Abstract
The European Society of Gastrointestinal Endoscopy (ESGE) and United European Gastroenterology present a short list of key performance measures for endoscopic ultrasound (EUS) and endoscopic retrograde cholangiopancreatography (ERCP). We recommend that endoscopy services across Europe adopt the following seven key and one minor performance measures for EUS and ERCP, for measurement and evaluation in daily practice at centre and endoscopist level:
This present list of quality performance measures for ERCP and EUS recommended by the ESGE should not be considered to be exhaustive; it might be extended in future to address further clinical and scientific issues.
Introduction
The European Society of Gastrointestinal Endoscopy (ESGE) and United European Gastroenterology (UEG) have identified quality of endoscopy as a major priority. The rationale for this priority and the methodology of the quality initiative process have been described elsewhere. 1 The aim of the ESGE pancreatobiliary endoscopy working group was to identify a list of key performance measures for endoscopic ultrasound (EUS) and endoscopic retrograde cholangiopancreatography (ERCP) that would be universally applicable. As with previous ESGE performance measures,2,3 the focus was on metrics that met the following requirements: proven impact on clinically relevant outcomes or quality of life; well-defined, and amenable to simple and robust measurement; and applicability to all levels of endoscopy services. This paper describes the methodological process utilized 1 and reports the agreed list of key performance measures for pancreatobiliary endoscopy.
Methodology
The multistep process of the methodology for developing performance measures has been described previously. 1 During initial meetings of the working group, a PICO approach (where P stands for population/patient, I for intervention/indicator, C for comparator/control and O for outcome) was used to define clinically relevant questions. Systematic literature searches were then performed by an expert team of methodologists. This in turn led to the development of performance measures in a consensus process.
The PICOs and the clinical statements derived from these were modified or excluded during iterative rounds of discussion of the working group members during a Delphi process. 4
In total, working group members participated in two rounds of voting to agree on performance measures in predefined domains and on their respective thresholds, discussed below. Statements were modified during the process and ultimately discarded if agreement was not reached after two voting rounds. The agreement that is given for the different statements refers to the last voting round in the Delphi process. The threshold for agreement was set at 80% throughout the process. The key performance measures were distinguished from minor performance measures on the basis of the ISFU criteria 1 (importance, scientific acceptability, feasibility, usability and comparison with competing measures) and expressed by mean voting scores. We used the grading of recommendations assessment, development and evaluation (GRADE) system to assess the quality of the available evidence. 5
Performance measures for pancreatobiliary endoscopy
Using the evidence derived by the literature search group and input from the working group members, a total of 10 clinical statements addressing 8 potential performance measures, grouped into five of the seven predefined quality domains, were formulated. Over the course of two voting rounds, a consensus agreement was reached for eight statements regarding eight performance measures; seven are considered to be key performance measures and one a minor performance measure. The development process for performance measures can be reviewed in the Supporting information (available online).
We used the highest mean voting scores to identify seven key performance measures for five of the seven quality domains (Figure 1). As mentioned above, the remaining performance measure was considered to be a minor performance measure. The pre-procedure domain and management of pathology domain each had two performance measures. All performance measures were deemed valuable by the working group members and were obtained after a rigorous process, as described above. The use of appropriate endoscopy reporting systems is crucial for facilitating data retrieval on identified performance measures.
6
The domains and performance measures chosen by the pancreatobiliary working group. EUS-FNA: endoscopic ultrasound-fine needle aspiration; ERCP: endoscopic retrograde cholangiopancreatography; PEP: post-ERCP pancreatitis; N/A: not available.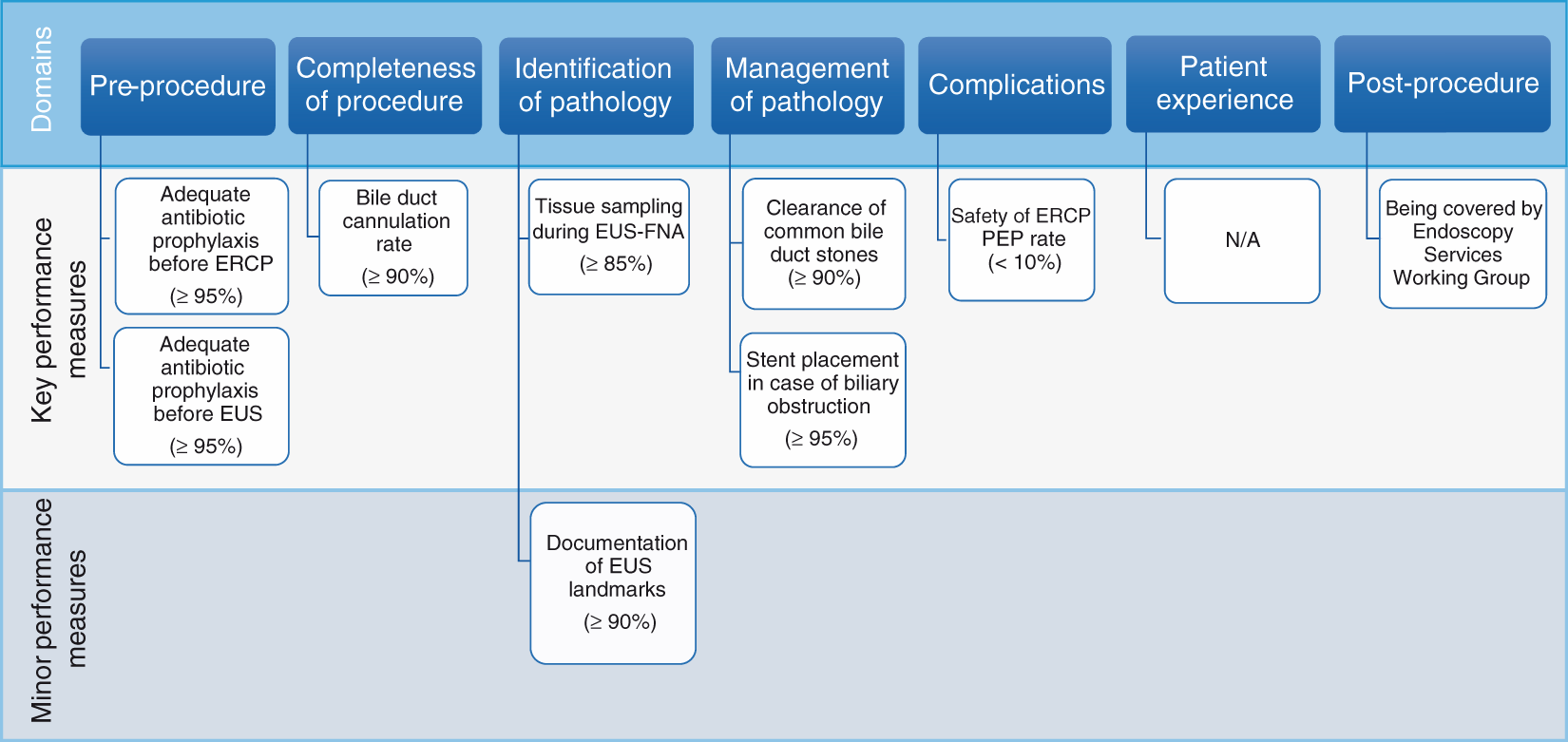
All the performance measures are presented below, according to domain, using the descriptive framework developed by the quality improvement committee (QIC) and with a short summary of evidence for the ISFU criteria. Each table describes a performance measure, the level of agreement during the modified Delphi process (scores), how the performance measure should be calculated, and recommendations supporting its adoption. The tables also note the desired thresholds.
The minimum number needed to assess whether the threshold for a certain performance measure has been reached can be calculated by estimating the 95% confidence intervals (CIs) around the predefined threshold for different sample sizes.3,7 As with previous ESGE performance measures, for issues of practicality and to simplify implementation and auditing, we suggest that at least 100 consecutive procedures (or all of them if fewer than 100 procedures are performed) should be measured to assess a performance measure. However, continuous monitoring is the preferred method of measurement.
1 domain: pre-procedure
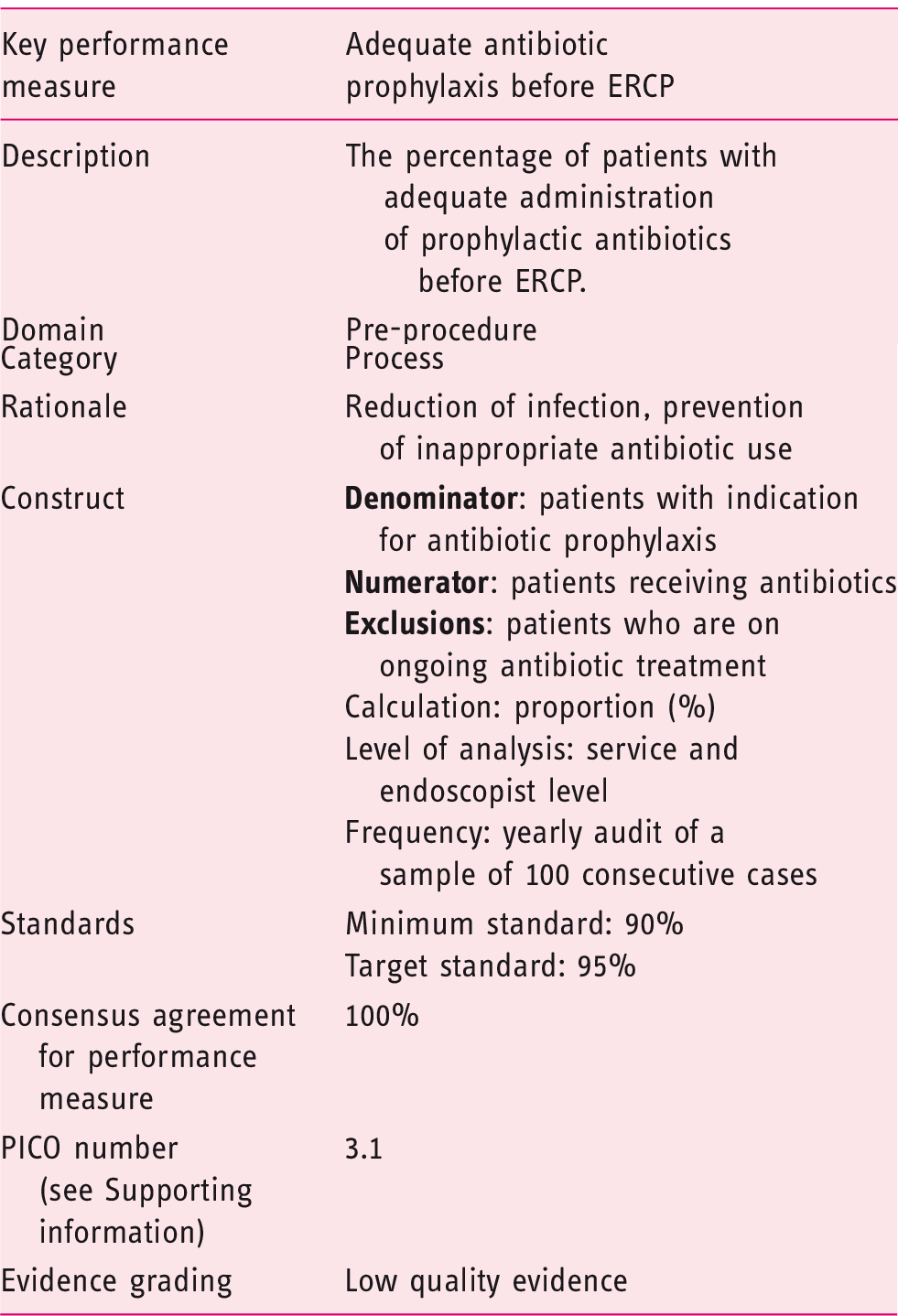
The acceptance of this performance measure is based on agreement with the following statement:
Routine antibiotic prophylaxis is not recommended for ERCP in unselected patients. Antibiotic prophylaxis should be given before ERCP for the subgroup of patients with predicted incomplete biliary drainage, e.g. those with primary sclerosing cholangitis (PSC) and hilar tumours, to immunocompromised individuals and to patients with pancreatic pseudocysts communicating with the pancreatic duct (Statement number 7.2).
Adherence to recommendations on prophylactic antibiotics before ERCP 8 should be monitored and reasons for deviation documented. The indication for antibiotic prophylaxis should be recorded in the endoscopy report.
Routine antibiotic prophylaxis is not recommended for ERCP in unselected patients as prophylactic antibiotics do not significantly reduce cholangitis in this setting. A systematic review of randomized controlled trials (RCTs) 9 reported that antibiotics did not significantly prevent cholangitis in unselected patients.
A Cochrane systematic review of RCTs 10 concluded that prophylactic antibiotics reduced cholangitis; however, in patients in whom biliary obstruction was relieved, there was no benefit in using prophylactic antibiotics.

The acceptance of this performance measure is based on agreement with the following statement:
Prophylactic antibiotic administration should be performed before EUS-guided puncture of cystic lesions in ≥ 95% of cases (Statement number 8.1).
The percentage of patients with administration of prophylactic antibiotics before EUS-guided puncture of cystic lesions should be at least 95% (minimum standard). In general, antibiotic prophylaxis should be used; the reason for any deviation (patient intolerance, patient preference etc.) should be reported.
The rate of infectious complications following EUS-guided puncture of cystic lesions is low.11,12 There are no systematic reviews or RCTs comparing antibiotics with no antibiotics before EUS-guided puncture of cystic lesions, although one study compared two regimens of antibiotics, 13 and two retrospective cohort studies14,15 focused exclusively on pancreatic cystic lesions. However, the study by Kwok and colleagues, 13 in which 117 patients were screened over an 11-month period, lacked statistical significance since only 22% of screened patients could be enrolled. The observed rate of cyst infection was zero. An adequately powered study to test noninferiority of withholding antibiotics in this setting would likely be logistically challenging since the authors calculated that inclusion of between 614 and 2450 patients would be needed. Current ESGE 16 and American Society for Gastrointestinal Endoscopy (ASGE) 8 guidelines recommend the use of prophylactic antibiotics for the EUS-guided puncture of cystic lesions, although data are equivocal. 14 In addition, the use of prophylactic antibiotics might not be free of adverse events.
2 domain: completeness of procedure

The acceptance of this performance measure is based on agreement with the following statement:
In patients with normal anatomy and native papilla, bile duct cannulation should be achieved in at least 90% of cases using all available techniques (Statement number 1.1).
Technical success at biliary ERCP is predicated on successful deep cannulation of the desired duct. Success or failure of cannulation should be documented in the post-procedure report for all cases. In certain clinical scenarios, e.g. pyloric or duodenal stenosis and post-surgical altered anatomy, conventional ERCP may be impossible and such cases are not included in this performance measure. In addition, patients with prior sphincterotomy should not be included in the calculation of cannulation rate. There are a number of potential determinants of successful cannulation of a native papilla, including endoscopist experience and case mix. The literature predominantly reports outcomes from academic centres, where case mix and experience may differ from other settings. The included studies reported cannulation rates from 70.5 to 100%,17–43 with a median of 96% and mean of 91.4%. The consensus of the working party was that a competent ERCP practitioner should achieve a cannulation rate in excess of 90% with a target standard of 95% at expert centres. ESGE guidance on different techniques is available. 44
During the voting process (second voting round), members of the pancreatobiliary working group discussed whether this performance measure (bile duct cannulation rate) should be extended and be adopted to both duct systems in the pancreatobiliary system, the common bile duct and the pancreatic duct, by stating ‘cannulation rate of desired duct’. However, to our knowledge, there are no data that would support adopting such a performance measure.
3 domain: identification of pathology

The acceptance of this performance measure is based on agreement with the following statement:
In patients with solid lesions undergoing EUS-FNA, the frequency of obtaining a full diagnostic tissue sample should be ≥ 85% (Statement number 5.1).
The percentage of patients in which a full diagnostic tissue sample, meaning a tissue sample allowing an accurate diagnosis, is obtained in EUS-FNA of solid lesions should be documented. The frequency of successful EUS-FNA of a solid lesion should be at least 85% (minimum standard); the ESGE proposes a target standard of 90%.
Since the evidence is of very low quality, this recommendation is to be considered as expert opinion. Although the evidence is scarce as regards the available literature,45–56 we consider the clinical issue of successful tissue sampling to be a major element in EUS. Based on the impact of EUS-fine needle puncture, whether performed as aspiration (FNA) or biopsy (FNB), we feel that this clinical quality indicator must be used as a key performance measure.

The acceptance of this performance measure is based on agreement with the following statement:
Appropriate landmarks should be documented in ≥ 90% of cases in patients undergoing EUS (Statement 6.1).
The components of a complete EUS investigation will vary depending on the indications for the procedure. However, in many cases, the visualization and documentation of standardized landmarks give a measure of the quality of the procedure. Documentation of the appropriate landmarks includes detailed description in the patient record of the endosonographic findings of the EUS procedure and, ideally, procedure quality will be enhanced by image documentation of normal or diseased landmarks. Such reporting forms the basis of the quality indicator. Although EUS is not indicated for staging of metastatic tumours, which might have been previously documented by other imaging modalities, there are clinical settings in which EUS may be indicated nevertheless, for example if therapeutic decision making is based on EUS findings, or if EUS-FNA is used to obtain a full diagnostic tissue sample (see domain above, identification of pathology), which may change the further management of the patient.
Landmarks to be assessed at endoscopic ultrasound according to the indication for the procedure.
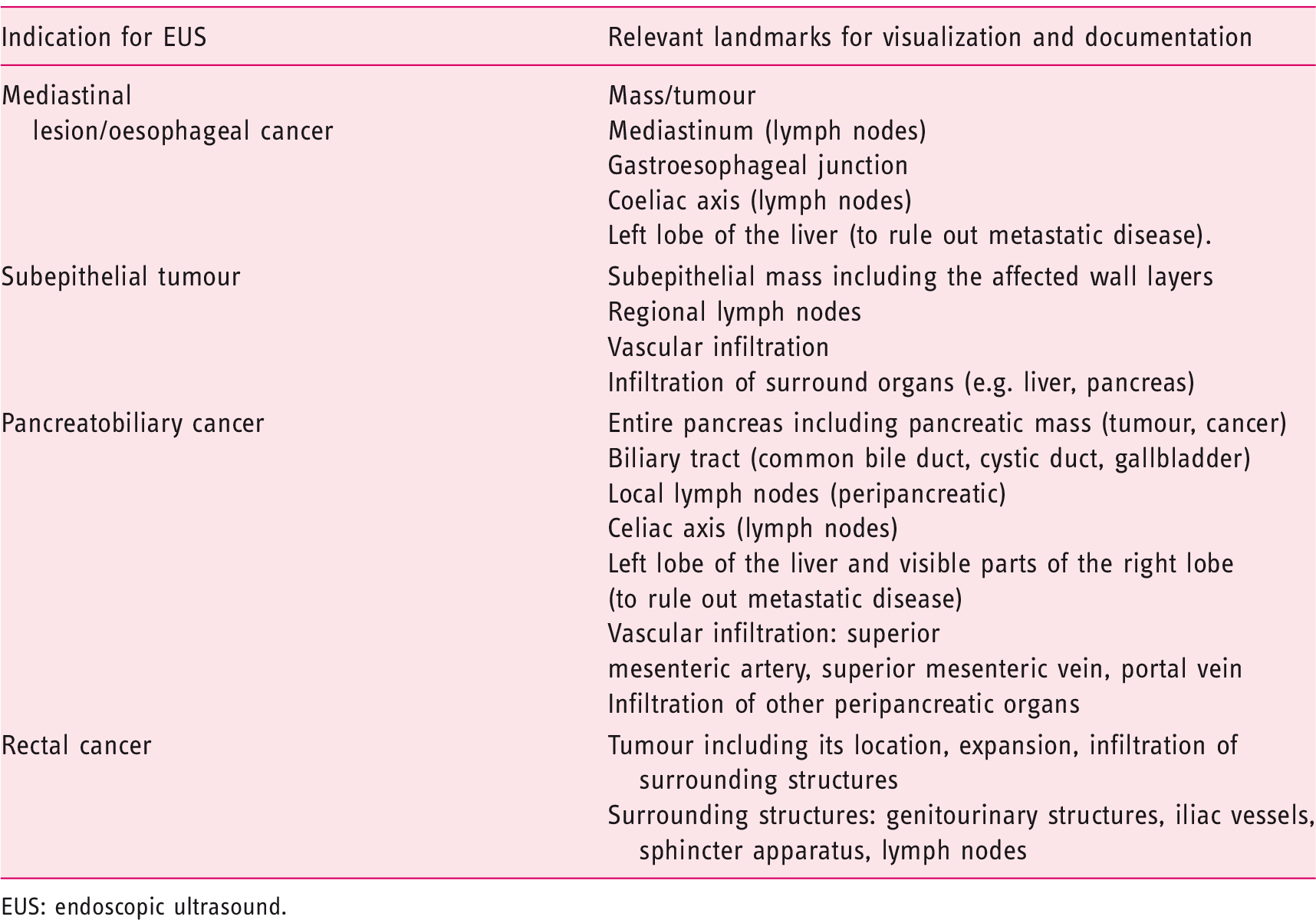
EUS: endoscopic ultrasound.
In 2015, an ASGE–American College of Gastroenterology task force published a work on quality indicators for EUS. 58 The authors stated that inclusion of the indication for EUS in the procedural documentation for all cases is a useful quality measure for two reasons. First, it may provide a justification for the procedure, serving as a means of tracking compliance with accepted indications. Second, the indication puts the procedure report into a context wherein reporting of certain EUS landmarks and finding characteristics should logically follow. For example, a detailed description of the pancreatobiliary system may not be necessary when the indication for EUS is staging of oesophageal cancer. If the indication for the EUS examination is staging of oesophageal cancer, certain landmarks should be included (uT-stage and uN-stage, including coeliac axis visualization). The exception to this is in the case of failed passage of a stenosed stricture when the tumour cannot be safely passed.
4 domain: management of pathology

The acceptance of this performance measure is based on agreement with the following statement:
After successful cannulation, stent placement should be achieved in ≥ 95% of cases in patients with biliary obstruction below the hilum (Statement number 3.1).
This statement refers to placement of plastic or metal stents. Subhilar strictures are the type most commonly encountered in daily practice. Stent placement in patients with obstruction below the hilum is technically less challenging than placement for obstruction at or above the hilum, with high success rates reported.59,60
Indications include failure to clear bile duct stones, and the presence of biliary strictures of benign or malignant origin. Competent ERCP practitioners should achieve successful subhilar stent placement in at least 95% of cases.

The acceptance of this performance measure is based on agreement with the following statement:
After successful cannulation, clearance of bile duct stones < 10 mm should be achieved in ≤ 90% of cases (Statement number 2.1).
The endoscopy report should provide details about the size, number and position of stones in the bile duct, and whether they were successfully cleared from the duct. All relevant findings, such as the presence of a stricture, should also be recorded.
A range of techniques and devices, including balloon/basket extraction, balloon dilation of the ampulla and mechanical lithotripsy, are available for clearance of stones from the bile duct with high success rates reported for stones smaller than 10 mm in size.61,62 Competent ERCP practitioners should be able to achieve a duct clearance rate in excess of 90%.
5 domain: adverse events and harms
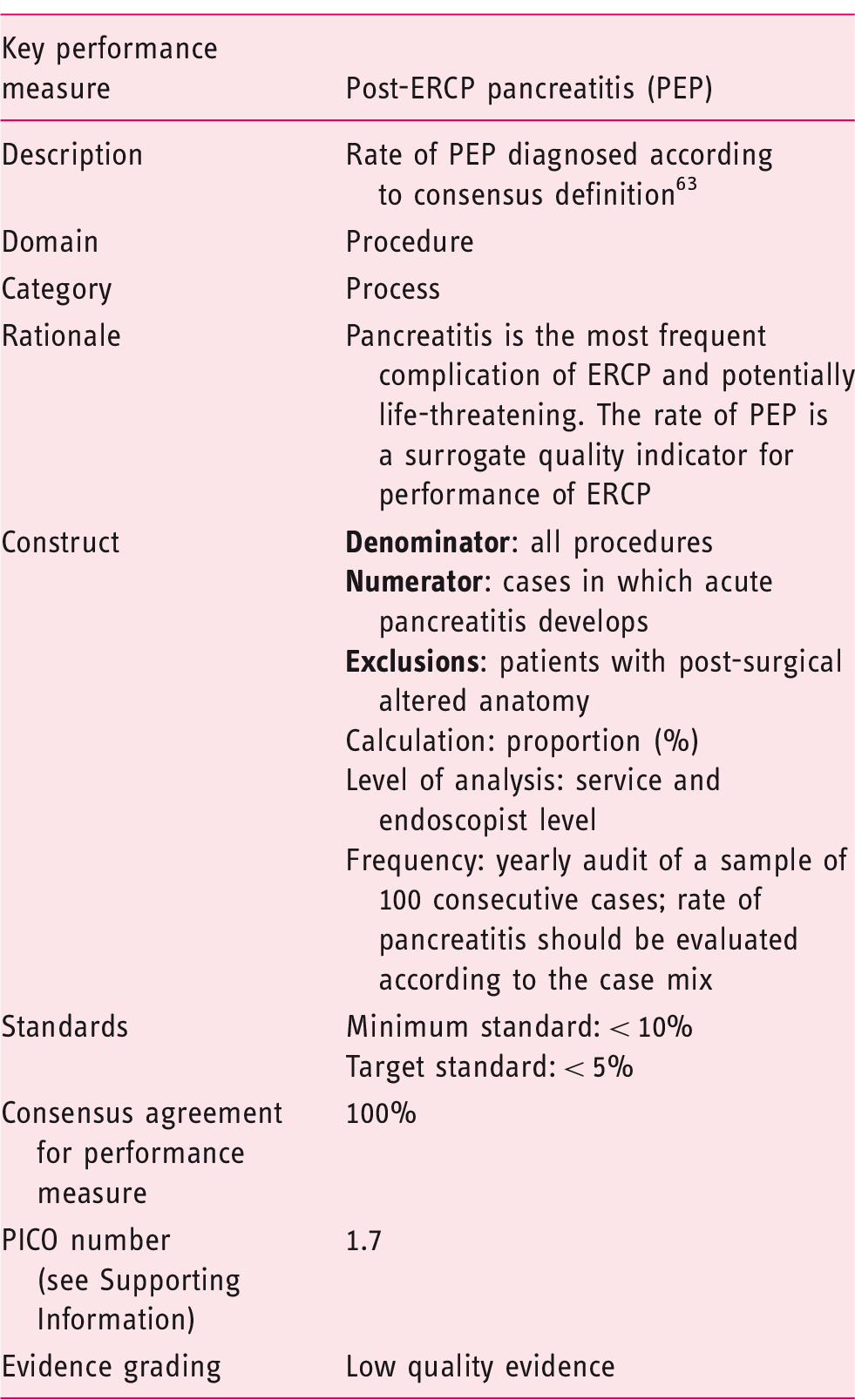
The acceptance of this performance measure is based on agreement with the following statement:
The rate of post-ERCP pancreatitis should be < 10% (Statement number 4.1).
PEP is the most common adverse event following ERCP and is therefore the most appropriate indicator of adverse event rate. There are a number of well-recognized risk factors, including female sex, normal bilirubin and previous PEP. A recent systematic review of RCTs documented an overall PEP rate of 9.7%, with a rate of 14.7% in high risk patients. 64 Large observational studies have reported rates of between 2.7 and 5.1%.65–68 A minimum standard of < 10% adverse event rate (pancreatitis) is therefore recommended, with a target standard of 5%. At audit, the rate of pancreatitis should be evaluated in terms of case mix. ESGE recommends PEP prophylaxis using rectal nonsteroidal anti-inflammatory drug (NSAID) administration for all patients in whom a contraindication does not exist, and consideration of placement of pancreatic duct stents in high risk cases. 69 The working group suggests the documentation of use of rectal NSAIDs and prophylactic pancreatic duct stenting, to facilitate root cause analysis in severe cases of pancreatitis and to investigate reasons why this performance measure might not be reached.
General conclusions, research priorities and future prospects
Research priorities identified by the pancreatobiliary working group for quality improvement performance measures: endoscopic retrograde cholangiopancreatography.

ERCP: endoscopic retrograde cholangiopancreatography; NSAIDs: nonsteroidal anti-inflammatory drugs.
Research priorities identified by the pancreatobiliary working group for quality improvement performance measures: endoscopic ultrasonography.

ERCP: endoscopic retrograde cholangiopancreatography; EUS: endoscopic ultrasonography: FNA: fine needle aspiration; FNB: fine needle biopsy; GI: gastrointestinal; MRCP: magnetic resonance cholangiopancreatography.
This manuscript, like the other ESGE quality improvement papers, is a working document that will be used, it is hoped, by national member societies to determine which performance measures can feasibly be monitored in the setting of their countries and which measures are relevant. The first task now is to implement these new performance measures into endoscopy practice throughout Europe on a national basis. This is in order to determine the value of setting performance measures, to allow audit against such measures and, in the light of audit findings, to permit responsive adaptation of performance measures in the future.
The implementation of performance measures is important to identify services and individual endoscopists with lower performance levels. Obviously, there are no legal implications associated with the ESGE QIC initiative since these documents are not guidelines, but are rather guidance on how quality can be monitored for all aspects of GI endoscopy.
The aim of setting performance measures is to improve the quality of endoscopy, and we encourage individual endoscopists, as well as heads of endoscopy units, to implement these performance measures without delay. Since the techniques of ERCP and EUS belong to the most sophisticated endoscopic examinations, with a flat learning curve, performance measures should be put in place as soon as possible to monitor endoscopist and endoscopy unit performance. At a unit level, this may mean investing in hardware to accommodate a more efficient auditing process.
Through such feedback, measures can be taken to improve quality, to rise above the proposed minimum thresholds. This should not be considered as a ‘1984’-like scenario with the goal of penalizing specific endoscopists, but rather as a tool to improve patient outcomes, and provide training and assistance to endoscopists where needed. A second barrier may be the perceived financial implications of establishing a quality control system. The aim is to encourage hospital management to support the implementation of these performance measures in endoscopy services. We think that in an era where hospital accreditation is becoming more important, hospital administrations will be more inclined to support such actions.
Performance measures to be included in the future for quality improvement in endoscopic retrograde cholangiopancreatography and endoscopic ultrasonography.

ERCP: endoscopic retrograde cholangiopancreatography; EUS: endoscopic ultrasonography; NSAIDs: nonsteroidal anti-inflammatory drugs.
Supplemental Material
Supplemental material for Performance measures for endoscopic retrograde cholangiopancreatography and endoscopic ultrasound: A European Society of Gastrointestinal Endoscopy (ESGE) Quality Improvement Initiative
Supplemental Material for Performance measures for endoscopic retrograde cholangiopancreatography and endoscopic ultrasound: A European Society of Gastrointestinal Endoscopy (ESGE) Quality Improvement Initiative by Dirk Domagk, Kofi W Oppong, Lars Aabakken, Laszlo Czakó, Tibor Gyökeres, Gianpiero Manes, Peter Meier, Jan-Werner Poley, Thierry Ponchon, Andrea Tringali, Cristina Bellisario, Silvia Minozzi, Carlo Senore, Cathy Bennett, Michael Bretthauer, Cesare Hassan, Michal F Kaminski, Mario Dinis-Ribeiro, Colin J Rees, Cristiano Spada, Roland Valori, Raf Bisschops and Matthew D Rutter in United European Gastroenterology Journal
Footnotes
Supporting information
Acknowledgements
The authors gratefully acknowledge the contributions from the following: Dr Stuart Gittens, of ECD Solutions, for the development and running of the web platform; Iwona Escreet and all at Hamilton Services for project administrative support; the Scottish Intercollegiate Guidelines Network for hosting the critical appraisal module; and the Research Foundation-Flanders (FWO) for providing funding for Professor Raf Bisschops. UEG supplied co-funding and additional project governance to this endeavour.
Declaration of Conflicting Interests
C. Bennett owns and works for Systematic Research Ltd, and received a consultancy fee from ESGE to provide scientific, technical and methodological expertise for the present project (2014–2018). R. Bisschops has received speaker’s fees from Covidien (2009–2014) and Fujifilm (2013); speaker’s fee and hands–on training sponsorship from Olympus Europe (2013–2014); speaker’s fee and research support from Pentax Europe; and an editorial fee from Georg Thieme Verlag as co–editor of
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
In brief
The European Society of Gastrointestinal Endoscopy (ESGE) and United European Gastroenterology (UEG) present a short list of seven key and one minor performance measures for endoscopic ultrasound and ERCP. A systematic and scientifically based methodology was applied to substantiate the proposed measures with available evidence where possible. Adoption of these performance measures in all endoscopy services across Europe is recommended.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
