Abstract
Background
The diagnostic processes for chronic abdominal conditions are challenging. Despite their tendency for diagnostic tests in patients with irritable bowel syndrome (IBS) symptoms, clinicians are encouraged to make a positive diagnosis based on symptom criteria without alarm signs. We explored how European physicians diagnose and manage patients suffering from IBS.
Methods
We conducted a vignette-based survey to evaluate the diagnostic approaches in four standardized patients with IBS with constipation (IBS-C), IBS with diarrhea (IBS-D), inflammatory bowel disease (IBD) and chronic constipation (CC). General practitioners (GP, n = 104), gastroenterologists (GE, n = 100) and IBS experts (n = 25) from five European countries participated.
Results
Experts showed the highest rates of correct diagnoses (88%–92%) for all cases except CC (only 60%) and were more prone to a positive diagnosis (64%/68% in IBS-C/CC), whereas GEs and GPs tended toward a diagnosis by exclusion (63%/63% and 62%/60% in IBS-C/CC). In the CC vignette, conducting tests was more frequent than prescribing treatment among 44% experts, 63% GEs and 36% GPs. The diagnosis of IBD presented little difficulty for any of the participants.
Conclusions
This study highlights the difficulties in confidently diagnosing chronic functional bowel conditions, especially for non-experts, whereas IBD caused little difficulty. Differentiating between IBS-C and CC seemed particularly challenging, even for experts.
Introduction
Functional bowel disorders are identified only by symptoms. They include irritable bowel syndrome (IBS), functional bloating, functional constipation, functional diarrhea, and unspecified functional bowel disorders. Inflammatory bowel disease (IBD) and IBS represent two conditions characterized by chronically recurring symptoms of abdominal pain or discomfort and alterations in bowel habits. IBS is a symptomatic motility and sensory disorder of the lower gastrointestinal tract, characterized by abdominal pain or discomfort associated with irregular bowel movements and the absence of detectable structural abnormalities. 1 IBD is characterized by inflammation or ulcerations in the small and/or large intestine, with signs that reflect the inflammatory process such as rectal bleeding, diarrhea, fever, and weight loss, occasionally associated with extra-intestinal manifestations. 2 The major types of IBD are Crohn’s disease, a transmural disease of the gastrointestinal mucosa that can affect the entire gastrointestinal tract from the mouth to the anus, and ulcerative colitis, affecting colonic mucosa only. Functional constipation, also called chronic constipation (CC) in many European countries, is another functional bowel disorder that presents as persistently difficult, infrequent, or seemingly incomplete defecation, which do not meet IBS criteria. 1
The diagnostic processes for functional bowel disorders and the differentiation between functional and IBD can be challenging and uncertain for a variety of reasons. With regard to functional bowel disorders, these include the lack of consistent biological markers, leaving clinicians to rely on patient symptoms alone to make the diagnosis and the non-specificity of the cardinal symptoms of abdominal pain or discomfort as well as the heterogeneity of patients. 3 Despite the tendency of clinicians to order diagnostic tests in the face of IBS symptoms, the Rome III criteria encourage clinicians to make a positive diagnosis on the basis of symptom criteria without alarm signs (e.g. fever, gastrointestinal bleeding, weight loss, anemia, abdominal mass), also emphasizing that for diagnosing functional bowel disorders, laboratory tests are rarely helpful. 1
To better understand current diagnostic decision making in IBS, we performed a vignette survey to explore how physicians diagnose and manage patients suffering from IBS. As the overall objective of the survey was to provide for a comprehensive description of physicians’ practices in the IBS diagnostic process, no hypothesis was posed prior to the survey setup. This description also includes the level of confidence to set up a diagnosis, to communicate this diagnosis outcome to the patient, to select the appropriate test as well as the confidence to interpret the results of tests and to communicate test results to the patient.
Additional assigned objectives were to measure physicians’ beliefs about whether IBS is a diagnosis of exclusion, a positive diagnosis or a combination of diagnoses (exclusion and positive), assessing the diagnostic approach in IBS, namely type of diagnostic tests to be performed and the appropriateness of available diagnostic tests.
Lastly, level of awareness with diagnostic criteria and use of guidelines among physicians were also included in the overall assessment.
Methods
Vignette survey design
This was an online vignette-based questionnaire to evaluate the diagnostic approaches in four standardized patients demonstrating typical cases of irritable bowel syndrome with constipation (IBS-C), irritable bowel syndrome with diarrhea (IBS-D), inflammatory bowel disease (IBD) and functional constipation (CC). The survey was conducted in five European countries (France, Germany, Italy, Spain and the United Kingdom (UK)) between February and March 2013. IBS Experts, gastroenterologists (GEs) and general practitioners (GPs) were invited to participate through an email with cover letter and a link to the survey, using an online questionnaire platform system. Those agreeing to participate were to answer a 30- to 40-minute questionnaire online. The survey was totally anonymous and the name of the sponsor has not been revealed to participants. The data management of the study was processed in France, and the overall study and data management were covered by the French Data Protection Regulation (CNIL – number 1493177).
The four vignettes were developed based on the Rome III criteria 1 in concert with IBS experts and survey design specialists, to ensure clinical face validity, comprehensibility and comprehensiveness. The vignettes displayed symptom patterns, physical examination, patient’s personal and family history and diagnostic results.
The IBS-C vignette described a 57-year-old woman with longstanding intermittent abdominal pain improving after bowel movements, constipation with hard stools and straining and no evidence of alarm signs or symptoms. The IBS-D vignette described a 38-year-old man with longstanding frequent, loose stools, lower abdominal cramping that improved with stool passage, and no evidence of alarm signs or symptoms. The CC vignette described a 42-year-old woman with longstanding constipation, bloating, hard stools and straining, and one or two bowel movements per week. The IBD vignette described a 20-year-old man with abdominal pain and urgent, occasionally bloody diarrhea for three months, which had recently worsened, accompanied by tiredness and significant weight loss. The full vignettes are presented in the Appendix. The vignettes were accompanied by a series of stand-alone questions pertaining to clinical decision making, overall knowledge and perception of IBS, and awareness and use of guidelines.
For each vignette, participants were asked to make a diagnosis and answer questions on the investigation and further management of each case. An overall exploration of participants’ perception, attitude and diagnostic approach to IBS was also collected. Most questions were presented with a series of closed-ended questions providing a list of answer choices. These types of questions were commonly in the form of multiple choices, either with one answer or with “check-all-that-apply” options (type of diagnosis, type of test to be conducted, type of treatment to be recommended). The questionnaire also included questions related to level of confidence to set up a diagnosis, to communicate the diagnosis to the patient, to interpret and communicate test results to patients as well as appropriateness of a test to be conducted. These were presented in an ordinal scale format, where the respondents could decide where to rate their answer along the scale continuum. Some open-ended questions in the form of “Other, please specify” were also offered to ensure that physicians could include other responses not initially anticipated during the design of the survey.
Sampling frame
Sample of IBS experts
GEs recognized as experts in IBS were identified based on publication in peer-review journals, participation as a key speaker in conferences, training or teaching activities given to peers, and participation in scientific advisory boards. Recruitment was stopped once five experts in each country accepted participation, leading to a total of 25 experts in this group.
Sample of GEs
We surveyed a random sample of 100 GEs from the membership directory of each country’s medical association, using a random number generator. In case the random selection process identified a GE already included in the experts group, the selection process was repeated to identify a second individual to avoid duplicates between samples. To be qualified for the survey, GEs had to have at least three years in practice, spend at least 60% of their time in a hospital or clinic and to weekly see 10 or more IBS patients. Recruitment was stopped once 20 GEs in each country completed the questionnaire.
Sample of GPs
We surveyed a random sample of GPs from the membership directory of each country’s medical association, using the same process as for the GE group. To be qualified, GPs had to have at least three years in practice, spend at least 70% of their time in a hospital or clinic and to weekly see five or more IBS patients. Recruitment was stopped once 20 GPs in each country completed the questionnaire, leading to a total of 104 GPs.
Evaluation and statistical analyses
Sample size determination
For each of the two groups, GEs and GPs, a sample size of 100 was required to set the maximum 95% confidence interval for the estimation of proportions to ±10%.
The expert sample size was limited to 25 because these experts are extremely difficult to recruit.
Diagnosis assessment and perception of the case vignettes
Following each vignette, physicians were asked the following question: “Based on the information you have just read, do you consider this patient to be more likely …?” Physicians selected among six options: “a patient with irritable bowel syndrome (IBS),” “a patient with inflammatory bowel disease (IBD)”, “a patient with chronic constipation (CC),” “a patient with functional abdominal pain,” “Other (please specify),” “Don’t know.” Those who selected IBS were asked to specify if they were referring to IBS-C or IBS-D. For analysis, we calculated the rates of “correct” and “incorrect” diagnosis and assessed the rates of “do not know.” The next question assessed the level of confidence toward the presumed diagnosis at that stage, using a standard nine-point RAND/University of California Los Angeles (UCLA) Appropriateness Scale (RAS) with the following interpretation: scores 1–3 = “generally not confident,” 4–6 = “neither not-confident nor confident,” 7–9 = “generally confident.” 4 Then, physicians were asked how they would “proceed to set up a diagnosis at that stage,” choosing between “a positive diagnosis,” “a diagnosis of exclusion” or “do not know.” Participants were then asked what would be the next action: collect further clinical information or refer to a specialist, conduct tests and/or prescribe a treatment, and the kind of tests or treatments prescribed. In case of tests recommended, participants rated the appropriateness of a range of laboratory, radiographic, and endoscopic tests using RAS and their level of confidence to interpret the test results. For the analysis of the different RAS assessments, we compared mean RAS scores among physicians using analysis of variance, considering a p value <0.05 as significant.
Overall knowledge and perception of IBS
Stand-alone questions, separated from the vignettes, investigated whether participants believed the diagnosis of IBS to be primarily positive, or by exclusion or both and whether they used guidelines. Participants were also asked to rate their level of awareness of the Rome I, II and III criteria, their overall perception of IBS diagnosis (useful, complex, time-consuming, challenging, uncertain) and their interest regarding information on IBS (sharing experience with peers via seminars, more information about guidelines, more information about diagnostic tests) using a standard nine-point RAS.
General belief about IBS diagnosis
We posed a stand-alone question separate from the clinical vignette, as follows: “Based on your clinical experience, do you consider IBS, in general, to be…?” Physicians had the choice for a single answer between four options: “IBS is primarily a diagnosis of exclusion (i.e. one or more diagnostic tests should be performed before diagnosing IBS),” “primarily a positive diagnosis,” “a combination of both (diagnosis of exclusion and positive exclusion),” “Not sure/Do not know.”
We first calculated the proportion of respondents in each group who endorsed IBS as a diagnosis of exclusion, and then compared proportions among groups using a χ2-test.
Knowledge about yield of common diagnostic tests in IBS
Data indicate that the yield of common diagnostic tests in IBS, including colonoscopy, stool studies, inflammatory markers, and thyroid function testing, among others, is low.5,6 It may be possible that some physicians overestimate or underestimate the diagnostic yield of common diagnostic tests in IBS, and that knowledge about test yield may provide diagnostic decision making. We therefore posed, for each vignette, a series of questions to assess knowledge with diagnostic testing yield in IBS conditions and willingness to prescribe or recommend tests with confidence to the patient with the presumed diagnostic.
Physicians viewed a list of 12 common diagnostic tests and an option for an open-ended space to add additional tests not anticipated in the list shown. Mean number of tests prescribed was assessed between the groups using analysis of variance and a p value <0.05 as significant. Differences in physicians’ outcomes (in terms of type of tests prescribed and number of tests) were examined using Student’s t-tests (when comparing groups of physicians with correct diagnosis vs. group of physicians with incorrect diagnostic) and analysis of variance when comparing the three groups (experts, GEs, GPs). A p value for all tests was displayed at a significance level of 0.05.
Results
Sample characteristics
Demographic and practice-pattern information of participants
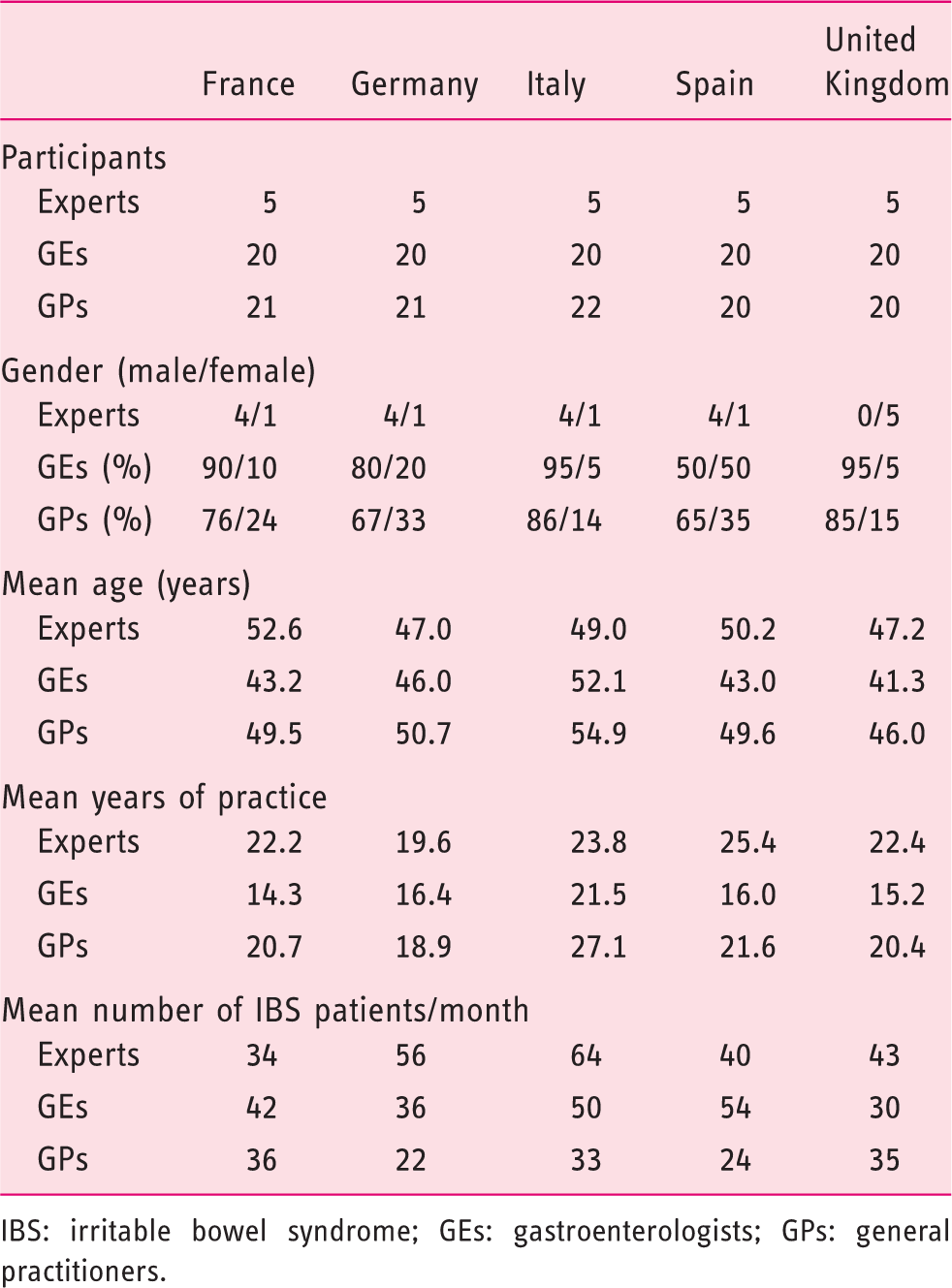
IBS: irritable bowel syndrome; GEs: gastroenterologists; GPs: general practitioners.
Diagnosis assessment and perception
Ability to make a diagnosis for each patient vignette varied among specialties (Figure 1). Experts showed the highest rates of correct diagnoses (88%–92%) for all vignettes except for the CC vignette, for which the rate of correct diagnosis was 60%. Levels of unknown diagnoses were higher in the GP group, especially for IBS-C and IBS-D.
Ability to diagnose irritable bowel syndrome with constipation (IBS-C), with diarrhea (IBS-D), inflammatory bowel disease (IBD) and chronic constipation (CC) on the basis of patient history and physical examination findings in patients’ vignette. Participants were asked: “Based on the information you just read, do you consider this patient to be more likely…?” The figure depicts the percentages by group giving a correct or incorrect answer or responding “Unsure — need more information.” GEs: gastroenterologists; GPs: general practitioners.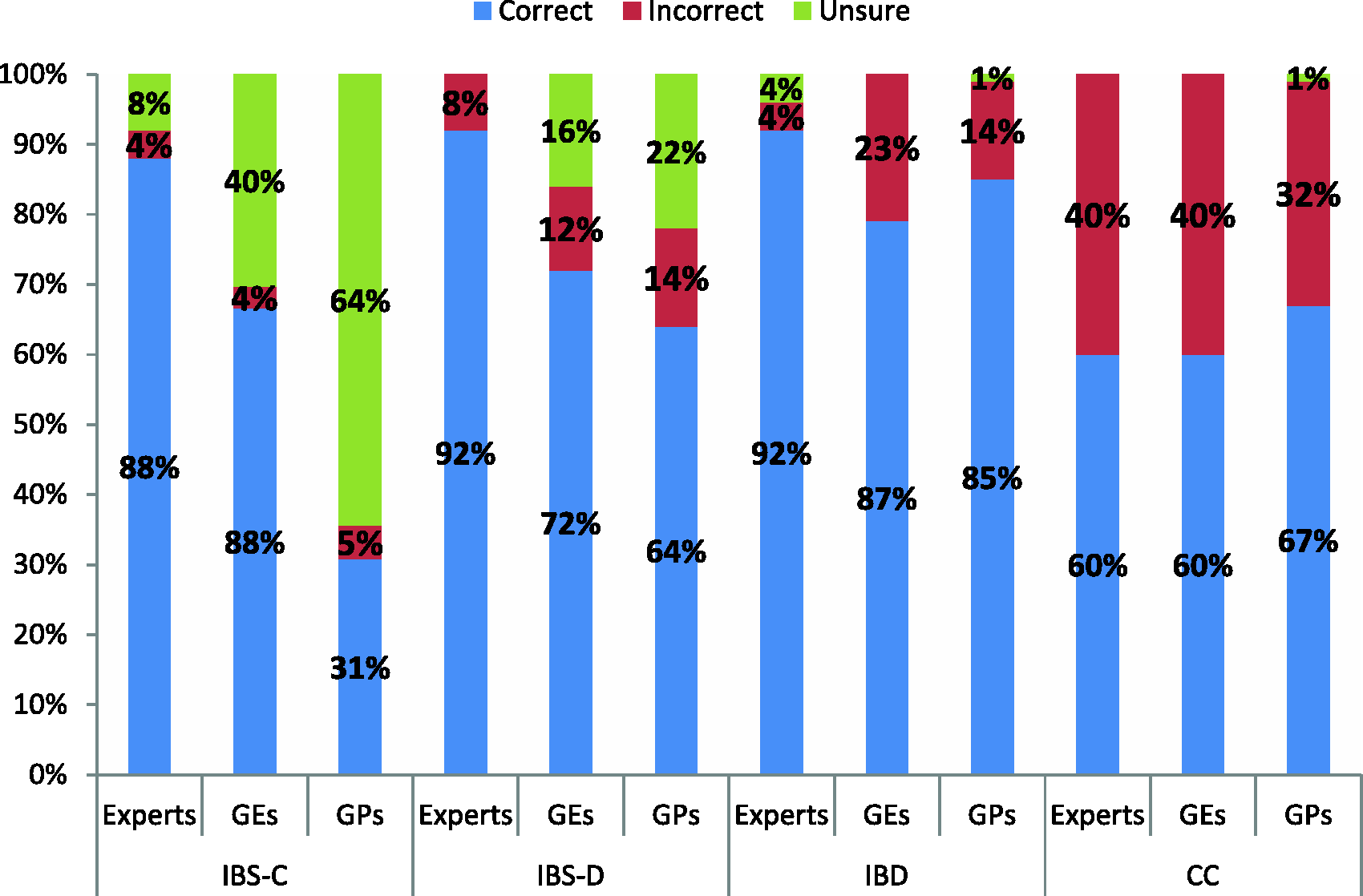
Ability to make a diagnosis for each vignette, by country
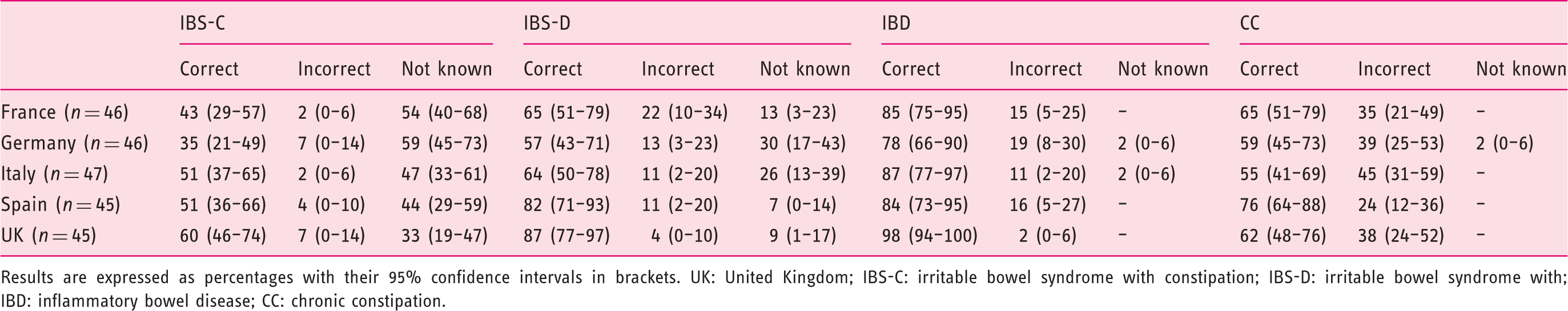
Results are expressed as percentages with their 95% confidence intervals in brackets. UK: United Kingdom; IBS-C: irritable bowel syndrome with constipation; IBS-D: irritable bowel syndrome with; IBD: inflammatory bowel disease; CC: chronic constipation.
Assessment of IBS-C and CC
Diagnosis assessment and perception
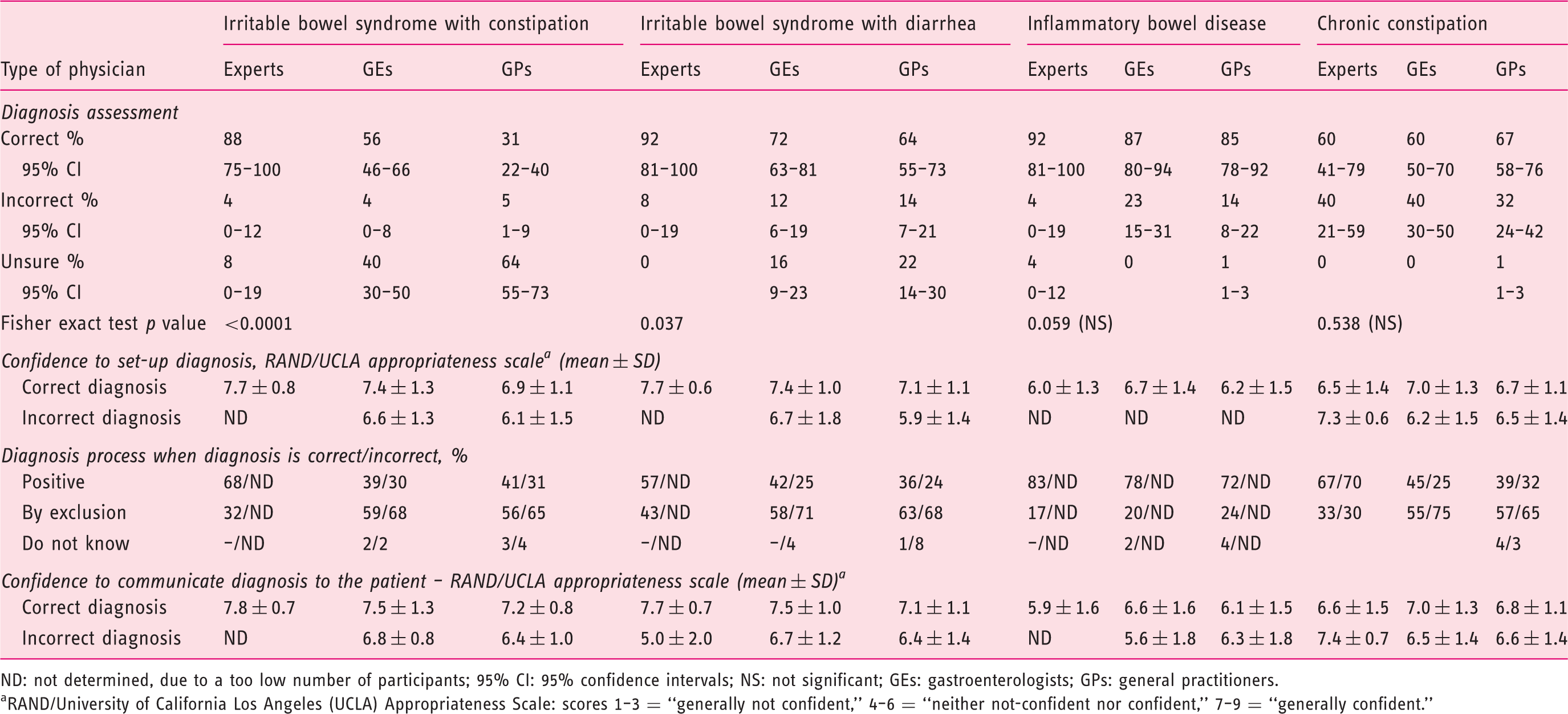
ND: not determined, due to a too low number of participants; 95% CI: 95% confidence intervals; NS: not significant; GEs: gastroenterologists; GPs: general practitioners. aRAND/University of California Los Angeles (UCLA) Appropriateness Scale: scores 1–3 = “generally not confident,” 4–6 = “neither not-confident nor confident,” 7–9 = “generally confident.”
Patient’s management
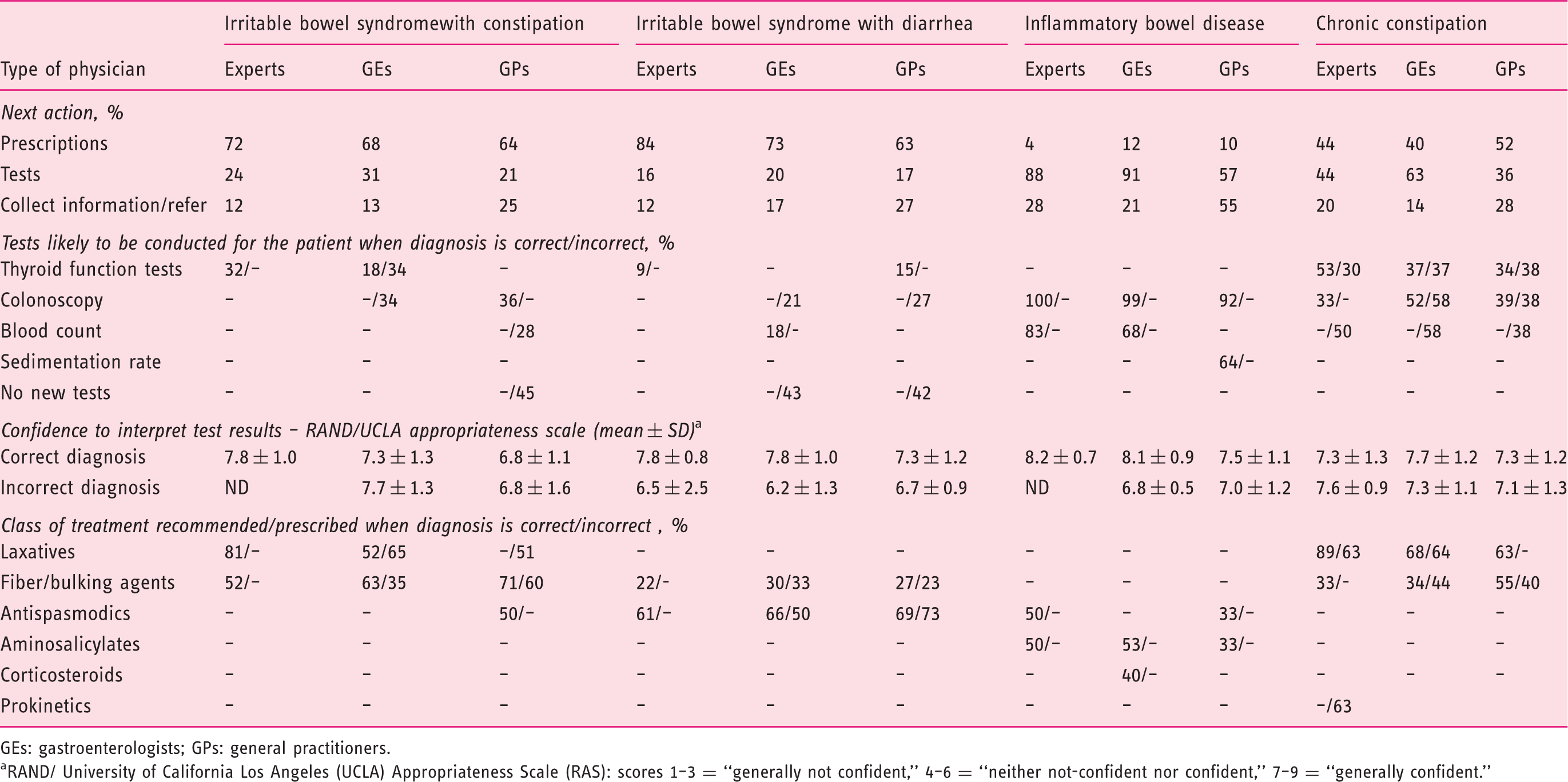
GEs: gastroenterologists; GPs: general practitioners. aRAND/ University of California Los Angeles (UCLA) Appropriateness Scale (RAS): scores 1–3 = “generally not confident,” 4–6 = “neither not-confident nor confident,” 7–9 = “generally confident.”
For the IBS-C vignette, prescribing a treatment as next action was more frequent than requesting tests. Irrespective of correct or incorrect diagnostic setup, fiber and bulking agents were prescribed by 50% to 60% of physicians, laxatives by 77% of experts and 58% of GEs, antispasmodics by 41% of experts and GPs. A test was requested by 24% experts, 31% GEs and 21% GPs, all being generally confident in how to interpret them. Some GEs and GPs also recommended a colonoscopy even though the patient has presented with normal ileo-colonoscopy results.
For the CC vignette, conducting a test was more important than immediately prescribing a treatment, mainly thyroid function test and colonoscopy, all physicians being generally confident in how to interpret them. A treatment was prescribed by 40% to 52% of physicians, mainly laxatives by experts and GEs.
Assessment of IBS-D and IBD
Based on the IBS-D vignette, 16% of GEs and 22% of GPs were unable to make a diagnosis, this proportion being very low for the IBD vignette (Tables 3 and 4). For IBD, 68% to 80% physicians had a positive diagnosis approach, whereas the proportion of IBS-D diagnosis by exclusion was still high with GEs (62%) and GPs (64%), irrespective of correct or incorrect diagnostic setup. When the diagnosis was correct, the mean level of confidence to set up a diagnosis and communicate it to the patient was always above 7 and similar between physicians’ groups for the IBS-D vignette, whereas IBD led to less confidence, 5.9 to 6.7, because most physicians said they would wait for the result of investigation before making a diagnosis.
After IBS-D diagnosis, a medical treatment was the next action for 84% experts, 73% GEs and 63% GPs, mainly antispasmodics, then fiber/bulking agents. Tests were requested by 16%–20% of physicians. For the IBD vignette, the next action after diagnosis was to conduct tests (colonoscopy in all cases); GPs were 57% likely to request a test and 55% likely to collect more information or refer the patient to a GE.
Overall knowledge and perception of IBS
IBS is considered by experts as mainly a combination of positive and exclusion diagnosis, whereas GEs and GPs as mainly a diagnosis of exclusion (Figure 2). Most physicians refer to guidelines, with the proportion among experts being higher than GEs or GPs. While experts would rather refer to international guidelines, GEs and GPs are more inclined to refer to national guidelines (Figure 2). The Rome III criteria are the most prevalent diagnosis criteria; the share of awareness among experts is overriding and is significantly higher compared to other groups (96% vs. 73% GEs; and vs. 15% GPs). Awareness mean score is significantly higher for experts (8.4 vs. 7.1 GEs and vs. 3.3 GPs), with more than half of them scoring the highest score: 9 (median =9 for “know very well”). Whatever criteria tested, the level of awareness among GPs is significantly lower compared to GEs or to experts.
Overall knowledge and needs in irritable bowel syndrome. Participants were asked: For diagnosis process: “Based on your clinical experience, do you consider IBS, in general, to be?” For the interest regarding information on IBS: “To what extent would you be interested in the following aspects using a scale of 1–9, where 1 means ‘not at all’ and 9 ‘very’?” Percentages presented represent the rates of participants who gave a score of 7–9. For reference to guidelines: “During IBS diagnosis process, if you refer to guidelines, which guidelines do you generally refer to?” The figure depicts the percentages by group for each possible answer. IBS: irritable bowel syndrome; GPs: general practitioners; GEs: gastroenterologists.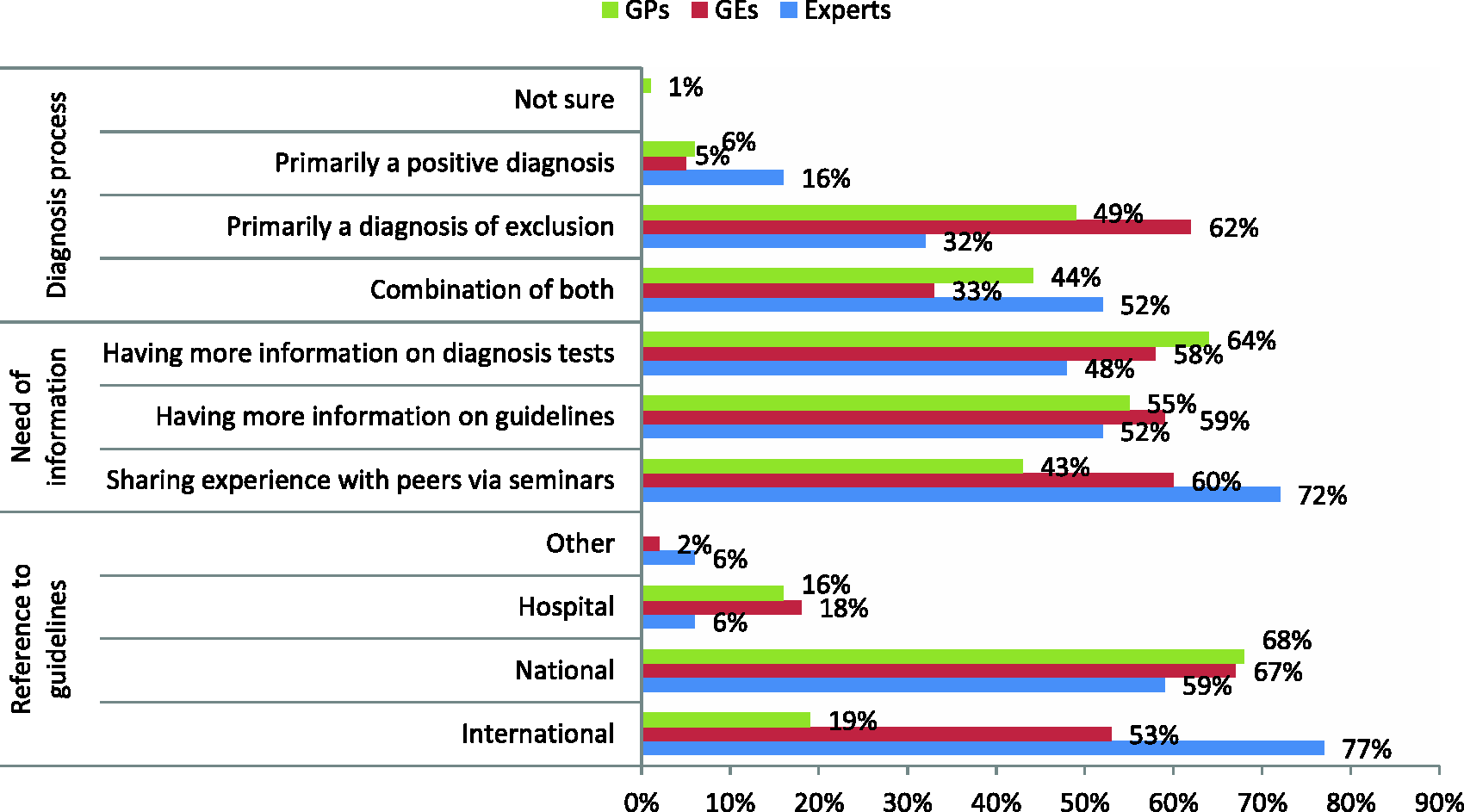
Experts and GEs show substantial interest toward sharing experience with peers via seminars. GPs expressed less interest in sharing experience than in having more information on diagnosis tests (Figure 2).
The IBS diagnosis process (Figure 3) is overall perceived as very useful. This perception is reinforced among experts. For most physicians, experts or non-experts, the diagnostic process appears to be time consuming, complex and challenging. In a significant proportion, GPs associate the IBS diagnosis process with uncertainty.
Overall perception of diagnosis process. Participants were asked: “Please assess your overall perception of IBS diagnosis process, using a scale from 1 to 9, where 1 means ‘not at all’ and 9 ‘very.’” The figure depicts the percentages by group for each possible answer. Percentages represent the rates of participants who gave a score of 7–9. IBS: irritable bowel syndrome; GPs: general practitioners; GEs: gastroenterologists.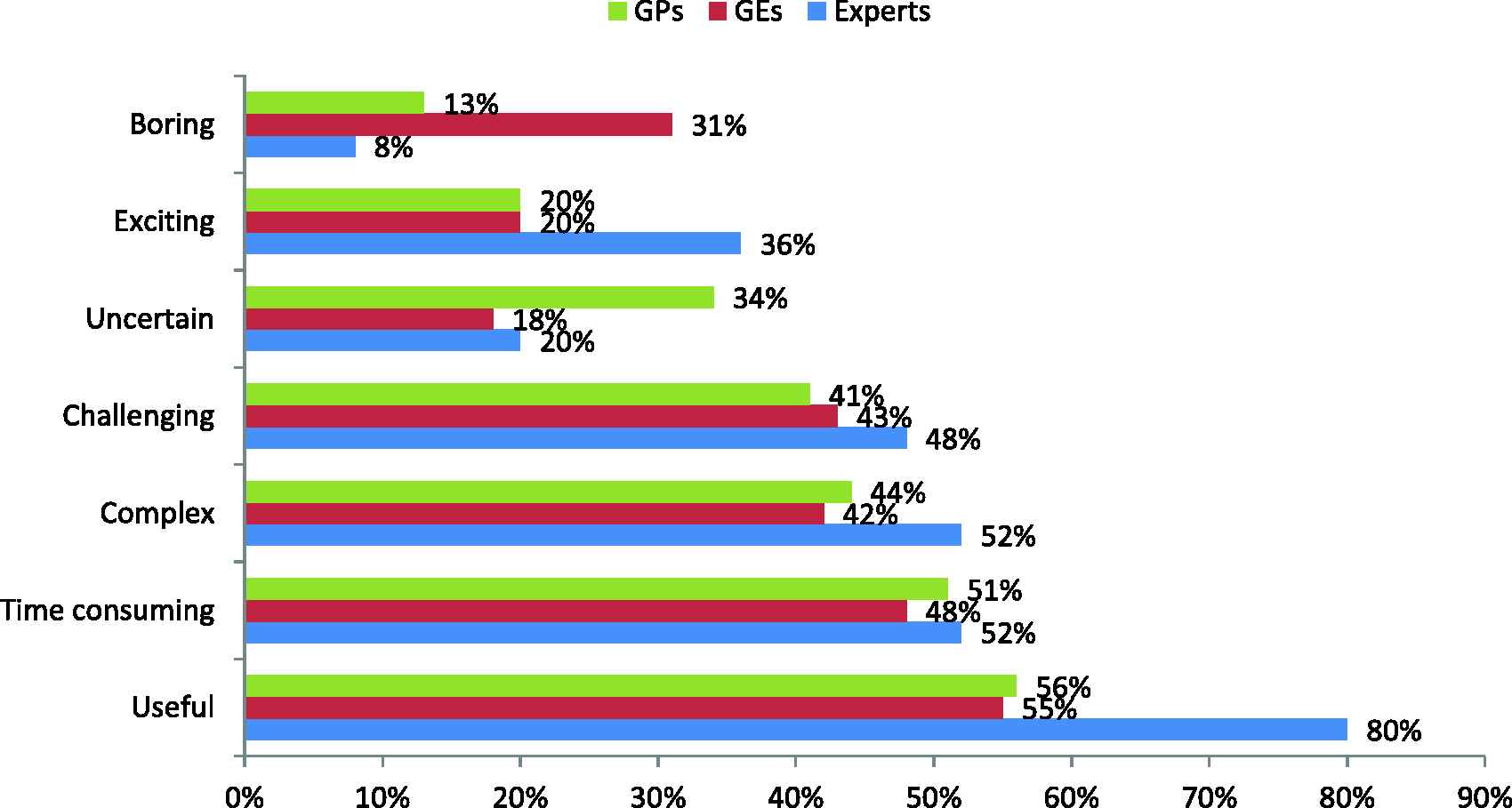
Discussion
The diagnostic processes for chronic abdominal conditions and differentiation between functional and IBDs remain challenging. Knowledge of real-life diagnostic process allows for evaluation of the conformity of practices with the current guidelines and their applicability.
This study highlights the difficulty experienced by GPs, GEs and experts in confidently diagnosing conditions such as IBS. Participants generally regarded the IBS diagnostic process as time consuming, complex and challenging.
Concerning IBS knowledge, GPs were less aware of guidelines and they overall regarded IBS as a diagnosis of exclusion. Among GEs, however, there was a discrepancy between guidelines and clinical practice: Despite a high rate of awareness of the Rome III guidelines, IBS was still perceived to be a diagnosis of exclusion. In contrast, most experts with high guidelines awareness regarded IBS as a positive diagnosis or a combination of a positive diagnosis and diagnosis by exclusion.
Except for experts, a significantly high proportion of physicians were unable to make a diagnosis based on information provided in the patient vignette, especially for the IBS-C patient vignette and to some extent IBS-D, which is indicative of a general lack of confidence in making the diagnosis. On the other hand, experts and GEs were more likely to misdiagnose CC as IBS-C than GPs. The latter may actually reflect a more general view of these two disorders to be different manifestations of the same underlying disease, mostly considered as IBS. In fact, several studies have shown a large overlap between these two disorders,7–13 and it may often be neither possible nor necessary to differentiate between the two. Still, current diagnostic guidelines such as the Rome III criteria do differentiate between the two disorders, where absence of abdominal pain would be the main differentiating sign of CC versus IBS-C. The awareness of this differentiation may also be of importance for the use of medical therapies, where some drugs are developed and approved only for CC and others only for IBS-C.
With regards to diagnostic approaches, experts were more likely than GEs and GPs to endorse a positive approach to the diagnosis of IBS, IBD or CC, whereas GEs and GPs would rather adopt a diagnosis by exclusion approach indicating an important discrepancy between the practice of experts and “generalists.” However, despite a high rate of exclusion diagnoses, the need for additional investigations remained rather low for IBS-C and IBS-D. This may partly be explained by the fact that the IBS patients in the vignettes have already presented with diagnostic workups so that the physicians could have rated their diagnosis as “diagnosis of exclusion” at that stage. But this could also partly reflect a different attitude toward the meaning of a “positive diagnosis.” To an expert, a positive approach typically means emphasizing the certainty of the diagnosis to the patient but still accepting the need for some investigation if necessary, whereas the generalist is more likely to prefer exhaustive investigation followed by a less certain diagnosis: “The tests are all negative therefore it is probably IBS.” This discrepancy may reflect valid concerns about the incomplete understanding of the pathophysiology of IBS. Indeed, an increasing body of evidence suggests molecular and cellular alterations at the mucosal level, changes in gut flora, disturbances of super-ordinated regulatory systems, and increased prevalence of psychological co-morbidities. 14
IBD, as an inflammatory process, was easily recognized, but colonoscopy and blood tests were deemed necessary to confirm the diagnosis. The CC case presented with few prior test results and probably therefore led 40% to 45% of experts and GEs to request colonoscopy and thyroid testing, underlining once more the difficulty experienced by physicians and their fear of misdiagnosing a more severe condition.
It is of note that the level of confidence in the different diagnoses and in communicating the diagnosis to patients was similar among experts, GEs and GPs. All physicians were generally confident in announcing the diagnosis to the patient except for IBD, for which they would wait for investigation results.
Interestingly, there were some country-related variations in the ability to make a diagnosis based on the information in the vignettes. This may reflect differences in health care systems, specifically differences in national guidelines and practice approaches as well as the availability and costs of diagnostic tests. However, this was not specifically assessed in our study, which is a limitation of this survey. Hence, it would be interesting for future, most likely not purely survey-based studies to additionally assess health care system-related factors including cost issues to be able to compare cost-per-case estimations among the different countries.
Another possible limitation of this survey is that responses may not reflect the real-life diagnosis-making process. The true process of care may theoretically be better evaluated by direct observation, but in this approach, physicians tend to alter their practice when they know they are being observed. Survey-based clinical vignettes have been validated as an accurate surrogate for both chart abstraction and standardized patients, 15 and are thus widely recognized to be a valid, reliable, practical, and cost-effective technique to assess process of care.
Another limitation is that we were unable to compare participant and non-participant characteristics, no master files being available for each of the surveyed countries, therefore the sample representativeness and a generalizability of our results may be questioned. However, the comparability between countries was established.
Finally, our distinction of expert and non-expert, although based on explicit criteria, may fail to acknowledge the fact that many community providers who manage IBS on a daily basis might have a higher clinical experience than some academic opinion leaders.
Overall, this study highlights the difficulties in confidently diagnosing chronic bowel conditions. Where an inflammatory disease caused little difficulty, all functional symptoms were regarded as difficult to diagnose, especially by non-experts. Differentiating between IBS-C and CC seemed particularly challenging even with reference to guidelines. Such criteria as Rome III are extremely useful for research purposes but are complicated and may not be easily applicable in a normal clinical setting. While guidelines may not necessarily improve diagnostic ability, they may partly influence the perception of the diagnostic approach. High rates of diagnostic resource use may be driven by potentially modifiable knowledge deficits, beliefs, and attitudes regarding IBS.
Footnotes
Acknowledgment
The authors thank Dr N Debbas, MediAxe, France, for writing assistance, funded by Kantar Health.
Funding
This work was supported by Almirall.
Conflicts of interest
V Andresen has received lecture fees from Almirall, Abbvie, Aptalis, Ardeypharm, Norgine, Shire, Mundipharma and Falk; and is a consultant for Almirall, Astra Zeneca, Norgine and Shire.
P Whorwell has received financial support for research from Danone; lecture fees from Abbott, Danone, Shire and Almirall; and is a consultant for Norgine, Danone, Shire and Almirall.
J Fortea is an employee of Almirall.
S Auzière has received financial support for research from Almirall and is a consultant for Kantar Health.
