Abstract
Background
EndoFaster is novel device able to perform real-time ammonium measurement in gastric juice allowing H. pylori diagnosis during endoscopy. This large study aimed to validate the accuracy of EndoFaster for real-time H. pylori detection.
Methods
Consecutive patients who underwent upper endoscopy in two centres were prospectively enrolled. During endoscopy, 4 ml of gastric juice were aspirated to perform automatic analysis by EndoFaster within 90 seconds, and H. pylori was considered present (>62 ppm/ml) or absent (≤62 ppm/ml). Accuracy was measured by using histology as gold standard, and 13C-urea breath test (UBT) in discordant cases. Accuracy, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) were calculated.
Results
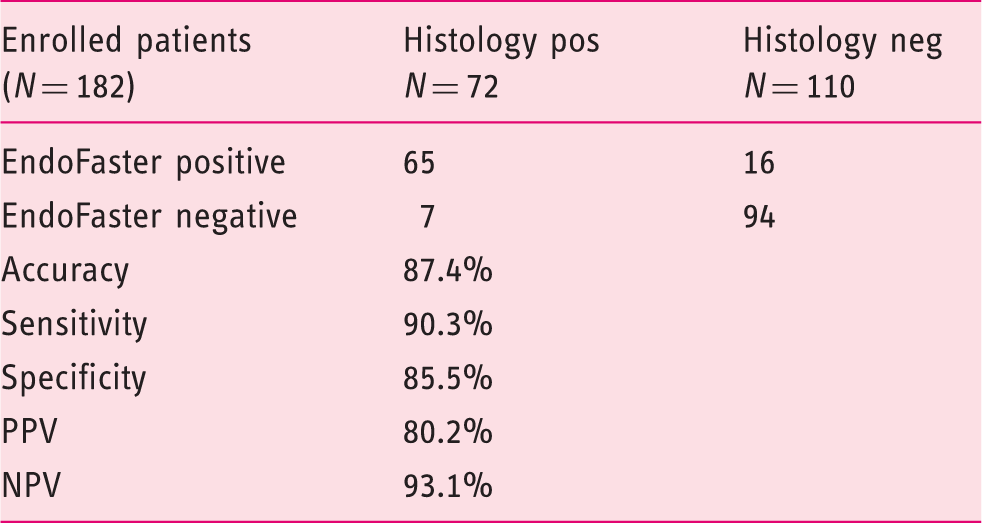
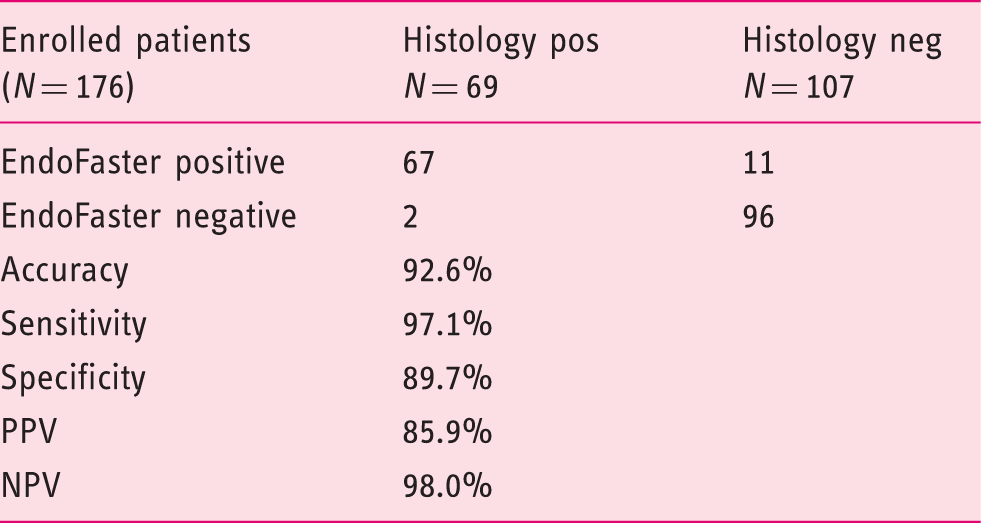
Overall, 189 patients were enrolled, but in seven (3.4%) the aspirated gastric juice amount was insufficient to perform the test. The accuracy, sensitivity, specificity, PPV, and NPV were 87.4%, 90.3%, 85.5%, 80.2%, 93.1%, respectively, and 92.6%, 97.1%, 89.7%, 85.9%, 98.0%, respectively, when H. pylori status was reclassified according to the UBT result in discordant cases.
Conclusions
This study found a high accuracy/feasibility of EndoFaster for real-time H. pylori diagnosis. Use of EndoFaster may allow selecting those patients in whom routine gastric biopsies could be avoided.
Introduction
Helicobacter pylori infection invariably causes chronic active gastritis, which is the first step starting the carcinogenetic cascade of gastric cancer. 1 Indeed, such an infection accounts for most cases of gastric cancer, the risk being about 6-fold greater in infected than in uninfected subjects, with a population attributable fraction as high as 74.7%. 2 Similarly, H. pylori is the main cause of low-grade B-cell lymphoma of the stomach, and peptic ulcer disease.3–5 Therefore, H. pylori infection is widely involved in the pathogenesis of the most relevant gastroduodenal diseases, which are distinctly infrequent when the infection is absent.
Current European guidelines suggest to search for H. pylori infection by using upper endoscopy with biopsies in dyspeptic patients with an increased risk of gastric cancer (over a local age cut-off point) as well as in those patients with alarm symptoms (bleeding, anaemia, weight loss, persisting vomiting, and dysphagia).
6
However, the prevalence of H. pylori infection is decreasing in Western countries. A recent US nationwide study found that H. pylori prevalence is less than 15% in those patients who underwent upper endoscopy.
7
Therefore, H. pylori infection is absent in the majority of patients who underwent upper endoscopy and histological examination. The availability of an accurate test able to disclose H. pylori presence during endoscopy could allow to select those patients deserving biopsy mapping of gastric mucosa from those in whom biopsies are most likely useless, time-consuming, and costly. A novel device (EndoFaster21-42) able to perform real-time – i.e. during endoscopy – ammonium measurement in gastric juice has been introduced (Figure 1). Some preliminary data showed that EndoFaster accurately predicts H. pylori presence on gastric mucosa with a sensitivity of 96.7% and specificity of 94.3%, with an accuracy equal to that of 13C-urea breath test (UBT).8,9
The EndoFaster.
This large, prospective study aimed to validate the performance of such a tool for real-time H. pylori detection in patients who underwent upper endoscopy.
Methods
Study population
A prospective study was conducted in two Endoscopy Units (Rome, Milan). Consecutive patients who were referred from their General Practitioners for upper endoscopy due to dyspeptic symptoms were included in the study. Patients were excluded when one of the following conditions was present: proton pump inhibitor or antibiotic therapies in the previous 4 weeks, previous H. pylori eradication therapy, gastric surgery, neoplasia, liver cirrhosis, severe renal, cardiac, or respiratory disease as well as those on anticoagulant treatment. The study was approved by the local Ethical Committees (Rome: N = P/1182/CE/2012; Milan: N = CE ICH 205). All patients gave written informed consent to participate.
Endoscopic procedure
All patients underwent conventional white-light upper endoscopy with video-endoscopes. Standard biopsy sampling according to updated Sydney System was performed. 10 In detail, two biopsies were taken from the antrum (within 2 cm from the pylorus; anterior and posterior wall), two from the angulus, and two from the gastric body (anterior and posterior wall). The main endoscopic findings observed during endoscopy were reported on a standardized endoscopic report.
EndoFaster
The study has been conducted with EndoFaster 21-42 (NISO Biomed S.r.l, Turin; Italy), an innovative device which performs automatic gastric juice analysis during upper endoscopy and communicates the result in real time. The device is interposed between the endoscope and the suction system, utilizing 4 ml of the gastric juice aspirated at the beginning of endoscopy. H. pylori diagnosis is based on the determination of ammonium concentration, as a consequence of the urease activity of the bacterium within 30–90 seconds, that is during endoscopy.8,9 The test was considered positive for H. pylori infection when the ammonium concentration was 62 ppm/ml and negative when ≤62 ppm/ml. As compared with the prototype (MT 21-42) used in previous studies,8,9 the EndoFaster 21-42 has an improved performance since it requires a lower volume of gastric juice for performing the test. In addition, the actual cut-off 62 ppm simply depends on a new parameter – that is a corrective for the temperature – introduced in the software of the new version of device. Therefore, the 62 ppm cut-off of the device exactly corresponds to 60 ppm of the prototype. For the purpose of our study, EndoFaster analysis was performed before histological sampling.
H. pylori assessment
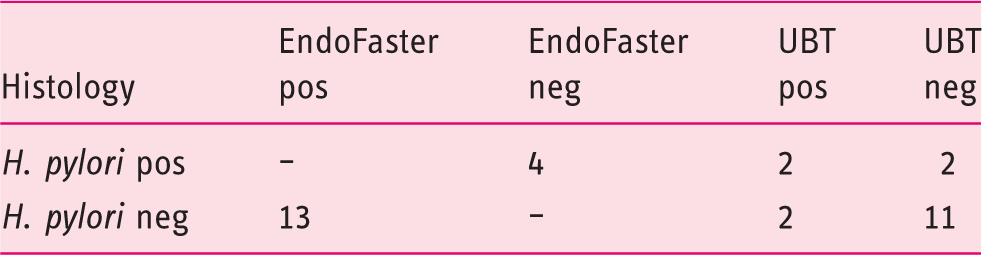
Histological assessment was performed for both detection of H. pylori infection and evaluation of gastritis. In detail, biopsies were stained with haematoxylin–eosin for gastritis assessment and with the modified Giemsa staining for detection of H. pylori. In cases of disagreement between EndoFaster and the histological examination for H. pylori infection, a UBT, with a cut-off value of 3.5‰ according to the manufacturer’s recommendations, was performed and the infection status was accordingly reclassified.
Statistical analysis
The EndoFaster accuracy, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) for H. pylori assessment were calculated by using histology as gold standard, and with the UBT for the divergent cases. In order to assess the sample size, we assumed a 30% H. pylori prevalence and an unfeasibility of the EndoFaster analysis in 10% of cases (i.e. insufficient gastric juice amount). Thus, 180 patients were needed to show an EndoFaster accuracy for H. pylori diagnosis of 90% with a 95% confidence interval (CI) between 86% and 94%.
Results
Accuracy of EndoFaster for H. pylori diagnosis with only histology as gold standard
13C-Urea Breath Test (UBT) results in discordant cases
Accuracy of EndoFaster for H. pylori diagnosis following bacterial status re-classification according to UBT
Discussion
H. pylori infection remains the major factor involved in both neoplastic and non-malignant gastroduodenal lesions, which are infrequent when the infection is absent.2–5 Indeed, international guidelines advise to search for and to treat the infection in dyspeptic patients. 6 Luckily, the prevalence of H. pylori in developed countries is decreasing, so that probability of detecting the infection at endoscopy is less than 20–35% according to recent data, or even less when considering only those patients without macroscopic lesions.7,11 Therefore, the majority of gastric biopsies currently performed are negative, time-consuming, costly and, most likely, clinically worthless. This undesirable circumstance may be resolved when an accurate test is able to disclose H. pylori presence during endoscopy. The proposed use of a new ultra-fast rapid urease test did not solve the problem since biopsies are still required to perform the test, and an accurate result is achieved only after 5 minutes, that is, when the endoscopic examination has generally ended. 12 On the contrary, EndoFaster was introduced to allow a real-time detection of H. pylori in the stomach.7,8 The present study clearly showed a very high accuracy of this tool for real-time diagnosis of the infection. Overall, by applying EndoFaster, biopsies would have been avoided in 94 (51.6%) patients with an EndoFaster-negative result, without significantly overlooking the infection, the NPV being as high as 98%. The risk of such a procedure is to miss precancerous conditions (atrophy/intestinal metaplasia) in uninfected patients. However, a recent meta-analysis found that diffuse intestinal metaplasia – which is the only precancerous condition deserving a 3-year interval follow-up according to current European Guidelines 13 – was overall present in 13% (9–17%) of patients, with values widely ranging in different countries. 14 Indeed, in a recent nationwide Italian study, diffuse intestinal metaplasia was present in only 3% of patients, and it was significantly associated with the H. pylori. 11 Therefore, the use of EndoFaster would be valuable to select those patients with H. pylori infection deserving gastric biopsies, allowing gastric sampling to be avoided in the majority patients who are uninfected. 15 However, a cost-effective analysis study taking into account the prevalence of diffuse intestinal metaplasia in H. pylori-negative patients in different populations is needed to test the consistency of such an approach. As far as the cost is concerned, near 20 euros is the charge of a single test performed by using an EndoFaster device which is supplied on loan for use.
In conclusion, this large study established the high accuracy of EndoFaster in detecting H. pylori infection. Such a tool could be useful to select those patients in whom gastric biopsies are probably useless.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
