ESD in Western Countries makes the learning curve long and difficult to be completed in a reasonable period of time, even in referral centers. This reflects the different skills and it might be in part responsible of the differences of the performances when comparing the present series with larger, Eastern series.
This study presents some limitations. The main limitation of the present study is the small sample size. Although the sample size is similar to that of previous European studies, this study seems to be hardly comparable when looking at East. On the other hand, only few studies report Western experiences on the endoscopic treatment of gastric lesions. Therefore, the present study gives a new, additional picture of the gastric ESD in regions where the incidence of gastric neoplasia is low and where, probably, the endoscopic skill for ESD needs to be improved. The small number of the two groups (i.e. standard and extended indications) makes the comparison difficult. As a consequence of the small sample size the difference of most of the evaluated parameters was not significant. Finally, although patients were prospectively collected, the retrospective design and the lack of a formal power calculation are a clear limitation. About this, it needs to be underlined that although the differences between standard and extended criteria group failed to reach statistical significance a formal power calculation was not performed and with just 24% of cases meeting standard criteria it is likely that the study is underpowered to show any difference.
In conclusion, the results of the present study suggest that ESD for EGC is a safe and effective technique when performed by experienced Endoscopists also in Western setting. When comparing the outcomes of the ESD in patients with standard and extended indication there is no significant difference in terms of complete resection, complications and local recurrence rates. The small sample size of the present trial limits the generalizability of the results. Larger Western studies are needed to clearly define the role and the outcomes of ESD in regions at low incidence for gastric neoplasia.
Footnotes
Declaration of conflicting interests
None declared.
Ethics approval
This study was approved by the Ethical Committee of our Hospital (Prot n 0040189/16).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
All patients were informed about the endoscopic technique, including advantages and disadvantages, and possible complications. All patients signed an informed consent.
