Abstract
Objective
The quantification of basophil activation by flow cytometry is a useful tool for the assessment of immediate-type responses to food allergens and the prediction of clinical tolerance in food allergy patients. The aim of this study is to investigate how the analysis of allergen-induced CD63 up-regulation by flow cytometry can be effective in monitoring the acquisition of clinical tolerance by specific oral desensitization in food allergy. To our knowledge, this is the first study to examine this topic.
Materials and methods
Three male patients affected by cow’s milk allergy underwent successful oral desensitization to cow’s milk. In order to monitor the acquired clinical tolerance that occurred after treatment, we performed laboratory tests for total and specific IgE, specific IgG4 and the Basophil Activation Test (BAT) both at baseline and at the end of the desensitization protocol.
Results
Using a fluorescent enzyme immunoassay, the comparison of specific cow’s milk antibodies before and after treatment showed a decrease of specific IgE levels, without reaching normal values, and an increase of specific IgG4 levels. A complete suppression of cow’s milk proteins (α-lactoalbumin, β-lactoglobulin and casein) induced CD63 regulation was observed in all three reported cases.
Conclusions
Using flow cytometry, food allergen-specific basophil responses could be monitored in order to identify an acquired tolerance induced by desensitization treatment. Although further studies are needed to develop this important new topic, it was interesting to note that the BAT seemed to be more sensitive and characterized by a close correlation with clinical tolerance.
Keywords
Introduction
Cow’s milk allergy is the most common food allergy in children, affecting approximately 2.5% of children under 3 years of age. In particular, researchers estimate the occurrence of gastrointestinal symptoms between 5% and 20%, depending on the diagnostic criteria and the study setting. 1 In this context, it is necessary to perform an accurate differential diagnosis to avoid several diagnostic mistakes, especially at a pediatric age. Fortunately, a loss of food hypersensitivity may occur in a high percentage of patients by the fifth year. 2 The quantification of basophil activation by flow cytometry (Basophil Activation Test – BAT) has proven to be a useful tool for the assessment of immediate-type responses to food allergens mediated by IgE3,4 and the prediction of clinical tolerance in food-allergic patients. 5
The first therapeutic choice for food allergy is avoidance of the responsible food by elimination diet, 6 but this approach is not always possible, especially for basic foods (such as milk or egg), because they have an essential role on psychophysical wellbeing and their complete exclusion from the diet is sometimes very difficult (for example, in the case of hidden allergens).
A specific oral desensitization treatment could be considered a good alternative for those patients who did not acquire a clinical tolerance and cannot avoid the ingestion of incriminated food. Successful immunotherapy has been correlated with changes in cytokine production (IL-4 and INF- γ) 7 and allergen-specific IgE and IgG4 antibodies.8–10 Analyzing the data from the literature, a significant decrease in specific IgE and a significant increase in specific IgG4 levels is possible to note at the end of oral food allergy desensitization8,9; and this appears to be in line with a switch from a Th2 to a Th1 response.
The aim of this study is to investigate how the analysis of allergen-induced CD63 up-regulation using flow cytometry can be effective in monitoring the acquisition of clinical tolerance by specific oral desensitization in food allergy. The lack of updated data in the literature on the role of the BAT in monitoring the efficacy of oral desensitization treatment in food allergy prompted us to undertake this study.
Methods
We enrolled three male patients, ranging from 10–11 years in age, with documented IgE-mediated cow’s milk allergy. In early childhood, they had experienced several episodes of urticaria, angioedema, asthma and abdominal pain that occurred after accidental ingestion of milk proteins.
The diagnosis of food allergy was based on their clinical history and a positive allergy work-up, including allergy tests (skin prick tests and in vitro laboratory tests), the BAT and a double-blind placebo-controlled food challenge (DBPCFC), considered the gold standard in food-allergy testing. The skin prick tests, considered positive with a wheal reaction >3 mm in diameter, were performed by using a commercial extract of milk protein (Alk Abellò, Madrid, Spain) and fresh cow’s milk (prick-by-prick method). Specific IgE and IgG4 (ImmunoCAP System, Phadia, Uppsala, Sweden; specific IgE values equal to or greater than 0.35 kUA/l was considered positive) to cow's milk proteins (α-lactoalbumin, β-lactoglobulin, and casein) were determined in all samples using a fluorescent enzyme immunoassay (FEIA), according to the manufacturer's instructions.
Basophil activation was assessed by flow cytometry. 11 The BAT was performed using fresh whole blood derived from the three patients affected by cow's milk allergy, within 3 hours after blood withdrawal. Aliquots (100 µl) of heparinized whole blood were incubated for 10 minutes at 37℃ with 20 µl of basophil stimulation buffer; and then with 50 µl of α-lactoalbumin, 50 µl of β-lactoglobulin and 50 µl of casein (100 ng/ml, Buhlmann). The negative and positive controls were obtained as only basophil stimulation buffer and 20 µl of anti-IgE (0.5 mg/ml, Pharmingen), respectively. We used the CD63-FITC/CD123-PE/HLA-DR-PerCP reagent mixture (BD Biosciences) for gating activated basophils by a FACSCanto flow cytometer (BD Biosciences).
In order to establish the cut-off values for each cow’s milk protein, we previously conducted dose-finding experiments using five patients suffering from IgE-mediated cow's milk allergy and five healthy controls. We used the following increasing concentrations for each allergen protein (α-lactoalbumin, β-lactoglobulin and casein): 0.0001 ng/ml, 0.01 ng/ml, 10 ng/ml and 100 ng/ml. Based on the results obtained from the dose-finding experiments, a concentration of 100 ng/ml was considered the best for discriminating milk-allergic patients from the control group and chosen for subsequent experiments. ROC (Receiver Operating Characteristic) analysis generated a threshold limit value of 13% (sensitivity 75%, specificity 100%), 10% (sensitivity 67%, specificity 100%) and 16% (sensitivity 80%, specificity 100%) for α-lactoalbumin, β-lactoglobulin and casein, respectively.
Patient characterization
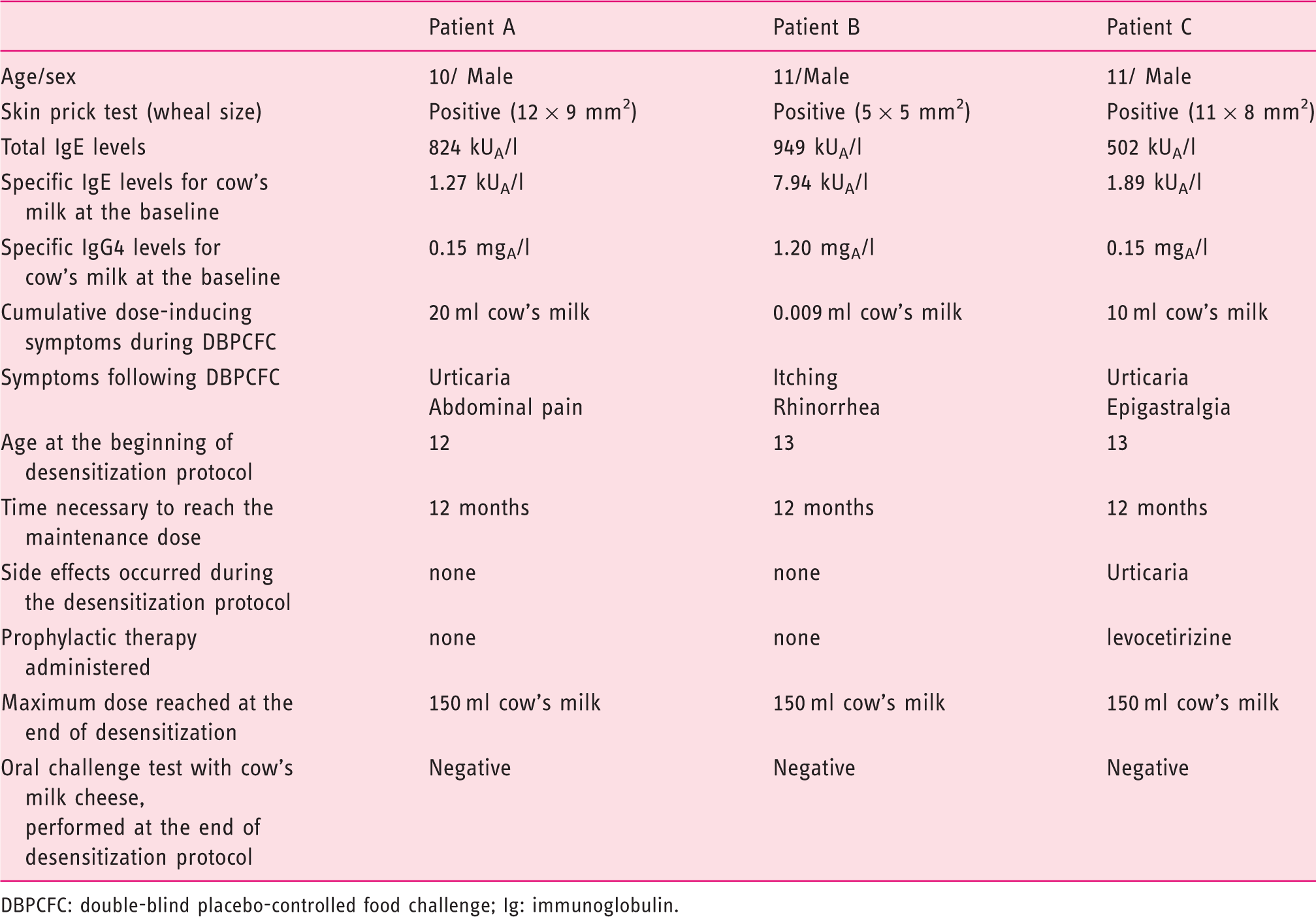
DBPCFC: double-blind placebo-controlled food challenge; Ig: immunoglobulin.
Based on the positive allergy work-up, we decided to carry out a sublingual oral desensitization treatment with cow’s milk, performed according to standardized protocols.6,7 All the patients were asked for written informed consent, according to the rules of the ethical committee of our university, which was countersigned by their parents. We started with doses diluted in water, administered via the sublingual route, and then undiluted for up to a maximum dose of 150 ml of pasteurized whole cow’s milk. The starting solution of the protocol was prepared by placing 0.04 ml of cow’s milk in 100 ml of water and then increased gradually reducing the amount of water or increasing the drops of milk. The first dose of the desensitization protocol administered was equivalent to 0.04 ml of the starting solution.
The patients underwent the desensitization treatment at home and were followed in a day-hospital regimen, every 15 days. According to the protocol, their parents were trained in the medical treatment of allergic reactions and were equipped with an emergency kit: autoinjectable epinephrine, betamethasone and clorphenamine. In order to evaluate the immunological changes that occurred during the immunotherapy, we performed further laboratory tests (total and specific IgE, specific IgG4 and the BAT) at the end of our protocol.
Results
Laboratory data
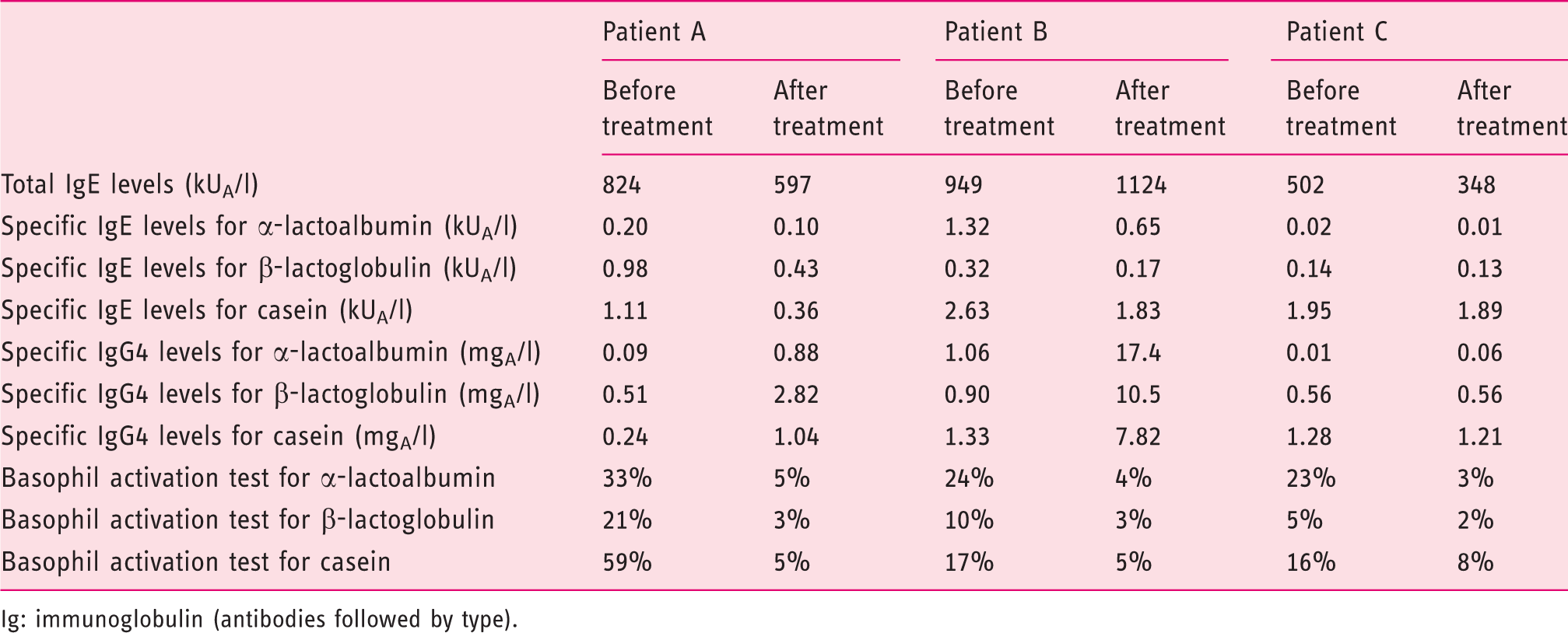
Ig: immunoglobulin (antibodies followed by type).
Patient A reached the maximum dose of cow’s milk after 12 months, without side effects. The patient showed a trend of decreasing specific IgE antibody, without reaching normal values, and increasing specific IgG4; whereas the BAT that started from a positive value of all the three types of milk proteins tested (α-lactoalbumin, β-lactoglobulin and casein) showed a decrease in basophil activation below the established cut-off values, after 12 months of treatment. Oral food challenge performed at the end of the desensitization protocol with cow’s milk cheese was negative.
Patient B became tolerant to the maximum dose after 12 months of treatment and there were no adverse effects reported. The detection of milk protein-specific IgE and IgG4 in the serum of this patient showed a similar trend as reported in the case mentioned above; and he also did not experience a decrease in specific IgE below the cut-off value commonly used. Using BAT, we observed a reduction of the α-lactoalbumin- and casein- induced CD63 expression level after 12 months of treatment. When the desensitization protocol was completed, the patient had a negative oral food challenge to cow’s milk cheese.
Patient C needed 12 months to tolerate the maximum dose of the protocol, but at the dose of 6 ml of pure solution he developed a mild urticarial reaction; so we decide to continue the protocol, administering an antihistamine (levocetirizine 5 mg per day) until he reached the final dose of 150 ml. Four weeks after the discontinuation of oral antihistamine therapy, we performed laboratory tests using a FEIA for determining specific IgE and IgG4 in serum and using flow cytometry to monitor allergen-specific basophil responses. Using FEIA, we observed that the patient achieved subtle modifications in IgE and IgG4 levels, in comparing laboratory data before and after the desensitization treatment. The patient, as had happened previously, showed decreased basophil activation to milk protein, reaching values lower than the cut-off points mentioned above. Based on the negative oral food challenge to cow’s milk cheese, the patient (as well as two other subjects) was able to reintroduce dairy products in his diet. Representative BAT flow cytometry plots before and after desensitization treatment are shown in Figure 1.
Representative BAT flow cytometry plots before (a) and after (b) desensitization treatment.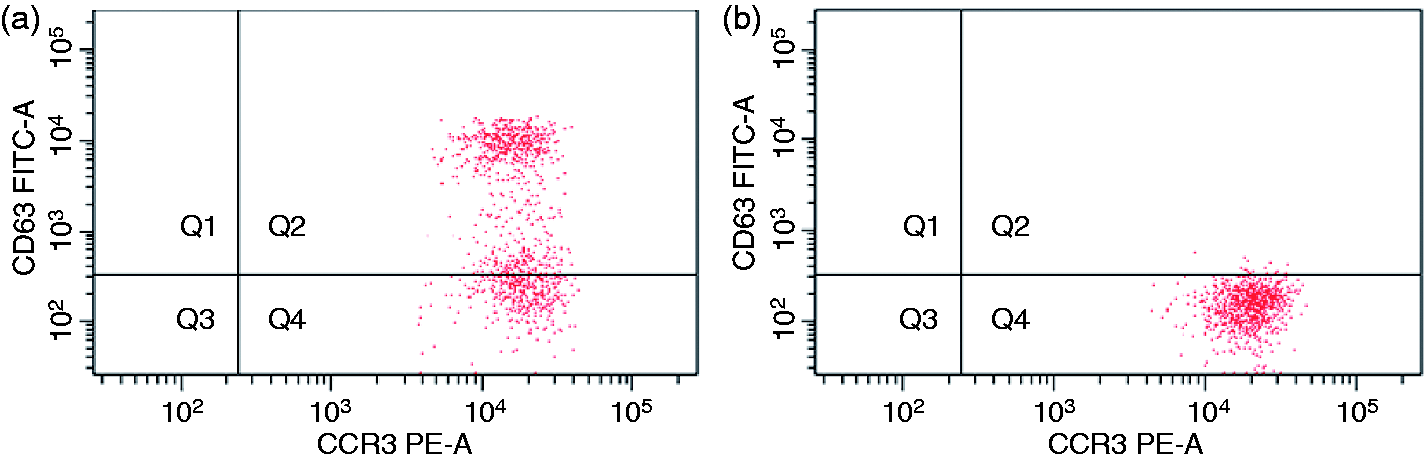
Discussion
Oral desensitization represents an alternative and safe approach in food allergic patients,7–10 although the immunological mechanisms involved are not yet completely known. The recent development of the BAT represents a new opportunity for the diagnosis of food allergies. Several studies demonstrate the comparable sensitivity and specificity of BAT to current testing for food allergy. The effectiveness of the desensitization to food allergens underlines the importance of defining how tolerance induction should be monitored. The advantage of a biological test such as BAT over specific IgE measurement for food allergy is in specificity. Basophils are known to be an early and abundant source of Th2 cytokines. Allergen immunotherapy modulates Th2 responses to allergens and, in this context, Wanich et al. demonstrate a significant relationship between clinical tolerance acquired by milk-allergic children and extrinsically-suppressed allergen-specific effector cell reactivity. 11 Based on recent acquisitions, the identification of basophil activation seems to be a promising approach for monitoring allergic inflammation. The quantification of basophil activation by flow cytometry has proven to be a reliable and valid candidate marker to monitor subcutaneous venom immunotherapy. Ebo et al. 13 demonstrated a significantly decreased CD63 expression (p < 0.04) after 6 months of specific venom immunotherapy and even lower venom-induced CD63 expression (p < 0.001) in patients treated for at least 3 years.
In the current study, we investigated the clinical relevance of BAT for monitoring the acquired clinical tolerance induced by food oral immunotherapy. We observed a reduction of cow’s milk protein-induced CD63 expression level in our patients at the end of the cow’s milk desensitization protocol. Although our patients reached values comparable to those found in the healthy group, the limited number of milk-allergic patients used to calculate the optimal cut-off of positivity during the dose-finding experiments sets limits to our study. Using FEIA data, all three patients showed a decrease in allergen-specific IgE without reaching negative values and an increase in specific IgG4 levels. There is an association between the acquisition of a clinical tolerance to cow’s milk and the reduction of basophils’ reactivity to milk proteins.12
According to our knowledge, this is the first study in which flow cytometry was used during a cow’s milk desensitization protocol. Our data show how it is possible to monitor allergen-specific basophil responses using flow cytometry, in order to identify an acquired tolerance induced by oral food desensitization. Although further studies are needed to develop this important new topic, it was interesting to note that the BAT seems to be more sensitive and characterized by a close correlation with clinical tolerance. In the future, the application of BAT to define immunological modifications could lead to the identification of an in vitro profile characteristic for successful immunotherapy.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest
None declared.
