Abstract
Background
Treatment of gastric cancer is based on accurate staging. Emerging methods, such as PET-CT, are increasingly being used for this purpose. Our aim was to analyze the results of EUS and PET-CT in staging and restaging our patients with gastric cancer, comparing both of them with the histological results.
Methods
Patients with confirmed gastric cancer were prospectively enrolled. Inclusion criteria for the final analysis included only patients who finally received a surgical resection. All patients underwent preoperative TNM staging by means of EUS and PET-CT within 21 days prior to the surgical treatment.
Results
A total of 256 patients were included. The overall EUS accuracy for T staging was 78% and 80.2% in restaging. EUS showed its best accuracy when distinguishing T1–T2 tumors vs. T3–T4, with an increased accuracy in restaging. Regarding N staging, the overall accuracy of EUS was 76.2%, and 72.5% for PET-CT (
Conclusions
EUS performed better than PET-CT in gastric cancer N staging and restaging. EUS accuracy in this setting is still suboptimal and probably more than one single diagnostic procedure should be used.
Introduction
Gastric cancer is one of the most common malignant tumors in the gastrointestinal tract, and the second most common cause of cancer-related deaths worldwide. 1 The treatment of gastric cancer comprises different options based on preoperative staging tests. The main way to achieve a curative result is radical surgery. However, new approaches, which comprise endoscopic therapy for early gastric cancer and neoadjuvant therapy for patients with advanced disease, have become the mainstay in the management of these patients. 2 In this setting, precision in preoperative staging is essential for the individualized stage-dependent treatment of gastric cancer and for the prognosis of these patients.
Endoscopic ultrasonography (EUS) and multislice spiral computed tomography (MSCT) are the most common techniques for the preoperative staging of gastric cancer patients. EUS has shown to be very useful for detecting the depth of gastric cancer invasion, with a T staging accuracy of approximately 80%–90%. 3 In this regard, some meta-analyses have reported a high accuracy, especially for differentiating T1–2 from T3–4 gastric cancers, with an overall sensitivity of 86% and a specificity of 91%. However, the performance of EUS for the detection of mucosal and submucosal invasion of early gastric cancer is not accurate enough, and has been so observed in a recent meta-analysis 4 and in previous reports in which EUS did not show a significant advantage over conventional endoscopy for early gastric cancer staging. 5
The endosonographic assessment of lymph node (LN) metastasis (N staging) is usually based on their morphological characteristics, echogenicity and size. Nevertheless, exclusive echographic criteria have shown a poor reliability, and a lower performance for N staging has been reported.4–6 The advent of EUS-fine-needle aspiration (FNA) has improved the accuracy of EUS for N staging to a promising level, although more data are needed to definitely establish the role of EUS-FNA in this setting. 7
Nonetheless, in view of the available evidence and the current guidelines, the real distinctions with a clinical impact on patients are, apart from early gastric cancers, in which conventional endoscopy could be enough and EUS probably has a small role, 8 the ones between T1–2 vs T3–4 and N0 vs N-positive gastric cancers. 9 Furthermore, a recent report has shown that early cancer can be accurately adequately assessed with simple criteria for conventional endoscopy, with accuracy comparable to that of EUS. 10
However, some other methods have been tested and used in clinical practice to improve the preoperative assessment of these patients. For instance, multidetector row computed tomography (MDCT) can actually be considered the standard of care for the detection of distant metastases, 9 and has shown results comparable with EUS. 8 The addition of positron-emission tomography (PET) to MDCT, with the current PET-CT technology, offers a promising option for the global staging of gastric cancer, given that the isolated PET has already emerged as a promising imaging modality. 11 With the increasing evolution of these techniques and expanded clinical experience, studies comparing the most accurate and efficient methods in this setting are needed. Therefore, we decided to analyze the results of both methods in our patients, when staging new patients with gastric cancer, and in patients restaged after neoadjuvant therapy, a clinical scenario in which solid evidence is still lacking. Moreover, we analyzed our results in a real clinical setting, studying our usual staging algorithm for gastric cancer. Few studies have analyzed EUS results in restaging, only acknowledging this procedure has some value when downstaging was observed, as a marker of good prognosis.12,13
Methods
Study design and population
This was a prospective series of consecutive patients referred to our unit for gastric cancer staging or restaging, under a strict protocol approved by the gastrointestinal cancers committee. Inclusion criteria included patients who finally received surgical resection and who had a confirmed gastric cancer. Patients who had undergone endoscopic mucosal resection (EMR) and patients with widespread metastatic disease or who had not yet received a surgical resection were excluded from the study. All patients received preoperative TNM staging by means of EUS and PET-CT within 21 days prior to the surgical treatment. (See Figure 1.)
Study design.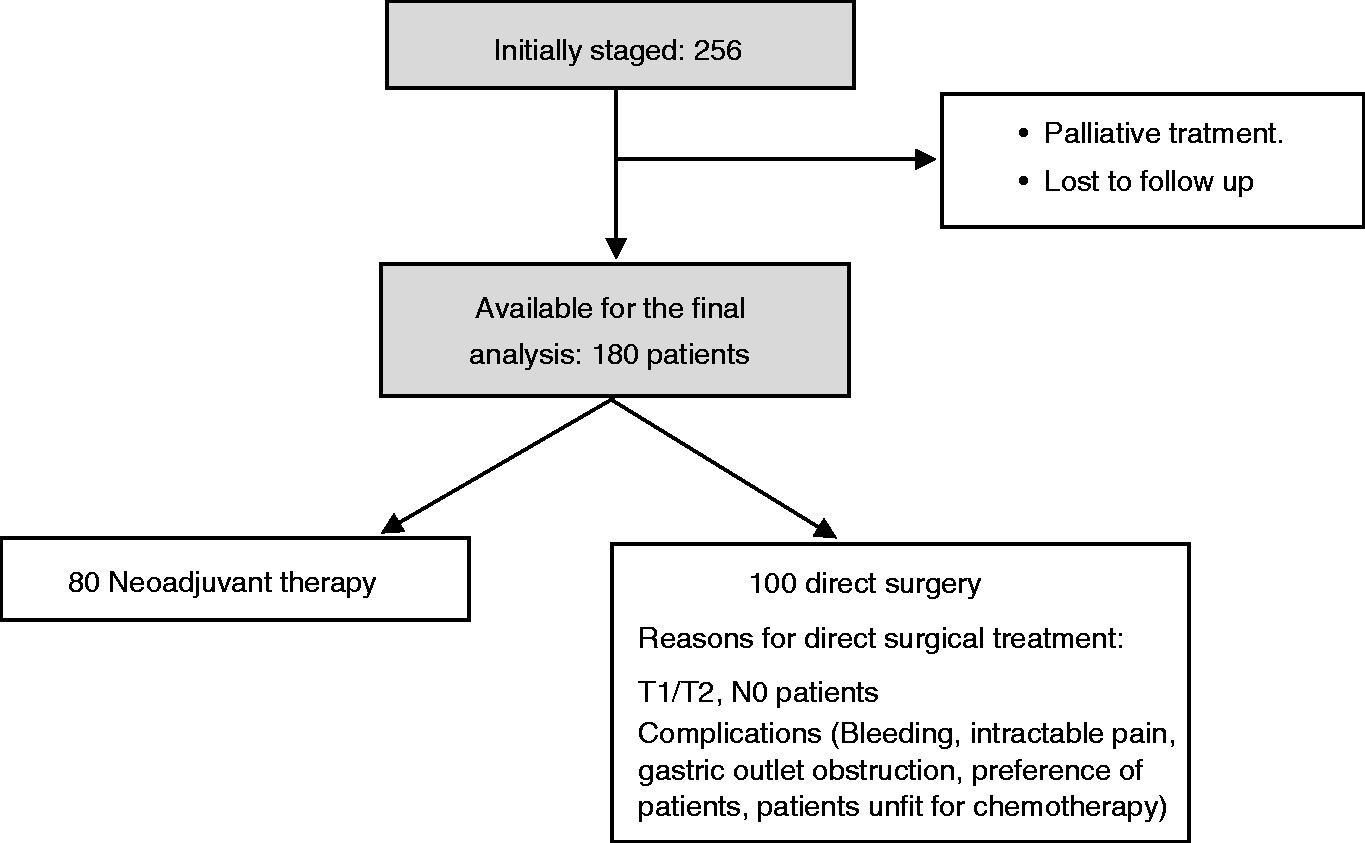
EUS
All EUS were performed by two experienced ultrasonographers (ER-C, JGM-C), who perform more than 300 procedures per year. We always began the staging with a radial echoendoscope (GF-UM160; Olympus Europe) reserving curvilinear echoedoscope for FNA (UCT-180-OL5; Olympus). Every patient received propofol sedation guided by the endoscopist and by a trained nurse.
A systematic complete US evaluation was performed in each patient. We began the EUS exam in the descending portion of the duodenum, except in patients with gastric outlet obstruction. We usually inspect the stomach during endoscope retrieval, after instilling water inside it and with the aid of a balloon over the transducer tip. Every structure and LN was studied. Also, mediastinal and upper abdominal LNs were systematically evaluated. Local tumor infiltration was established using the five-layer structure of the gastric wall. 14 EUS assessment of the N stage was based on the number of metastatic perigastric LNs. A lymph node metastasis was determined using two or more of the following criteria: (1) size greater than 5 mm, (2) round shape, (3) hypoechoic pattern and (4) smooth border. 15 For TNM staging we used the seventh International Union Against Cancer (UICC) classification. 16 When a complete examination was impossible because of a significant stenosis, we staged the areas at the reach of the echoendoscope, explaining our limitations in the endoscopic report.
PET-CT imaging
All patients fasted for at least four hours before the fluorodeoxyglucose (FDG)-PET-CT study. The blood glucose concentration was measured before FDG administration, and the study was performed only if the glucose concentration was <200 mg/dl. All FDG-PET-CT imaging was performed with a hybrid PET-CT scanner (Biograph LSO 2; Siemens Medical Solutions, Malvern, PA, USA). The CT component of the PET-CT studies was performed without administration of an intravenous (iv) contrast agent when risk factors of contrast-induced nephropathy were suspected. CT images (5 mm slices) were obtained from the base of the skull through the proximal thighs at 130 kVp and 110 mA. Emission PET images were obtained over the same anatomic extent beginning 45 to 60 minutes after the administration of 15 to 20 mCI FDG, with imaging times of two to four minutes per bed position, depending on patient weight. PET-CT images were reviewed for abnormal FDG uptake at the primary tumor site, LN regions, and distant sites. The radiologist provided information about N staging but information about T staging was not usually available, because of the low local accuracy previously experienced in this area. Only in obvious cases of other organs invasion (cT4b) was this information added to the radiology report.
Data collection
Information regarding patients’ demographic data, comorbidities, current medications, clinical presentation, laboratory tests, and endoscopic findings was collected. Interventions were recorded, including the procedures performed for staging, neoadjuvant therapy, and surgery.
Data analysis
Statistical analysis was carried out using the software PAWS Statistics 17.0 (SPSS Inc, Chicago, IL, USA). The results of preoperative staging using EUS and PET-CT were compared to the postoperative histological diagnosis. The chi-square and the Kappa tests were used to evaluate the consistency between the EUS and histopathological staging of gastric cancer. Chi-square and Fisher exact tests were used to compare EUS and PET-CT as appropriate. A
Ethics
The local ethics committee had previously approved the protocol and the database of our study. Every patient signed the informed consent for each procedure and to be included in the database. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Results
Patient characteristics
Patient characteristics.

EUS: endoscopic ultrasonography; PET: positron-emission tomography.
Staging and restaging
Accuracy of EUS and PET vs histology.
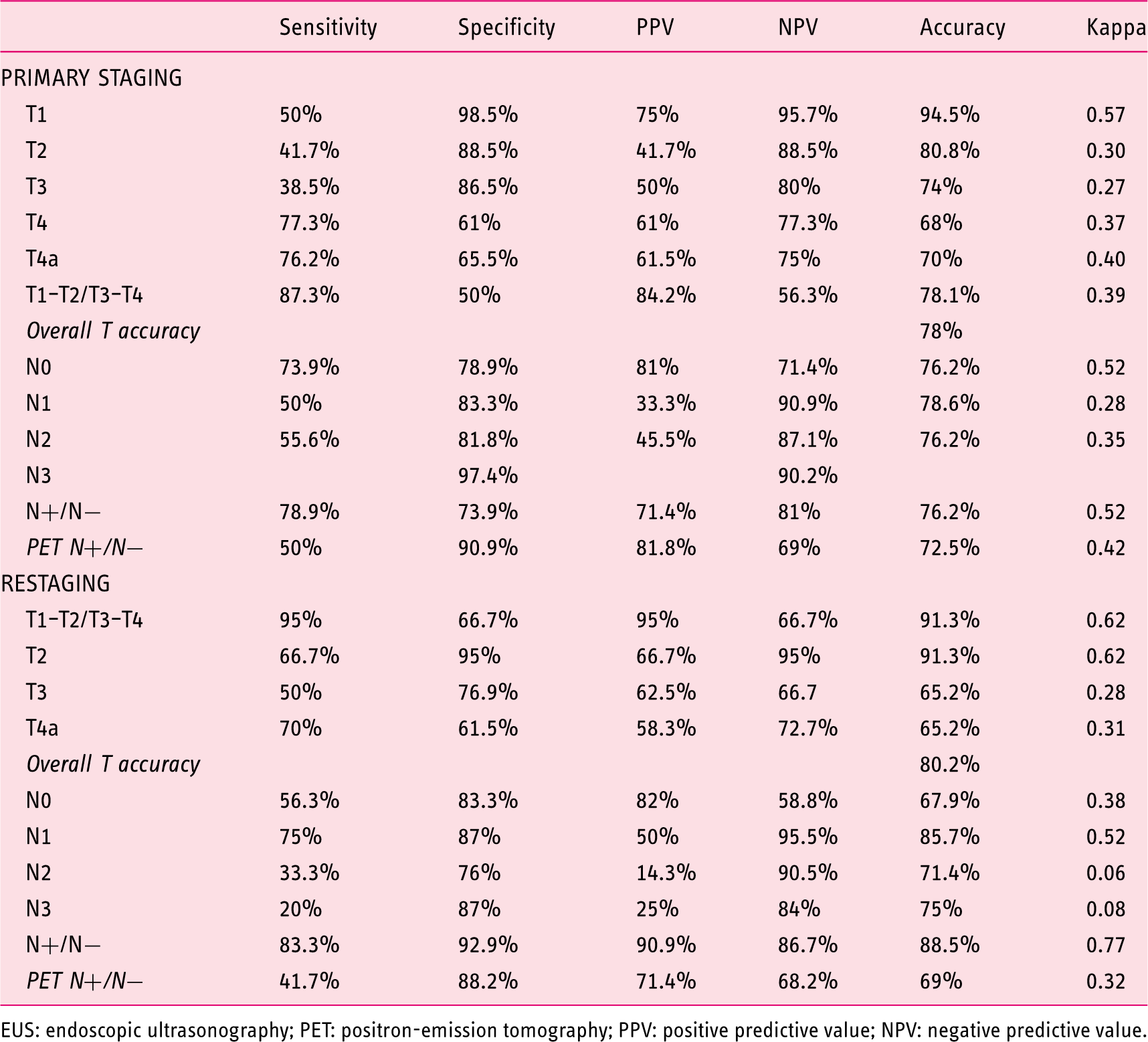
EUS: endoscopic ultrasonography; PET: positron-emission tomography; PPV: positive predictive value; NPV: negative predictive value.
When considering the accuracy of EUS for different categories, it was always better when distinguishing T1–T2 tumors vs. T3–T4 tumors. However, it showed an added advantage in restaging, with a higher sensitivity and specificity than for primary staging.
Nineteen patients not submitted to neoadjuvant therapy were overstaged in T stage and 15 in N stage. Understaging was observed in 14 patients regarding T stage and in 18 patients regarding N stage.
Considering LN involvement, although we follow the TNM classification in our daily practice, we only considered for this study purpose the positivity or negativity of the LNs explored in each case, as it is the main issue when making decisions about patient management.
10
The accuracy of EUS was 76.2%, very similar to what was observed for PET-CT (72.5%), but with statistical differences (
When studying the performance of both techniques in restaging we found that EUS had better performance when considering the main staging differences that can change patients’ management. In this regard, the accuracy in distinguishing T1–T2 tumors vs. T3–T4 tumors was 91.3%, better than in the first staging. Indeed, similar results were found for N staging with an accuracy of 88.3% for N-positive vs. N-negative distinction. By contrast, PET-CT showed an accuracy of 69% for LN involvement when restaging gastric cancer, inferior to what was found for EUS and for the initial staging (
Discussion
Our results, obtained from real clinical practice, showed that the overall accuracies of EUS and PET-CT for preoperative N staging were 76.2% and 72.5%, with significant differences between both techniques. The overall accuracy of EUS for T staging was 78% and 80.2% for restaging. More important, our results show a significant advantage of EUS over PET-CT in restaging, even in our series, in which the vast majority of suspicious LNs were not sampled.
Similarly to what has been described in a previous meta-analysis, EUS can be considered clinically relevant with regards to T staging because it offers important information for the distinction between early to intermediate (T1–T2) and advanced (T3–T4) primary gastric tumors. Our study confirms these findings, showing a much better performance in the diagnosis of advanced tumors than the early ones, in which the specificity was poor. However, when each T stage was independently considered, accuracy was better for T1 and T2 stages (Table 2).
Following the National Comprehensive Cancer Network (NCCN), 9 the most relevant data in gastric cancer staging are the difference between T1–T2/T3–T4 and LN positivity. It is obvious that visceral metastases are better addressed by PET-CT, even more when, as it happens in our hospital, an MDCT as part of the PET-CT. This has been previously showed in other reports, 17 and was not a study goal.
Regarding N staging, our results show a better performance of EUS than PET-CT, with significant differences, something that has already been described but with fewer differences between both procedures observed in previous reports. 18 We have not performed FNA in most of the cases because we usually find technical difficulties with regards to primary tumor interposition and because in the evolving of our practice we have found an increasing reliability of EUS on malignant characters when a primary tumor is present. 19 Thus, only with the endosonographic characteristics of the LNs was EUS able to improve PET-CT results. The lack of a greater difference, which was, indeed, achieved in a previous study in which this topic was addressed, may be explained because of the higher rate of FNA performed by those authors. 18 However, our data are very similar to what has been reported by highly experienced groups. 20 Indeed, previous reports have shown no differences in N staging between EUS and CT.17,21–23 Yet, fewer studies have compared EUS and PET-CT, in which the sensitivity of FDG-PET alone for N staging was generally low.11,24,25 Moreover, in another paper 26 the technical advances achieved by PET-CT have also failed to offer clear advantages. Indeed, even for metastatic disease, the role of PET-CT is still to be confirmed. 8
Nevertheless, in our view, the most important result of our series is the accuracy in restaging. EUS has offered little value in this setting, adding to the workup only a prognosis improvement in patients in whom downstaging was observed.
13
By contrast, our results, which come from a bigger series than the previous one by Park et al.,
13
offer a higher accuracy in N staging, near 90% for the detection of positive LNs, and significantly better than PET-CT. Except for N0, for which EUS precision was lower, accuracy for the rest of the stages was high, over 70% in all of them. When comparing to PET-CT, EUS was significantly more accurate for the detection of positive LNs, something previously not described and that supports the use of this procedure in every restaged patient before surgery. Moreover, EUS accuracy was higher in this setting to that observed in the previous paper by Park et al.,
13
and this could be explained by the technology differences between their series and ours. Indeed, our echoendoscope is the Olympus GF-UE160-AL5, which compared to the GF-UM-200 used by Park et al. offers a better imaging quality. Another reason for our relatively high restaging accuracy could be the amount of previous information provided from the previous PET-CT and EUS that offers the endosonographer a highly precise map of the former disease extension that could have led to a performance bias, which is somewhat likely in a clinical series like ours
Nevertheless, we still have to acknowledge that the accuracies of either EUS or PET-CT are suboptimal. This leads to a considerable proportion of patients undergoing wrong, burdensome and even life-threatening therapies. In this particular aspect we agree with most of the studies previously published, and probably more than one single method should be used for staging and restaging gastric cancers.2–4,13,17
We acknowledge the limitations of this study: First, it was developed in a single institution, an academic center with all the procedures performed by two experienced endoscopists and therefore results might not be applicable to other settings; second, few LNs were sampled by FNA because of technical difficulties; third, in patients with kidney disease or high risk of contrast-induced nephropathy, radiologists did not use iv contrast in the CT scan performed with the FDG-PET because they didn’t considered it essential in their protocol, and this could have limited their accuracy.
In conclusion, EUS and CT scan accuracies can still be considered reasonable, but not perfect, and both methods should be offered to every patient for gastric cancer staging. The addition of PET to CT does not offer additional information that could justify its routine use for this condition.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
