Abstract
Aim
The aim of this article is to evaluate the clinical and cost implications of failed endoscopic hemostasis in patients with gastroduodenal ulcer bleeding.
Methods
A retrospective claims analysis of the Medicare Provider Analysis and Review (MedPAR) file was conducted to identify all hospitalizations for gastroduodenal ulcer bleeding in the year 2012. The main outcome measures were to compare all-cause mortality, total length of hospital stay (LOS), hospital costs and payment between patients managed with one upper gastrointestinal (UGI) endoscopy versus more than one UGI endoscopy or requiring interventional radiology-guided hemostasis (IRH) or surgery after failed endoscopic attempt.
Results
The MedPAR claims data evaluated 13,501 hospitalizations, of which 12,242 (90.6%) reported one UGI endoscopy, 817 (6.05%) reported >1 UGI endoscopy, 303 (2.24%) reported IRH after failed endoscopy and 139 (1.03%) reported surgeries after failed endoscopy. All cause-mortality was significantly lower for patients who underwent only one UGI endoscopy (3%) compared to patients requiring >1 endoscopy (6%), IRH (9%) or surgery (14%), p < 0.0001. The median LOS was significantly lower for patients who underwent only one UGI endoscopy (four days) compared to patients requiring >1 endoscopy (eight days), IRH (nine days) or surgery (15 days), p < 0.0001. The median hospital costs were significantly lower for patients who underwent one UGI endoscopy ($10,518) compared to patients requiring >1 endoscopy ($20,055), IRH ($34,730) or surgery ($47,589), p < 0.0001.
Conclusions
Failure to achieve hemostasis at the index endoscopy has significant clinical and cost implications. When feasible, a repeat endoscopy must be attempted followed by IRH. Surgery should preferably be reserved as a last resort for patients who fail other treatment measures.
Introduction
Peptic ulcer disease is the most common cause of non-variceal upper gastrointestinal bleeding (UGIB), which is a common medical emergency requiring hospitalization. 1 Despite improvement in endoscopic and pharmacologic therapies, bleeding continues or recurs in more than 10% of patients after initial endoscopic hemostasis. 2 Re-bleeding, a significant predictor of mortality, may be treated by repeat endoscopic therapy, interventional radiology-guided hemostasis (IRH) or transarterial embolization (TAE) and/or surgery.3,4 The 2012 United Kingdom National Institute of Clinical Excellence (NICE) guideline recommends that unstable patients who re-bleed after endoscopic treatment should be offered selective TAE and if this is not promptly available, the patient should be referred for urgent surgery, which carries a morbidity and mortality rate of 55% and 30%, respectively.2,5 While repeat attempts at endoscopic hemostasis reduce the need for surgery and its associated adverse events, studies comparing TAE and surgery have reported conflicting outcomes.3,4,6,7 Although TAE was found to reduce the need for surgery in some reports, a recent systematic review of nine studies involving 711 patients found that re-bleeding rates were higher for TAE than surgery with no significant difference in mortality between techniques.4,6,7 A major reason for these conflicting findings is that all studies involved a small cohort of patients. In addition to increased morbidity, the treatment of re-bleeding is costly with significant financial implications to the health care system.
To our knowledge, the clinical and cost implications of failed endoscopic hemostasis in patients with peptic ulcer bleeding have not been examined in a national context. To answer this question we examined the MedPAR claims file to compare all-cause mortality, total length of hospital stay (LOS), hospital costs and payment for patients with peptic ulcer bleeding in whom hemostasis at index endoscopy was unsuccessful and who subsequently underwent repeat endoscopy, interventional radiology-guided or surgical hemostasis.
Methods
MedPAR data source
A retrospective claims analysis was conducted using the most current 2012 MedPAR file and the Provider of Services file. The MedPAR file contains complete inpatient hospitalizations’ claims for the entire United States Medicare population, or 56.8 million covered lives. These claims report patient demographic information such as age, sex, diagnosis and comorbidities. In addition, information regarding the hospitalization is provided including volume, LOS, diagnostic testing, therapeutic procedures and the charge for each hospitalization. In order to analyze the demographics of the hospitals where the claims of interest were generated, the MedPAR file was linked to the Provider of Services file, which contains geographic and demographic information for all hospitals.
Patient population
The study population consisted of all patients who had claims for receiving a blood transfusion and underwent an UGI endoscopy for gastroduodenal ulcer bleeding. Claims were included based on the combination of primary diagnosis and procedure. The following International Classification of Diseases, ninth revision, clinical modification (ICD-9-CM) diagnoses codes were used to identify patients with a gastroduodenal ulcer bleed: 531.0 (acute gastric ulcer bleeding), 531.4 (chronic gastric ulcer bleeding), 532.0 (acute duodenal ulcer bleeding) and 532.4 (chronic duodenal ulcer bleeding). In addition, the claim had to have the ICD-9-CM procedure code 44.3 (endoscopic control of gastric or duodenal ulcer bleeding) reported with ICD-9-CM procedure code 99.04 (transfusion of red blood cells).
A subgroup analysis was then conducted to identify patients who subsequently required (a) more than one endoscopy, ICD-9-CM code 44.3 (endoscopic control of gastric or duodenal ulcer bleeding) reported more than one time together with ICD-9-CM code 99.04 (transfusion of red blood cells); (b) IRH identified using ICD-9-CM code 39.77 (temporary therapeutic endovascular occlusion of vessel) or ICD-9-CM code 44.44 (transcatheter embolization for gastric or duodenal bleeding) reported together with ICD-9-CM code 99.04 (transfusion of red blood cells); (c) surgical hemostasis reported with ICD-9-CM code 43.6 (partial gastrectomy with anastomosis to the duodenum) or ICD-9-CM code 43.7 (partial gastrectomy with anastomosis to the jejunum) or ICD-9-CM code 44.01 (truncal vagotomy) or ICD-9-CM code 44.02 (highly selective vagotomy) or ICD-9-CM code 44.03 (other selective vagotomy) or ICD-9-CM code 44.42 (local excision of other lesion or tissue of stomach) reported together with ICD-9-CM code 99.04 (transfusion of red blood cells).
Outcome measures
The outcomes evaluated compared all-cause mortality during hospitalization, hospital LOS, hospital costs and hospital payments for patients who underwent blood transfusion and required one endoscopy, >1 endoscopy, IRH following failed endoscopy or surgical hemostasis following failed endoscopy of gastroduodenal ulcer bleeding. A secondary analysis was then conducted to analyze the demographics of the hospitals in which the procedures were performed.
Statistical analysis
All analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC). Discrete data are reported as frequencies and continuous data as medians. Using a chi-square test, the patient demographics and comorbidities reported on the claims were compared across the cohorts. Covariates were not adjusted for in the analysis of health resources because of limitations associated with using Medicare data.
Results
Baseline demographics and comorbid conditions of patients
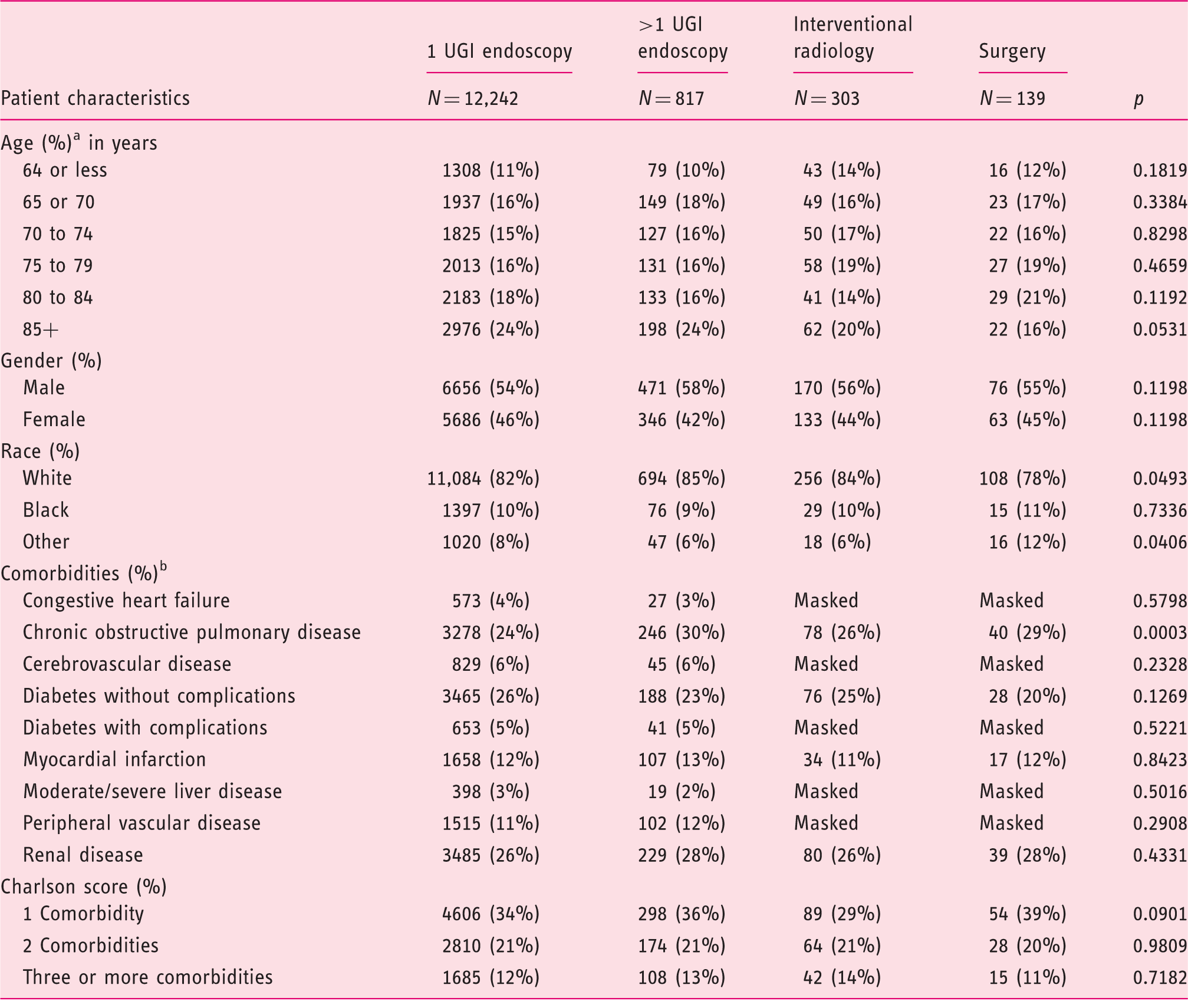
Mean/median years not calculated, as age in years was available only as categories.
Any cell with a volume of 1–10 is masked per HIPAA and CMS data use agreement. To avoid inputting the values, other cells may be masked.
UGI: upper gastrointestinal; HIPAA: Health Insurance Portability and Accountability Act; CMS: Centers for Medicare and Medicaid Services.
There was a significant difference in rates of all-cause mortality between the four cohorts: one UGI endoscopy 3%, >1 UGI endoscopy 6%, IRH following failed endoscopy 9% and surgery following failed endoscopy 14%; p < 0.0001 (Figure 1). The median aggregate hospital days or the total LOS from admission to discharge was significantly lower for patients who required only one UGI endoscopy compared to the other cohorts: four days vs. eight days for >1 UGI endoscopy, nine days for IRH and 15 days for the surgical cohort; p < 0.0001 (Table 2).
Mortality rates of the treatment cohorts following failed hemostasis at index endoscopy. Summary of the main outcome measures All-cause mortality = death prior to discharge. Costs are total claim costs weighted by 19 cost groups. Payments are total payments present on the claims (diagnosis-related group (DRG) price + outlier amount).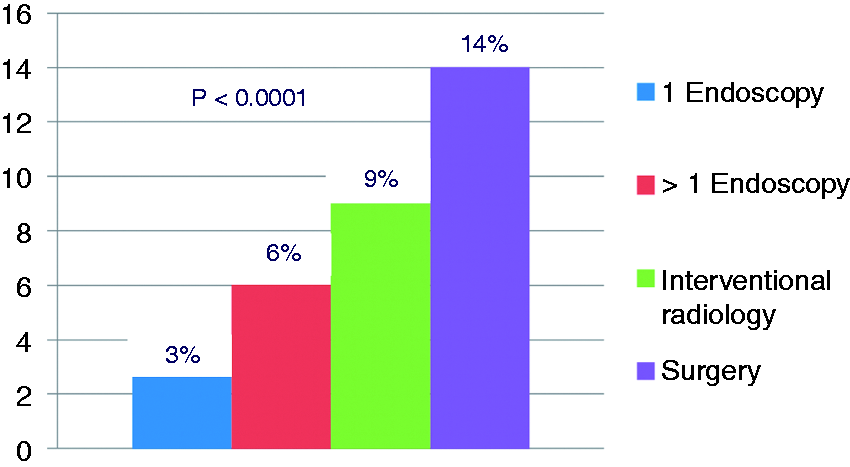
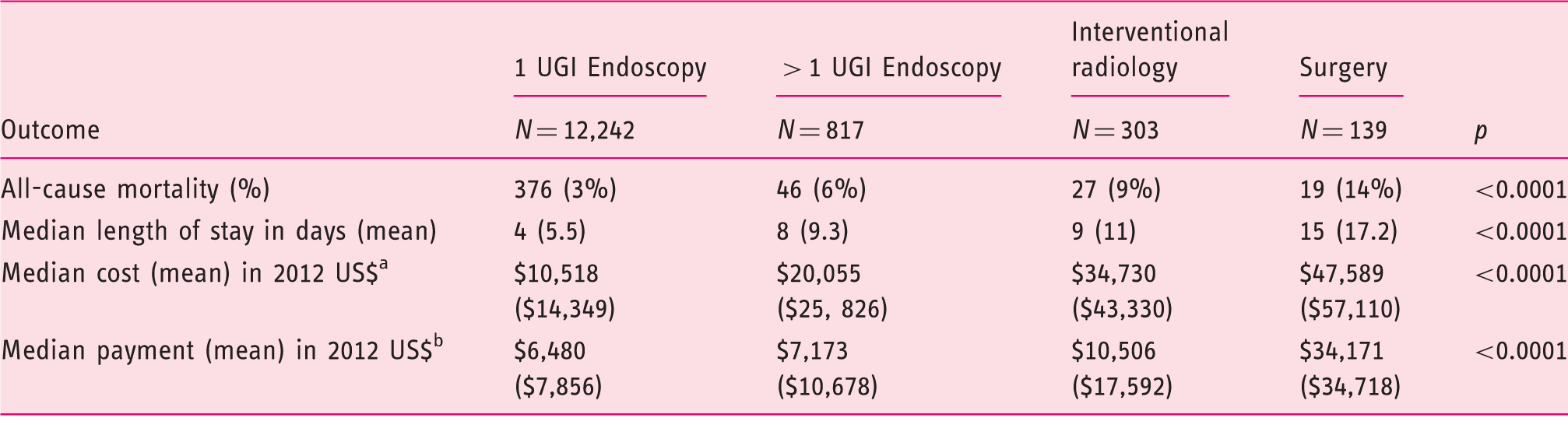
The median cost per claim for a single UGI endoscopy was significantly lower than that for >1 UGI endoscopy, IRH and surgery: US$ 10,518 vs. $20,055 vs. $34,730 vs. $47,589; p < 0.0001. Also, the median Medicare hospital payment was significantly lower for claims that included one UGI endoscopy when compared to >1 UGI endoscopy, IRH and surgery: US$ 6480 vs. $7173 vs. $10,506 vs. $34,171; p < 0.0001 (Table 2).
A total of 2080 hospitals were represented in the claims: 762 (37%) were academic and 1318 (67%) non-academic. While more patients in non-academic hospitals underwent a repeat UGI endoscopy than in academic hospitals, 53 vs. 47%, p < 0.0001, significantly fewer patients underwent surgery in academic than non-academic hospitals, 45 vs. 55%, p < 0.0001. A significantly larger number of patients underwent IRH in academic than non-academic hospitals, 68 vs. 32%, p < 0.0001.
Discussion
This study demonstrates that following a failed attempt at endoscopic hemostasis, subsequent treatment options are associated with a significantly prolonged length of hospital stay, increased all-cause mortality and had higher costs.
Bleeding recurs in 10%–20% of patients with peptic ulcer hemorrhage after hemostasis has been established by endoscopy.2,3 A recent systematic review identified active bleeding at endoscopy, large ulcer size, posterior duodenal and lesser gastric curvature ulcers as independent predictors of re-bleeding. 8 Although substantial data have been published on risk stratification in UGI bleeding, evidence-based guidelines for managing patients after initial hemostasis do not exist. An area of particular interest is the use of second-look endoscopy after initial hemostasis but the concept remains controversial because of conflicting data, high costs and the fact that this algorithm will involve a large number of unnecessary procedures. 9 In a randomized trial of 92 patients with re-bleeding randomized to repeat endoscopy or surgery, significantly higher adverse events were encountered in the surgical cohort. 3 Also, endoscopic retreatment reduced the need for surgery without increasing mortality. In the present study, 6% of the patient cohort underwent a repeat endoscopy but with mortality that was twice as high as those patients managed with a single endoscopic treatment.
IRH involves the use of vasoconstrictive agents or selective TAE, which is considered more effective. 10 While the choice of the best embolic agent is debatable, most centers use coils, gelatin sponge, gelfoam, cyanoacrylate glue or a combination of particles to achieve hemostasis. The technique is generally recommended for unstable patients who re-bleed after endoscopy or have bleeding uncontrolled by endoscopy. 5 It is especially used in patients with multiple medical comorbidities with the principal advantage being avoidance of major surgery in critically ill and unstable patients. Although not widely available initially, TAE is now more commonly practiced and available in most larger teaching hospitals worldwide. 11 In a retrospective study that compared TAE with surgery as salvage therapy for peptic ulcer bleeding, TAE reduced the need for surgery without increasing the overall mortality and was associated with fewer adverse events. 6 In a recent systematic review of nine studies that compared the clinical outcomes of patients treated with TAE or surgery, patients treated with TAE had higher rates of re-bleeding but with no difference in mortality between techniques. 4 In the present study we found a significant difference in mortality between the IRH and the surgical cohorts, 9% vs. 14%.
In a retrospective study of 10,428 patients who were admitted with peptic ulcer bleeding, the 30-day mortality rate was found to be 6% and 80% of patients died from severe comorbid disease and not the failure to control hemorrhage. 12 Although the administrative nature of the MedPAR database precludes specific evaluation such as underlying coagulopathy, there was no significant difference in the number of underlying comorbidities between the four cohorts. In the present study, all four cohorts were mutually exclusive with no overlap: i.e. after an initial UGI endoscopy patients underwent either further attempts at endoscopic hemostasis, IRH or surgery.
When examining the demographics of the hospital in which these claims were generated, we observed that after a failed endoscopic attempt, hemostasis under radiological-guidance was the preferred treatment approach at academic medical centers compared to either a repeat attempt at endoscopy or surgery, which were the preferred options in non-academic centers. It is likely that a majority of non-academic medical centers do not have emergent access to interventional radiology facilities and hence rely on repeat endoscopy or surgery as salvage treatment measures.
There was a significant difference in median hospital costs for the four treatment approaches with endoscopy being least expensive and IRH and surgery being more costly. Although the median Medicare hospital payment was highest for the surgical cohort, the costs of surgery were also higher, resulting in a hospital impact of a loss of $13,418 per claim. Similarly, IRH resulted in a median loss of $24,224 per claim and repeat endoscopy by $12,872 per claim. For claims for which patients were treated with only one UGI endoscopy, the hospital impact was a loss of $4038 per claim.
There are several limitations to this study. One, the database does not capture individual components of a treatment and hence the specific nature or timing of interventions undertaken are unknown. Two, details of pharmacological treatment or blood transfusion that is administered is unknown. Finally, the database also precludes propensity score matching or any modeling based on patient comorbidities.
As we move to a health care system focused on improving outcomes and containing costs, these data are very important. For example, for institutions that have selected to participate in Medicare’s bundled payment initiative, specifically for gastrointestinal hemorrhage, depending on the program model selected, a single payment is provided to the provider to cover several aspects of the continuum of care for GI hemorrhage cases. In this type of payment system, it is critical for providers to use the most effective endoscopic approach and devices as the first-line treatment to prevent patients from cascading to less effective and more costly approaches. The American College of Gastroenterology (ACG) practice guidelines recommend a repeat endoscopy for patients with recurrent bleeding with application of hemostatic therapy for those with high-risk stigmata of hemorrhage. 13 The ACG practice guidelines recommend that epinephrine injection should not be used alone. Thermal therapy with bipolar electrocoagulation or heater probe and injection of sclerosant is recommended to decrease further bleeding, need for surgery and mortality. 13 Placement of clips is also recommended as a treatment option, as they decrease further bleeding and the need for surgery. Therefore, endoscopists must be familiar with the variety of and most successful treatment options and have adequate training in all hemostatic techniques such as injection therapy, coaptive coagulation and hemostatic clips to optimize clinical outcomes. Hands-on training in models must be encouraged for endoscopists with inadequate experience and for those unfamiliar with the use of new hemostasis accessories.
Health care systems should track the success of their physicians for initial endoscopic hemostasis and ensure that those who are treating ulcer bleeding have success rates commensurate with the published literature and societal guidelines. Training should be provided for those who fall short of expected outcomes. Additionally, if initial endoscopic hemostasis fails, repeat endoscopy should be considered as second-line treatment for re-bleeding rather than moving directly to interventional radiology and surgery. To obtain optimal outcomes and minimize costs to the health care system and facilities, interventional radiology and surgery should be reserved for extenuating circumstances.
Footnotes
Acknowledgments
S Varadarajulu is the guarantor of this manuscript. All authors have reviewed and approved this manuscript, and their contributions are listed below.
A Roy: study design, data analysis, drafting of the manuscript.
M Kim: study design, acquisition of data, analysis, drafting of the manuscript.
R Hawes: concept and design, analysis, drafting of the manuscript.
S Varadarajulu: concept and design, analysis and interpretation, drafting of the manuscript.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
