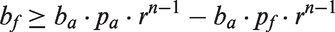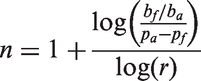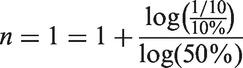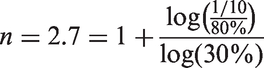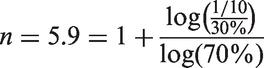Abstract
Background and aims
In complex endoscopies, the initial attempt is often associated with the highest success and subsequent attempts to achieve the same outcome have a higher failure rate. An attending physician needs to decide how the ultimate success may become compromised by letting a fellow start the procedure. A decision analysis is aimed to shed light on this issue.
Methods
A formula is derived to calculate the point in time when to switch the instrument between an attending and fellow physician. This time point is determined by the ratio of benefit to the patient over benefit to the fellow, the difference in probability of achieving success by an attending versus a fellow, and the decline in probability of success during consecutive procedural attempts.
Results
The attending should undertake the first attempts at doing the procedure, if the fellow is inexperienced and the difference in success rate between attending and fellow is still large, if the procedure is risky, and if the benefit of a successful outcome outweighs the benefit of a teaching experience to the fellow. Vice versa, fellows should take the lead, if they have become well trained and the difference in their procedural success rate compared with the attendings’ has grown relatively small. The fellow should also be trusted to lead in all instances where the risk to the patient is small.
Conclusions
Such rules can serve as general guidance when to pass the endoscope to a fellow physician. Medical decision analysis is helpful to enlighten complex situations during training of fellows.
Introduction
A large body of literature deals with teaching of endoscopic skills to future gastroenterologists. Professional societies have published lists of required skills and recommendations for training in various endoscopic procedures.1–5 A variety of different tools has been developed to assess skill advancement and outcome of training. 6 Most tools are based on the measurement of procedure-specific metrics that that are thought to reflect endoscopic competence. 7 For endoscopic retrograde cholangiopancreatography (ERCP), for instance, such metrics include cannulation of the desired duct, sphincterotomy, stone clearance, and stent placement. 8 In colonoscopy, the metrics include cecal intubation, insertion time to the cecum, and polyp detection rates.9,10 As the accomplishment of different metrics improves with the overall number of procedures, many teaching recommendations have centered on procedure numbers that fellows should have accomplished before their proficiency becomes accredited.10,11 Although the number of procedures seems important, obviously, competence in endoscopy involves more than achieving a minimal number.13–15
Learning how to scope occurs by observing others, by instruction, and mostly by doing procedures oneself. Fellows have reported that they learned endoscopy best hands-on by performing procedures themselves.16,17 Through repetition the fellow ultimately obtains a feeling for the most effective means to hold and move the instrument and for how various organs react to a multitude of different manipulations. During a fellow’s training, the patient may experience a slightly longer procedure with more discomfort and a less than perfect outcome. Two teaching scenarios arise, depending on the type of procedure being taught. For standard procedures, such as esophagogastroduodenoscopy (EGD) or colonoscopy, the attending physician may always let the fellow start the procedure and decide to take over only when the fellow begins to struggle or the patient becomes uncomfortable. For complex procedures or difficult interventions, however, there may be an initial window of opportunity that narrows over time. In such instances, the attending may try first before handing the instrument to the fellow. The present analysis deals primarily with the second scenario.
Gastroenterologists in general and interventional endoscopists in particular, who have perfected their manual skills during a prolonged professional period, find it difficult or even painful to watch a beginner fumble a delicate maneuver. After an unsuccessful initial attempt by a fellow, subsequent attempts to achieve the same outcome may have a lower probability of success or fail altogether. During ERCP, for instance, after multiple manipulations, the papilla will become edematous and more difficult to cannulate. A papillotomy cut too long is impossible to reverse. A stent deployed too low or too high with respect to a stricture is associated with a higher failure rate or becomes wasted altogether. Similar exigencies apply to placement of percutaneous endoscopic gastrostomy (PEG) tubes, dilation of strictures, removal of large colonic polyps, banding of varices, or various other means of hemostasis in the gastrointestinal tract. By letting the fellow perform the procedure the attending physician has to assess to what extent the ultimate success may become compromised. The potential compromise in procedural benefit to the patient has to be weighed against the training benefit to the fellow. When is the best point in time to switch and let the fellow do the procedure? A simple decision analysis may shed some light on this issue.
Methods
The methods serve to derive a formula for the point in time when to switch between endoscopy done by an attending versus fellow physician. In probability theory, economics and medical decision analysis alike, an uncertain benefit is generally weighted (multiplied) by the probability of its occurrence to yield an expected benefit value. Any comparison between different benefits weighted by varying probabilities of occurrence is based on comparing the magnitudes of their respective expected values. Similarly, any difference between two benefits associated with varying probabilities is calculated as the mathematical difference of their respective expected values.18–20 Let pa and pf be the probability of achieving success with a given endoscopic intervention done by an attending physician or fellow, respectively. With each consecutive attempt this initial probabilities falls to a fraction r. The procedural benefit to the patient after a successful intervention is ba. The expected benefit exp of the first attempt by an attending is
When both sides of the equation are equal, this algebraic equation can be solved for the threshold value of n:
Although it may look complicated, the formula is actually easy to compute using a spreadsheet or any hand-held calculator. The outcome n indicates the running number of the attempt, when the attending physician should let the fellow take over and try finishing the procedure. This outcome is determined by three factors: first, the (inverse) ratio of benefit to the patient over benefit to the fellow ba/bf ; second, the difference in probability of achieving success by an attending versus a fellow pa – pf ; and third, the decline in probability of success r during consecutive procedural attempts. Because the two benefit values enter the equation only as a ratio, there is no need to specify any benefit in absolute terms but only as a relative measure of benefit to patient over benefit to the fellow. In all subsequent calculations, a tenfold ratio of patient over fellow benefit is assumed.
Results
In this first paragraph, I provide some examples of how the formula from above helps to decide when to pass the endoscope from attending to fellow physician. Assuming a tenfold ratio of patient over fellow benefit, a 10% difference in probability of achieving success by an attending versus a fellow, and a 50% decline in success probability during consecutive procedural attempts yields
For illustrative purposes, consider the example of a 76-year-old icteric female with serious cardiovascular comorbidities and a 1 cm gall stone in her common bile duct. The probability of success for a well-placed papillotomy is assumed to vary by pa – pf = 80% between the attending and fellow physician. The first cut seems the most crucial step and, by comparison, a second attempt may be only r = 30% as successful. As shown by the calculation below, the fellow should not perform the papillotomy at least during the first two attempts:
As another example, consider a 2 cm flat adenoma in the cecum of an otherwise healthy 60-year-old male. The probability of success is assumed to vary by pa – pf = 30% between the attending and fellow physician. The success rate drops by 30% among consecutive attempts at polypectomy, with r = 70%. As suggested by the calculation below, the attending should probably complete the polypectomy him/herself and not switch the instrument before the sixth attempt:
As baseline for the subsequent sensitivity analyses, I assumed a tenfold ratio of patient over fellow benefit, a 40% difference in probability of achieving success by an attending versus a fellow, and a 80% success probability of consecutive procedural attempts. In the four panels of Figure 1, these variables were then varied over a broad range. As the expected benefit of consecutive procedures drops for the attending, as well as the fellow, the difference between their expected benefits shrinks (Panel A). An increase in the difference between the success probability of attending versus fellow shifts the point of when to let the fellow do the procedure towards later attempts (Panel B). The ratio of patient benefit over fellow benefit reflects on the severity of the underlying medical condition and the consequences of potential failures. A rising ratio shifts the point of switching the instrument towards later attempts (Panel C). If the initial success rate rapidly declines (with r ≪ 1), the difference in the expected benefit of procedures done by the attending versus fellow also rapidly declines, and the fellow could take over sooner rather than later. If the decline occurs only slowly, a substantial difference between attending and fellow is maintained longer, and the fellow should take over later rather than sooner (Panel D).
Panel A. Decline in the expected benefit with increasing number of trials. Panel B. Influence of the difference between the success probability of attending versus fellow on the number of procedural attempts before the switch (and letting the fellow take over). Panel C. Influence of the ratio of patient benefit over fellow benefit on the number of procedural attempts before the switch. Panel D. Influence of the general decline in success rate on the number of procedural attempts before the switch.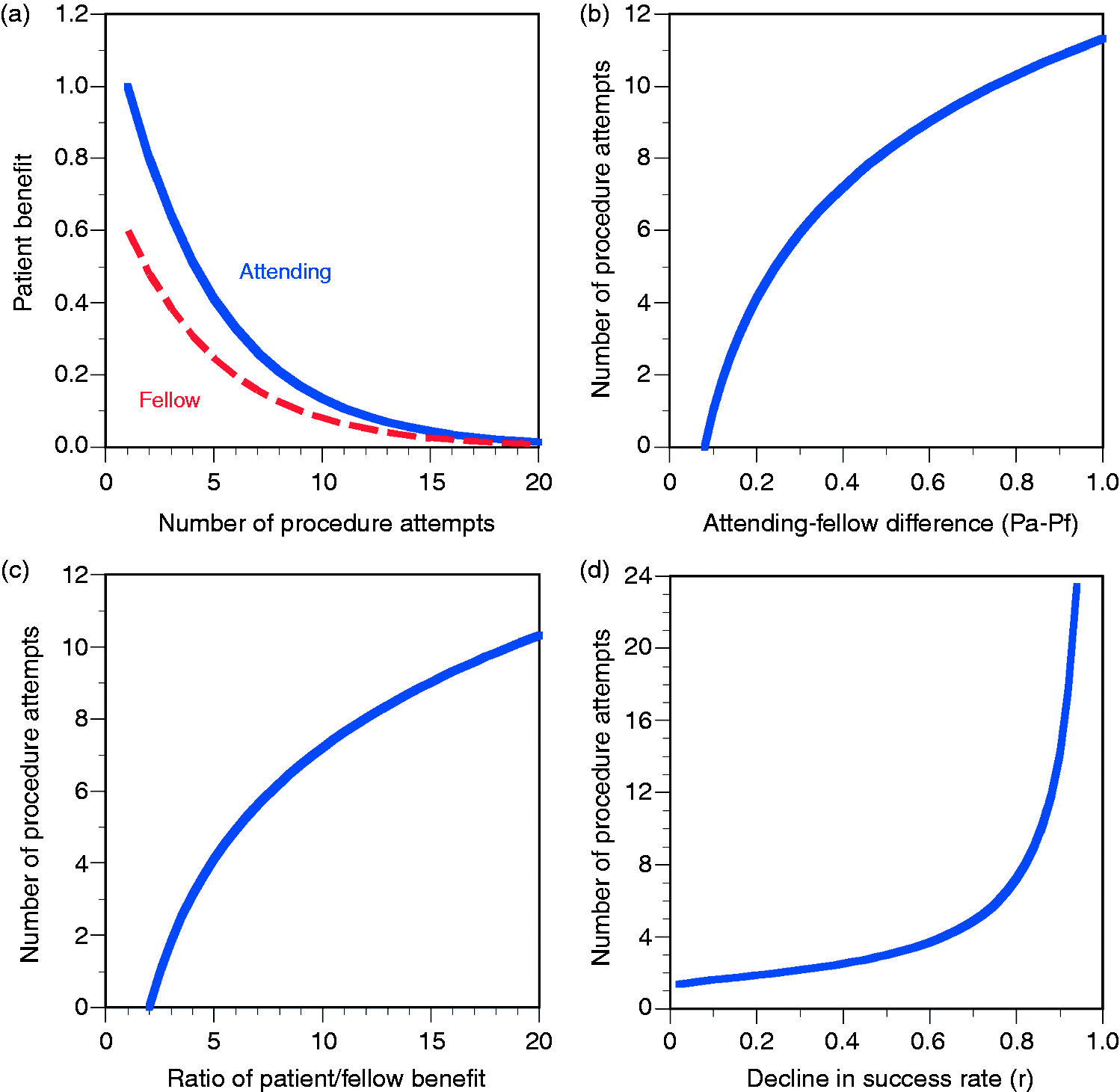
Discussion
A decision analysis was used to assess the factors that determine the time point when the attending endoscopist should let a fellow physician take over performing a potentially complex endoscopic procedure. As the decision analysis illustrates, the time point of when to let a fellow take over depends on three parameters, that is, the difference in the success rate of attending versus fellow, the ratio of patient over fellow benefit, and the decline in success rates among consecutive procedural attempts.
When should the attending undertake the first attempts at doing the procedure? First, when the fellow is inexperienced and the difference in success rate between attending and fellow is still large. Second, when the procedure is risky with a narrow window for success and the benefit of a successful outcome outweighs the benefit of a teaching experience to the fellow. Vice versa, fellows should take the lead, first, if they have become well trained and the difference in their procedural success rate compared with the attendings’ has grown relatively small. Second, the fellow should be trusted to lead in all instances where the consequences of initial failure are negligible. On one hand, a rapid deterioration of the preconditions for a successful procedure renders the initial interventional attempts more important and preference should be given to an initial attempt by the attending. On the other hand, since all subsequent attempts are less likely to be successful by the attending or the fellow, such circumstances may also favor to let the fellow attempt the procedure sooner rather than later.
These rules are meant to serve as general guidance on how to decide when to pass the endoscope to a fellow physician. As one would not expect a single type of endoscopic procedure to be successful in all types of gastrointestinal disease, similarly, one must not expect a single model to cover the variety of all scenarios that arise in daily practice. Mathematical modeling of medical decision making is helpful in conceptualizing physician behavior and understanding how interacting factors influence the outcome of medical interventions. However, mathematical models only represent an abstraction and simplification of reality. They cannot capture the contribution of every clinical detail, such as age, gender, comorbidity, medical history, or local exigencies, which affect individual decision making. Attending physicians are not expected to pull out a computer and use the present formula during a busy endoscopy schedule. Instead, the model should be perceived as a means to enlighten and understand a complex situation that often arises during training of fellows.
Footnotes
Disclosure of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.