Abstract
Barrett’s oesophagus is a precursor lesion for oesophageal adenocarcinoma, which generally has a poor prognosis. Patients diagnosed with Barrett’s oesophagus therefore undergo regular endoscopic surveillance to detect neoplastic lesions at a curable stage. The efficacy of endoscopic surveillance of Barrett’s oesophagus patients is, however, hampered by difficulties to detect early neoplasia endoscopically, biopsy sampling error, inter-observer variability in histological assessment and the relatively low overall progression rate. Efficacy and cost-effectiveness of Barrett’s surveillance may be improved by using endoscopic and clinical characteristics to risk-stratify Barrett’s patients to high- and low-risk categories. Recent national and international surveillance guidelines have incorporated Barrett’s length and presence of low-grade dysplasia in the advised surveillance intervals. In this review we will discuss endoscopic characteristics that may be associated with neoplastic progression in Barrett’s oesophagus and that may be used to tailor surveillance in Barrett’s patients.
Introduction
Barrett’s oesophagus (BO) is a premalignant condition induced by longstanding gastro-oesophageal reflux, which causes the squamous epithelium to be replaced by columnar epithelium containing intestinal metaplasia (IM). 1 Patients with non-dysplastic Barrett’s oesophagus (NDBO) have a reported annual risk of 0.6% for oesophageal adenocarcinoma (OAC) and 1% for high-grade dysplasia (HGD) and OAC combined. 2 The overall five-year survival for oesophageal cancer ranges from 15% to 25%, and the best outcomes are associated with disease diagnosed in the early stages. 3 Progression to oesophageal adenocarcinoma is a gradual multistep process, from NDBO to low-grade dysplasia (LGD), HGD and finally cancer may arise. 1 Currently, endoscopic surveillance of BO with biopsies is advised to detect early neoplasia at a curable stage. Early mucosal neoplasia and even low-risk superficial submucosal carcinomas can be treated with minimally invasive endoscopic techniques. 4 Guidelines suggest surveillance every two to five years for NDBO and 6–12 months for LGD.5–8 Endoscopic surveillance, however, suffers from several shortcomings, such as difficulty to detect early neoplasia endoscopically, biopsy sampling error, inter-observer variability between pathologists, and questionable cost-effectiveness given the low overall risk of malignant progression.
Improving cost-effectiveness requires more objective methods for risk stratification, allowing identification of high-risk patients who would benefit from endoscopic surveillance or prophylactic eradication of their BO and identification of low-risk patients in whom surveillance intervals may be prolonged or surveillance stopped.
Much international research effort is currently aimed at identification of a panel of biomarkers, to allow for objective risk stratification. Although there have been some promising results, at present none of these panels have been validated in large cohorts prospectively. Therefore their use in routine daily practice is not yet advised.5–8 However, previous studies have demonstrated an association between certain endoscopic and clinical factors and malignant progression that may be used for risk stratification in Barrett’s patients. Examples are the length of the BO segment,9–16 the presence of erosive oesophagitis at endoscopy,17,18 presence of a hiatal hernia,15,19–21 LGD,22–26,28–30 gastro-oesophageal reflux symptoms, smoking habits, body mass index (BMI) and use of proton pump inhibitors (PPIs), statins and non-steroidal anti-inflammatory drugs. 15
The recently published American College of Gastroenterology clinical guidelines on diagnosis and management of BO already advise endoscopic therapy for confirmed LGD in BO or endoscopic surveillance every 12 months. 7 The recent British Society of Gastroenterology (BSG) guidelines also advise to offer prophylactic ablation to patients with a repeat and confirmed diagnosis of LGD, or shorten surveillance intervals to every six months.5,6 The BSG guidelines also incorporated the length of the BO without dysplasia into their advice to perform surveillance every three to five years for BO <3 cm or every two to three years for BO ≥3 cm.5,6
In this review we will discuss what is known on endoscopic characteristics and their association with malignant progression in BO, and how these characteristics can be used to tailor endoscopic surveillance in Barrett’s patients. An overview is presented in table 1.
Endoscopic risk factors for neoplastic progression
Length of the Barrett’s segment
Barrett’s length varies greatly among individuals, but once diagnosed, Barrett’s length appears to be rather stable in affected individuals. 9 Multiple studies have recognised the length of the Barrett’s segment to be a significant risk factor for developing oesophageal adenocarcinoma. In 1992, Iftikhar et al. first described how patients with OAC had a longer median Barrett’s segment than patients without dysplasia. 10 Several studies hereafter have shown that the length of the Barrett’s segment is associated with an increased risk for progression.10–16
In a meta-analysis from 2012 evaluating the incidence of OAC in non-dysplastic BO in 57 studies, an incidence of 0.33% per year was found. When looking at patients with a BO segment <3 cm, the incidence was significantly lower (0.19%). 11 In another meta-analysis including six studies involving 1218 patients, Thomas et al. found that patients with neoplastic progression had a significant longer Barrett’s segment than patients without neoplastic progression (p < 0.002). 12 Wong et al. showed that a BO > 3 cm is an independent predictor for progression to dysplasia (odds ratio (OR) 1.2, 95% confidence interval (CI) 1.07–1.34). 13 A study by Wani et al. including 1204 patients with non-dysplastic BO found that there was a significant difference in incidence of HGD/OAC when BO length exceeded 6 cm (yearly incidence of OAC 0.09% (95% CI 0.03–0.24) vs. 0.65% (95% CI 0.33–1.25) in patients with BO >6 cm). 14 In a prospective trial with 713 patients, Sikkema et al. found a risk ratio of progression to HGD/OAC of 1.11 (95% CI 1.01–1.20) per centimetre of Barrett’s segment. 15 In a recent German study, patients were stratified to long (≥3 cm), short (≥1 to <3 cm) and ultra-short (<1 cm) BO. 16 The number needed to treat, i.e. the number of patients undergoing endoscopy to find one patient with HGD/OAC, was 450 for long-segment BO, 3440 patients with short-segment BO and 12,365 patients with an ultra-short segment BO. 16 The results from these studies strongly suggest that the length of the Barrett’s segment is correlated with the risk of neoplastic progression. It appears therefore logical to adjust the interval for surveillance to the extent of the Barrett’s segment.
Oesophagitis
Continuous reflux can lead to inflammation of the oesophagus, which may lead to DNA damage, resulting in dysplasia. 17 Oesophagitis has been associated with OAC as demonstrated by a few studies that were not specifically focused on Barrett’s patients. 18 The association of oesophagitis in Barrett’s patients and risk of neoplastic progression was described in the previously mentioned prospective surveillance study by Sikkema et al. 15 In this study the presence of oesophagitis was associated with an increased risk of neoplastic progression using multivariable regression analysis, adjusted for age and sex (relative risk (RR) 3.5; 95% CI: 1.3–9.5). The presence of oesophagitis at baseline or during surveillance therefore was an independent predictor of progression to HGD or OAC. Sikkema et al. also looked at the grade of oesophagitis; however, it was not possible to find an association between the severity of the oesophagitis and the risk of neoplastic progression because of the relatively small number of patients with oesophagitis.
Presence of a hiatal hernia
In patients with a hiatal hernia the lower oesophageal sphincter and the diaphragmatic pinch are separated, which weakens the gastro-oesophageal closing mechanism and increases gastric reflux into the oesophagus. Up to 65–87% of BO patients have a hiatal hernia upon endoscopy.15,19–21 A case-control study comparing endoscopic findings in BO patients with HGD/OAC to patients with non-dysplastic BO found that Barrett’s patients with HGD/OAC more often had a hiatal hernia (84%) than non-dysplastic patients (65%), p = 0.000. Using multivariate logistic regression, the length of the hiatal hernia increased the risk of neoplastic progression with an OR of 1.2 (95% CI 1.04–1.39, p = 0.013) per centimetre. 19 Anaparthy et al. did not find the presence of a hiatal hernia to affect the rates of progression to HGD/OAC in their multicentre BO surveillance study in 1175 patients. 20 Another study reviewing endoscopic data of 109 BO patients showed that the presence of a hiatal hernia was not a significant parameter in predicting higher pathological grade at index endoscopy (OR 5.168, p = 0.061). However, if present, the size of the hiatal hernia was strongly predictive of higher pathologic grades at index endoscopy, and 34% of individuals with HGD had hernias >4 cm, compared to 10% of individuals with intestinal metaplasia alone. 21 The prospective study by Sikkema et al. did not show length of hiatal hernia to be a significant risk factor for progression in univariate analysis (RR 0.9, 95% CI 0.7–1.2, p = 0.821). 15 However, most of these studies did not adjust for Barrett’s length.
Presence of LGD
The reported risk of neoplastic progression for patients diagnosed with LGD varies widely. In some studies, the reported neoplastic progression rates for LGD are comparable to those found in patients with NDBO.22–24 However, in other studies in which the LGD diagnosis was confirmed by an expert pathologist, the reported risk to develop HGD or OAC was as high as 27% after a median follow-up of two years and eight months.25,26
Recently, a meta-analysis found that BO surveillance cohorts with a high rate of LGD diagnosis report low rates of progression to HGD or OAC, whereas a much higher rate of neoplastic progression from LGD is reported in cohorts in whom LGD is diagnosed less frequently. 27 The most important issue seems to be the reliability of the baseline LGD diagnosis.
Especially in community settings, over-diagnosis of LGD may result in underestimation of the risk of neoplastic progression. Curvers et al. investigated the natural history of LGD in a large community-based cohort of BO patients. 28 After the original histological diagnosis was reviewed by two expert gastrointestinal pathologists, the LGD diagnosis was confirmed in only 15% of patients. The LGD diagnosis was down-staged in the remaining 85% of cases. The rate of neoplastic progression was 13.4% per patient-year for patients with confirmed LGD, compared with 0.49% per patient-year for patients who were down-staged to NDBO.
Another comparable study by Duits et al. retrospectively examined the prognostic value of an expert pathology panel for reviewing the LGD diagnosis of community hospital pathologists. 29 Similarly to the aforementioned study, they demonstrated that only a small percentage (27%) of the original LGD diagnoses were confirmed, while the remaining 73% of cases were down-staged. After a median follow-up of 39 months the risk of progression to HGD or OAC was 9.1% per patient-year in the confirmed LGD group. In contrast, patients with a down-staged diagnosis to NDBO or indefinite for dysplasia had a neoplastic progression rate of 0.6% and 0.9%, respectively.
Endoscopic surveillance has been recommended by international guidelines for Barrett’s patients with LGD.5–8 However, taking the high risk of neoplastic progression in patients with confirmed LGD in consideration, this might no longer be the adequate strategy for this group of patients. A randomised trial by Phoa et al. compared endoscopic surveillance with prophylactic radiofrequency ablation (RFA) in Barrett’s patients with confirmed LGD. 30 In the control group the rate of progression was 11.8% per patient per year, similar to the studies mentioned above. Prophylactic ablation reduced the risk of progression to HGD or OAC by 25.0% (1.5% for ablation vs. 26.5% for surveillance). The multivariable analysis demonstrated the following three independent predictors of progression in the control group: the number of years since the diagnosis of BO, the number of endoscopies with dysplasia prior to inclusion, and circumferential Barrett length in centimetres.
These recently published papers resulted in revision of the National Institute for Health and Care Excellence (NICE) guideline on ablative therapy for the treatment of BO in 2014. The guideline now includes LGD as a valid indication for RFA.5,6
Future directions
Next to identification of endoscopic and patient characteristics that may be used for risk-stratification of BO patients, much international research effort is aimed at identifying objective biological markers to predict the risk for neoplastic progression in patients with BO. 31 Despite promising results in this field, none of these biological markers or panels have been prospectively validated, and are thus not yet available for routine use in clinical practice. In the future, however, they will surely be part of BO management. Furthermore, recent genome-wide association studies have demonstrated that there might be a genetic base in development of OAC.32,33 In the future, a multi-disciplinary approach to patients with BO may be used, incorporating endoscopic, patient, genetic and biomolecular characteristics to tailor surveillance and to select patients who would benefit from prophylactic treatment of their BO.
Overview of studies describing risk factors for neoplastic progression in Barrett’s oesophagus.
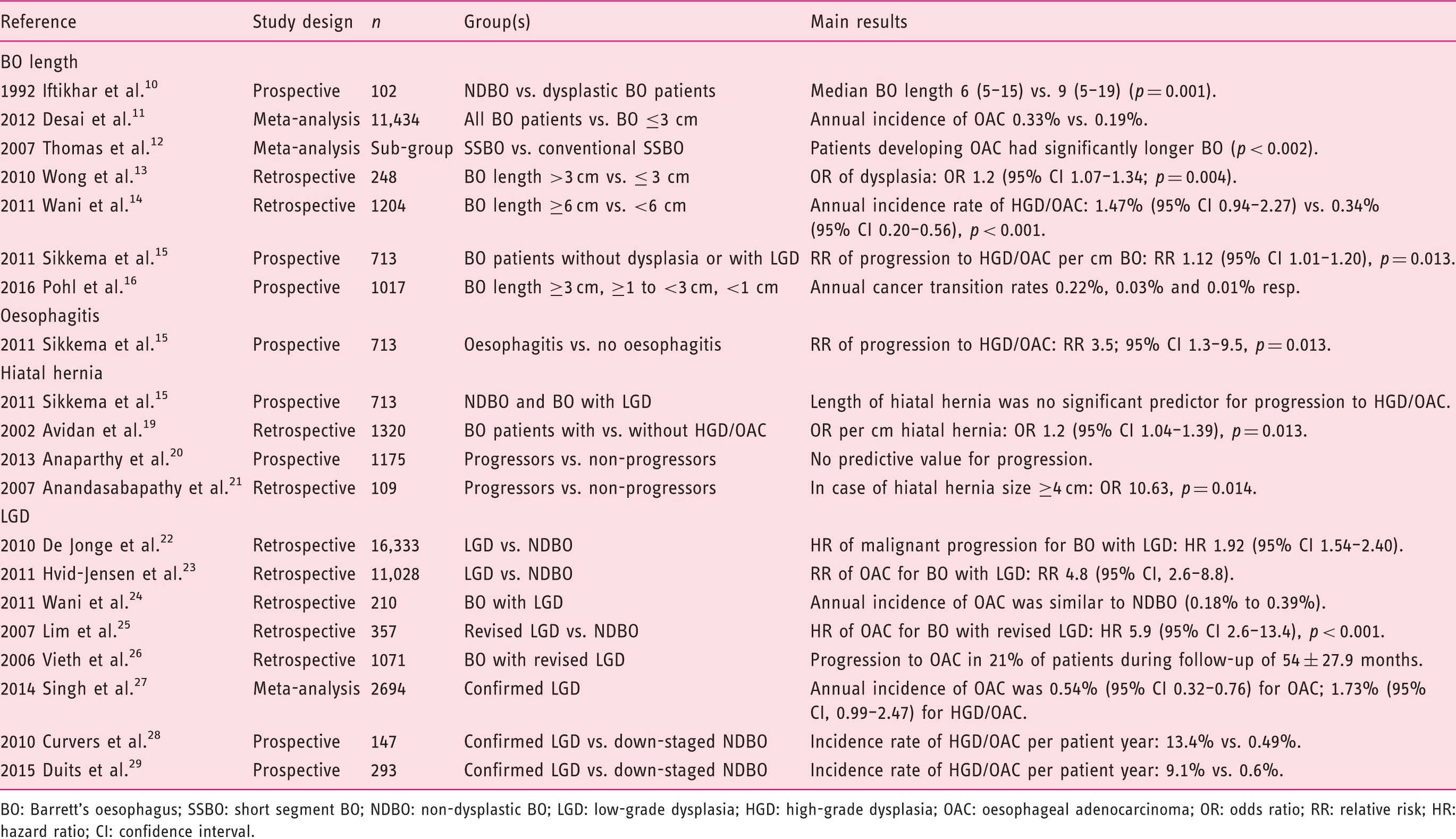
BO: Barrett’s oesophagus; SSBO: short segment BO; NDBO: non-dysplastic BO; LGD: low-grade dysplasia; HGD: high-grade dysplasia; OAC: oesophageal adenocarcinoma; OR: odds ratio; RR: relative risk; HR: hazard ratio; CI: confidence interval.
Conclusion
Endoscopic Barrett’s surveillance at regular intervals is recommended to detect neoplasia at an early and curable stage. Efficiency of current endoscopic surveillance programs may be improved by identifying patients at risk for developing neoplasia. Barrett’s surveillance can be tailored by using endoscopic characteristics. As discussed in this review, longer Barrett’s segments (>3 cm) carry an increased risk of progression compared to short-segment BO. Furthermore, presence of LGD in biopsies, confirmed by an experienced gastro-intestinal (GI)-pathologist, is associated with a significant risk of developing HGD/OAC and warrants endoscopic surveillance every 6–12 months or prophylactic ablation using radiofrequency ablation.
Footnotes
Acknowledgement
The authors were invited to write this review as a result of the 2015 UEG Week top abstract prize.
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
