Abstract
Background
Desmoid tumours represent a major complication of familial adenomatous polyposis. Our aims were to study the factors associated with the development of desmoid tumours in familial adenomatous polyposis patients, and to describe presentation and management of desmoid tumours.
Methods and patients
We reviewed all patients with familial adenomatous polyposis followed at our institution between 1965–2013, with either identified adenomatous polyposis coli gene mutation, or a personal and family history suggesting adenomatous polyposis coli-related polyposis. Response to treatment of desmoid tumours was assessed by Response Evaluation Criteria In Solid Tumor (RECIST) criteria.
Results
A total of 180 patients with familial adenomatous polyposis were included with a median follow-up of 19 years since diagnosis. Thirty-one (17%) patients developed 58 desmoid tumours, a median (range) 4.7 (0.8–41.6) years after their diagnosis of familial adenomatous polyposis. The only factor significantly associated with occurrence of desmoid tumours was the type of surgery: 12 (12%) desmoid tumours in 104 patients treated by colectomy, versus 19 (25%) desmoid tumours in 76 patients treated by proctocolectomy, p = 0.027. The localisation of desmoid tumours was: mesenteric (n = 25), abdominal wall (n = 30) or extra-abdominal (n = 3). Nineteen patients underwent 36 surgical procedures for desmoid tumours. Recurrence occurred in 26 (72%) cases and the recurrence-free survival was 2.6 (95% confidence interval (CI), 0.2–5.9) years. Thirteen patients received 27 medical treatments over a median 14 months. Objective response was observed in four (15%) patients and the median progression-free survival was nine (95% CI, 1.1–16.9) months.
Conclusion
If confirmed, colectomy (versus proctocolectomy) should be performed in adenomatous polyposis coli-related familial adenomatous polyposis patients to avoid desmoid tumours. We show that there is a high prevalence of post-surgical recurrence and the low efficacy of available medical treatments for desmoid tumours.
Introduction
Familial adenomatous polyposis (FAP) is a hereditary condition transmitted in an autosomal dominant fashion, which is characterised by the appearance of numerous adenomatous polyps in the colon and rectum. This condition results from germline mutation of the adenomatous polyposis coli (APC) gene on chromosome 5 and accounts for 1% of cases of colorectal cancer. 1 For patients with FAP, progression to colorectal cancer is inevitable and cancer occurs at an average age of 39 years, 2 which turns surgical treatment into the first and only choice of therapy.
Desmoid tumours (DTs), from the Greek word desmos (tendon-like), are benign monoclonal fibroblastic proliferations arising in musculoaponeurotic structures. 3 They do not metastasise but can display aggressive biological behaviour characterised by infiltrative growth and a high recurrence rate after resection. They are relatively rare lesions representing <3% of all soft tissue tumours with a reported annual incidence of 0.2–0.5 per 100,000 persons.4–6 In contrast, the association of DTs and FAP is relatively frequent. According to Gurbuz et al., a patient with FAP is at a 852-fold increased risk of developing a DT compared with the general population. 7 The exact aetiology of DT development is not known, but several factors contribute to their development and growth, such as genetic predisposition,8–11 hormonal factors and trauma (i.e. surgery)7,10,12–14 which may partially explain the high proportion of DTs in patients with FAP. There are currently no national or international guidelines for the management of DTs, and no recommended first-line systemic treatment. However, some DTs exhibit self-limiting behaviour and show growth arrest or spontaneous regression and, therefore, a wait-and-see approach seems to be an appropriate option for these selected patients. Otherwise, therapy consists of surgery, radiotherapy and/or systemic approaches in various non-standardised combinations.15–24 Surgery is the main treatment strategy, but is associated with a high risk of recurrence and function loss especially for patients with mesenteric DT.
In this context, we therefore conducted a retrospective study to evaluate the frequency and risk factors associated with the development of DTs in FAP patients, describe presentation of DTs, and report outcomes after treatment of DTs.
Patients and methods
Patients with FAP
We retrospectively reviewed all FAP patients, identified from a national prospective database (clinical trial no.: NCT01987518), treated and regularly followed in our tertiary centre between 1965–2013. The medical records of all patients were reviewed carefully for data on demographics (age, sex, previous medical history), diagnosis and pattern of FAP lesions (number and stage of colorectal tumours, duodenal tumours), surgical procedure data (age at surgery, laparoscopy versus laparotomy, anastomosis type), pathology, complications and outcomes.
DTs
The diagnosis of DTs was done firstly by radiology, then confirmed by pathology for those who underwent surgery. No biopsy of DT was routinely recommended in this very specific situation of FAP. DT patients were divided into two groups according to tumour location: mesenteric DT and extra-mesenteric DT. Treatment was categorised as either surgery or systemic therapy or, if no treatment was received over the follow-up period, ‘wait and see'. For patients who underwent surgery for DTs, recurrence and the time to recurrence or last known status is reported, and recurrence-free survival (RFS) was calculated (from DT surgery to the date of DT recurrence or death from any cause). Systemic treatments were only prescribed in cases of DT progression and/or symptomatic DT, and when surgery was not an option. Responses to systemic treatments were evaluated according to the Response Evaluation Criteria In Solid Tumor (RECIST) criteria, 25 as complete response (CR), partial regression (PR), stable disease (SD) and progressive disease (PD). Progression-free survival (PFS) was calculated from initiation of each systemic treatment to the date of disease progression according to RECIST criteria or death from any cause, whichever occurred first.
Statistics
Categorical variables were expressed as percentages, and compared by the Chi-square test or with Fisher’s exact test. Unpaired parameters were calculated by means of t tests or analysis of variance (ANOVA) tests. Continuous variables were expressed as median with range. Occurrence of DT and survival curves (RFS and PFS) were assessed using Kaplan-Meier analysis. Results are given as median survival (95% confidence interval (CI)). A p value of <0.05 was considered statistically significant.
Results
Patient characteristics
Patient characteristics.
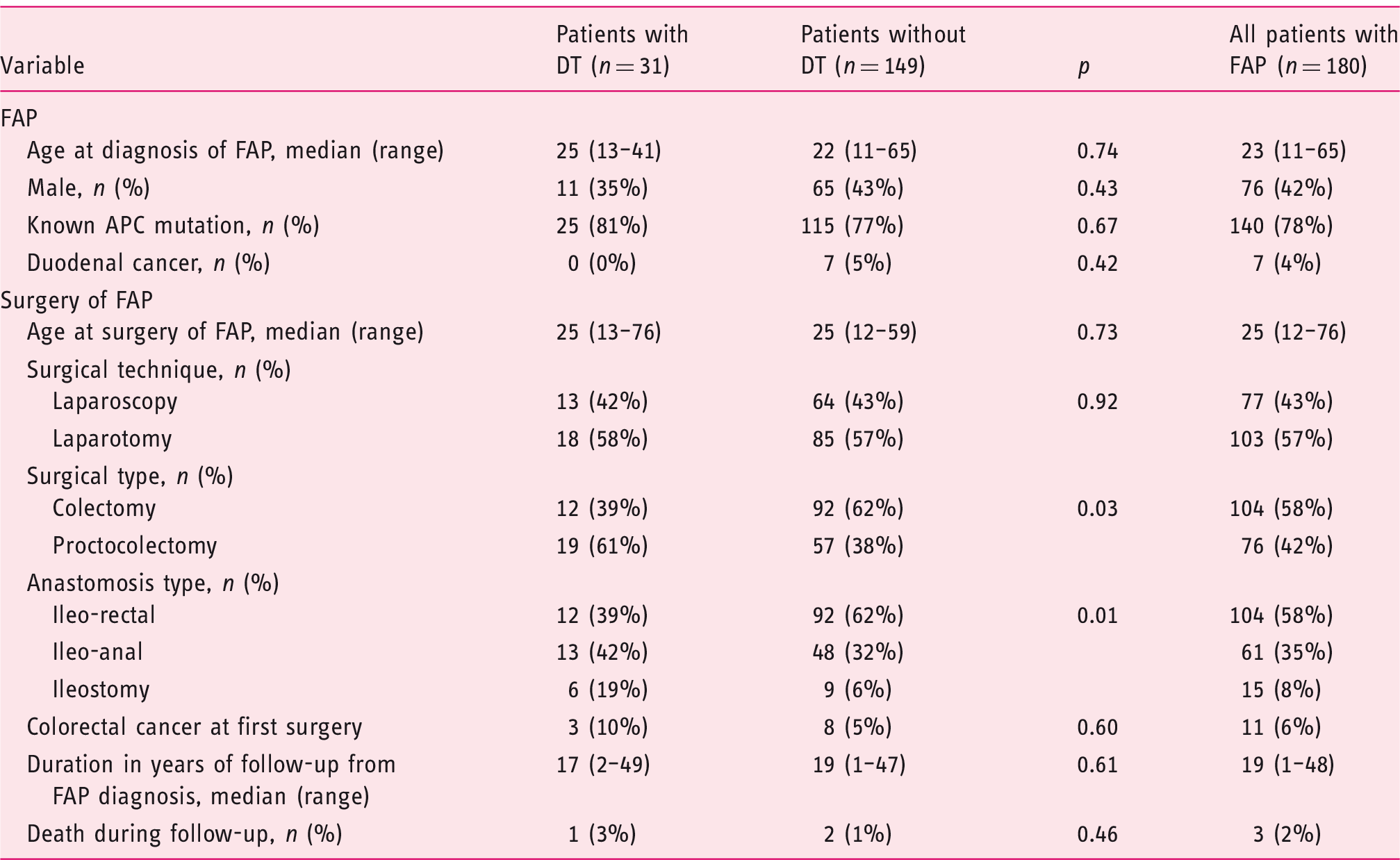
APC: adenomatous polyposis coli; DT: desmoid tumour; FAP: familial adenomatous polyposis.
Frequency and risk factors of DTs in FAP patients
During follow-up, 31 (17%) patients with a FAP developed at least one DT, after a median (range) 4.7 (0.8–41.6) years after colonic ± rectum surgery. Of the 104 patients who underwent a colectomy with rectal preservation, 12 (11% of those concerned) patients developed a DT, as compared to 19/76 (25%) patients who underwent a proctocolectomy (p = 0.027) (Table 1). Occurrence of DT in the 180 FAP patients over time, according to the type of surgical resection (colectomy versus proctocolectomy) is shown in Figure 1. The prevalence of DTs was 12/104 (12%) in patients with ileorectal (IR) anastomosis, 13/61 (21%) in patients with ileoanal (IA) anastomosis, and 6/15 (40%) in patients with terminal ileostomy.
Occurrence of desmoid tumours in familial adenomatous polyposis (FAP) patients during follow-up, according to the type of surgical resection (colectomy versus proctocolectomy).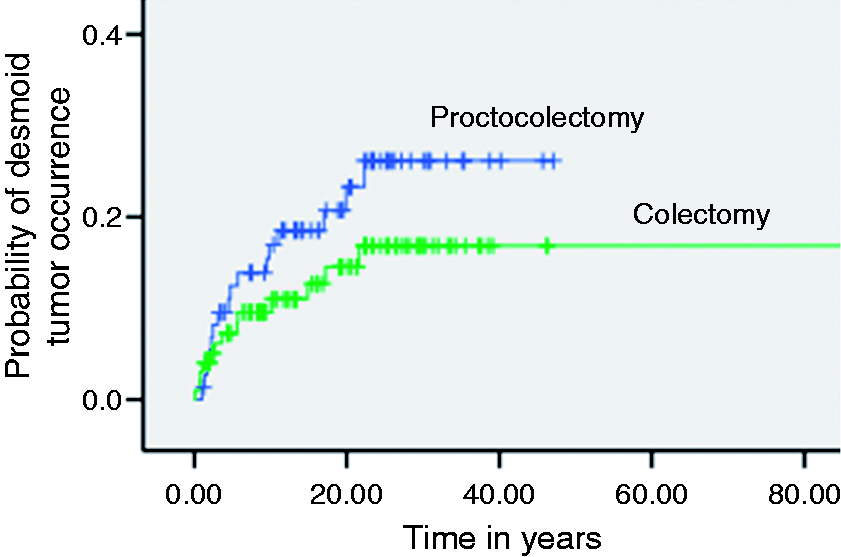
Presentation and management of DTs
DTs occurred in 31 FAP patients, 11 males and 20 females. Median (range) age at time of DT diagnosis was of 33 (16–69) years. A total of 58 DTs were diagnosed: 25 mesenteric and 33 extra-mesenteric DT (abdominal wall in 30, breast in two, and gluteal muscle in one). The median (range) DT size was 4.4 (1–25) cm. There was no significant difference between genders for age at DT occurrence (31 years for females versus 36 years for males, p = 0.37), mean number of DTs (1.6 for females versus 1.9 for males, p = 0.45), and median DT size (2.8 cm for females versus 4.3 cm for males, p = 0.19). The median (range) follow-up between diagnosis of DT to the last known status was nine (0.1–31) years. One death related to DTs was recorded (due to hepatic failure after embolisation of DT).
Three patients with eight (five mesenteric and three extra-mesenteric) DTs had stable disease during the entire follow-up period without any treatment (median duration 7.6 years).
Thirty-six surgical procedures were performed in 19 patients for 40 DTs (22 mesenteric, 10 extra-mesenteric, and four patients with both mesenteric and extramesenteric DTs); six of these patients were treated by systemic therapy (one mesenteric and five extra-mesenteric DTs) before surgery. After a median (range) follow-up of 9.8 (1.4–46.7) years subsequent to DT surgery, recurrence occurred in 26/36 (72%) cases, without significant difference between mesenteric DTs (77%) and extra-mesenteric DTs (50%), p = 0.12. The median (95% CI) recurrence-free survival (RFS) was 2.6 (0.2–5.0) years (Figure 2).
Recurrence-free survival (RFS) of 30 surgical procedures (33 desmoid tumours (DTs)) in 24 patients, according to the localisation of DTs (mesenteric versus extra-mesenteric DTs).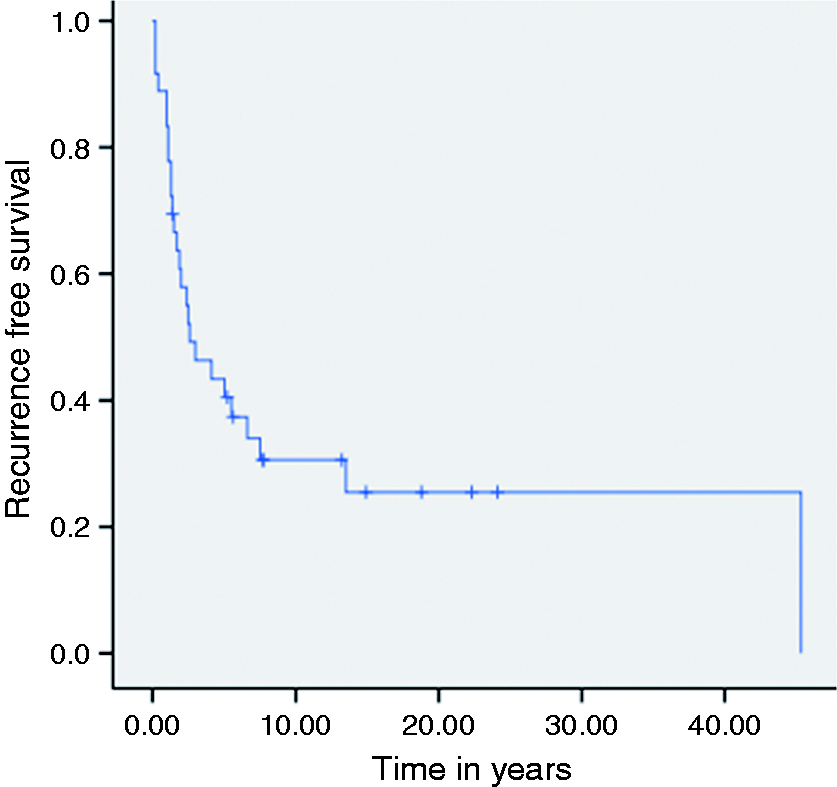
Response to the 27 systemic treatments of desmoid tumours given in 13 patients.

CI: confidence interval; RECIST: Response Evaluation Criteria In Solid Tumor.
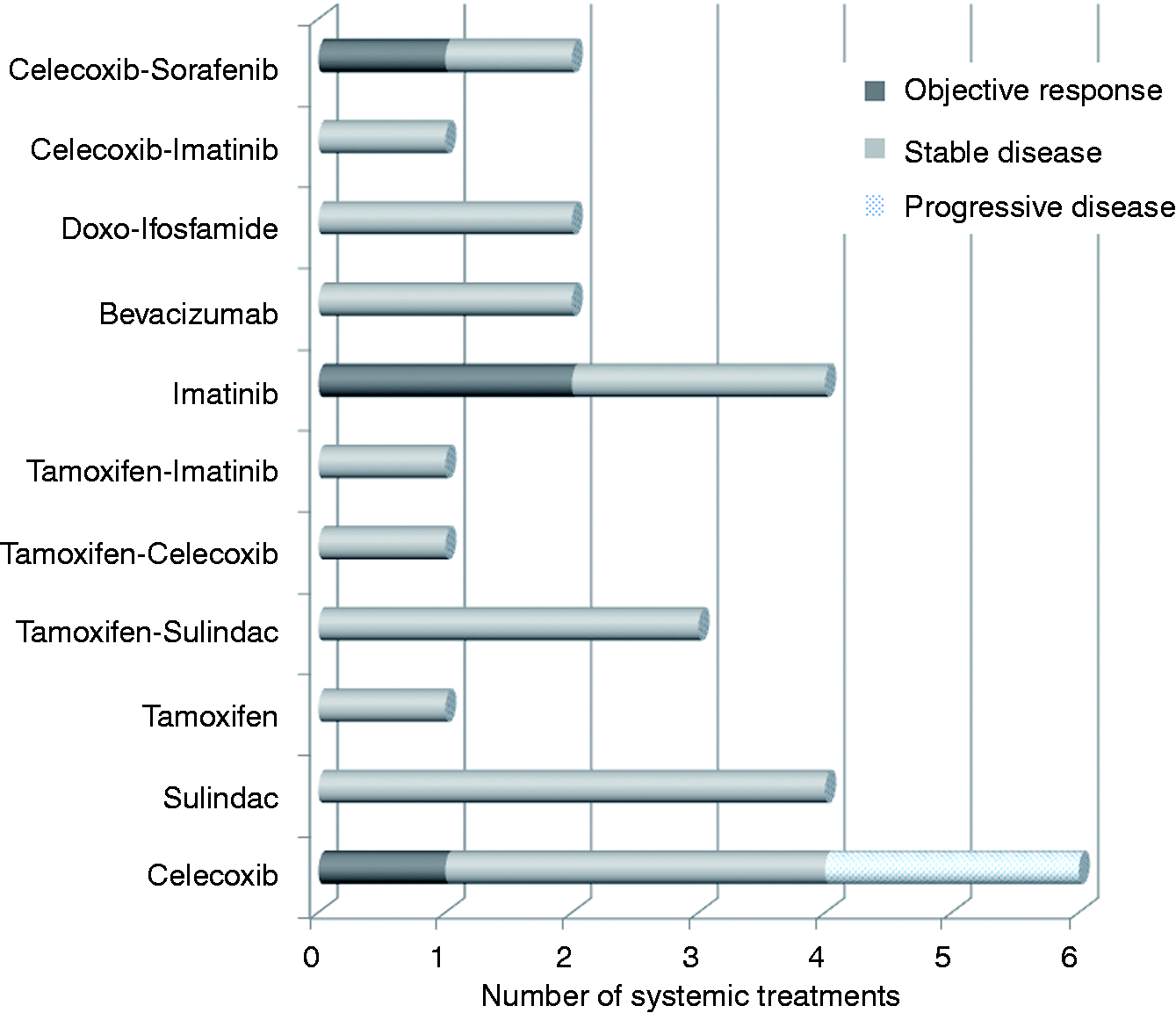
Efficacy with best responses assessed by Response Evaluation Criteria In Solid Tumor (RECIST) criteria of 27 systemic treatments given in 13 patients.
Discussion
In the present study, the prevalence of DTs was 17%, but may be slightly underestimated owing to the short postoperative follow-up of some young patients. It does, however, concord with that reported in the literature (9–26%), with more recent publications reporting higher prevalence.7,26–31
When comparing the characteristics of FAP patients with and without DTs, there was no significant difference found in sex, age at FAP diagnosis, age at colonic surgery, APC mutation rate or surgical technique (laparotomy versus laparoscopy). However, the main observation of this retrospective study, was a significant difference between the extension of colon and rectum resection and further development of DTs: there was a significant two-fold higher probability of DT occurrence among patients who underwent proctocolectomy as compared to those who underwent sub-total colectomy. It is also of note that patients harbouring a definitive ileostomy and proctocolectomy had the highest prevalence of DTs. The physiological basis for this difference needs further investigation but one hypothesis may be that extensive surgery increases local inflammation and favours the inflammatory process leading to the development of DTs. Interestingly, there was no risk difference between patients with laparotomy versus laparoscopy. Because of study limitations (size of the population and retrospective analysis), larger studies are needed to confirm this result. We are now running a national French database register (clinical trial no.: NCT01987518) that could help answer this important question. However, given the improved ability of endoscopic aggressive follow-up of the rectal stump to decrease the risk of secondary proctectomy, 32 the lower functional results of proctocolectomy, 33 and the fertility issue in women, 34 this is a new argument in favour of simple colectomy in a majority of FAP patients.
The second part of the study aimed to describe the presentation and management of 58 DTs in 31 FAP patients. The median age at DT diagnosis in our study (33 years) was comparable as that reported in previous studies (31–34 years).26,28,31 There was no difference between genders regarding the rate of DT occurrence. We also did not confirm the results reported by Schiessling et al. 26 showing a higher number of DTs per patient, and a larger total DT size in male patients. Treatment of DTs in FAP patients remains a difficult challenge, with poor progress in comparison with the prevention of colorectal and duodenal cancers that has highly improved over the last 20 years. Simple surveillance remains a recommendation in patients with stable and non-life-threatening disease, 35 but these represented only three of our 31 patients and they had a relatively short follow-up. The study found that surgery remains a major option in DT of FAP patients, but recurrences are frequent (nearly three-quarters of patients). Thus, the two options (extensive surgery or medical treatments) should be considered. Conclusions about medical systemic treatments of DT in FAP patients are difficult to make because of the wide range of treatments used. Several different regimens were used in this study, which were discussed in multidisciplinary tumour board specialised in sarcoma. Most of these drugs (tamoxifen, non-steroidal anti-inflammatory drugs, sorafenib, imatinib) are reported alone or in combination in European Society for Medical Oncology (ESMO) guidelines. 3 Bevacizumab is not listed in these guidelines, but has shown some activity. 36 However, objective response was rare, but most DTs were stabilised while they were progressive before treatment.
In conclusion, the main result of this study is the link between a specific type of surgery in FAP patients and the development of DTs. Regarding treatment efficacy, our disappointing results regarding medical treatment justifies the urgent need for randomised, prospective studies in FAP patients. In this setting, at least the prospective registration of treatments and results is required to improve the knowledge and management of FAP disease.
Footnotes
Acknowledgements
The authors wish to thank Philip Robinson (DRCI: Direction de la Recherche Clinique et de l'innovation, Hospices Civils de Lyon), for his help in editing the manuscript.
Declaration of conflicting interests
None declared.
Funding
This work was made possible by a grant from the French National Cancer Institute (INCA).
