Abstract
Barrett’s oesophagus is a pre-malignant condition associated with the development of oesophageal adenocarcinoma. Currently white light endoscopy and biopsy is the mainstay diagnostic tool. Yet this approach is troubled by issues related to cumbersome biopsy sampling, biopsy sampling errors and cost. Therefore in order to overcome such adversity, there needs to be evolutionary advancement in terms of diagnosis, which should address these concerns and ideally enhance risk stratification in order to provide timely management in real time. This review highlights the current endoscopic tools aimed to enhance the diagnosis of Barrett’s oesophagus and its subsequent progression.
Keywords
Background
Barrett’s oesophagus (BO) is a pre-malignant condition associated with chronic inflammation, tissue damage and ultimately malignancy. The replacement of the stratified squamous lining by columnar epithelium within the distal oesophagus is the hallmark pathological change as a result of longstanding gastro-oesophageal reflux.
The diagnosis has been deemed by global consensus to be determined by four random quadrant biopsies. Such intervention is typically operator dependent and a skill that is not uniform in practice. In addition such an approach can appear overbearing particularly due to the fact that evidence highlights a low annual risk of Barrett’s related adenocarcinoma (AC) (0.12%). 1
The epithelial changes associated with BO have been well described. In the USA, BO is confirmed only on the presence of columnar-lined epithelium in addition to goblet cells, namely intestinal metaplasia (IM). 2 In the UK, the existence of goblet cells is not however required to confirm a diagnosis of BO. 3
Diagnostic avenues
The use of white light endoscopy (WLE) is the initial step in gastro-oesophageal junction visualisation and the currently recommended gold standard approach. Despite this, drawbacks do exist and include limited image resolution, and dependency on the operator’s skill. As a result, biopsy sampling is necessarily typically random in nature which adds significantly to the procedural cost. Furthermore, sampling errors can lead to an inaccurate diagnosis and ultimately long-term management. To aid detection, several imaging-enhanced technologies have emerged in the hope of improving the diagnostic algorithm.
Chromoendoscopy
Chromoendoscopy in the diagnosis of BO is not new. Here dyes are applied topically during the endoscopic procedure to aid in enhancement of tissue architecture. Examples of dyes used include methylene blue (MB) which is predominantly absorptive, acetic acid, and indigo carmine which concentrates between epithelial cells.
Sharma et al. utilised MB-based chromoendoscopy for the detection of IM in patients with short segment BO and observed a detection rate of 61% as compared to 42% in the control group. In addition patients that underwent MB chromoendoscopy were noted to require fewer biopsies. 4 Another study of 43 patients demonstrated that the average number of biopsies per patient was significantly lower, with the proportion of specialised columnar epithelium in each specimen significantly higher when using MB. Furthermore, dysplasia or cancer was observed in significantly more MB specimens (12% vs 6% p = 0.004). 5 A randomised prospective cross over trial comprising 57 patients, observed that the use of MB-directed biopsies diagnosed significantly more specialised IM (75%), compared with a random biopsy technique (75% vs 68% p = 0.032). There was, however, no significant difference in the diagnosis of dysplasia or carcinoma. Interestingly, a meta-analysis of nine studies comprising 450 patients noted that there was no significant incremental yield with MB over random biopsy for the detection of specialised IM, dysplasia and high-grade dysplasia and/or early cancer. 6
Acetic acid (AA)-guided chromoendoscopy was employed during a 31-patient prospective randomised trial and helped to detect a significantly higher percentage of tissue containing specialised columnar epithelium (78%) compared to a random biopsy approach (57%). 7 Fortun et al. also utilised AA in 64 patients and observed clear visualisation of epithelial pit patterns within BO as well as a higher yield of specialised IM and dysplasia. 8 Another study noted that AA-based chromoendoscopy yielded a sensitivity of 95.5% and specificity of 80% for the detection of neoplasia with a significant improvement in detection as compared to WLE (p = 0.001). 9 Tholoor et al. undertook a retrospective cohort study aiming to investigate the effectiveness of AA in a BO surveillance programme. Results noted that AA detected more neoplasias than conventional protocol mapping biopsies and required 15 times fewer biopsies per neoplasia detected. 10 Pohl et al. undertook a prospective study analysing the rate of high-grade intra-epithelial neoplasias or early carcinomas that are macroscopically occult to enhanced visualisation with high-resolution endoscopy and AA chromoendoscopy. Findings concluded patient-related sensitivity and specificity rates of 96.7 and 66.5%. 11
Indigo carmine chromoendoscopy (ICC) is an alternative dye-based approach. Three types of mucosal patterns have been described with this intervention, namely a ridged/villous or circular pattern for IM and an irregular/distorted pattern for high-grade dysplasia. In this regard ICC enables identification of IM and high-grade dysplasia but not low-grade dysplasia. 12 A 28-patient prospective, randomised crossover study of high resolution endoscopy (HRE) plus ICC or narrow band imaging concluded a sensitivity for high-grade dysplasia/early oesophageal cancer of 93% and 86% for HRE-ICC and HRE-narrow band imaging (NBI) respectively. Furthermore ICC and NBI observed a limited number of additional lesions occult to high-resolution endoscopy, however such lesions did not alter the sensitivity for identifying patients with high-grade dysplasia/early oesophageal cancer. 13 In contrast, Curvers et al. aimed to compare the benefits of IC, AA or NBI in reference to standard WLE and noted no improvement in inter-observer agreement or diagnostic yield of early BO-related neoplasia. 14
Variation in the outcome of chromoendoscopy has been noted and can be attributed to non-uniform concentrations/volumes of dye used in addition to contact time. Furthermore, the variation in sensitivity and specificity, as well as inter-observer variability has meant there is as yet no guaranteed consensus in terms of this approach.15–17
Virtual chromoendoscopy
NBI
The principle of NBI is based on the fact that the higher the wavelength of light, the greater the surface penetration. Therefore blue light penetration is superficial in nature, with red light allowing for much deeper surface assessment. The NBI-based endoscopy system comprises the typical xenon light source with red, green and blue illumination, coupled with modification centred on the use of narrow band filters along with an increased contribution of blue light. It is well-known that haemoglobin has a maximum absorptive wavelength in the blue part of the spectrum which is the fundamental reason why NBI is able to analyse vascular structures with greater accuracy (see Figure 1). In addition, NBI suffers from less light scattering as compared to standard white light endoscopy resulting in higher resolution images. Wolfson et al. in a prospective blinded tandem study of 65 patients with BO noted higher grades of dysplasia using NBI as opposed to standard WLE. In addition, NBI-directed biopsies confirmed dysplasia in more patients (57%) compared with biopsies taken using standard resolution endoscopy (43%). Furthermore, a greater number of biopsies were needed using WLE compared to NBI (mean 8.5 vs 4.7 p < 0.001).
18
An additional randomised crossover trial of 123 BO patients also noted that NBI required fewer biopsies per patient (3.6 vs 7.6 p < 0.0001). In addition NBI detected a greater proportion of dysplastic areas (30% vs 21%, p = 0.01).
19
Kara et al. analysed the use of autofluorescence imaging (AFI) followed by NBI in 28 BO patients with suspected or endoscopically treated high-grade intra-epithelial neoplasia (HGIN). Results demonstrated that 47 suspicious lesions were detected with AFI: 28 contained HGIN and 19 were false positives. Utilising NBI, 14 of the false positives were deemed not suspicious, leading to a false positive rate reduction from 40% to 10%.
20
White light endoscopy evidence of Barrett’s oesophagus followed by narrow band imaging (NBI) highlighting surface anatomy and vasculature (image obtained from the National University Hospital, Singapore following appropriate consent).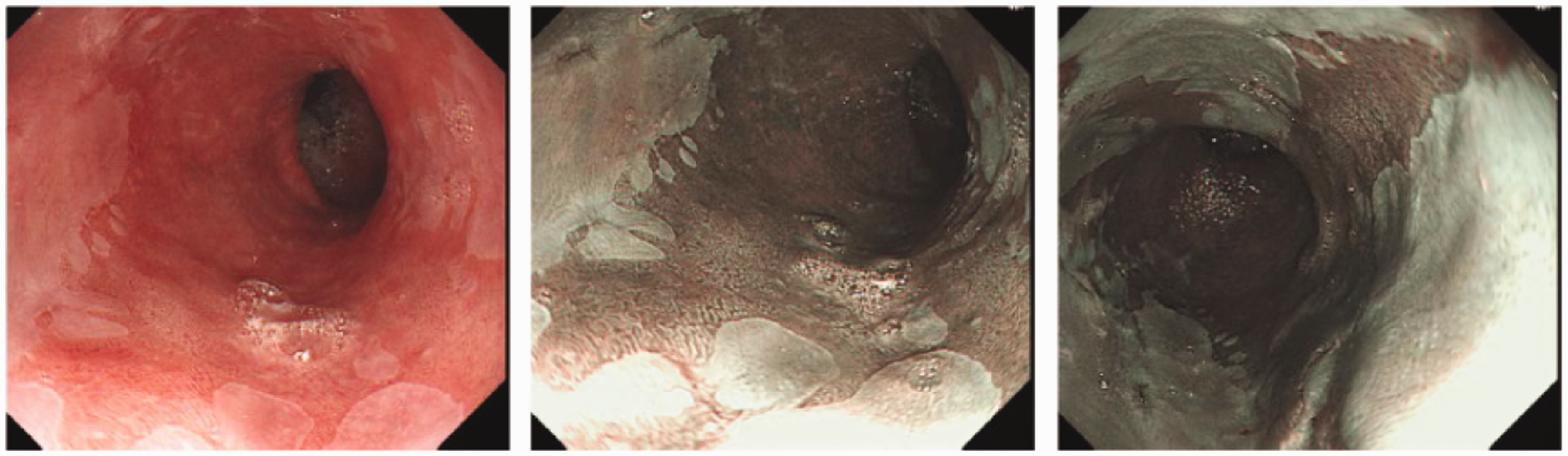
NBI has helped give rise to a pattern classification system for BO patients. A single-centre study of 63 patients with BO noted that specialised IM was characterised by villous/gyrus forming patterns in 80%, which were primarily regular with regular vascular patterns, or a flat mucosa with regular normal-appearing long branching vessels in 20% of cases. HGIN was associated with irregular/disrupted mucosal patterns, irregular vascular patterns and abnormal blood vessels, with 85% of cases having two or more abnormalities. In the case of HGIN, NBI demonstrated a sensitivity of 94%, specificity of 76%, positive predictive value of 64% and negative predictive value of 98%. 20 An additional study of 51 patients, utilised NBI to grade BO patients with a ridge/villous pattern for IM with a sensitivity, specificity and positive predictive value of 93.5%, 86.7% and 94.7% respectively An irregular/distorted pattern for high-grade dysplasia demonstrated a sensitivity, specificity and positive predictive value of 100%, 98.7% and 95.3% respectively. 21 Goda et al. undertook a study of 58 patients and demonstrated that patients with BO-related AC had evidence of irregular patterns in both the fine mucosal and capillary patterns with NBI. Those with specialised IM had a cerebriform fine mucosal pattern (sensitivity 56%, specificity 79%) or ivy- or deoxyribonucleic acid (DNA)-like capillary pattern (sensitivity 77%, specificity 94%). 22
Mannath et al. undertook a meta-analysis of eight studies including 446 patients and concluded that NBI was accurate for the diagnosis of high-grade dysplasia in BO on the basis of irregular mucosal pit patterns/irregular microvasculature, with a high sensitivity but poor specificity for specialised IM. 23
Overall NBI has been shown to enhance the mucosal surface architecture and vasculature. Ease of use is achieved through simple switch activation on the endoscope and no additional patient preparation is required, unlike chromoendoscopy which relies on necessary dye utilisation. Furthermore such enhanced visualisation methods can allow the operator to reduce the number of random biopsies taken, which increases cost effectiveness. Downsides do exist and include the fact that no agreed consensus exists for mucosal and vascular pattern classifications, as well as limited data comparing inter- and intra-observer variability. Therefore NBI should be used as a biopsy stratifying tool, where regularly appearing areas can effectively be left alone and not biopsied. 24
Flexible spectral imaging colour enhancement (FICE)
FICE was introduced in 2005 as a tool to enhance mucosal surface contrast in particular vascular aspects without the need for dyes. Furthermore it is able to produce images at a more distant view without the need for significant magnification. Osawa et al. utilised a transnasal FICE approach to determine a diagnosis of BO and noted that the palisade vessels could be more clearly visualised in BO mucosa with transnasal FICE than with conventional endoscopy. 25 Camus et al. undertook a prospective study combining AA and FICE in the detection of Barrett’s epithelium and associated neoplasia. Results concluded that patients with high-grade dysplasia showed an irregular mucosal pattern in 14% using high definition WLE and in 100% using AA-FICE combined. 0% of irregular vascular patterns were identified using high definition WLE, while AA and FICE combined visualised such patterns in 86% of cases. 26
i-Scan
i-Scan-based endoscopy incorporates three software modalities: surface enhancement focusing on delineating edges and lesion borders and contrast and tone enhancement focusing on subtle mucosal irregularities. Hancock et al. undertook a case series on the use of i-Scan and noted its potential in the diagnosis or therapy of BO with dysplasia or oesophageal AC. 27 Hoffman et al. compared AA with i-Scan imaging for the detection of BO and noted that the yield of targeted biopsies was significantly greater with both i-Scan (66% vs 21% p = 0.009) and AA (57% vs 26% p = 0.012). 28 Large scale studies on the use of i-Scan are currently however lacking.
Autofluorescence imaging (AFI)
AFI-based endoscopy relies on excitation of fluorophores such as collagen and elastin within tissue, with the subsequent detection of emitted light. As a result molecules endogenous to the tissue in question can be utilised for potential sub type differentiation.
Initial proof of concept studies showed promising results. Curvers et al. utilised AFI during a prospective cohort study and observed confirmation of early neoplasia in autofluorescence positive areas. 29 An additional study of 36 patients observed that all patients with evidence of high-grade dysplasia were classified accurately following the use of autofluorescence. 30 Holtz et al. analysed 22 patients through autofluorescence and concluded that the combination of 395 nm excitation with an r3 intensity ratio enhanced the discrimination of non-dysplastic tissue from early neoplasia. 31
Subsequent comparative results lent further support. Borovicka et al. undertook a multi-centre randomised trial and observed an AC/high-grade dysplasia detection rate of 12% with AFI as compared to 5.3% with conventional endoscopy. 32 A more recent study utilising third-generation AFI demonstrated an increased detection of high-grade intra-epithelial neoplasia/early cancer from 47% to 89%. 33
However, these promising results were not borne out by other insights. A study of 50 patients noted that the sensitivity for high-grade dysplasia/early stage cancer detection was 85% for standard endoscopy and only 69% for AFI-based endoscopy. 34 Similarly, Boerwinkel et al. in a study of 211 patients observed 39 with evidence of high-grade intraepithelial neoplasia or intramucosal cancer: 23 detected by standard WLE, 11 by a random biopsy approach and five detected by AFI, concluding that AFI had minimal effect on the diagnosis of early stage neoplasia. 35 In addition, a 42-patient study, where 14 had evidence of high-grade dysplasia or early oesophageal AC demonstrated a sensitivity, specificity and negative predictive value of 50%, 61% and 71% for AFI compared with 71%, 46% and 76% for NBI. 19 A multi-centre trial of 87 patients undergoing endoscopic tri-modal imaging (ETMI) and standard video endoscopy (SVE) demonstrated no significant difference in the histological yield between ETMI and SVE. ETMI was shown to have a significantly higher targeted yield compared with SVE secondary to AFI (p = 0.02), but the yield of targeted biopsies of ETMI was significantly inferior to the overall yield of SVE. 36
The current consensus regarding AFI is one of high sensitivity in the detection of dysplasia with the potential risk of false positives, therefore branding it predominantly as a red flag technique during surveillance.
Endocytoscopy (EC)
EC provides real-time visualisation of the cellular components of the surface epithelial layer. Visualisation requires pre-staining of subcellular structures with contrast agents and diagnosis is based on the size, density and cell arrangement as well as the size and shape of the nuclei, nucleus to cytoplasm ratio and staining pattern. 37 Pohl et al. concluded complete inability to adequately assess EC images in 49% of the pre-marked areas with magnification×450 and in 22% with magnification×1125. At most, 23% of images with lower magnification were interpretable to identify characteristics of neoplasia, and 41% with higher magnification. Inter-observer agreement was fair at best (kappa from<0 to 0.45). Positive and negative predictive values for HGIN or cancer were 0.29 and 0.87, respectively, for magnification×450, and 0.44 and 0.83, respectively, for magnification×1125. 38
Confocal laser endomicroscopy (CLE)
CLE is a technique also applicable in the diagnosis of BO, allowing for real-time microscopic imaging (see Figure 2). In general, the process relies on tissue illumination and subsequent detection of reflected fluorescent light passing through a confocal pinhole aperture which helps to improve spatial resolution. Tissue fluorescence is provided courtesy of contrast agents, primarily fluorescein injected intravenously.
Columnar-lined epithelium and dark mucin in goblet cells noted on confocal endomicroscopy (image reproduced with permission from Cellvizio.net).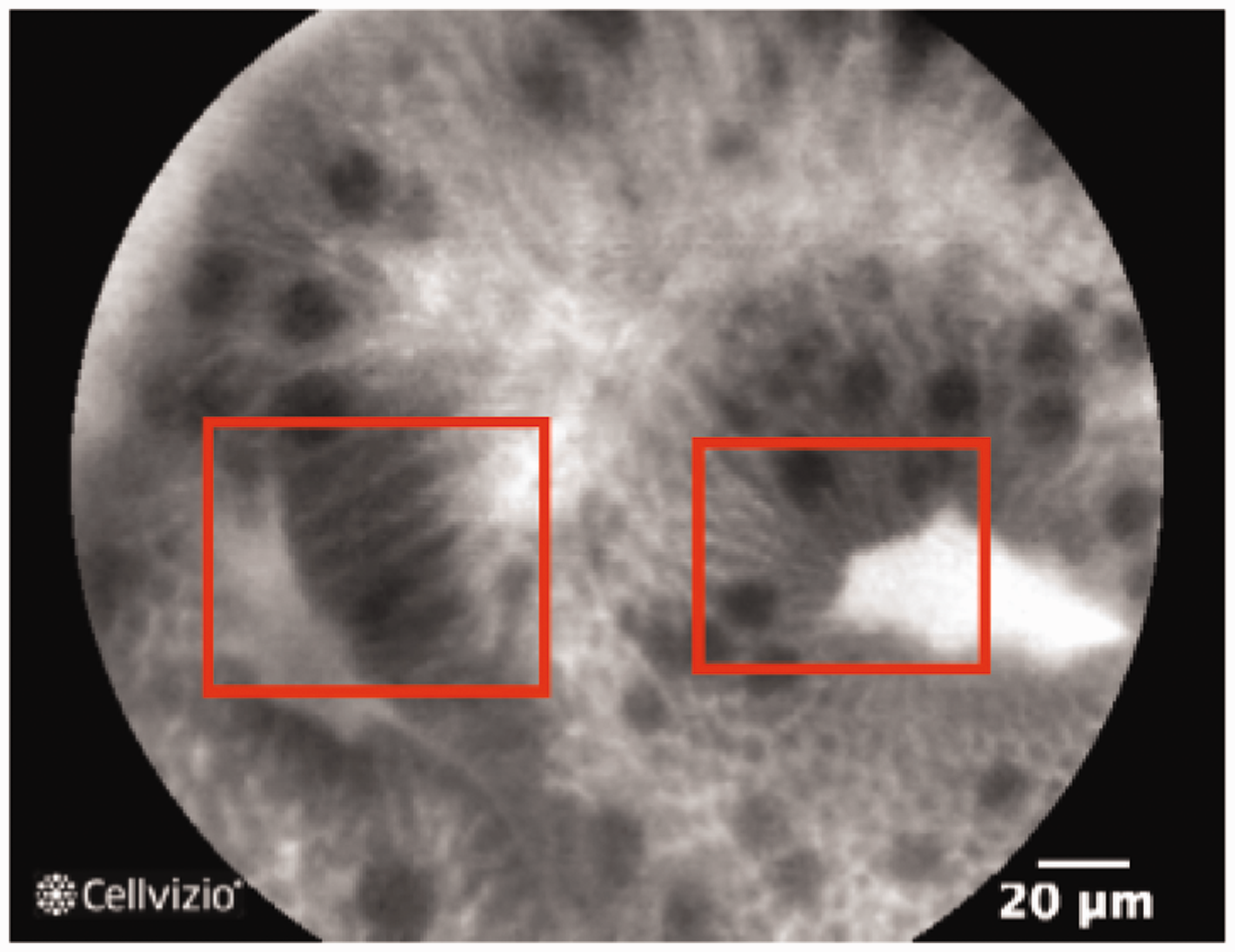
Two endoscopic systems exist employing the confocal approach, one which is integrated into the distal tip of the endoscope (Optiscan) and the other an external flexible probe which passes through the working channel of the endoscope (Mauna Kea Technologies). Both allow for magnification up to 1000× greater than normal with the former providing a depth of up to 250 microns.
A multi-centre study of 101 BO patients presenting for surveillance or endoscopic treatment of high-grade dysplasia/AC demonstrated a sensitivity and specificity for high definition white light endoscopy (HD-WLE) of 34.2% and 92.7%, respectively, compared with 68.3% and 87.8%, respectively, for HD-WLE or probe-based CLE (pCLE) (p = 0.002 and p < 0.001, respectively). The sensitivity and specificity for HD-WLE or NBI was 45.0% and 88.2%, respectively, compared with 75.8% and 84.2%, respectively, for HD-WLE, NBI, or pCLE (p = 0.01 and p = 0.02, respectively). 39 Bertani et al. undertook a study of 50 patients and observed that the dysplasia detection rate was significantly higher using CLE as compared to HD WLE (p = 0.04). 40 A further multi-centre trial of 192 BO patients highlighted that HD-WLE and CLE resulted in a fewer number of mucosal biopsies and a higher diagnostic yield for neoplasia (34% vs 7%, p < 0.0001) compared with HD-WLE. In addition, HD-WLE and CLE increased the sensitivity for neoplasia detection to 96% from 40% (p < 0.0001) with a treatment plan change in 36% of patients. 41
Wallace et al. analysed 40 sites of BO tissue in a prospective, double-blind study of 11 endoscopists and noted a sensitivity for the diagnosis of neoplasia of 88% and specificity of 96%, with a substantial agreement on diagnosis (86%, kappa 0.72). 42 Another study concluded that the use of CLE in 63 patients allowed for BO and neoplasia to be predicted with a sensitivity of 98.1% and 92.9% respectively, and a specificity of 94.1% and 98.4% respectively. 43 In contrast, Jeyasekera et al. analysed 50 patients for the detection of high grade dysplasia/intramucosal cancer and noted a lower sensitivity and specificity using CLE as compared to WLE and NBI (CLE 75.7%/80%, WLE 79.1%/83.1%, NBI 89%/80.1%). 44
The primary advantage of CLE is in its application for targeted biopsies of abnormal tissue minimising the need for random sampling. One of the major disadvantages of pCLE however is the limited number of probe uses which hinders cost effectiveness. In addition, as the probe occupies the same biopsy channel of the endoscope, it has to be removed before histological sampling, which may hinder the targeted biopsy approach. In this regard it is recommended that suction of areas of interest should be undertaken as a guiding point. Furthermore, CLE requires a long learning curve in order to acquire the skill needed to interpret the images with sufficient accuracy. 45 It is also important to note that patients allergic to fluorescein are unable to undergo confocal endomicroscopy.
Optical coherence tomography (OCT)
OCT is an endoscopic diagnostic tool which enables cross-sectional tissue images to be obtained as a result of the backscattering of light. 46 Analysis of backscattering comes courtesy of the principle of low coherence interferometry where incident light typically at the near infrared range is split and directed either towards the tissue in question or a positioned mirror. The backscattered tissue light is combined with the reflected mirror source, and measurement of backscattering at different depths is obtained through alteration of mirror position. Through such methodology, it is estimated that a scanning depth of 1–3 mm is achieved.
Poneros et al. utilised endoscopic OCT in 121 patients and characterised specialised IM by the absence of the layered structure of normal squamous epithelium and the vertical ‘pit and crypt’ morphology of gastric mucosa, the presence of a disorganised architecture with inhomogeneous tissue contrast and an irregular mucosal surface as well as the presence of submucosal glands. Prospective analysis revealed a sensitivity of 97% and specificity of 92%. 47 Another study utilised OCT to determine intramucosal carcinoma and high-grade dysplasia in BO, and noted a sensitivity and specificity of 83% and 75% respectively. 48 Qi et al. utilised OCT to develop a computer-aided algorithm for the diagnosis of dysplasia in BO and demonstrated a sensitivity of 82%, specificity of 74% and accuracy of 83%. 49 A further study observed a sensitivity of 66% and specificity of 57% for the diagnosis of specialised IM on the basis of an irregular architecture and good image penetration. 50 An additional prospective double-blinded study of 33 patients noted a sensitivity of 68%, specificity of 82%, positive predictive value of 53%, negative predictive value of 89% and diagnostic accuracy of 78% during endoscopic OCT detection of dysplasia. 51 More recently a feasibility study of volumetric laser endomicroscopy courtesy of Nvision was performed, with real-time images obtained of the mucosa and submucosa in 87% of BO patients. 52
OCT has also been employed during a radiofrequency ablation (RFA) treatment response study of 33 patients which demonstrated that the presence of OCT visible glands soon after RFA correlated with the presence of residual BO at follow-up with a sensitivity of 83.3%, specificity of 95% and accuracy of 90.6%. 53
OCT has the potential to enhance the operator’s morphological understanding of tissue in real time enabling real-time treatment instigation. Yet at present studies demonstrate variation in both sensitivity and specificity which may be due to the limited scanning depth and subjectivity of operator interpretation.
Molecular biomarkers
Endoscopy incorporating molecular biomarkers is a promising avenue in the diagnosis of BO. Recent work has targeted the potential for lectins as a diagnostic tool with a highly significant correlation between wheat germ agglutin fluorescence and the degree of dysplasia (p = 0.0002). 54 Sturm et al. developed the ASY* peptide labelled with fluorescein (FITC) and applied this conjugate topically to 25 oesophageal cancer patients. Results demonstrated a 3.8-fold greater fluorescence intensity for oesophageal cancer patients compared with BO and squamous epithelium with a sensitivity of 75% and specificity of 97%. 55 Another study, utilised FITC labelled to the peptide sequence SNFYMPL on resected BO specimens and demonstrated specific binding for dysplasia which was statistically significant (p < 0.01). 56 More recently, AFI was utilised on a three-biomarker panel of aneuploidy, p53 and cyclin A demonstrating a diagnostic accuracy for high-grade dysplasia/early cancer superior to AFI-negative areas (p < 0.001). 57
Endoscopic-based biomarker imaging is a novel field and could further tackle the problem of missed lesions secondary to insufficient visualisation and operator skill. However, BO is plagued by limited specific cell surface biomarker targets and we hope that further research will shed more light on this translational intervention.
Closing remarks
This review provides insight into the importance of astute endoscopic performance in determining a diagnosis of BO and subsequently AC. Despite the gold standard of WLE, additional platforms do exist to enhance the diagnostic process such as NBI and CLE. More recently OCT and a molecular-based approach through fluorescent biomarker conjugation have been explored in the hope of further enhancing diagnosis. The Preservation and Incorporation of Valuable endoscopic Innovations (PIVI) initiative courtesy of the The American Society for Gastrointestinal Endoscopy (ASGE) recommends a need for imaging systems that help to limit the need for biopsy sampling with development of technologies that have a per patient sensitivity of >90% and specificity of 80% for the detection of high-grade dysplasia/AC. 58 A large scale review and Delphi consensus for the management of BO conditionally recommends the use of HRE with targeted biopsy and against the use of advanced endoscopic imaging for routine use. 59 We look forward to further movement in this direction.
Summary points
Chromoendoscopy (CE): dye application for tissue surface enhancement. Varied sensitivities/ specificity and inter-observer agreement. NBI: enhanced mucosal surface architecture and vascular imaging with no dye use. Allows for fewer random biopsies. No consensus on mucosal and vascular pattern classification. FICE: enhanced mucosal surface contrast without dyes. Limited study data on effectiveness. i-Scan: surface, contrast and tone enhancement focused on lesions and mucosal irregularities. Large scale studies on effectiveness lacking. AFI: relies on natural tissue fluorescent properties. High sensitivity for dysplasia but associated high false positives. EC: subcellular structure analysis. Poor image interpretation accuracy and use of dyes needed. CLE: histological imaging in real time enabling targeted biopsy sampling. Image interpretation requires a long learning curve. Associated contraindications with fluorescein. OCT: cross-sectional imaging tool. Sensitivity and specificity variation with limited scanning depth. Molecular based biomarkers at endoscopy: imaging based on cell surface markers. Limited biomarker targets currently.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
