Abstract
Introduction
Radiofrequency ablation (RFA) is a well-validated treatment of dysplastic Barrett's esophagus. Other indications of endoscopic RFA are under evaluation.
Results
Four prospective studies (total 69 patients) have shown that RFA achieved complete remission of early esophageal squamous intra-epithelial neoplasia at a rate of 80%, but with a substantial risk of stricture. In the setting of gastric antral vascular ectasia, two prospective monocenter studies, and a retrospective multicenter study, (total 51 patients), suggest that RFA is efficacious in terms of reducing transfusion dependency. In the setting of chronic hemorrhagic radiation proctopathy, a prospective monocenter study and a retrospective multicenter study (total 56 patients) suggest that RFA is an efficient treatment. A retrospective comparative study (64 patients) suggests that RFA improves stents patency in malignant biliary strictures.
Conclusions
Endoscopic RFA is an upcoming treatment modality in early esophageal squamous intra-epithelial neoplasia, as well as in gastric, rectal, and biliary diseases.
Keywords
Introduction
Radiofrequency ablation (RFA) allows the destruction of unwanted tissue by heat. It is a widely used treatment of cardiac arrhythmia, cancer, varicose veins, and uterine bleeding. RFA has become a standard of care for dysplastic Barrett's esophagus (BE) ever since interventional endoscopy devices were developed.1,2
This review focuses on the modalities of use and the performances of endoscopic radiofrequency (RF) in various diseases of the digestive tract, specifically in emerging indications such as early esophageal squamous neoplasia gastric antral vascular ectasia (GAVE), radiation proctopathy (RP), and biliary strictures. Peroperative (laparoscopy or laparotomy) and percutaneous RFA techniques are not addressed here.
We searched through the NCBI PubMed system database by using individual free word or MeSH term “radiofrequency” cross-referenced with other relevant terms, as well as a manual search and review of reference lists, up to December 2014.
Biophysical principles of endoscopic radiofrequency ablation
All current endoscopic RFA catheters use an electric generator connected to bipolar electrode arrays to deliver energy to the tissue (Figure 1). Electricity travels in the radiofrequency range (450–500 kHz) between the alternating positive and negative poles along the electrodes arrays, locally delivering energy (heat) around the RF electrode. This results in protein coagulation and destruction of micro-vessels, with consequent tissue death (ablation). For effective tissue destruction, endoscopic disposable electrodes are applied in direct contact with the targeted tissue. A pre-determined spacing and geometry of the electrical contacts and pre-set parameters (dose-energy, power) of the generator aim to achieve a consistent ablation depth. Typically, when applied against the digestive wall, the ablation goes as deep as the muscularis mucosae (700–800 microns deep). Current therapeutic clinical protocols (energy power, energy density, number and duration of impacts) have been determined based on the results of dosimetric animal and pre-clinical human studies that are not detailed herein.3−11
Biophysical principles and two different types of radiofrequency catheters. (a) Schematic view of energy traveling along the alternating positive and negative poles through the electrodes arrays of a RF catheter. (b) Close-up view of over-the-scope RF probe (13 mm × 20 mm) used for focal ablation (Halo90, Covidien GI Solutions, Sunnyvale, California, USA). (c) Close-up view of over-the-wire RF probe (31 mm diameter) used for biliary and pancreatic ablation (Habib™ EndoHPB, EMcision Ltd, London, UK).
Emerging indications of endoscopic radiofrequency in the esophagus
Endoscopic radiofrequency is a well-validated treatment of dysplastic BE.1,2 It has also been evaluated as a treatment of early esophageal squamous cell carcinoma (ESCC). Early ESCC is usually addressed by endoscopic ablation techniques, such as endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD), or by surgical resection. Pouw et al. first reported the complete eradication of ESCC with surrounding high-grade intra-epithelial neoplasia (HGIN) in a 66-year-old patient. 12 Bergmann et al. published a single-center, prospective evaluation of 29 Chinese patients with 3 cm or larger esophageal Lugol unstained lesions containing flat-type ESCC (one patient) or HGIN (10 patients) or moderate-grade intra-epithelial neoplasia (MGIN, 18 patients), treated by RF. 13 After a thorough chromo-endoscopic and histological evaluation, all patients had a circumferential RF session (10–12 J/cm2, 40 W/cm2, twice, with or without scraping of the ablated tissue). At 3 months of follow-up, four patients with residual Lugol unstained lesion (two MGIN, two HGIN) were retreated by focal RF, according to protocol. At 12 months, the single patient with ESSC had persistent HGIN. Overall, complete response (as defined by the absence of MGIN or worse) rates were 86% at 3 months and 97% at 12 months. Interestingly, absence of intra-epithelial neoplasia was achieved in 83% of patients at 12 months. Four patients (14%) developed esophageal strictures, with favorable outcome after 2–4 sessions of balloon dilatation. They had received double-impacts of 12 J/cm2 RF regimens (with or without scraping). 13 Most recently, Wang et al. have reported on the efficacy of RF (circumferential initially, then focal if any residual lesion at 3 months) for the treatment of ultralong (>10 cm) ESCC (four patients) and HGIN (three patients). Six patients (86%) had a complete histological response at 6 months, and two patients (29%) required endoscopic dilatations for symptomatic structuring after treatment. 14
A prospective case series from Van Vilsteren et al. 15 evaluated the safety and efficacy of RF (median two sessions) for the treatment of ESCC (three patients) or HGIN (10 patients). Interestingly, nine patients had undergone an EMR of non-flat Lugol unstained lesions in the initial evaluation. During RFA sessions, complications occurred in three patients: two mucosal lacerations (where previous EMR had been performed), and one submucosal hematoma. Three stenosis occurred during follow-up (one of which was complicated by perforation after balloon dilation). The absence of squamous intra-epithelial neoplasia was assessed by chromo-endoscopy and biopsies during a median follow-up of 17 months in all patients. 15 A prospective cohort from the United Kingdom provided less enthusiastic results in 20 patients with HGIN (12 patients) or ESCC (eight patients) followed during a median period of 24 months after RFA (1–3 sessions, median 1 session). Five of them had an EMR of non-flat lesions before RFA. While 10 patients (50%) had no signs of dysplasia after 1 year of follow-up, six patients (30%) progressed to invasive cancer during the same period. One additional patient developed a cancer after the end of the protocol. Four patients (20%) developed esophageal strictures, which resolved with balloon dilation. 16 Other multimodal non-surgical approaches of ESCC, combining RFA, EMR and ESD, have been proposed by others, with encouraging results. 17
Main studies evaluating endoscopic circumferential (+/− focal) radiofrequency ablation (RFA) for esophageal squamous cell intra-epithelial neoplasia (IEN) and early esophageal squamous cell carcinoma (ESCC)
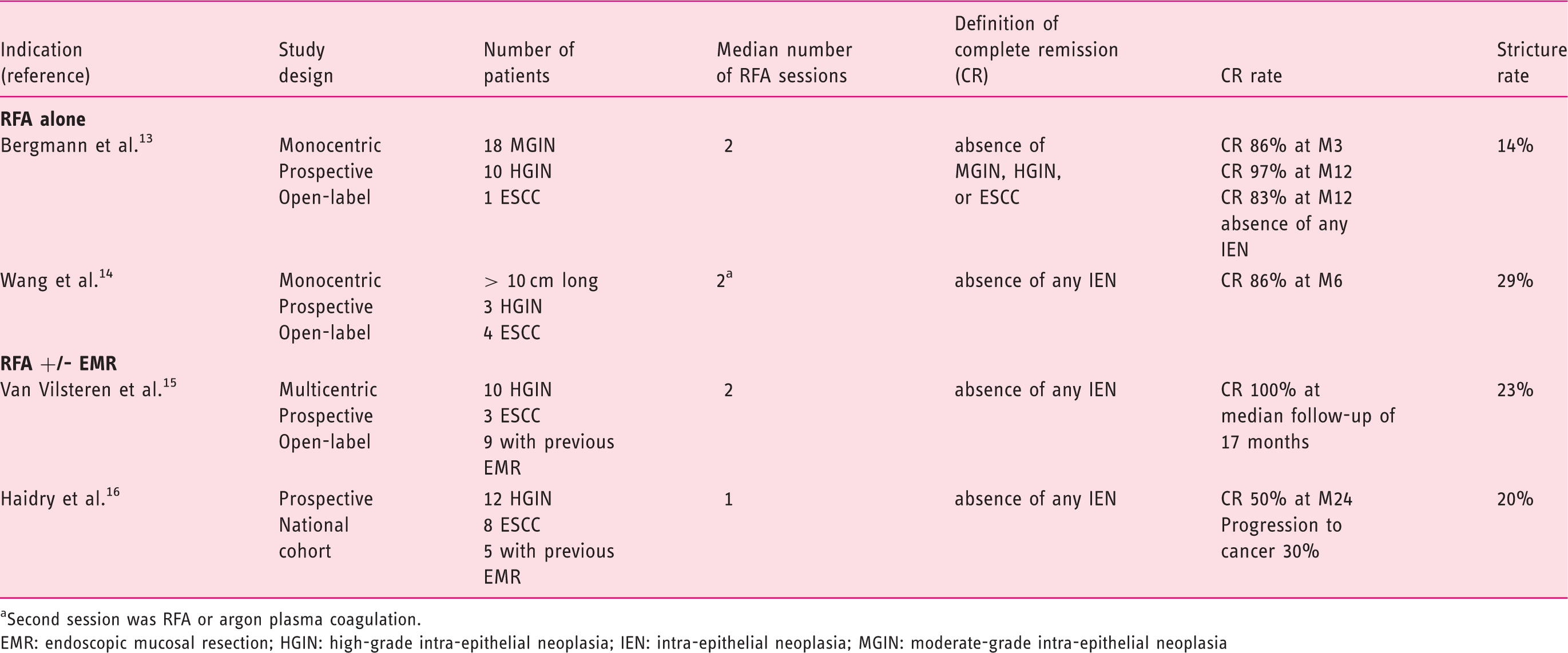
Second session was RFA or argon plasma coagulation.
EMR: endoscopic mucosal resection; HGIN: high-grade intra-epithelial neoplasia; IEN: intra-epithelial neoplasia; MGIN: moderate-grade intra-epithelial neoplasia
Emerging indications of endoscopic radiofrequency in gastric diseases
Gastric antral vascular ectasia
Gastric antral vascular ectasia is a rare cause of gastrointestinal bleeding. Endoscopic presentation consisting of antral erythematous stripes forming a radial pattern around the pylorus, also called “watermelon stomach”, is highly specific. 23 Iron replacement, red blood cell (RBC) transfusion, and treatment of underlying conditions (including cirrhosis, diabetes, scleroderma, and chronic renal diseases) are the mainstay of conservative management. Pharmacologic treatments currently have no definitive place in the therapeutic strategy. 24 Antrectomy is considered the only cure, but it has a 10%, comorbidity related, mortality rate.25,26 Endoscopic therapy mostly uses thermal techniques, aiming to achieve a widespread eradication of mucosal capillary ectasia in the antrum, with subsequent re-epithelialization, without causing damage deeper than the superficial submucosa. Argon plasma coagulation (APC) is the preferred endoscopic treatment of GAVE on account of being easy to use, relatively safe, and at an acceptable cost. However, it requires multiple sessions,27,28 and overall, discontinuation of transfusion dependence is currently achieved in only one-third of patients. 23
RFA has strong benefits when it comes to achieving ablation of these broad but superficial stomach lesions. The endoscopic procedure is typically performed as follows (Figure 2). An over-the-scope (OTS) RF electrode pivoting probe (Halo90, 20 × 13 mm or Halo90ultra, 40 × 13 mm, Covidien GI Solutions, Sunnyvale, California) attached to a cap placed at the distal end of an endoscope is applied on the pathologic mucosa and bleeding areas. Two consecutive impacts of energy are delivered in the same mucosa area with a pre-set energy density of 12–15 J/cm2 and a power density of 40 W/cm2. The coagulum is not scraped to promote haemostasis, like in the case of BE. Double-impacts are repeated until complete eradication of the lesions is achieved. The endoscope is repeatedly withdrawn to clean the probe, and to rotate the cap in order to facilitate access to the whole antrum circumference. A minimal time interval of 6 weeks is usually chosen between two RFA sessions. Patients are kept on proton-pump inhibitors in between.
Endoscopic view of focal radiofrequency ablation (RFA) for gastric antral vascular ectasia (GAVE). (a) Antrum before treatment. (b) Over-the-scope (OTS) focal ablation catheter (Halo90, Covidien GI Solutions, Sunnyvale, California, USA) placed at a 6 o'clock position. View of the antrum after application of the first pair of radiofrequency ablation pulses (whitened area). (c) OTS focal ablation catheter at a 9 o'clock position. View of the antrum after multiple applications of radiofrequency ablation pulses. (d) OTS focal ablation catheter at a 12 o'clock position. View of the pylorus after multiple applications of radiofrequency ablation pulses. E. Antrum after circumferential RF applications.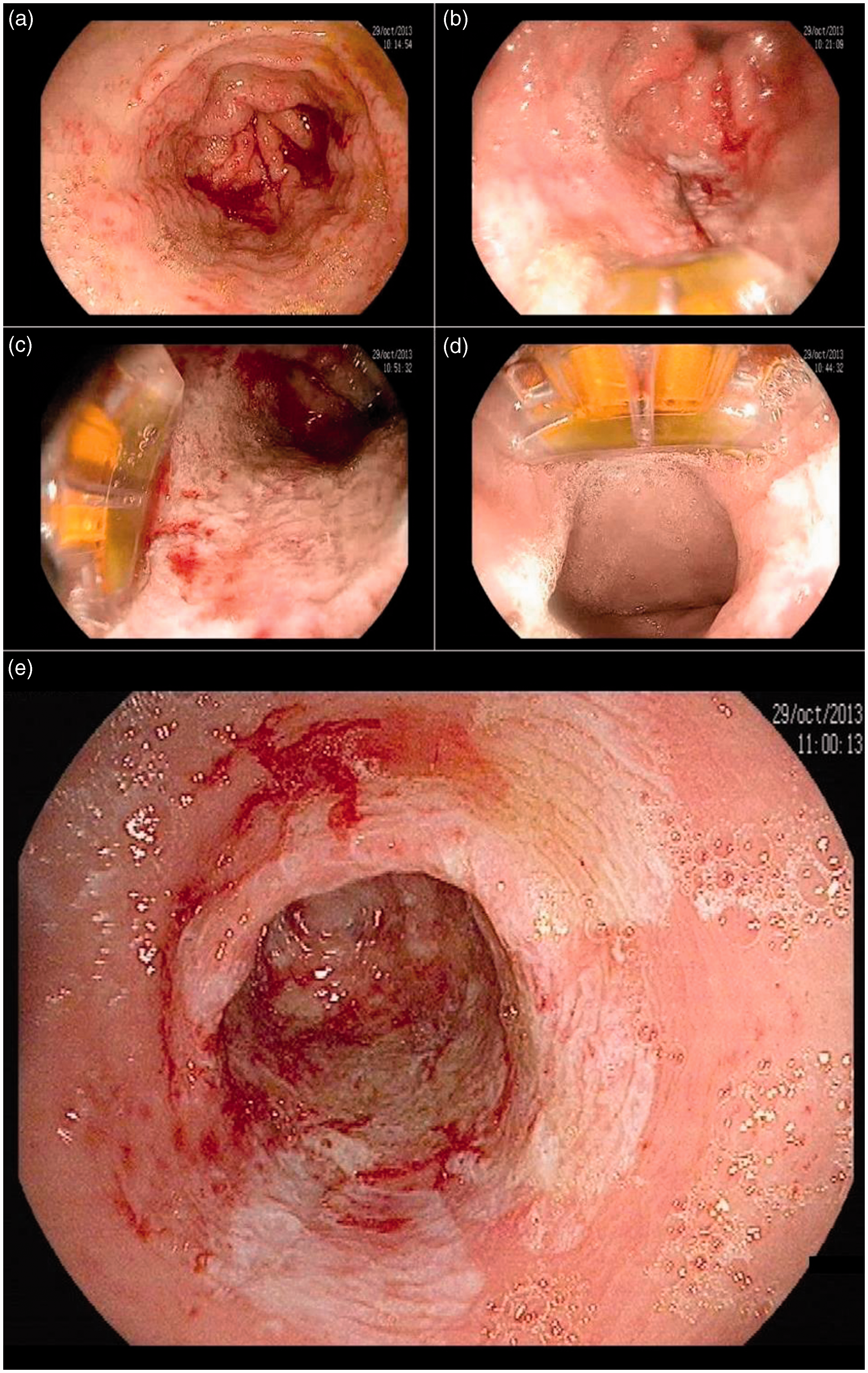
Main studies evaluating endoscopic radiofrequency ablation for hemostasis of gastrointestinal diseases
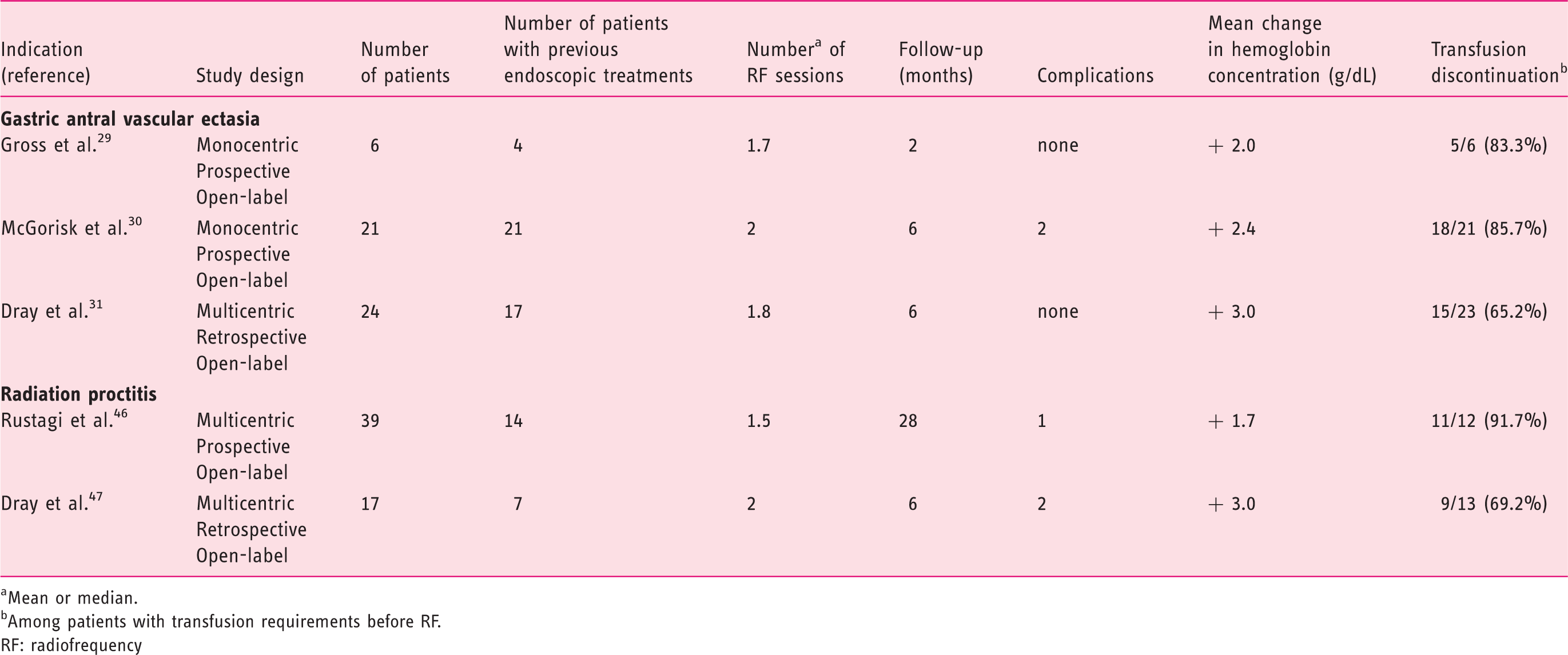
Mean or median.
Among patients with transfusion requirements before RF.
RF: radiofrequency
Approximate prices of radiofrequency ablation (RFA) systems and catheters
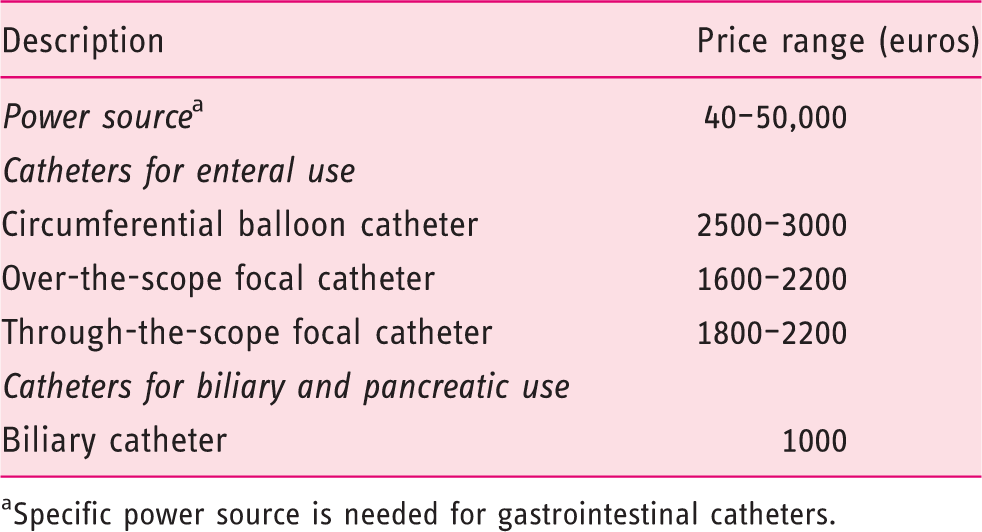
Specific power source is needed for gastrointestinal catheters.
Other indications in the stomach
RFA has been used in other gastric conditions presenting with superficial and broad lesions. Three patients presenting with gastric dysplasia were treated with two RFA sessions. Extensive biopsy follow-up within 18 months after treatment displayed negative results for dysplasia. 32 However, this original approach cannot be recommended at this stage. A combined treatment of RFA and APC has been reported efficacious in a patient presenting with hemorrhagic radiation gastropathy for whom hyperbaric oxygenotherapy had failed. 33 Although anecdotal, these reports provide information on the feasibility and safety of RFA as second-line treatment of several gastric diseases.
Emerging indications of endoscopic radiofrequency in rectal diseases
Radiation proctopathy
Chronic RP occurs years after pelvic radiation therapy, in 5–20% of patients. 34 Rectal bleeding, diarrhea, and pain are the usual symptoms. RP is due to ischemic endarteritis of the submucosal arterioles, submucosal fibrosis, and mucosal angiectasia. A symptomatic treatment is sufficient in most cases. However, patients presenting with anemia and/or abundant bleeding require a specific treatment. 34 These elderly patients often present with anticoagulants and antiplatelet drugs which worsen the bleeding and make it more difficult to treat. The local treatment of RP consists in an ablation of the abnormal mucosal micro-vessels of the rectum, allowing subsequent re-epithelialization. Endoscopic APC is a widely available, easy to use, relatively safe and cost-effective treatment of RP.35−37 Notwithstanding, APC often requires multiple sessions and fails in up to 20% of cases, specifically in the setting of active bleeding, extensive lesions, and/or distally located lesions (ano-rectal junction).35,36 Complications occur in 19 to 47% of cases. They are mostly benign (colon distension, rectal pain, tenesmus), but may be severe (perforation, ulceration, fistula and strictures).34,37,38 In refractory cases (especially in the setting of distally located lesions), topical application of formalin can be discussed but its local morbidity is an issue. In the study by de Parades et al., 39 formalin application was effective in 23 out of 33 patients (13 had complete cessation of bleeding, and 10 had only minor bleeding). Six anal or rectal non-malignant strictures occurred (18%). Anal incontinence worsened or appeared in nine patients (27%), but 33% of all patients had received radiation in the setting of anal cancers, which probably enhances the risk of incontinence to a higher degree. 39 Indeed, in one prospective series focusing on RP following radiation therapy above the anal sphincter (for prostate cancer), formalin application tolerance was as good as APC tolerance (no worsening of fecal incontinence in either group, no development of stenosis). 40
RFA has recently been evaluated in the setting of RP (Table 2). The idea is to accomplish superficial and controlled ablation of the superficial mucosal lesions (Figure 3). Initial case reports (11 patients overall) have provided encouraging results.41−45 Since then, two short retrospective series have been published.46,47
Endoscopic view of focal radiofrequency ablation (RFA) for radiation proctitis. (a) and (b): Retroflexion view of the rectum before treatment, showing multiple telangiectasia. (c) Through-the-scope (TTS) focal ablation catheter (Channel, Covidien GI Solutions, Sunnyvale, California, USA) placed against the targeted rectal pathological tissue. (d) View of the rectum after a pair of radiofrequency ablation pulses (whitened area). (e) TTS focal ablation catheter is applied against the targeted rectal pathological tissue, next to the previously treated area. (f) Retroflexion view after circumferential RFA of the rectum. (g) Retroflexion view of the rectum 3 months after RFA treatment, with barely visible telangiectasia.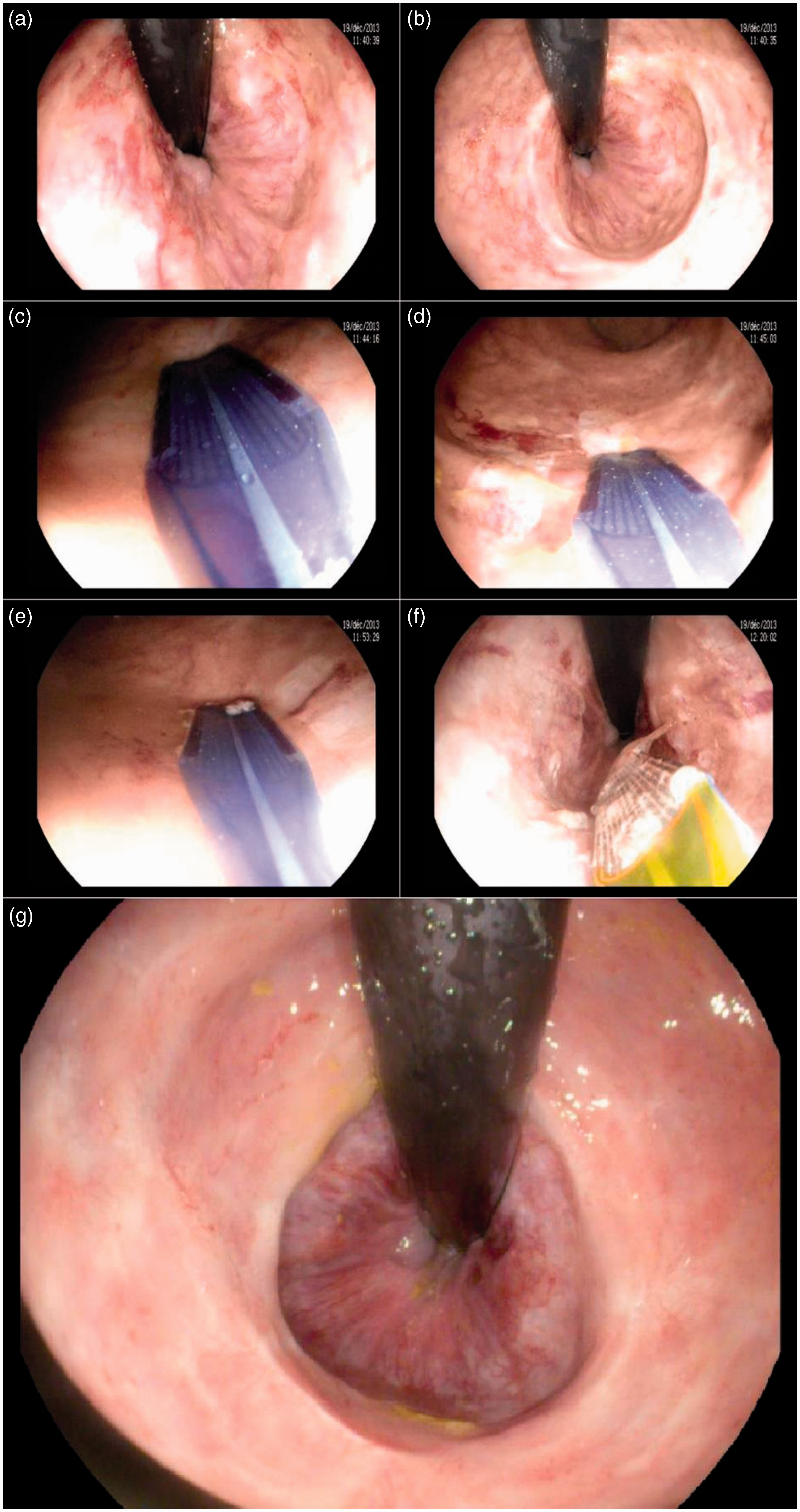
In the multicenter open-label study by Rustagi et al., 46 39 male patients with hemorrhagic RP were treated with RFA (72% undergoing a single session). Double-impacts were delivered per site (density 12 J/cm2, power 40 W/cm2), without scraping of the coagulum, until up to 270° of the rectum’s circumference was treated in the same session, in order to prevent rectal stenosis. Fourteen patients (39%) had received a previous endoscopic treatment for RP (mostly APC). Mean follow-up was 28 months. Bleeding stopped in all 39 patients (100%) and endoscopic RP severity score improved in 38 patients (96%), with subsequent increase in mean hemoglobin level from 11.8 g/dl to 13.5 g/dl. Discontinuation of RBC transfusion in 11 out of 12 patients (92%) as well as discontinuation of iron therapy in 14 of out 17 patients (82%) was observed. Treatment was well tolerated in most cases (but a significant arterial bleeding controlled by endoscopic clipping in a single patient). 46
In an open-label, retrospective, multicenter study, 47 we included 17 patients (12 males) with chronic hemorrhagic RP. Seven patients had undergone a previous treatment (APC in most cases). One to four RFA (mean 1.8) were performed, with pre-set energy density of 12 to 15 J/cm2 and power density of 40 W/cm2. Two patients developed an asymptomatic rectal ulceration, and two other patients received additional APC treatment during follow-up because of recurrent bleeding. Global and bleeding symptom scores significantly decreased in 16 patients (94%). In the 13 patients who were transfusion-dependent before RFA, the mean number of RBC packs over 6-month periods decreased from 7.6 prior to RFA to 0.9 in 10 patients, and remained unchanged in the three remaining patients. Discontinuation of transfusion was achieved in 9 out of these 13 patients (69.2%). The mean hemoglobin level increased in all 17 patients, from 8.3 g/dl to 11.3 g/dl overall.
These two series, in accordance with other case reports, suggest that RFA is feasible, quick and easy to perform, safe, and efficient, for the treatment of RP. Based on these preliminary results, we believe that double-impacts delivered per site at energy density of 12 J/cm2 are efficacious in most cases. Although there is no study comparing RF and APC yet, it seems that mucosal ablation depth is better controlled with RF, and that subsequent iatrogenic rectal ulcerations are less frequent with this innovative technique. In most cases, rectal RFA does not require a general anesthesia. A complete bowel cleansing using an oral preparation is warranted, as it prevents from the risk of colonic explosion due to fermentation gas and avoids additional rectal mucosal injury by enema tubes. Much like in gastric indications, using through-the-scope RFA catheters may prevent time-consuming and uncomfortable scope withdrawals and reinsertions. As proposed by Rustagi et al., circumferential treatment of the rectum should be discouraged due to the risk of rectal stenosis. 46 An important unsolved question concerns the cost-effectiveness of RFA, given how expensive RFA devices are (Table 3) and how inexpensive APC and formalin application are. Prospective, comparative and medico-economic studies with long-term follow-up would be of interest. Until such studies are conducted, RFA seems appropriate in patients for whom APC has failed, noticeably when contra-indications to other techniques exist.
Other indications in the rectum
Vavra et al. evaluated the use of RF in the setting of palliative care, in patients presenting with rectosigmoid cancer. 48 An Endoblate® RF probe (EMcision Ltd., London, United Kingdom) was used for tumor destruction in 10 patients. The probe was placed into the tumor, through the operating channel of a transanal endoscopic microsurgery system. Ablation (1–4 W, until 10% increase of impedance was achieved) was performed, and was immediately followed by surgical resection. There were no peroperative complications. Pathological assessment showed that 60% to 99% (mean 82%) of the tumor mass was destroyed. In two additional patients with refractory bleeding, RFA alone (without surgery, one and two sessions, respectively) stopped the hemorrhaging, without complication or relapse after 3 weeks of follow-up. This pilot study illustrated a smart translation from a percutaneous (liver lesions) to an endoscopic (luminal lesions) RF procedure, with promising results. 48
Emerging indications of endoscopic radiofrequency in biliary and pancreatic diseases
Biliary tract diseases
Self-expandable metal stents (SEMS) are the current standard of care for the palliative management of malignant biliary strictures. 49 Although metal stents have improved bile duct patency compared with plastic stents, occlusion often occurs within 6–8 months of stent placement due to tumor growth, sludge, or biofilm formation. RFA, because of its theoretical local effect on tumoral tissue, has recently been tested in terms of its ability to improve SEMS patency. 50
The procedure requires a disposable, bipolar, over-the-wire RFA catheter, suitable for endoluminal delivery of RF into the biliary tree (Habib™ EndoHPB, EMcision Ltd, London, UK). It consists of an 8 French (2.6 mm) catheter with a 180-cm long working length that can be deployed through endoscope working channels with a diameter of at least 3.2 mm (Figure 1). After biliary sphincterotomy, followed by a cholangiogram and by balloon dilation when appropriate, the probe is advanced up to the biliary stricture and ablation is performed. Once RFA is performed, a SEMS is placed according to routine technique.
Over the last 3 years, four studies including over 100 patients, most presenting with cholangiocarcinoma (>75%), have been published, documenting the technical feasibility and short-term success of RFA.7,9,10,51 Most of the data consist of retrospective case series with a limited number of patients, and with malignant biliary strictures of various etiologies. A recently published study attempted to overcome some of these limitations. Sharaiha et al. analyzed retrospectively 64 patients with malignant biliary strictures (cholangiocarcinoma and pancreatic cancer). 11 Patients who underwent RFA with metal stenting were compared with matched patients who were treated conventionally with metal stenting alone. Although overall technical success and SEMS patency rates were the same in both groups (100%), with no difference in terms of mean number of endoscopic retrograde cholangiographies (2.26 ± 1.00 in the RFA group vs. 1.94 ± 1.27 in the control group, p = 0.84), RFA was found to be a significant and independent predictor of survival in patients with end-stage cholangiocarcinoma and pancreatic cancer (hazard ratio 0.29, p = 0.01).
In the setting of a malignant disease with limited treatment options, this modality may prove to be beneficial compared with stenting alone. Randomized controlled trials and quality of life assessments are warranted to confirm these findings. One case of partial liver infarction was reported in a patient with a Bismuth IV stage Klatskin tumor after RFA procedure. 51 No other serious complications have been reported so far. According to our knowledge, the endobiliary RFA technique has not been tested so far for clearance of occluded SEMS in malignant biliary obstruction.
Hu et al. reported on nine patients presenting with refractory benign biliary stricture (post-operative stricture, liver transplant, and chronic inflammation). 52 Intraductal bipolar RFA was delivered at a power of 10 W for 90 s per strictured segment, followed by balloon dilatation with/without stent placement. All patients had an immediate stricture improvement after RFA with no severe adverse event. During follow-up (median of 12.6 months), benign biliary stricture resolution without the need for further stenting was achieved in four patients. This pilot study suggests that endobiliary RFA may be an interesting therapy option in refractory benign biliary strictures.
Overall, endobiliary RFA seems to improve SEMS patency in malignant biliary obstruction. Direct comparison of endobiliary RFA and dynamic phototherapy would be of interest to determine which technique is the best to be combine to SEMS placement, to prolong biliary stent patency in biliary malignant obstruction. 53
Pancreatic diseases
In the setting of obstructive tumors, such as unresectable locally advanced pancreatic cancers, RFA could, in theory, be considered as well. The histological effect and risk of post-operative complications have been addressed in in vivo animal models by laparotomy or with a prototype RFA probe (0.01 mm diameter) adjusted to a 19 G EUS-needle.54−56 Pancreatic EUS-RFA appeared to be feasible, effective, and relatively safe in a porcine model, but no human case has been reported yet.
Emerging indications for endoscopic ultrasound radiofrequency for lymph node ablation
Using the same principle as for advanced pancreatic cancers, EUS-RFA has recently been evaluated for mediastinal lymph nodes destruction in a non-survival porcine model. A total of 18 lymph nodes in eight pigs were ablated. No peroperative complication was reported and upon autopsy, no signs of damage to the surrounding tissue were observed. The area of lymph node ablation ranged between 8.0% and 53.2% (mean 17.6%), demonstrating feasibility but calling for substantial technical improvement. 57
Conclusion
RFA is a treatment modality which has recently gained interest in gastrointestinal endoscopy. Endoscopic RFA has rapidly become a standard of care in dysplastic BE. It is also an upcoming treatment modality in esophageal ESCC (Table 1), and in several gastrointestinal diseases, noticeably for hemostasis of extended vascular lesions of the stomach and the rectum (Table 2), 58 and to achieve prolonged biliary stent patency. Although some dosimetric studies have been conducted for biliary RF,4−11 we have found only one dosimetric study for RP 3 and none for GAVE. Such pre-clinical studies would be of interest to determine the most accurate parameters for treatment, such as energy density and the number of impacts. Although cost-effectiveness is an issue, clinical long-term effectiveness is to be addressed first. Indeed, all studies performed to date have included small numbers of patients, with short-term follow-up, and with very heterogeneous approaches to selection and end points.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
X Dray and G Rahmi have received consultancy fees from Covidien, GI Solutions.
