Abstract
Background
Hypermobile Ehlers-Danlos syndrome (hEDS) and hypermobility spectrum disorders (HSDs) are heterogeneous disorders of the connective tissue associated with widespread chronic pain and co-occurring autonomic dysfunction. No disease-specific treatment has been identified. Unlicensed cannabis-based medicinal products (CBMPs) are increasingly prescribed for chronic mental and physical conditions.
Methods
Project Twenty21 is a longitudinal, observational registry study producing real-world evidence on the safety and effectiveness of CBMPs prescribed by specialist consultants at UK-based private clinics. This study reports baseline characteristics and three-monthly follow-up patient-reported health outcomes assessed using clinically validated questionnaires for up to 12 months in 121 patients with hEDS/HSD enrolled in Project Twenty21.
Results
Co-morbidity was common, with participants reporting a mean of 6.16 ± 3.8 secondary conditions. Prescription opioid analgesic use was reported by 54 participants (45%), with a statistically significant reduction in mean morphine equivalent observed between baseline and three-month follow-up (p < 0.001). Statistically significant improvements in mood/depression (p = 0.010) and sleep quality (p = 0.031) were observed between baseline and at each follow-up interval for the 12-month study duration with moderate effect sizes. Statistically significant improvements in quality-of-life (p = 0.001), pain severity (p < 0.001) and interference (p < 0.001) were observed at each follow-up interval between baseline and nine-month follow-up with small–moderate effect sizes. Adverse events were reported by six participants (5%) and were predominantly assessed as mild.
Conclusions
CBMP treatment is associated with improved symptomology, quality of life, and functionality in addition to reduced opioid requirement in hEDS/HSD patients. Future research should aim to improve clinical care, mechanistic understanding and refine effective CBMP formulations.
Introduction
Hypermobile Ehlers-Danlos syndrome (hEDS) and hypermobility spectrum disorder (HSD) are heritable disorders of connective tissue which share phenotypes of joint hypermobility, skin hyperextensibility and tissue fragility (Copetti et al., 2019). hEDS/HSD has an estimated prevalence of one in 500 (Demmler et al., 2019). The hEDS/HSD phenotype is clinically heterogeneous and multi-systemic, encompassing arthralgia, joint instability, soft tissue injury, co-occurring neurodevelopmental conditions and dysfunction of the cardiovascular, autonomic, gastrointestinal, and gynaecological systems. Chronic pain impacts up to 90% of patients (Chopra et al., 2017) and can be nociceptive, neuropathic and nociplastic (central sensitization) in origin.
Altered tissue mechanics are reported in hEDS/HSD (Palmer et al., 2020). Damage to the peripheral Aδ fibres and C fibres due to abnormal stretching and pressure has been associated with the prevalence of co-occurring small fibre neuropathy and autonomic disorders (Igharo et al., 2023). Repeated joint injury results in central sensitisation, hyperalgesia and deficient endogenous pain inhibitory control (De Wandele et al., 2022; Leone et al., 2020; Scheper et al., 2017).
No disease-specific pharmacological treatment has been identified for hEDS/HSD (Börsch et al., 2024). Given the considerable unmet clinical need, alternative treatment approaches are increasingly explored. Since the legalisation of cannabis-based medicinal products (CBMPs) in the UK in 2018, real-world evidence suggests that patients are predominantly obtaining these to treat chronic pain and anxiety disorders (Lynskey et al., 2023). Increasing reports of clinical use in hEDS/HSD cohorts suggest potential benefit in managing chronic pain and improving health-related quality of life (Dar, 2021; Dickinson et al., 2025). A survey of 500 hEDS/HSD patients found that 36.9% reported cannabis use for symptom management (Demes et al., 2020). Another survey of 24 hEDS patients found 48% had trialled cannabis, with 80% reporting therapeutic benefit (Doyle and Halverson, 2022).
In addition to the emerging evidence of clinical effectiveness, a role for cannabinoids in the treatment of hEDS/HSD is biologically plausible. Active components of cannabis include phytocannabinoids such as Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD). The major pharmacological target of phytocannabinoids is the endocannabinoid system, providing various potential therapeutic targets for symptom management in hEDS/HSD (Figure 1) (Bourke et al., 2022). THC is a partial agonist of cannabinoid receptor type 1 (CB1) and cannabinoid receptor type 2 (CB2). CBD is a negative allosteric modulator of CB1 and a partial agonist of CB2. Endocannabinoid signalling is implicated in the activity-dependent regulation of physiological processes, including mood, pain, gastrointestinal function, stress and immune and inflammatory response. Notably, treatment with CBD, THC and CB1 and CB2 receptor agonists has consistently demonstrated attenuation of pain-related behaviours in rodent neuropathic and inflammatory pain models (Finn et al., 2021).

Potential therapeutic targets within the endocannabinoid system for treatment of symptoms associated with hEDS/HSD. CB1 (red) are ubiquitously distributed throughout regions implicated in peripheral, spinal and supraspinal pain modulation, while CB2 (blue) are primarily distributed in peripheral tissues, microglia and immune response cells (20). Cannabinoids bind to CB1 and CB2 at peripheral nerve endings and skeletal muscle tissue. In the enteric nervous system, CB1 and CB2 are distributed on extrinsic sensory afferents, epithelial cells of the gut wall, mast cells, macrophages and lymphocytes. In the spinal cord, CB1 and CB2 are expressed on the dorsal root ganglion. CB1 is also expressed at the dorsal horn, while CB2 is present on neighbouring glial cells. Within the brain, CB1 is expressed in the prefrontal cortex, anterior cingulate cortex, somatosensory cortex, insula cortex, basal ganglia, amygdala, PAG, RVM, cerebellum, thalamus and hippocampus (21). CB1 are also expressed on oligodendrocytes, microglia and astrocytes. The widespread expression of the endocannabinoid system provides various potential therapeutic targets for symptom management in hEDS/HSD. Figure produced in BioRender.com.
The increasing reports of CBMP use in this cohort and speculated therapeutic mechanisms warrant further research. The aim of this paper is to capitalise on real-world evidence collected as part of Project Twenty21 (T21) (Sakal et al., 2022), a prospective registry study of patients prescribed CBMPs, to report baseline characteristics of hEDS/HSD patients seeking treatment and any changes in clinical outcomes for up to 12 months.
Methodology
Recruitment
T21 is a longitudinal, multi-centre, observational, UK-based registry study launched in August 2020 to produce real-world evidence on the safety and effectiveness of prescribed CBMPs (Sakal et al., 2022). Patients were invited to participate when making an appointment to obtain CBMP treatment at participating private clinics. Whilst no inclusion or exclusion criterion was applied for patients to participate in T21, patients required an established diagnosis and evidence of two failed licensed treatments due to guidance stipulating the prescription of unlicensed medicines. Prescribing doctors retained clinical discretion to not prescribe CBMPs, to prescribe from a formulary containing a range of CBMP formulations or to prescribe any CBMP legally available in the United Kingdom. Based on the Medical Research Council decision tools, Research Ethics Committee review and approval were not required. However, informed written consent was obtained from all participants for the use of their data for research purposes.
Participants who enrolled in the study before February 2022 were classified as one of the following ‘primary’ conditions via self-report: anxiety disorders, chronic pain, multiple sclerosis, post-traumatic stress disorder, Tourette's syndrome, epilepsy, substance use disorder and attention-deficit hyperactivity disorder. Participants were asked to self-report secondary conditions from a list of chronic health conditions or via free-text. In February 2022, study procedures changed so that patients receiving a prescription for CBMPs for the treatment of any primary condition were eligible to enrol. Participants entered their primary and/or secondary conditions from a list of chronic health conditions or via free-text. In this analysis, we included participants who identified ‘hypermobile Ehlers Danlos syndrome’, ‘hEDS’, ‘hypermobility syndrome’, ‘hypermobility spectrum disorder’, ‘HSD’, ‘hypermobility’, ‘joint laxity and subluxations due to hypermobility’, ‘hypermobile joints’ or ‘joint hypermobility syndrome’ as a self-reported primary or secondary condition between study commencement and December 2023. Due to the observational nature of the study, there was no control group. As a secondary analysis of an existing observational data set, the sample size was pre-determined.
Baseline characteristics
The sociodemographic and health characteristics of participants, all assessed via self-report at baseline, included age (years), gender (classified as female, male or non-binary), ethnicity, primary condition, any prior cannabis use with the intention of medical treatment, and the mean number of secondary conditions was reported.
Prescribed cannabis products
Participants self-reported the specific CBMPs prescribed at baseline and each follow-up appointment. The total number of distinct CBMPs prescribed over the 12-month study duration and the mean number of CBMPs prescribed to each participant during a consultation are reported. Products were classified based on their consumption method (inhalation vs. oil) and composition (‘CBD-dominant’, ‘THC-dominant’ or ‘CBD–THC balanced’).
Adverse events and dependency
Participants were asked to identify the CBMP they were using by name and to report any product-specific adverse events they experienced via free-text at each three-month follow-up, for up to 12 months. Additionally, participants were asked whether they had submitted a Yellow Card report. The Yellow Card Scheme is a UK-wide pharmacovigilance system whereby practitioners and patients can report adverse events related to the use of any prescribed medication or medical device to the Medicines and Healthcare products Regulatory Agency (McLernon et al., 2010). We reported the percentage of participants who submitted a yellow card report or an adverse event over the study duration, in addition to the type and severity of adverse events, classified as ‘mild’, ‘moderate’ or ‘severe’ via participant self-report.
Participants were asked to complete the 10-question cannabis-based medicines questionnaire (CBM-Q) to assess problematic medical cannabis use and dependency (Schlag et al., 2021). Participants rated the occurrence of seven domains from 0 (‘never’) to 4 (‘everyday’), including issues with thinking clearly and unfinished tasks related to CBMP use, use of CBMP to relax and external sourcing of CBMPs, in addition to side effect development and effectiveness over the duration of treatment, up to a total summed score of 37. The mean and standard deviation of the total score were reported at each follow-up interval. Assessments of other drug use were not undertaken.
Patient-reported health outcomes
Participants completed clinically validated questionnaires at baseline and at each three-month follow-up appointment. Quality-of-life, sleep and mood/ depression measures were completed by all participants, whilst chronic pain measures were only completed by participants who reported chronic pain or hEDS/HSD as a primary condition. Full descriptions of measures used are provided in Supplemental Materials (Table S1), including:
Quality of life assessed using EQ-5D-5L (Foundation, 2019). Mood and depression were assessed using the patient health questionnaire-9 (PHQ-9) (Kroenke et al., 2001). Sleep quality assessed using items from the Pittsburgh Sleep Index (Buysse et al., 1989). This modified assessment has been previously shown to have excellent psychometric properties in the T21 sample (Lynskey et al., 2023). Chronic pain severity and interference were assessed using the brief pain inventory (BPI; Cleeland and Ryan, 1994).
Prescription medication use
Participants were asked to disclose all current prescription medications at baseline and each follow-up via free-text. The percentage of patients prescribed medication and the mean number of prescribed medications at baseline are reported.
Opioid medications were manually identified from reported current prescription medications. The percentage of participants prescribed opioids at baseline was reported. Daily mean morphine equivalent (MME) was calculated for each participant reporting opioid use based on the frequency of opioid use multiplied by the dosage and the potency of the reported oral opioid (Table S2 in Supplemental material). The mean MME at baseline and three-month follow-up was reported.
Statistical analyses
All analyses were undertaken using RStudio (team, 2025).
An unpaired two-tailed t-test of the number of secondary conditions reported by hEDS/HSD patients and non-hEDS/HSD patients enrolled in T21 was undertaken. The mean and standard deviation of each patient-reported health outcome measure at treatment commencement and at each three-month follow-up, for up to 12 months, were reported. Effect size was estimated using Cohen's d (95% confidence interval (95% CI)) between baseline and three-month follow-up, whereby 0.2, 0.5 and 0.8 are indicative of small, medium and large effect sizes, respectively (Sullivan and Feinn, 2012). To model change in patient-reported health outcome measures during the 12-month period of observation, a repeated-measures Gaussian generalised linear model with full mixed effects was performed. A p-value under 0.05 was considered statistically significant. Post hoc power analyses of generalised linear models were undertaken using the ‘pwr’ package. Due to the variability of questionnaires completed by participants based on their primary condition and the varying attrition rate at each follow-up, missing data were omitted to maintain consistency, and the number of participants included in the analysis at each follow-up was reported. When spelling errors were apparent in the free-text reporting of medication use and adverse events, entries were manually reviewed and interpreted.
To model change in MME between baseline and three-month follow-up, a Gamma generalised linear model with inverse link function was performed. Due to typos in the free-text submission of opioid dosages, reducing the sample size, we imputed the mean MME of the same opioid taken by other participants across the duration of the study. A sensitivity analysis was undertaken, with missing data excluded.
Results
Sample characteristics
Between August 2020 and 31 December 2023, 4230 participants had enrolled in T21. Of these, 121 individuals identified hEDS/HSD as a primary or secondary condition at baseline/treatment commencement. The mean age of the hEDS/HSD sample was 36.8 ± 11.4 years, ranging from 19 to 68 years (Table 1). The majority of patients were female (76.0%), whilst 21.5% were male and 2.5% were non-binary. The sample was predominantly white (80.2%). Participants were seeking treatment for a range of primary conditions, most notably Ehlers-Danlos syndrome (47.1%) and other chronic pain conditions (39.7%). Co-morbid symptomatology was common, with participants reporting a mean of 6.16 ± 3.8 secondary conditions (range 0–20). Compared with patients enrolled in Project Twenty21 with conditions other than hEDS/ HSD who had a mean number of 3.46 ± 2.9 secondary conditions, patients with hEDS/HSD had a significantly higher number of secondary conditions (p < 0.001). The majority (87.6%) of participants were taking other pharmaceutical medications to treat their medical condition at baseline, reporting a mean of 5.1 ± 4.2 medications (see Table S3 in Supplemental material). Prior cannabis experience was common, with 62.0% of participants reporting current use of cannabis with the intention to treat their medical condition.
Characteristics of hypermobile Ehlers-Danlos syndrome/hypermobility spectrum disorder (hEDS/HSD) patients at treatment initiation.

Prescribed CBMPs
A total of 532 prescriptions for 107 distinct CBMPs were recorded over the 12-month study duration. Participants received a mean of 1.85 ± 0.88 (mean ± standard deviation) prescriptions (range = 0–6) at each consultation. The majority of CBMPs were THC-dominant (63%), followed by THC–CBD balanced (27%) and CBD-dominant (6%). Inhaled flower via vaporisation was the predominant (69%) formulation, followed by oil via oral/sublingual administration (27%). The product constituents and route of administration were missing for 4% of prescriptions.
Adverse events
Over the 12-month study duration, adverse events were reported by a total of six individuals (5.0%) who together reported 24 adverse experiences (Table 2). The most commonly reported experience was tachycardia (reported four times), followed by dry mouth, depression and anxiety (each reported three times). The majority (75%) of adverse events were assessed as mild, whilst 25% were rated as moderate. No Yellow Card reports were reported to be submitted. Problematic CBMP use (Table 3), assessed via the CBM-Q, did not significantly change over the duration of the study.
Number of self-reported adverse events over study duration and ratings of their severity.

Mean total cannabis-based medicines questionnaire (CBM-Q) score over the 12-month study duration.

Patient-reported health outcomes
Chronic pain
Chronic pain severity, assessed via change in the BPI pain severity score, decreased from a mean score of 5.76 ± 1.43 (mean ± standard deviation) to 4.76 ± 1.50 between baseline and nine-month follow-up (Table 4), reaching statistical significance at all follow-up intervals in this time period (p < 0.05). However, statistical significance was not maintained at the 12-month follow-up. Chronic pain severity score reduced with a small–moderate effect size of 0.47 (95% CI [0.16, 0.78]) between baseline and three-month follow-up. Furthermore, current pain severity reduced with a moderate effect size of 0.54 (95% CI [0.23, 0.84]), which was statistically significant for the 12-month period of observation (p < 0.05). Small–moderate effect sizes ranging from 0.24 to 0.46 were observed for pain ratings at worst, least and mean in the last 24 h. Patient-reported relief obtained from pain treatments and medications increased from a mean score of 40.94 ± 25.05 to 56.43 ± 26.02 during the period of observation, reaching statistical significance between six- and 12-month follow-ups (p < 0.05).
Comparison of self-reported health outcomes at baseline and up to 12-month follow-up.
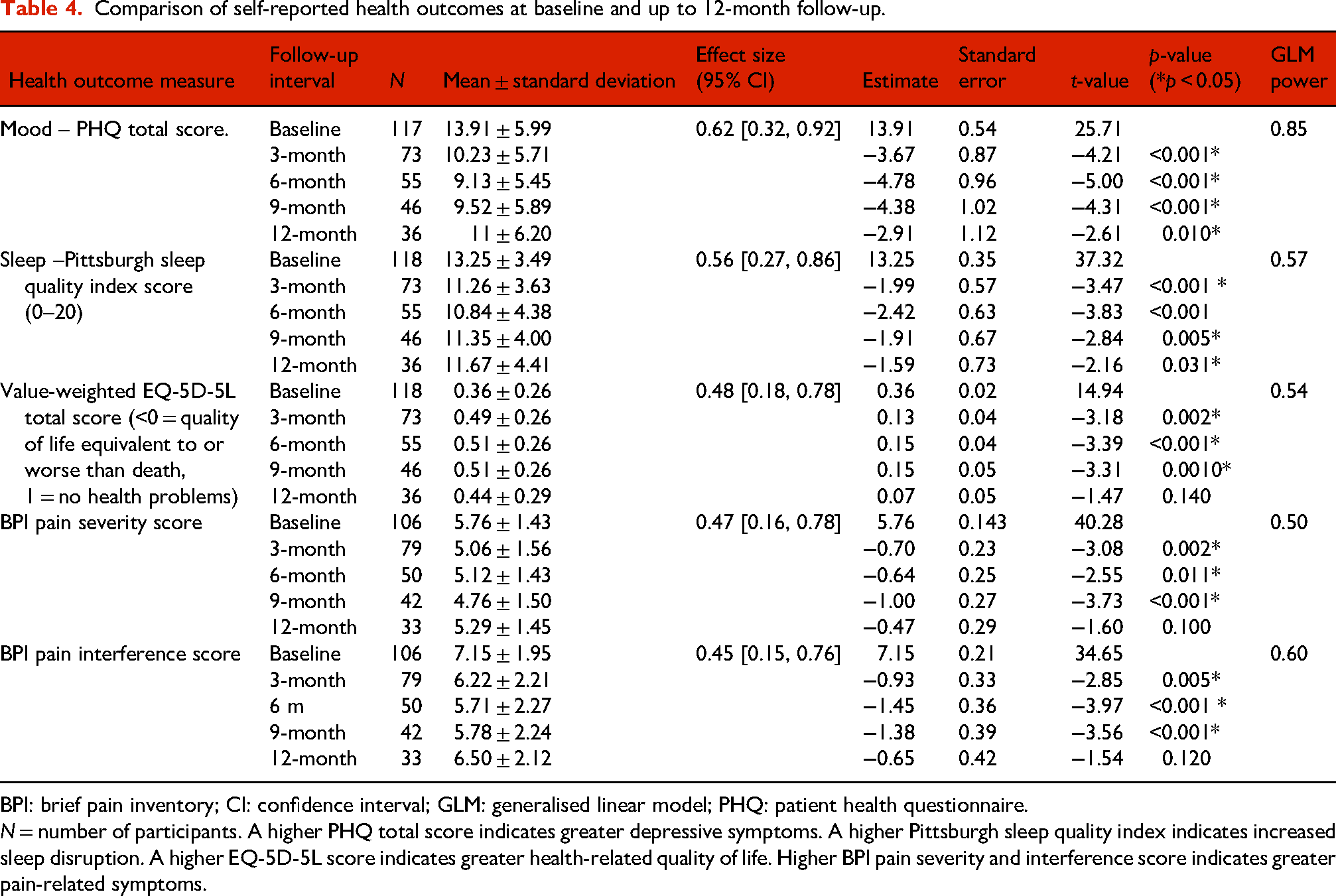
BPI: brief pain inventory; CI: confidence interval; GLM: generalised linear model; PHQ: patient health questionnaire.
N = number of participants. A higher PHQ total score indicates greater depressive symptoms. A higher Pittsburgh sleep quality index indicates increased sleep disruption. A higher EQ-5D-5L score indicates greater health-related quality of life. Higher BPI pain severity and interference score indicates greater pain-related symptoms.
The interference of chronic pain on general activities, assessed via the BPI pain interference score, reduced from a mean score of 7.15 ± 1.95 to 5.71 ± 2.27 between baseline and nine-month follow-up, reaching statistical significance at all follow-up intervals during this period (p < 0.050). However, this significance was not sustained at the 12-month follow-up. Overall pain interference score decreased with a small effect size of 0.45 (95% CI [0.15, 0.76]) between baseline and three-month follow-up. Similarly, small effect sizes ranging from 0.26 to 0.49 were observed across all assessed pain interference domains, most notably associated with improvements in enjoyment of life, sleep and undertaking general activities.
Mood/depression
Improvements in mood and depression assessed via change in total PHQ-9 score compared with baseline were observed at all follow-up time periods for up to 12 months (p < 0.050) (Table 4). The total PHQ-9 score at baseline was 13.91 ± 5.99 (mean ± standard deviation), indicating moderate clinical depression. At six- and nine-month follow-up, the mean total PHQ-9 score was below 10.00, suggestive of symptomatic improvement into the range of mild depression. Improvement in mean overall PHQ-9 score was observed between baseline and three-month follow-up with a moderate effect size of 0.62 (95% CI [0.32, 0.92]).
Sleep quality
Improvements in sleep quality scores assessed via change in total sleep score were observed for all follow-up time periods for up to 12 months compared to baseline (p < 0.050) (Table 4). The total sleep quality score at baseline was 13.25 ± 3.49 (mean ± standard deviation), indicating significantly disrupted sleep. Total sleep quality score reduced to 11.26 ± 3.63 at three-month follow-up with a moderate effect size of 0.56 (95% CI [0.27, 0.86]), suggesting decreased symptomatology related to sleep disruption. Within the assessed sleep domains, a moderate effect size of 0.63 (95% CI [0.33, 0.93]) was observed in the effect of improved sleep pattern reducing interference on daily activities. Small effect sizes ranging from 0.22 to 0.48 were observed related to improvements in falling and staying asleep, in addition to waking up too early.
Quality of life
Health-related quality of life, assessed using the value-weighted EQ-5D-5L score, increased from 0.36 ± 0.26 to 0.51 ± 0.26 between baseline and nine-month follow-up (Table 4), meeting statistical significance during this period (p = 0.001). Statistical significance was not met at the 12-month follow-up. A small effect size of 0.48 (95% CI [0.18, 0.78]) was observed between baseline and three-month follow-up, indicating a clinically meaningful improvement in quality of life. Small effect sizes ranging from 0.21 to 0.41 were reported across all quality-of-life domains, including mobility, self-care, pain and anxiety/depression. Improvement in ability to undertake usual activities was statistically significant at all follow-up periods up to 12 months (p < 0.05).
Prescribed opioid use
At baseline, 54 participants (44.6%) reported prescription opioid use to manage pain symptoms. At baseline, the daily MME used by these patients was 23.05 MMEs (Table 5). Change in MME met statistical significance at three-month follow-up of retained patients (p < 0.050), suggesting that treatment with CBMP may reduce prescription opioid requirement. The sensitivity analysis corroborated these findings.
Change in mean morphine equivalent with imputed mean values and sensitivity analysis with missing values excluded.

Discussion
We report significant improvements in sleep, quality of life, mood, pain severity and pain interference in hEDS/HSD patients during a 12-month period of observation following the prescription of CBMPs. A significant reduction in prescribed opioid use for the treatment of chronic pain was also observed. The observed improvements across a range of health outcome measures are a particular strength of the study design, highlighting the breadth of effects of CBMP treatment. The prevalence and severity of adverse events were minimal. Together, these findings provide real-world evidence supporting the safety and effectiveness of CBMPs for the management of symptoms associated with hEDS/HSD. As a disabling and multi-systemic condition with no identified pharmacological treatment, further research into the development of a CBMP licensed for the treatment of chronic pain symptoms associated with hEDS/HSD is warranted. Due to high rates of illicit cannabis use, we suggest that clinical and patient education related to medicinal cannabis be implemented as a harm reduction measure.
Research and development of treatments for hEDS/HSD have not received the attention and funding that they deserve (Börsch et al., 2024), signifying an urgent need for increased research in this disorder. It has been recognised that the condition is more prevalent than previously believed (Demmler et al., 2019) whilst being commonly associated with severe and impairing chronic pain (Voermans et al., 2010), prescription drug and opioid use (Schubart et al., 2019) and increased risk of psychiatric disorders (Cederlöf et al., 2016).
We report a predominantly young and female cohort, consistent with the existing literature assessing hEDS/HSD populations (Castori et al., 2010; Teran-Wodzinski and Kumar, 2023), whilst in contrast to non-hEDS/HSD patients enrolled in T21 who report a majority male population (Lynskey et al., 2023). The severity of the condition is evidenced in the current sample, where quality of life scores, assessed using the EQ-5D-5L, were remarkably low at baseline (0.36). In comparison to English women aged 35 to 39 who report a mean score of 0.85 (McNamara et al., 2023), this finding represents a disabled population with markedly poor health, which interferes with quality of life. At baseline, participant mean PHQ-9 scores were above 10, indicating an 88% specificity and sensitivity for meeting diagnostic criteria for major depression (Kroenke et al., 2001). High rates of co-morbidity were observed in this cohort, demonstrating the complex and multi-systemic nature of the condition (Halverson et al., 2023). Similarly, high rates of polypharmacy and opioid prescription were reported, which have previously been associated with negative health outcomes in hEDS/HSD patients (Upadhyaya et al., 2023).
Treatment with CBMPs was generally well tolerated, as adverse events were uncommon and predominantly mild in severity. The most commonly reported adverse events included tachycardia, dry mouth, anxiety and depression, consistent with previous reports of the mild and often self-resolving adverse effects observed in the assessment of CBMP safety (Almog et al., 2020; Lynskey et al., 2023). Whilst the risk of dependence following long-term CBMP use is potentially notable (Schlag et al., 2021), minimal CBM-Q scores were reported throughout the study duration. With no fatal overdose of cannabis ever reported in humans (National Academies of Sciences et al., 2017) and an excellent pre-clinical (Filipiuc et al., 2023) and currently reported safety profile, this adverse event profile compares favourably to commonly prescribed drugs for hEDS/HSD pain management such as opioids (Kurteva et al., 2021), tricyclic anti-depressants (de Farias et al., 2020) and nonsteroidal anti-inflammatory drugs (LaForge et al., 2023).
Improvements were observed in all assessed domains of symptomatology, suggesting that CBMP treatment was associated with both reduced perception of pain and improvements in functionality, sleep and quality of life. Treatment with CBMPs was associated with reductions in pain severity, with a statistically significant 1.00-point reduction in BPI severity score reported between baseline and nine-month follow-up. Other real-world evidence studies of chronic pain patients have yielded consistent, albeit greater changes in pain severity findings (Harris et al., 2022; Lynskey et al., 2023). This discrepancy may arise from the widespread pain of both chronic and acute onset experienced by hEDS/HSD patients and high rates of co-morbidity, resulting in particularly severe pain symptoms in this cohort. Correspondingly, whilst a reduction in opioid use was observed between baseline and three-month follow-up in our sample, we identified smaller reductions in MME following CBMP treatment in patients with hEDS/HSD compared to patients enrolled in T21 with other chronic pain conditions (Lynskey et al., 2023). Therefore, severe pain manifestations in hEDS/HSD may necessitate an ongoing opioid requirement.
THC-dominant flower for vaporisation was the most commonly prescribed CBMP composition, despite consensus recommendations for the dosing and administration of CBMPs suggesting that the prescription of CBD-dominant and balanced oils should initiate treatment (Bhaskar et al., 2021). Vaporisation is preferred for the management of breakthrough pain due to the rapid onset of action, which may alleviate acute symptoms of hEDS/HSD. Participants reported concurrent prescription of multiple CBMPs, suggesting that complementary cannabis products were being prescribed to meet individual patient needs.
Our findings corroborate a recent case series reporting significant improvements in sleep, pain, and health-related quality of life in hEDS/HSD patients prescribed CBMPs over an 18-month duration (Dickinson et al., 2025). Whilst the absence of a significant difference in BPI and health-related quality-of-life scores at 12-month follow-up observed in our study challenges the long-term effectiveness of CBMPs, the recent case series observed improvements up to 18 months. This discrepancy may arise from our approach of omitting missing data, whilst Dickinson et al. implemented a baseline-observation-carried-forward approach. Furthermore, we reported a predominance in the prescribing of cannabis flower, in contrast to high rates of cannabis-based oil use. Whilst our study relied on self-report, Dickinson et al. collected adverse events through self-reporting, direct questioning by researchers, or documentation by clinicians, which may have resulted in the increased number of adverse events described.
Strengths and limitations
Our findings provide an exploratory analysis that is resource-efficient in comparison to clinical studies such as randomised controlled trials (RCTs). However, there are significant limitations associated with real-world evidence. The lack of a placebo control group restricts conclusions about causation. Participants were not blinded, introducing performance bias and an increased likelihood of inaccurate and inflated reporting of patient outcomes (Hróbjartsson et al., 2014). There was also significant heterogeneity in the wide range of CBMP formulations prescribed, which restricts the ability to decipher the effects of individual products. Additionally, the heterogeneity of primary and secondary conditions of presenting patients introduces further confounding bias. As the diagnosis was based on self-report by the patient via free-text, diagnoses were not verified by clinicians, and the potential for response bias is notable. Therefore, the study sample presents a heterogeneous disease spectrum. This population is likely still representative of the often broad categorisation of chronic pain associated with hypermobility-related conditions due to ongoing challenges in clinical and research classification (Carroll, 2023). As reporting was based on patient self-report, attribution of adverse events specifically to a single cannabis-based medicinal product (CBMP) cannot be confirmed. There is potential for response bias and inaccuracies in distinguishing between product-related and unrelated adverse effects. Furthermore, the CBM-Q questionnaire used in this study has not been formally validated. Therefore, findings derived from this measure should be interpreted with caution, as psychometric properties have not yet been established.
As T21 enrolled self-funding patients obtaining treatment from private clinics, selection bias exists, and our findings may not be generalisable to low–middle socio-economic groups or wider National Health Service (NHS) populations. The reported high rates of current cannabis use further indicate response and selection bias. These patients may have improved tolerance for CBMP treatment compared with cannabis naïve patients and may be subject to recall bias when reporting health outcomes. Whilst a stratified analysis assessing differences in outcomes between participants with and without prior cannabis experience was not undertaken in this study, and represents a further limitation of this study. However, previous T21 analyses have reported minimal discrepancies in health outcome measures between cannabis naïve participants and participants reporting prior cannabis use (Lynskey et al., 2023). Additionally, accessing specialist pain management services may result in enhanced awareness of pain and treatment options, which may contribute to the reported improvements.
Due to the exploratory nature of the study aimed at describing longitudinal outcomes in a novel patient group, we did not control for multiple comparisons and report unadjusted p-values to avoid inflating Type II error. Whilst the omission of missing data contributes to potential attrition bias, we used a complete case approach as opposed to imputation techniques for missing data, as attrition patterns were heterogeneous. Although the sensitivity analyses corroborated our findings, the opioid analysis was limited by typos and missing dosages submitted as free-text by participants disclosing prescription medications. Whilst our findings were underpowered due to incomplete follow-up outcomes, our study benefited from a relatively substantial sample size considering the limited research into hEDS/HSD. Various uncontrolled factors likely contributed to missing patient follow-up, including unsustainable costs of treatment, treatment intolerance, lack of NHS funding, supply chain issues and stigmatisation (Beckett Wilson and Metcalf McGrath, 2023).
Future outlook
This analysis has identified a potential pharmacological treatment for currently unmet clinical needs associated with hEDS/HSD. The development of a placebo-controlled randomised controlled trial investigating a specific CBMP for the treatment of hEDS/HSD symptoms has been considered the gold standard for the assessment of safety and efficacy (Banerjee et al., 2022). However, there are challenges to the feasibility and study design of RCTs for CBMP treatment, including expense, lack of funding sources, non-patentability of specific whole-plant products and identification of an adequate placebo control. Furthermore, CBMPs are heterogeneous in pharmacology and formulation, with multiple products typically prescribed and titrated based on clinical response. Therefore, the outcomes of RCTs in isolation would be limited in identifying the most suitable CBMP for each clinical group and scenario. Additionally, common exclusion criteria applied in RCT study design, such as co-morbid illness and the prior or current use of cannabis and other pharmaceutical drugs, would exclude the majority of the hEDS/HSD participants in our analysis (Schlag et al., 2022). Considering the lack of disease-modifying or adequate symptomatic treatments for hEDS/HSD, future research should aim to improve mechanistic understanding and clinical outcomes, including via equitable access to NHS treatment.
Supplemental Material
sj-docx-1-dsp-10.1177_20503245251398260 - Supplemental material for Medicinal cannabis in the treatment of hypermobile Ehlers-Danlos syndrome & hypermobility spectrum disorder: Real-world evidence from Project Twenty21
Supplemental material, sj-docx-1-dsp-10.1177_20503245251398260 for Medicinal cannabis in the treatment of hypermobile Ehlers-Danlos syndrome & hypermobility spectrum disorder: Real-world evidence from Project Twenty21 by Lucy V Stafford, Michael T Lynskey, Anne K Schlag, David J Nutt and Jessica Eccles in Drug Science, Policy and Law
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AKS is Interim CEO, and ML is CRO of Drug Science. All Drug Science committee members are unpaid by Drug Science for their effort and commitment to this organisation. Drug Science would not benefit from the wider prescription of medical cannabis in any form.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
