Abstract
Background
Low bone mineral density (BMD) remains a frequent problem in patients with inflammatory bowel diseases (IBD). There is no general agreement regarding osteoporosis screening in IBD patients.
Methods
Cases of low BMD and disease characteristics were retrieved from 3172 patients of the Swiss IBD cohort study. Multivariate logistic regression analysis was conducted for predictive modeling. In a subgroup of 877 patients, 253 dual-energy X-ray absorptiometry (DXA) scans were available for validation.
Results
Low BMD was prevalent in 19% of patients. We identified seven predictive factors: type of IBD, age, recent steroid usage, low body mass index, perianal disease, recent high disease activity and malabsorption syndrome. Low BMD could be predicted with a sensitivity of 79% and a specificity of 64%, a positive predictive value (PPV) of 35% and a negative predictive value (NPV) of 93%. The area under the curve of the receiver operating characteristics was 0.78. In the validation cohort we calculated a PPV of 26% and an NPV of 88%.
Conclusion
We provide a comprehensive analysis of risk factors for low BMD and propose a predictive model with seven clinical variables. The high NPV of models such as ours might help in excluding low BMD to prevent futile investigations.
Abbreviations
American College of Gastroenterology American Gastroenterological Association bone mineral density body mass index British Society of Gastroenterology Crohn's disease Crohn's Disease Activity Index confidence interval C-reactive protein dual-energy x-ray absorptiometry European Crohn's and Colitis Organization extra-intestinal disease manifestation inflammatory bowel disease indeterminate colitis Modified Truelove and Witts Activity Index odds ratio receiver operating characteristic Swiss IBD cohort Ulcerative colitis
Introduction
Osteoporosis is an important clinical problem in patients with inflammatory bowel diseases (IBD).1–3 The fracture risk for IBD patients is increased by approximately 40%−60% compared to controls.4,5 Risk factors for decreased bone mineral density (BMD) in IBD patients include activity and severity of gut inflammation, systemic steroid usage, and intestinal malabsorption leading to calcium and vitamin D deficiency.1,6–13 In addition general risk factors for low BMD such as low body mass index (BMI), advanced age, smoking, alcohol intake and reduced physical activity also apply to IBD patients.6,8,11,14,15 However, not all risk factors have been consistently identified in all investigations and considerable controversy remains.
Low BMD is a powerful predictor of fracture risk, and a BMD one standard deviation below the age adjusted mean increases the relative fracture risk by 1.6–2.6. 16 BMD is usually determined using dual-energy X-ray absorptiometry (DXA). For the T score, measurements are compared to a healthy, young, adult, sex-matched reference population while the Z score in addition controls for age and ethnicity. 17 A T score ≤−2.5 defines osteoporosis in postmenopausal women and in men ≥50 years while osteopenia is diagnosed with a T score <−1 in this population. 17 In premenopausal women and younger men, osteoporosis cannot be diagnosed on BMD alone but a Z score of ≤−2 might be a clinically useful cut-off.17,18
Recommendations regarding osteoporosis screening in IBD patients according to current guidelines

IBD: inflammatory bowel diseases; DXA: dual-energy X-ray absorptiometry; CD: Crohn’s disease; UC: ulcerative colitis; BMI: body mass index.
We used data from the Swiss IBD cohort (SIBDC) study, a prospective long-term study of well-characterized IBD patients, to evaluate risk factors for osteoporosis/osteopenia. Based on these results we provide a model for prediction of low BMD. In a subset of patients we performed a chart review for validation.
Patients and methods
SIBDC is a prospective cohort study of IBD patients. Within the SIBDC database, epidemiological data, information regarding disease activity, past treatments, physical activity and IBD complications are prospectively collected, and data from 3172 patients were available. While general information regarding the presence of osteopenia/osteoporosis is recorded in the database, details of DXA scans such as T scores and Z scores are not.
Epidemiological characteristics of our patients with IBD
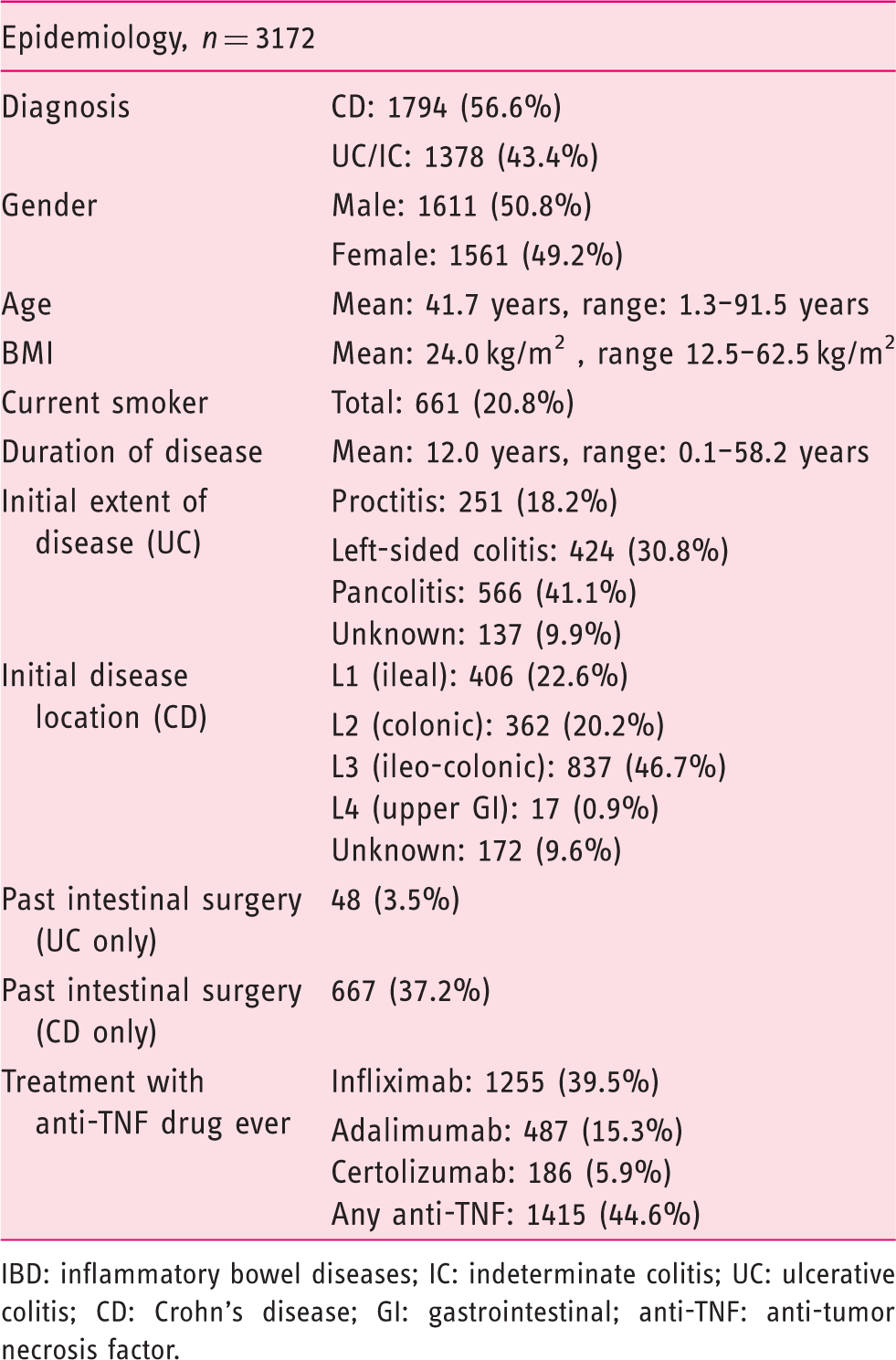
IBD: inflammatory bowel diseases; IC: indeterminate colitis; UC: ulcerative colitis; CD: Crohn’s disease; GI: gastrointestinal; anti-TNF: anti-tumor necrosis factor.
In six centers a chart review of all IBD patients was performed and 877 patient charts were reviewed for evidence of past DXA scans. All T scores and Z scores were retrieved. For the diagnosis of osteoporosis, cut-offs of −2.5 and −2 were chosen for the T score and Z score, respectively. For osteopenia, cut-offs of −1 > T score >−2.5 and −1 > Z score > −2 were used. The lowest scores for hip or spine were used.
For diagnosis or exclusion of osteoporosis and osteopenia during chart review, the following hierarchy was used: (1) T score (available for 229 out of 253 patients); (2) Z score (if no T score available, six patients); (3) diagnosis of osteoporosis/osteopenia in patient chart with clear reference to a DXA scan but no scores available (18 patients.)
Ethical considerations
The SIBDC study protocol has been approved by all ethics committees where patients were included. Data analysis was performed according to the Declaration of Helsinki.
Data analysis
Univariate and multivariate logistic regression analyses were used to determine the association with various potential risk factors for osteoporosis in IBD patients. For the multivariate models individual variables with a p value >0.157 were excluded in a step-wise approach. 26 For the predictive model of osteoporosis the multivariate analysis was performed to minimize the Akaike information criterion. The cut-off for a positive test was chosen to maximize the sum of the corresponding sensitivity and specificity (Youden’s criterion). For this analysis Stata software was used (StataCorp., 2015. Stata Statistical Software: Release 14. College Station, TX, USA: StatCorp LP).
Results
Risk factors for osteoporosis
For our analysis we used data from SIBDC, a large prospective cohort study of well-characterized Swiss IBD patients. Altogether, data for 3172 IBD patients could be retrieved; basic characteristics of our cohort patients can be found in Table 2. Our cohort thus represents a typical IBD population with respect to age, gender and distribution of disease severity. Presence of osteoporosis/osteopenia was mentioned for 616 patients (19.4%).
Univariate analysis of risk factors for low BMD in IBD patients

For the calculation of the activity index, the last measurement of Crohn’s disease activity index was divided by 500 or the last measurement of the modified Truelove and Witts severity index was divided by 21, respectively, and multiplied by 100.
BMD: bone mineral density; IBD: inflammatory bowel diseases; IC: indeterminate colitis; CD: Crohn’s disease; UC: ulcerative colitis; CI: confidence interval; BMI: body mass index.
Multivariate analysis for risk factors of low BMD
Predictive model/ multivariate logistic regression analysis of risk factors for low BMD in IBD patients

The model selection is based on the minimization of the Akaike Information criterion. To calculate the likelihood p of low BMD the following calculations need to be performed:
p = exp(s)/(1 + exp(s)); s = –2.299 (0.310) – 0.206 (0.115) * UC + 0.032 (0.003) * age – 0.074 (0.013) * BMI + 0.028 (0.004) * activity index + 1.471 (0.107) * steroid usage + 0.463(0.116) * perianal disease + 0.685 (0.220) * malabsorption. Values in brackets are the standard error. A value p > 0.16 is indicative of low BMD according to our model.
For the calculation of the Activity Index, the last measurement of Crohn’s disease activity index was divided by 500 or the last measurement of the modified Truelove and Witts severity index was divided by 21, respectively, and multiplied by 100.
BMD: bone mineral density; IBD: inflammatory bowel diseases; CD: Crohn’s disease; UC: ulcerative colitis; CI: confidence interval; BMI: body mass index.
To exclude confounding effects of preexisting osteoporosis/osteopenia on our model, we performed a sensitivity analysis, restricting our analysis to patients with new-onset of low BMD. For this secondary analysis 435 out of 616 patients (71%) with evidence of osteoporosis/osteopenia at enrollment were excluded. The resulting model was highly similar to the original model and six risk factors for low BMD were confirmed: BMI, steroids, perianal disease, malabsorption, age and last activity index. EIM showed significance only in our sensitivity analysis and IBD diagnosis lost significance (data not shown).
In another sensitivity analysis CD and UC/IC patients were considered separately: For CD identical risk factors were identified compared to the whole cohort (not shown). Risk factors for low BMD in UC/IC patients were also highly similar, only malabsorption syndrome was not significant anymore while presence of EIM was a positive risk factor for low BMD (p = 0.03, not shown).
Prediction of low BMD in IBD patients
We tested the power of our multivariate logistic model (Table 4) for the prediction of low BMD. The receiver operating characteristic (ROC) curve of our model is shown in Figure 1 and yields an area under the curve of 0.78. Using an optimal threshold (see Methods), we calculated a sensitivity of 79% and a specificity of 64% for low BMD in our cohort. In our patients our model had a positive predictive value of 35% and a negative predictive value of 93%. Globally, our model was able to classify 2074 out of 3108 patients correctly (67%). No cross-validation was performed.
The receiver operating characteristic (ROC) curve of our predictive model (compare with Table 4).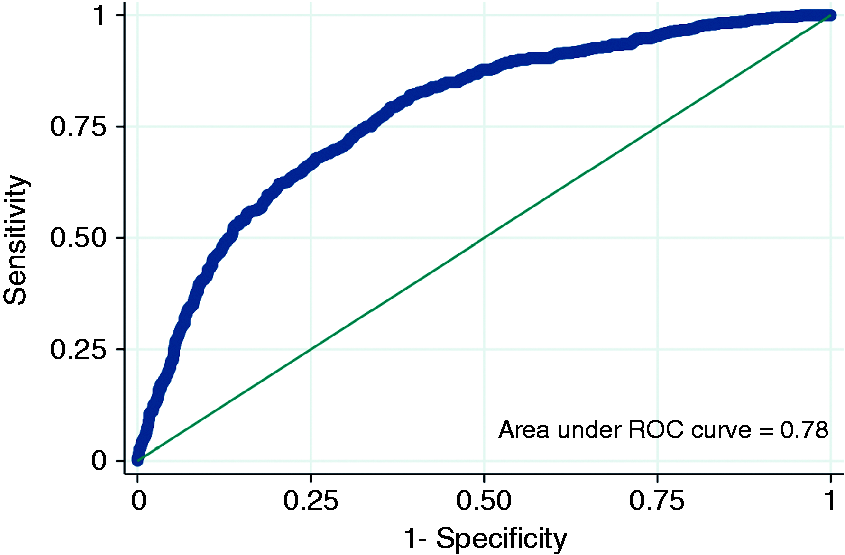
To validate our predictive model, we performed a chart review of 877 IBD patients and received data of individual DXA scans. Information regarding BMD was found for 253 patients. For these individuals a high degree of agreement with the SIBDC database was noted: In 132 patients with low BMD according to the SIBDC database information regarding DXA scans were available and the diagnosis was confirmed in 122 (92%). For five out of 10 patients with discordant results (i.e. low BMD according to the SIBDC database but a formally normal measurement) the discrepancy was likely due to T scores at the lower limit of normal (between −0.8 and −1.0). Similarly, normal BMD in the SIBDC database was confirmed in 122 of 169 patients (72%) by our chart review. In 47 discordant cases 10 showed borderline osteopenia (T score −1.1 to −1.3), four were performed before 2006, one showed normalization of osteoporosis in the latest DXA scan and one DXA scan was recent and possibly not covered by the latest data entry. No possible explanation for a discrepancy was noted in 31 cases (18%).
When the results of DXA scans were used to define low BMD, our model had a sensitivity of 71%, a specificity of 51%, a positive predictive value of 25% and a negative predictive value of 88%.
Discussion
In our study we provide a comprehensive analysis of decreased BMD in patients with IBD. We use disease- and patient-specific risk factors to provide a model for prediction of low BMD. Our analysis identifies high disease activity, perianal disease including fistulae, steroid usage, low BMI, malabsorption syndrome and age as risk factors for low BMD.
We find a correlation between classical disease activity scales such as CDAI and MTWAI and osteoporosis/osteopenia. In most previous studies no association between disease activity indices and low BMD was detected and only one publication described the Rachmilewitz Colitis activity index as a predictor for osteoporosis in a small number of UC patients. 9 Severe or persistently active disease has been established as a risk factor for osteoporosis/osteopenia;13,27 however, up to now neither CDAI nor MTWAI but different definitions for “severe” and “active” disease have been applied including biochemical criteria such as levels of C-reactive protein (CRP)13,27 or albumin, 28 need for ileum resection 27 and patient symptom questionnaires. 29
Our model also identifies perianal disease with fistulae, abscesses or fissures as a risk factor for low BMD. Patients with perianal disease differ from other IBD patients by having a younger age at onset of disease, 30 a more aggressive disease course, 31 a higher likelihood of EIM 32 and higher rates of recurrences after surgery. 33 Perianal disease therefore might be a strong marker for a long, severe and recurring disease course, which in turn would predispose to low BMD.
In IBD patients, a correlation of BMI and BMD6–8,14,15,29 has been noted in our analysis as well as in previous studies. Thereby, positive effects of a high BMI for stimulation of bone formation cannot be distinguished from a low BMI as a marker of severe inflammation or disease activity. In line with previous studies,1,6,10–13,27,34 we also identified malabsorption as a risk factor for low BMD. Malabsorption might lead to low levels of calcium and vitamin D as well as other nutrients, resulting in preferential bone resorption. One study even demonstrated a direct relationship between low vitamin D levels and osteoporosis in IBD patients. 12 Our study also confirmed steroid treatment as a risk factor for low BMD, in line with previous analyses.6–9,29,35
Several international guidelines regarding osteoporosis screening in IBD patients exist with only partially overlapping recommendations (Table 1). Furthermore, most guidelines leave ambiguity and neither the European Crohn’s and Colitis Organization (ECCO)21,22 nor the British Society of Gastroenterology (BSG)19,20 guidelines provide an objective definition for “persistent” or “continuous” activity that should trigger a DXA scan. Guidelines of the American Gastroenterological Association (AGA) and the American College of Gastroenterology (ACG) provide objective criteria 23–25 ; however, no criterion specifically applies to IBD.
Overutilization of DXA scans would clearly limit cost-efficiency of osteoporosis screening. Therefore, accurate and easy applicable recommendations would be desirable. All seven clinical parameters in our model are available in the office. While the positive predictive power of our model was modest at best (
The size of our study of 3172 patients compares favorably with previous studies that included up to 1230 patients. In addition, the high level of detail provided by the SIBDC database, which included detailed disease characteristics such as EIM and disease activity scores but also lifestyle factors such as physical activity and alcohol consumption, has not been provided by most previous studies. Since our results remained robust in a sensitivity analysis excluding all patients with low BMD at enrollment, risk factors of our model likely marks a disease dynamic directly leading to osteoporosis rather than identifying preexisting patient differences at baseline.
Our study has several limitations: (i) Our model needs validation in an independent cohort of IBD patients.(ii) No systematic testing of all cohort patients by DXA scans has been performed and our analysis might miss osteoporosis/osteopenia cases. However, chart review and validation of our analyses using available DXA data largely confirmed our results. (iii) Since the SIBDC database does not distinguish between osteoporosis and osteopenia, both conditions cannot be distinguished in our predictive model. (iv) No control group of non-IBD patients is available and our study cannot determine the increase in osteoporosis risk attributable to IBD. (v) Even though the SIBDC study represents patients treated in tertiary referral centers as well as in private practice, the cohort study is biased toward large referral centers and our data might not be representative for the Swiss population affected by IBD.
In summary, in a comprehensive analysis we propose a model for the prediction of low BMD using parameters available in the office, which after validation might facilitate the decision regarding osteoporosis screening. A model with a high negative predictive value might prevent unnecessary DXA scans.
Footnotes
Acknowledgments
We acknowledge all IBD patients participating in the SIBDC study for providing their personal medical history and current clinical symptoms, and regularly responding to the cohort questionnaires. Furthermore, we would like to acknowledge everybody from the data center of the SIBDC study for their tremendous work in data collection.
SRV, GR and BM designed the study, SS performed the chart review of IBD patients, JBR performed the statistical analysis, SS and BM contributed to the statistical analysis and wrote the paper, JBR, DF, LB, MS, JZ, NFQ, VP, SRV and GR reviewed and edited the paper for important intellectual content. All authors have seen and approved the final version of the manuscript.
Funding
This work was supported by research grants from the Swiss National Science Foundation to BM (Grant No. 32473B_156525), and the Swiss IBD Cohort (Grant No. 3347CO-108792).
Declaration of conflicting interests
None declared.
