Abstract
Background and objectives
This study aimed to test the safety and efficacy of Hemospray® for emergency control of acute variceal bleeding (AVB) due to portal hypertension in cirrhotic patients.
Patients and methods
This single-arm, prospective trial, conducted at two hospitals in Belgium and Egypt, included patients admitted to the emergency room with hematemesis and/or melena and known or suspected liver cirrhosis. All patients received urgent hemodynamic stabilization, octreotide (50 mcg bolus then 25 mcg/hour for 24 hours) and intravenous ceftriaxone (1 g/hour). Endoscopy to confirm AVB and Hemospray® application (if indicated) was performed within six hours of admission. Patients were kept under observation for 24 hours and underwent second endoscopy and definitive therapy (band ligation and/or cyanoacrylate injection in cases of gastric varices) the next day.
Results
Thirty-eight patients were admitted for suspected AVB, and 30 of these had confirmed AVB (70% male; mean age 59.5 years (range, 32.0–73 years)). Child-Pugh class C liver disease was present in 53.4%. Esophageal varices were observed in 83.4% of patients, gastric varices in 10%, and duodenal varices in 6.6%. Spurting bleeding at the time of endoscopy was observed in 43.4%. One patient developed hematemesis six hours after Hemospray® application and underwent emergency endoscopic band ligation. No major adverse events or mortalities were observed during 15-day follow-up.
Conclusion
Hemospray® application was safe and effective at short-term follow-up for emergency treatment of AVB in cirrhotic patients.
Introduction
Portal hypertension is a typical feature of liver cirrhosis. Esophageal varices are present in 30% to 60% of cirrhotic patients and variceal bleeding is a severe complication of portal hypertension. 1 Associated mortality related to acute variceal bleeding (AVB) is reported to be 20% within the first six weeks and rebleeding from varices occurs in 60% of patients within one year after the first acute episode. 2
Treatment of AVB, which has been clearly shown to positively influence outcomes, includes restricted transfusion (with a threshold hemoglobin level of 7–8 g/dl), 3 vasoactive drugs, antibiotics, and endoscopic therapy.4,5 The latter consists of variceal band ligation of esophageal varices and obturation of gastric varices with cyanoacrylate injection.6,7
Early treatment is universally recommended and is considered to be mandatory within 24 hours of admission. Better outcomes are reported in those patients who receive endoscopic therapy within 12 hours. 5 Although this is still a matter of debate, it seems reasonable that the earlier the bleeding is stopped, the better the expected outcome.8,9 This is, however, not always possible in daily practice, both because of the lack of treatment capabilities available in every center in an emergency setting, and due to the fact that endoscopic therapy in acute bleeding is technically demanding and not always successful. 10
Hemospray® (COOK Endoscopy, Winston Salem, NC, USA) is a novel hemostatic powder licensed for endoscopic hemostasis of non-variceal upper gastrointestinal (GI) bleeding that has been shown to be effective in preliminary studies for the management of patients with peptic ulcer bleeding, 11 including those on anticoagulant or antithrombotic therapy. 12 Recently, two case reports13,14 and a pilot study that included the first nine patients of this series 15 reported that hemostatic powder may be useful in emergency management of variceal bleeding as a bridge toward more definitive therapy. The major advantage of such treatment, particularly in the case of esophageal variceal bleeding, is that its application does not require technical expertise in therapeutic endoscopy since the powder is delivered in a pre-established manner from the cardia to the mid-third of the esophagus during withdrawal of the scope. 15 In order to confirm the published preliminary results and to assess the effectiveness of the powder in spurting esophageal varices, gastric varices and ectopic varices, we conducted a prospective multicenter study that evaluated the safety and effectiveness of Hemospray® application for emergency control of variceal upper GI bleeding.
Patients and methods
Ethics
The ethics committees of Erasme University Hospital, ULB, Brussels, Belgium, (B406201214760) and Theodor Bilharz Research Institute, Giza, Egypt (TBRI-IRB01/13), approved the protocol and the study was registered with Clinical-Trials.gov under number NCT01783899. All patients signed informed consent before inclusion.
Aim of the study
This prospective, bicentric, single-arm study was designed to evaluate the safety and efficacy of Hemospray® for achieving short-term hemostasis in patients presenting with AVB.
The primary outcomes of the study were the following:
The efficacy of Hemospray®, that is, endoscopic hemostasis, was defined as the absence of fresh hematemesis less than two hours after the application of the hemostatic powder and the absence of hemoglobin drop more than 3 g/dl in absence of blood transfusion, and clinical hemostasis, defined as the absence of a single episode of clinically significant rebleeding within the following 24 hours after Hemospray® application. All patients were followed up throughout the period of the acute bleeding episode (five days)
5
while intravenous infusion of octreotide was administered only for 24 hours after powder application because all patients have a second endoscopy and definitive treatment within the next 24 hours. Rebleeding at five days and survival at 15 days were also recorded. Safety was defined as the incidence of procedure- and treatment-related serious adverse events.
The hypothesis of the study was that the combination of Hemospray® with medical treatment in patients with proven ABV would allow for control of variceal bleeding in more than 90% of cases during the 24 hours following its application.
Patients
Eligible patients were older than 18 years of age, and must have had endoscopic confirmation of AVB defined as either active bleeding or fresh blood in the stomach with red signs on the varices, and no other identified cause of bleeding. 16 Exclusion criteria included: (a) non-variceal bleeding at the time of endoscopy, (b) inability to consent, (c) contraindication to undergo endoscopy, (d) already hospitalized for another illness, (e) pregnant or lactating, (f) patients with altered post-surgical anatomy of the stomach, (g) previously placed intrahepatic portosystemic shunt and (h) patients treated by other endoscopic or surgical modalities within 30 days prior to the intended application of Hemospray®.
Study design
Patients admitted to the emergency room with hematemesis and/or melena with known or suspected liver cirrhosis were included. Urgent hemodynamic stabilization was performed and patients received octreotide (50 mcg bolus at admission then 25 mcg/hour for a period of 24 hours only) and intravenous ceftriaxone (1 g/24 hours). Within six hours of admission, endoscopy was performed in every patient to confirm active variceal bleeding and then to apply the Hemospray®.
Following endoscopy, patients were kept under surveillance for 24 hours and another endoscopy with “definitive therapy” was performed the next day. This therapy consisted of band ligation and/or cyanoacrylate injection in cases of gastric varices, as previously described.17,18
Description of the device
The device used for powder application (Figure 1) consisted of an application catheter, which was passed through the working channel of a therapeutic gastroscope, a chamber containing approximately 21 g of powder, and a propellant CO2 canister. Hemostatic powder (TC-325) is a granular, mineral non-absorbable powder that produces hemostasis by increasing the concentration of clotting factors, activating platelets, and forming a mechanical plug on the injured blood vessel.
19
When the powder comes into contact with moisture in the GI tract, it becomes cohesive and adhesive, forming a stable mechanical barrier that adheres to and covers the bleeding site. As the powder is not absorbed or metabolized by mucosal tissue, there is no risk of systemic toxicity. The covering formed by the powder separates from the intestinal wall and is naturally eliminated from the GI tract.
11
Hemospray® device.
Endoscopic technique
After confirmation of AVB (actively bleeding varices or fibrin plugs and/or red streaks of the mucosa overlying the varices with presence of fresh blood within the lumen), a bleeding site was identified that encompassed the definitive or most probable source (esophageal, gastric, or duodenal varices). The hemostatic powder was then administered diffusely to cover the mucosa over the bleeding varices in order to obtain immediate endoscopic hemostasis.
For esophageal varices, the protocol for application was simplified further, and consisted of positioning the catheter at the level of the cardia in the center of the lumen and applying the powder continuously while pulling the endoscope backward over the 12–15 distal cm of the esophagus. Once bleeding was controlled (first application), the bleeding site was observed for three minutes under endoscopy. If bleeding recurred during this three-minute observation period, Hemospray® was reapplied until hemostasis was achieved (second application). All patients were kept under surveillance and monitored for 24 hours, with continuous infusion of octreotide (25 mcg/hour for a period of 24 hours) and institutional standard of care.
A second endoscopy was performed the next day following initial therapy with hemostatic powder for control and definitive endoscopic therapy.
Data analysis
Analysis was performed using SPSS 20.0.0 (SPSS Inc, Chicago, IL, USA). In the event of missing data values, data were not replaced. Data were expressed as percentages, means ± SD, or medians and ranges, as appropriate.
Results
Thirty-eight consecutive patients with known liver cirrhosis and suspected AVB were included in the study. Eight patients were excluded because the cause of their bleeding was determined not to be variceal (Figure 2).
A flowchart describing the study design and procedures.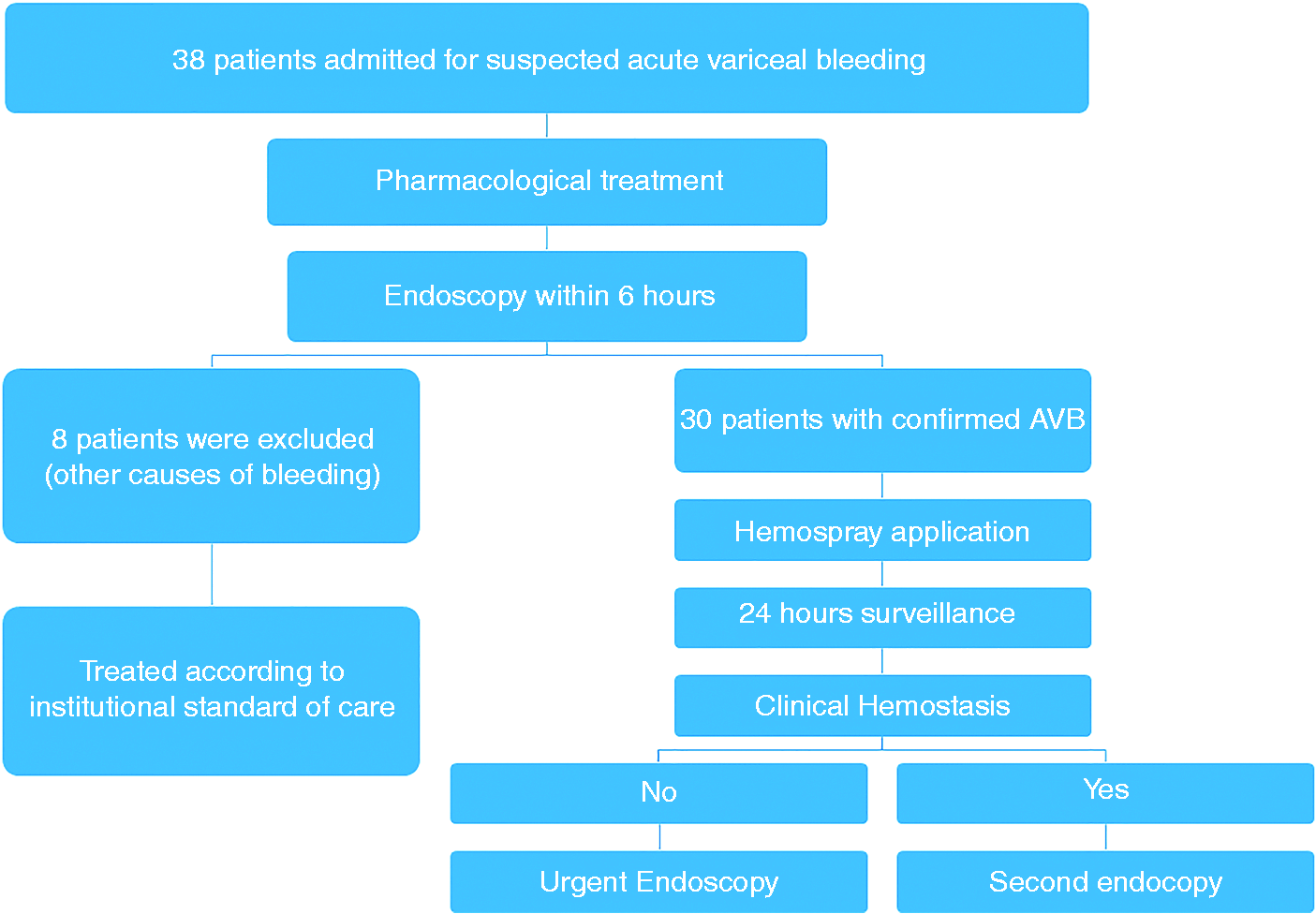
Baseline demographics of patients

Data are presented as medians for continuous variables and as numbers (percentages) for categorical variables. M: male; F: female.
Endoscopic procedure and follow-up
Endoscopy was performed under sedation without endotracheal intubation. Bleeding originated from esophageal varices in 83.4%, from gastric varices in 10%, and from duodenal varices in 6.6%. Spurting bleeding, defined as actively bleeding varices at the time of endoscopy, was observed in 13/30 (43.4%) and acute bleeding, defined as presence fibrin plugs and/or red streaks of the mucosa overlying the varices with presence of fresh blood within the lumen, in 17/30 (56.6%).
Hemodynamics profile of all patients before and after therapy
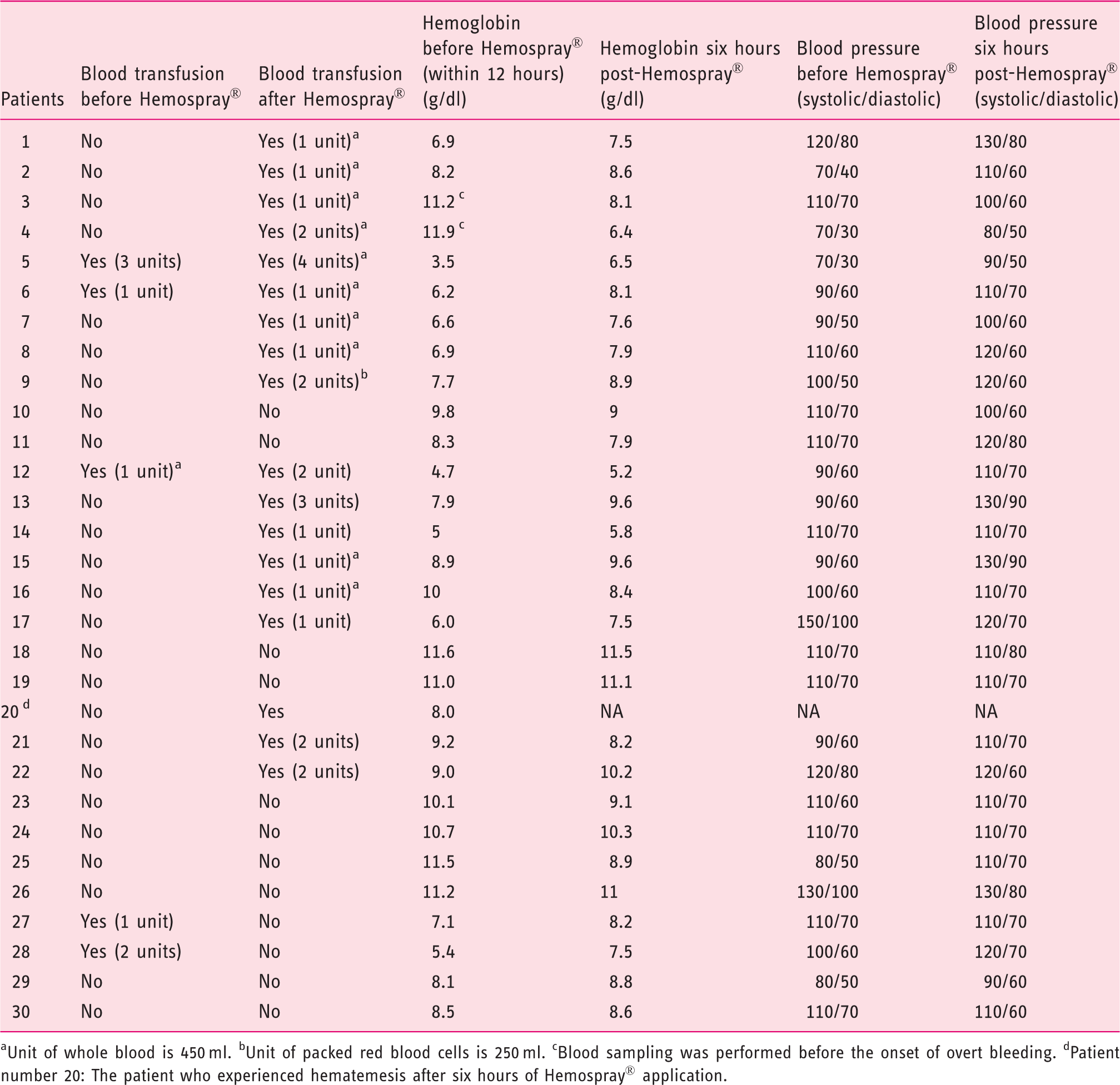
Unit of whole blood is 450 ml. bUnit of packed red blood cells is 250 ml. cBlood sampling was performed before the onset of overt bleeding. dPatient number 20: The patient who experienced hematemesis after six hours of Hemospray® application.
An elective band ligation was performed in patients with esophageal varices and cyanoacrylate injection in patients with gastric varices at the time of second endoscopy, while both patients with duodenal varices were treated with beta blockers and were followed up for three months without further bleeding.
Discussion
This prospective study shows that in cases of AVB, the endoscopic application of a hemostatic powder provides control of bleeding and stabilization of patients for the initial 24 hours, and changes their condition from an acute situation to an elective one that allows additional definitive therapy to be performed under optimal, non-emergency conditions.
Consensus guidelines 5 recommend that patients with ABV undergo endoscopic therapy within 12 hours of admission. The time from initial presentation to endoscopy and severity of underlying liver disease are predictors of rebleeding. 20 In patients presenting with hematemesis, six-week rebleeding and mortality rates are lower in patients undergoing endoscopic therapy within 12 hours of admission than in those for whom it is more delayed. 20
In daily practice, however, the picture is different and delays in performing endoscopy occur, often because of the lack of available expert endoscopists able to manage upper GI bleeding. A recent United Kingdom survey 21 reported that a very low rate (55%) of endoscopies were performed within 24 hours of admission and that standard endoscopic therapy was underused, particularly in AVB.
Development of a technique that allows physicians to obtain immediate hemostasis, that can be performed by any endoscopist, and does not require expertise in management of upper GI bleeding is therefore of major interest for its potential impact on the treatment and outcome of patients with ABV in daily practice.
This is even more vital in patients admitted with hematemesis in whom the importance of early endoscopy is paramount. 22 Indeed, risk factors for in-hospital mortality in patients with AVB not only include delayed endoscopy but also failure of first endoscopy, hematemesis, and severity of cirrhosis. If the application of hemostatic powder, through its simplicity, could reduce the proportion of delayed endoscopies, it would compare favorably with the best series reported to date 23 in terms of immediate hemostasis and failure to control bleeding. When comparing our results with those obtained by tertiary referral centers, 23 which used a combination of drug therapy and banding, our 100% rate of immediate control of bleeding and 3.3% rate of clinical failure over 24 hours suggests that, despite its simplicity, powder application might equalize the results of immediate hemostasis and offer an option in every center for patients to be treated later, once their condition has stabilized, by the most experienced endoscopist.
Recently, transjugular intrahepatic portosystemic shunt (TIPS) has been recommended in the early management of AVB, especially in patients with Child-Pugh class C cirrhosis or those with Child-Pugh C disease and persistent bleeding after endoscopy. 24 This requires availability of a TIPS procedure within 72 hours of admission and is not available in one of the centers that participated in this study. Moreover, when applied to patient populations where endemic AVB has been observed, such as the Middle East, there are currently no available resources to offer this treatment to every patient in whom it could be potentially indicated. However, even in optimal conditions, TIPS placement is associated with less morbidity in hemodynamically stable patients 25 and a technique that would allow centers to ensure that active bleeding has been controlled in every patient prior to undergoing TIPS is of major interest. In addition, the results we observed in our patients with Child-Pugh class C cirrhosis (92% hemostasis at 24 hours and a therapeutic failure rate similar to class B and A patients) may suggest that generalization of the use of hemostatic powder could provide a window that would allow clinicians more time in which to decide about mid- and long-term management of variceal bleeding and its impact on secondary prophylaxis.
Bleeding from duodenal varices is a rare presentation of portal hypertension that accounts for less than 3% of variceal bleeding and was observed in two cases in the current series. The treatment strategies currently used to control duodenal variceal bleeding are all either technically demanding or associated with procedure-related severe complications. Endoscopic obliteration using cyanoacrylate injection is an effective first-line treatment 26 but carries a major risk of portal or systemic embolization. 27 Endoscopic variceal band ligation is technically difficult in the duodenum and can cause severe ulcerations.28,29 Hemospray® could play a role in this instance as a bridge to secondary prophylaxis consisting of β-blockers or TIPS according to the severity of underlying liver disease.
Hemospray® is currently not approved for routine management of AVB since a theoretical concern is that its application with a CO2 cartridge delivers the powder at the end of the catheter with an outflow pressure of 15 mmHg that might be associated with a risk, never observed in the current and previous studies, of venous thromboembolization. This is, however, highly improbable since, by definition, Hemospray® is a noncontact method and, in the case of esophageal or esogastric variceal bleeding, it is delivered with the catheter positioned in the center of the lumen and pulled backward, making the risk of impact with a millimeter-sized bleeding site almost null.
In conclusion, management of AVB using hemostatic powder is feasible and safe. It offers immediate hemostasis using a simple, minimally operator-dependent technique in all cases and clinical and endoscopic hemostasis in >95% of cases until the next day. Further studies comparing strategies involving powder hemostasis are needed to confirm whether this technique might affect the paradigm of AVB management.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgments
Mostafa Ibrahim was responsible for study design, acquisition of data, analysis and interpretation of data, drafting of the manuscript, technical support and material support.
Ahmed El-Mikkawy and Haitham Abdalla were responsible for acquisition of data and technical support.
Ibrahim Mostafa was responsible for study supervision.
Jacques Devière was responsible for study design, interpretation of data, statistical analysis and study supervision.
