Abstract
Objective
Identify a coherent nomenclature for Cannabis sativa L. derived products and their analogues.
Design
Research undertaken in parallel to the three-year assessment of Cannabis derivatives by the World Health Organisation. The scope is limited to Cannabis products intended for human incorporation (internal and topical consumption). Primarily embedded in pharmacognosy, the study incorporates a wide range of scholarly and grey literature, folk knowledge, archives, pharmacopœias, international law, field pharmacy, clinical and herbal medicine data, under a philosophical scrutiny. Generic and Cannabis-specific nomenclatural frames are compared to determine the extent to which they coincide or conflict.
Results
All lexica reviewed use weak, ambiguous, or inconsistent terms. There is insufficient scientific basis for terms and concepts related to Cannabis at all levels. No sound classification exists: current models conflict by adopting idiosyncratic, partial, outdated, or utilitarian schemes to arrange the extraordinarily numerous and diverse derivatives of the C. sativa plant. In law and policy, no clear or unequivocal boundary between herbal and non-herbal drugs, nor natural and synthetic cannabinoids was found; current nomenclatures need updates. In science, the botanical Cannabis lexicon overlooks parthenocarpy, and wide disagreement remains as to the taxonomy and systematics of the plant; chemical research should address differences in kinds between synthetic cannabinoids; pharmacopœias include little information related to Cannabis, and disagree on broader classes of herbal medicines, virtually failing to embrace many known Cannabis medicines. Since existing products and compounds fail to be categorised in an evidence-based manner, confusions will likely increase as novel cannabinoid compounds, genetic and biotechnological modifications surge.
Conclusions
The lack of clarity is comprehensive: for patients, physicians, and regulators. This study proposes an update of terms at several levels. It points at gaps in morphological descriptions in botany and pharmacognosy and a need for a metaphysical address of cannabinoids. Methods of obtention are identified as a common criterion to distinguish products; the way forward suggests a mutually exclusive nomenclatural pattern based on the smallest common denominator of obtention methods. In the context of a swelling number of Cannabis products being consumed (be it via medical prescription, adult-use, ‘hemp’ foodstuff and cosmetics, or other purposes), this study can assist research, contribute to transparent labelling of products, consumer safety and awareness, pharmacovigilance, medical standards of care, and an update of prevention and harm reduction approaches. It can also better inform regulatory policies surrounding C. sativa, its derivatives, and other cannabinoid-containing products.
Keywords
“What’s the use of their having names”, the Gnat said, “if they won’t answer to them?”
“No use to them”, said Alice; “but it’s useful to the people that name them, I suppose. If not, why do things have names at all?”
– Lewis Caroll, Through the Looking-Glass, and What Alice Found There
Introduction
The World Health Organisation’s (WHO) Expert Committee on Drug Dependence (ECDD) is mandated under the International Drug Control Conventions (IDCC) to conduct ‘medical, scientific and public health evaluation of substances’. It advises in a manner that is ‘determinative as to medical and scientific matters’, on ‘whether or not the substances assessed should be placed under international control’ (ECDD, 2018; Riboulet-Zemouli, 2018; World Health Organisation, 2015). In 2015, the ECDD started a series of assessments of ‘cannabis-related drugs’. Despite significant efforts, goodwill, and resources, obstacles prevented the Experts from providing a fully clear, complete, and methodologically sound assessment. Among the barriers, a significant one relates to the ‘specific provisions and terms used in the [IDCC]’ (Room, 2013) and in particular the way Cannabis and its derived products are referenced. Primarily relied on at the national level, the nomenclature of the IDCC aggregates pharmaceutically distinct products under the same labels that correspond to abstract concepts rather than to classes of products found in real life. In science, the botanical ‘cannabis confusions’ (McPartland, 2018; McPartland and Guy, 2017; Watts, 2006) in the taxonomic ranking of the plant are echoed by unclear morphological designations of ‘cannabis’ and ‘cannabis resin’ in pharmacy, all within the context of a complex pharmacological activity. Aware of that lack of conceptual and terminological consistency, the ECDD included, in its outcome recommendations sent to the United Nations (UN) Secretary-General in 2019, elements suggesting a renewed, modernised, clarified terminology for ‘cannabis drugs’ in the IDCC.
Beyond international organisations, unscientific or connotated epithets such as ‘marijuana’ continue to be used, including in policy or research papers, without proper justification. The terms ‘industrial cannabis’, ‘industrial hemp’, and ‘hemp’ remain commonly employed as synonyms. However, in some parts of the world, ‘cannabis industry’ refers to companies undertaking activities related to adult-uses of Cannabis products (e.g. for recreational purposes). In contrast, in others, it applies only to fibre-related industries. The most studied active phytoconstituent of Cannabis is interchangeably called ‘THC’, ‘delta-9-THC’, and ‘dronabinol’, although these terms correspond to different chemical entities. Many other impractical terminological habits arise from the use of similar words to designate products that are essentially different and have no comparison in terms of pharmacological effects. The ‘fibres’ which compose the stems of Cannabis plants, used to produce paper, clothing, or concrete, are often confused with ‘protein fibres’ obtained from seeds. The word ‘oil’ refers to the non-psychoactive fatty oil obtained from seeds (popularly called ‘hemp seed oil’, ‘hemp oil’, or ‘cannabis seed oil’). Yet, it is also used to denote some preparations of extracted/concentrated active compounds (‘hash oil’, ‘butane honey oil’, ‘cannabis oil’, ‘Rick Simpson Oil’, etc.; see Chandra et al., 2017; Krawitz et al., 2018; Szendrei, 1997). The current increase in innovation and diversification in production, distribution, and transformation of ‘cannabis’ into medical, pharmaceutical, nutraceuticals, food, cosmetic, and adult-use products will only accentuate that tendency to confusion.
Jurisdictions around the world are increasingly reviewing policies on medicine-related or adult-use ‘cannabis products’, or on derivatives of Cannabis for which purposes are not related to psychoactivity (‘hemp’). The analysis of cannabinoid content is useful, but not sufficient to distinguish types of products: a traditional ‘hashish’ and an oromucosal spray can indeed have similar thresholds. Policies are expected to be grounded in consensual customs, conventions, and standards, when not directly based on substantial science. Nevertheless, the potential bias implied by weak lexica, a non-scientific nomenclature, and numerous sociocultural terminological variants, might hinder efficient decisions in the field of Cannabis policymaking. The need to establish definitions, categories, limits, and boundaries between the different products, preparations, and substances derived from Cannabis has only become more urgent after the WHO’s assessment. Beyond the IDCC, other standards of reference should allow for a better understanding of the subject matter over which regulators are starting to work. In continuation of the multidisciplinary work undertaken by the ECDD, a comprehensive public health approach guided by evidence, urges. This study reviews the scope, basis, and limitations of the nomenclatures and semantics currently used as references in the legal and public health debate on Cannabis, parsing their relevance and their gaps and discussing approaches towards possible updates.
Materials and methods
The problematisation and design of the study emerged during discussions held at the UN Commission on narcotic drugs meetings from 2014 onwards. A common conclusion was shared by patients, physicians, pharmacists, pharmacologists, toxicologists, chemists, biologists, botanists, UN and WHO staff, national or local health authorities, diplomats, parliamentarians, lawyers, traditional healers, farmers, and people crafting C. sativa derivatives in various contexts, from all corners of the world: difficulties in understanding one another due to critical linguistic inconsistencies with ‘cannabis-specific’ terms, and the extreme challenge to overcome this stalemate from a single science, discipline, field, or focus.
The initial workstream, which sought to propose an evidence-based nomenclature of Cannabis products, turned out to be insoluble before a first address of existing terms and concepts from the perspectives of the philosophies of science and language. Ontology, both traditionally as a metaphysical address of the nature and essence of ‘things’ and, in its modern understanding, as an applied discipline of knowledge management (Merrill, 2011) provided methodological guidance in this regard. ‘The big task for the new “ontology”’ as Smith (2003) notes, ‘derives from what we might call the Tower of Babel problem. Different groups of data- and knowledge-base system designers have their own idiosyncratic terms and concepts by means of which they build frameworks for information representation’ (158). The assessments of the ECDD, progressively unfolding, emphasised this problem (see Krawitz et al., 2018). Their work (data collection, preliminary documentation, meeting outcomes) served as the basis to identify areas where ‘cannabis’ ontologies and terminologies seemed inconsistent, insufficient, or lacking scientific background. Complementary research identified existing nomenclatures for C. sativa derivatives within international legal instruments and documentation from a wide range of institutions. These sources were also consulted concerning non-Cannabis herbal drugs (i.e. phytopharmaceuticals). Phytopharmaceutical nomenclatures and herbal drugs categorification criteria were searched in manuals and guides of herbal pharmacy, herbal medical practice, and pharmacognosy. Research was undertaken at Biblioteca de Catalunya, Jardí Botànic, and CRAI of the Universitat de Barcelona (Barcelona, Spain), Conservatoire et Jardin Botanique de Genève, Faculté de Médecine of the Université de Genève, Archives of the League of Nations, Libraries of the UN and of the WHO (Geneva, Switzerland), Dag Hammarskjöld Library (New York, USA, remotely), the library of the UN Office on Drugs and Crime (UNODC) (Vienna, Austria), and at the Académie Nationale de Médecine, Académie de Pharmacie, Académie des sciences, Muséum National d’Histoire Naturelle and Université de Paris (France). References used in the 2019–2020 courses of the universities visited were favoured. Drug codifying compendia and other relevant references were also consulted at this stage.
Pharmacopœias consulted.
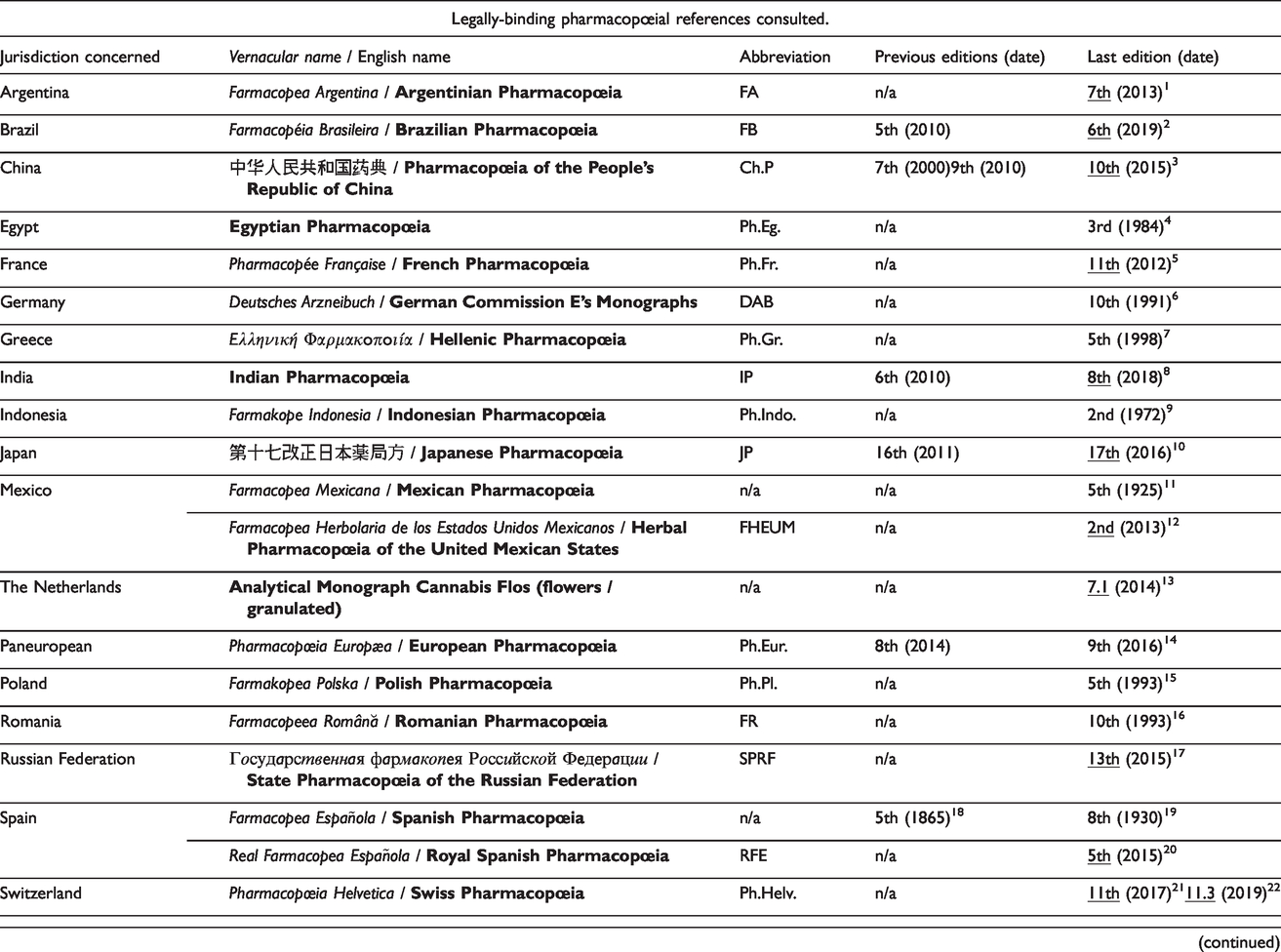
Pharmacopoeias underlined are those legally in force at the date of redaction of this article. References: 1 Administración Nacional de Medicamentos, Alimentos y Tecnología Médica, 2013; 2 Agência Nacional de Vigilância Sanitária, 2019; 3 Chinese Pharmacopoeia Commission, 2015; 4 Permanent Commission of the Egyptian Pharmacopœia, 1984; 5 Agence nationale de sécurité du médicament et des produits de santé, 2017; 6 Blumenthal et al., 1998; 7 Τσόκα and Βιολάκης, 1998; 8 Indian Pharmacopoeia Commission, 2018; 9 Departemen Kesehatan Republik Indonesia, 1972; 10 Ministry of Health, Labour and Welfare, 2016; 11 Unión Nacional de Farmacéuticos Científico-Cooperativa, 1925; 12 Comisión Permanente de la Farmacopea de los Estados Unidos Mexicanos, 2013; 13 Ministerie van Volksgezondheid Welzijn en Sport, 2019; 14 European Directorate for the Quality of Medicines, 2016; 15 Komisji Farmakopei Polskiej, 1993; 16 Institutul pentru Controlul de stat al Medicamentului și Cercetări Farmaceutice, 1993; 17 Министерство здравоохранения Российской Федерации, 2015; 18 Real Academia de Medicina de Madrid, 1865; 19 Real Academia Nacional de Medicina, 1930; 20 Agencia Española de Medicamentos y Productos Sanitarios, 2011; 21 Swiss Agency for Therapeutic Products, 2012, 22 2019; 23 General Council of Medical Education and Registration of the United Kingdom, 1914; 24 General Medical Council, 1968; 25 Medicines and Healthcare products Regulatory Agency, 2009, 26 2017; 27 United States Pharmacopeial Convention, 2019; 28 Cộng Hòa Xã Hội Chủ Nghĩa Việt Nam, 2002; 29 Brendler et al., 2010; 30 Organisation of African Unity, 1986; 31 Bellakhdar, 1997; 32 Oliver-Bever, 1986; 33 Organisation Ouest-Africaine de la Santé, 2013; 34 Ministry of Health and Family Welfare of India, 1989; 35 World Health Organisation, 2018b; 36 Sweetman, 2005; 37 Huang et al., 2009; 38 Willoughby et al., 1996; 39 Upton et al., 2014.
Literature searches were carried out up to 1 October 2019, in ProQuest, PubMed®, PMI, and Scopus™ using database-specific search strategies and appropriate keywords, truncation, symbols, and reference index terms, as appropriate. In addition, Google, DuckDuckGo, Archive.org, Google Scholar, and ResearchGate were employed to hand search additional articles and references. The door to serendipity stayed open.
The ‘Discussion’ section introduces neologisms: supplementary Appendix I exposes the methods followed to craft them.
There were several limitations in the research. The main limitation is contained in substance in the axis of research: the unsuitable, or nonexistent language and terminology for ‘cannabis’ derivatives. False cognates and other similar words relating to different concepts or different objects were overwhelmingly found between different authors or references, imposing the use of a vocabulary proper to the article as a manner of distinguishability (e.g. the use of phytopharmaceuticals, instead of herbal drugs whose meaning varies importantly among regions and areas of expertise). Another significant limitation is rooted in the Western embed of international pharmaceutical and medical standards; those could only partially be balanced due to language limitations.
A self-imposed limitation consisted in excluding from the scope of the article those products of C. sativa for which the purpose of use is not related to incorporation (internal or external consumption). C. sativa is a plant also used for the production of fibre, processed into paper, clothing, biocomposite materials, etc. These products are often processed in a manner that renders them unsuitable for incorporation, although they can be consumed (e.g. clothes are worn, paper is used, but not incorporated). Relying on the criteria of exemption by purpose present in the IDCC (i.e. products for which the purpose of use is not in relation to its potential psychoactive properties are exempt from drug control, see Riboulet-Zemouli, 2019) the products whose purpose is not that of human incorporation are excluded from the scope of this study. Because the products that can be incorporated are still numerous, the specific downstream criteria of the route of administration (which often vary among consumers, sometimes overlapping for the same product; and which are difficult to assess in a context of partial illegality) have been ignored, and left for further studies.
The IDCC, a set of treaties almost universally ratified, has shaped national laws, regulate research, and impose thorough obligations to Member States. Hence, the IDCC has been used as the paradigmatic point of departure for the study; however, all other classification criteria or existing Cannabis-related nomenclatures reviewed were given a comparable weight, and addressed with a similar approach, in an attempt to unearth a denominator.
The length of the article reflects the over three years of research involved in its production. The plan reflects the incremental aspect in which unfolded the research: it explores, from the most simple to the most sophisticated products, the limitations of existing terminology, why these limitations are rooted, beyond the terms, in conceptual confusions; the conclusion discusses lessons to be drawn, in view of possible new evidence-based terminologies to be proposed.
Results
The Home Medical Encyclopedia defines ‘cannabis’ as ‘[a]ny of the numerous psychoactive preparations derived from the hemp plant Cannabis sativa (such as hashish and marijuana)’ (American Medical Association, 1989: 230). Although commonly accepted, this is the perfect example of a circular definition: it explains the term ‘cannabis’ using this same word in the text of its definition (Kripke, 1980: 67–70, 1982). This ‘two cannabises’ approach is symptomatic of a fundamentally dichotomous vision of the word, used daily to designate two conceptually different concepts: either a plant genus, ‘Cannabis’, or a series of (mind-altering) products from that plant, ‘cannabis’ (Cherney and Small, 2016; Small, 2017: 1–5). This double-edged meaning affects and complicates the understanding of both ‘Cannabis’ as a plant and ‘cannabis’ as a product.
Polysemic acceptances of this word are profuse, worldwide, including in law. Legislations relating to ‘Cannabis’ and/or ‘cannabis’ often follow the codification of the IDCC (UNODC, 2013), particularly the Single Convention on Narcotic Drugs of 1961 as amended in 1972 (C61) and the Convention on Psychotropic Substances of 1971 (C71). This latter treaty does not directly mention ‘cannabis’; it comprises only pure compounds and includes dronabinol (ECDD, 2019: 41–44) as well as its stereochemical variants and isomers. Dronabinol is the international nonproprietary name (INN) for the (–)-trans stereoisomer of the delta-9 isomer of tetrahydrocannabinol (THC). Two confusions often arise (Figure 1): while the INN seems to only designate (–)-trans-delta-9-tetrahydrocannabinol, some references like United States Pharmacopœia (USP), recognise as dronabinol all four enantiomers of delta-9-tetrahydrocannabinol. For others, the word dronabinol is understood as referring only to delta-9-tetrahydrocannabinol obtained ex vivo. According to WHO, however, ‘dronabinol’ corresponds to (–)-trans-delta-9-THC, either obtained in vivo or ex vivo (ECDD, 2018: 33).
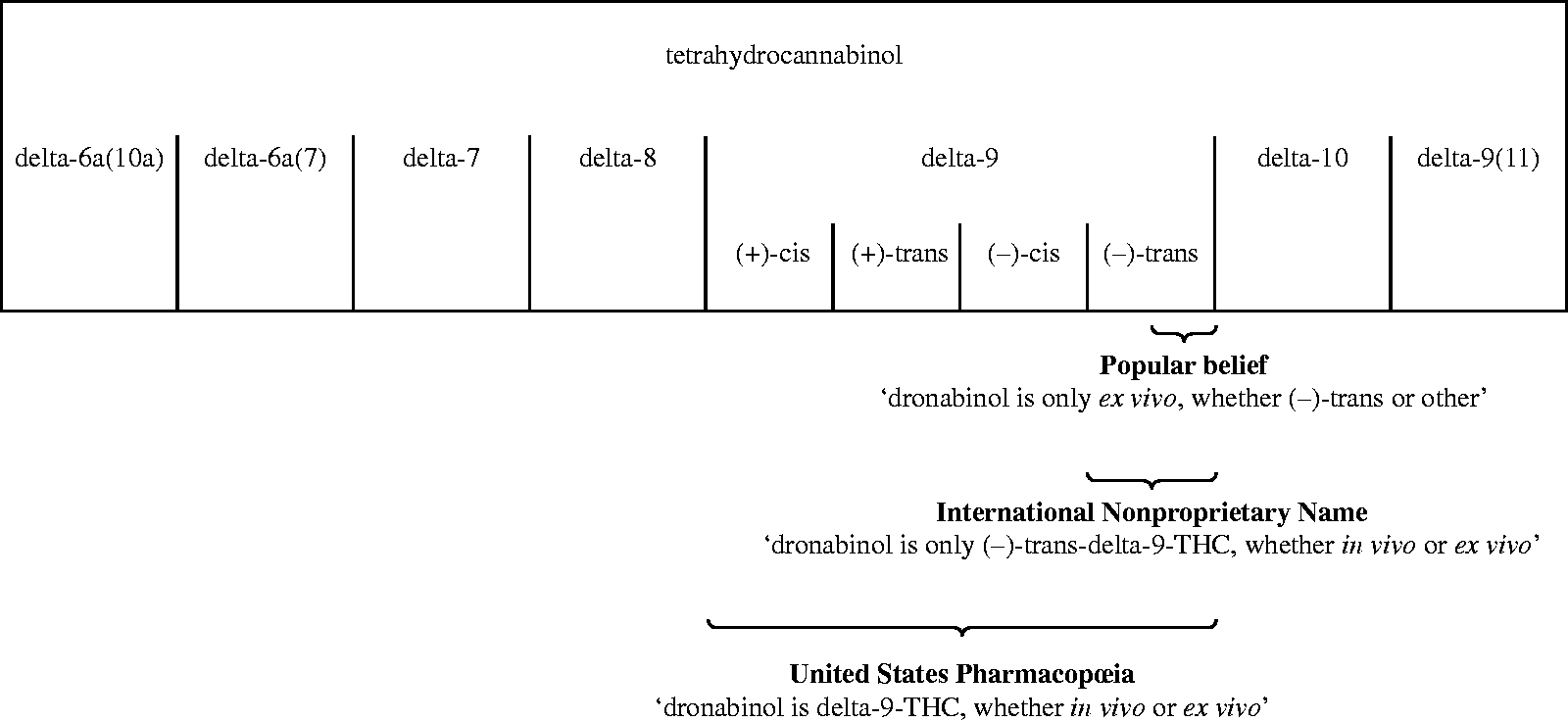
Conflicting ontologies: dronabinol and THC. THC: tetrahydrocannabinol.
The C61, on its side, includes four occurrences of the word in its Article 1 on definitions (UNODC, 2013: 24–26):
‘Cannabis plant’ that is ‘any plant of the genus Cannabis’ (art. 1[c]), ‘Cannabis’, defined as ‘the flowering or fruiting tops of the cannabis plant (excluding the seeds and leaves when not accompanied by the tops) from which the resin has not been extracted, by whatever name they may be designated’ (art. 1[b]). The Commentary on the C61 treaty, prepared by the office of the UN Secretary-General (hereinafter called Commentary, see UN, 1973), explains that ‘the term “cannabis” […] covers all tops including those which are not yet dried, as well as those of the male plants’ (UN, 1973: 2 §1). ‘Cannabis resin’ defined as ‘the separated resin, whether crude or purified, obtained from the cannabis plant’ (art. 1[d]). The Commentary further explains (UN, 1973: 5 §3) that ‘resin, however, becomes “cannabis resin” only when it is “separated” from the plant; without such separation, it remains a part of the cannabis plant, and if in the top part, of “canna bis”’. ‘Extracts and tinctures of cannabis’, for which no definition is provided.
The C61 treaty is a delicate equilibrium between the labour of merging several previous international legal instruments into a new, single text, and the geopolitical developments of the postwar era (Jelsma et al., 2014; Krawitz et al., 2018: 6–11; Riboulet-Zemouli, 2018). The limited knowledge of the active constituents of the plant at the time contributed such imprecise language. A background paper to ECDD’s 2012 meeting explains: ‘in the half-century since the 1961 Convention was adopted, there has been considerable developments in the terminology used’ (Room, 2013). Today the efficiency of this nomenclature, used globally for several decades, seems to be backed by weak evidence. It is, however, representative of a trend in regulatory terminological shortcuts, as well as in the legal polysemy of the very word cannabis. C61 defines drugs as ‘any of the substances in Schedules I and II, whether natural or synthetic’ (art.1(j)). Only ‘cannabis’, ‘cannabis resin’, and ‘extracts and tinctures of cannabis’ are listed in these Schedules (UN, 1961: 239). Consequently, ‘cannabis plant’ is not seen as a drug as per the C61 regulations.
‘Cannabis’ as a plant
Beyond the ancient uses of the word ‘cannabis’ to refer to this particular plant, modern science legitimises the word with Linnæus (1753: 1027; see also and Watts, 2006) describing a monogeneric plant under this name. At the higher family level, its belonging to Cannabaceae is nowadays generally accepted (McPartland, 2018; Stevens, 2001a onwards, 2001b onwards; Watts, 2006). However, although unrivalled at the genus level, the systematic classification of Cannabis at lower taxonomic levels continues to be an essential subject of controversy (Chopra and Chopra, 1957; Clarke and Merlin, 2013; Lynch et al., 2016; McPartland, 2018; Small, 2017; Stevens, 2001a onwards; Yang et al., 2013). Contemporary findings tend to show C. sativa as the only monospecific expression of the genus Cannabis; the rich genetic diversity responsible for the numerous types of C. sativa would be expressed at a lower level (in the taxonomic ranks of subspecies, variety, forma, or cultivar, see Lynch et al., 2016; McPartland, 2018). While no consensus arises, none of the findings matches the traditional distinction between drug-type and fibre-type Cannabis mimicked in folk language by the use of terms such as ‘marijuana’ and ‘hemp’. Hazekamp et al. (2010) even state that such a ‘distinction between the two types may have limited relevance for medicinal research’ (1037). Genetic diversity happening below the species rank reflects substantial variabilities of C. sativa crops, which might explain the reported use of ‘drug-type’ varieties for the obtention of fibres, and vice versa (Clarke and Merlin, 2013; Lynch et al., 2016; Szendrei, 1997). The UNODC (2009) finds: Chemical and morphological distinctions by which Cannabis has been divided into these subspecies are often not readily discernible, appear to be environmentally modifiable, and vary in a continuous fashion. For most purposes, it suffices to apply the name Cannabis sativa to all Cannabis plants encountered. (7)
‘Cannabis’ as phytopharmaceuticals
Surprisingly, evidence-based definitions of the word ‘cannabis’ understood as a series of products obtained from the C. sativa plant are absent from scientific literature. Besides the living plant, the word is also used to refer to one of the products of the harvest of C. sativa plants, a particular botanical part used for human consumption in relation to a sought-after pharmacological activity. This ‘cannabis’ is often referred to as buds, flowers, inflorescences, bracts, heads, or tops.
Additionally, an important number of extracts or transformed derivatives of the C. sativa plant, at different stages of elaboration, are commonly referred to as ‘cannabis’. In the 1950s, the Multilingual list of narcotic drugs under international control (UN, 1958: 13–14) defined ‘cannabis’ as both raw herbal parts, and more elaborate products such as confections, beverages, and preparations. Sixty years later, the WHO also considered a number of prepared, compounded, or elaborate products, mostly traditional ones, under the agenda item ‘cannabis’ rather than under that of ‘extracts and tinctures of cannabis’ (Cannazza and Citti, 2018a: 5, 2018b).
The C61 recognises differences between simple botanical ‘cannabis’, the processed ‘cannabis resin’, and ‘extracts and tinctures of cannabis’. However, no clear distinction between the two latter entities is mentioned in the Convention, the processes to obtain one or the other are sometimes similar (Table 2); the entities ‘cannabis’, ‘cannabis resin’, and ‘extract and tinctures’ are sometimes considered interchangeably (UN, 1973: 2 §5) even though they are listed in different Schedules and eventually subject to a distinct policy régime.
Nomenclature of methods of obtention of Cannabis products in the 1961 Convention on narcotic drugs.

The products in bold are those defined as ‘drugs’ in the Convention (Art. 1[j]), as of 2020.
In literature, neither simple ‘cannabis’ nor elaborate products are defined in a better way. In other international instruments, Cannabis and its derivatives are present under limited functional appellations, not always reconcilable with the ones used in the IDCC. For example, the World Trade Organisation’s Harmonised Tariff System contemplates a category (No. 1302.19) titled ‘Cannabis flower/Cannabis extract’ (Krawitz et al., 2018: 20).
Defining the basic botanical ‘cannabis product’
Scientifically identifying the ‘buds’, ‘tops’, ‘ganja’, ‘heads’, or ‘flowers’ is not an easy task, and consensus lacks on their precise botanical designation. C. sativa is an annual dioecious (although sometimes monoecious or hermaphrodite) flowering herb (Clarke, 1981; Evans, 2009) producing geniculate achenes as fruits (Evans, 2009; Upton et al., 2014: 8). Reproduction occurs through wind-dispersed pollen liberated from the flowers of staminate (i.e. male) plants (Chandra et al., 2017; Small and Antle, 2003). Chopra and Chopra (1957) described the parts of C. sativa used for the production of drugs as ‘flowers, leaves (and the resinous matter derived therefrom), fruit, young twigs, and bark of the stem’. Twenty-three years later, Kimura and Okamoto found that traces of active compounds were present at all stages of the plant’s life, in both staminate and pistillate (i.e. female) C. sativa, although mainly ‘contained in the parts in prosperous growth, and especially concentrated at the bractlet in the period when the seeds are at the peak of ripening’ (Kimura and Okamoto, 1970). They refer interchangeably to these botanical parts, containing the most active phytochemicals, as pistillate ‘tops’, ‘flowers’, ‘inflorescences’, or ‘fruits’. More accurately, the active ingredients (mostly phytocannabinoids, terpenoids, phenols) are biosynthesised inside the multicellular, glandular heads of epidermal multiseriate stalk trichomes (Chandra et al., 2017; Clarke, 1981; ECDD, 2018; Evans, 2009: 525–527, 553–557; Flemming et al., 2007: 8; Frank, 2018; Happyana et al., 2013; Heinrich et al., 2017: 18, 149–150; Turner et al., 1981). While these epidermal glandular trichomes (EGT) are also present in a much lesser extent on leaves, stem, and other parts of the plants (explaining the results of Kimura and Okamoto, 1970; see also Turner et al., 1981), their dense concentration around the reproductive parts of pistillate plants (the so-called ‘buds’ or ‘heads’) is a major factor in the choice of the parts to harvest and transform for psychoactive purposes. Hence, the most common and well-characterised phytoconstituents are found only in trace amounts outside of the EGT found on the leaves, flowers, and fruits (Jin et al., 2020).
‘Flowers’ is the name for the preferred harvested parts bearing phytopharmaceutical ingredients and is the most commonly encountered designation in the literature as well as references like USHP, Ph.Helv, UNODC (2009), or in the various ECDD (2018, 2019) reports. C61 accepts both flowering and fruiting tops in its definition but excludes seeds, which, however, are sometimes referred to as fruits, such as in Pharmacopœia of the People’s Republic of China (Ch.P) and Japanese Pharmacopœia (JP) (Chinese Pharmacopoeia Commission, 2015; Ministry of Health, Labour and Welfare, 2016).
Numerous references use imaginative formulations to mean that flowers have seeds, without directly verbalising ‘seeded flowers’. This would indeed oppose the basics of botany where seeded flowers are not an option. As early as 1894 it was noted that the materials used for the production of a local extract in India were ‘flower heads, which are now full of seed, discarding the coarser leaves’ (Indian Hemp Drugs Commission, 1894). 123 years later, a paper describes the botanical parts harvested for their psychoactivity, in Nepal, as the ‘mature seeded female inflorescences’ (Clarke, 2007). Creative alternatives, such as buds, bractlets, calyxes (Frank, 2018), ‘bracts which surround the ovaries’ (Dewick, 2012), or seedless floral clusters were found, reflecting a lack of consensus around the designation of these parts. The sentence describing the frontispiece photograph of Cannabis and Health (Graham, 1976: III) achieves the feat of referring to the same specimen simultaneously as a ‘fresh flowering top’ and a ‘developing fruit’. These curious phraseologies can be explained by the fact that, in traditional outdoor cultivation, staminate and hermaphrodite plants can occur in the field, thus resulting in the pollination of some flowers, and their transformation into fruits bearing seeds (Chopra and Chopra, 1957; Clarke, 2007). C. sativa farmers have developed strategies to avoid this and reduce the presence of seeds in harvestable crops, mostly by impeding pollination. Chopra and Chopra (1957) noted that an important operation of India’s 1950’s C. sativa farmers consisted of going ‘through the field cutting down all staminate plants’ for ‘preventing seed formation’. Hamayun and Shinwari (2004) explain that the early flowering stage of pistillate flowers allows for easy removal from the fields. They note that ‘even the young fruit of the female cannabis plant’ is used for the production of psychoactive products. The plants resulting from crops where such sexual selection has taken place are known as ‘sinsemilla’ (Clarke and Merlin, 2013; ECDD, 2018: 18), derived from Spanish sin semilla, literally meaning ‘without seed’. The UNODC (2009) explains: Cannabis with the highest level of THC is comprised exclusively of the female flower heads (“buds”) that remain unfertilized throughout maturity and which, consequently, contain no seeds. The production of sinsemilla requires identifying the female plants and ensuring that they are not exposed to pollen. (11)
The consideration of buds as flowers conflicts with the repeated mentions of a seemingly needed maturity of flowers to be harvestable. Flowers do not ripen: they wilt (senescence) and turn into fruits which, on their turn, do mature. Literature repeatedly suggests that the optimal moments for the harvest of seeds, and of content rich in cannabinoids, occur simultaneously. Dewick (2012: 120) explains that ‘resin is produced from the time flowers first appear until the seeds reach maturity’, as confirmed by Clarke (1981: 12). Kimura and Okamoto (1970) go in the same direction: although they do not refer to the word fruit, they distinguish ‘bractlet’ from ‘flowers’, the former appearing chronologically after the latter. They explain that bractlets bear a higher concentration of cannabinoids two months after analysing the staminate flowers, which are known to appear slightly before pistillate flowers and to have a maximum life duration of three weeks (Small and Antle, 2003). These observations could mean that pistillate flowers would survive five weeks or more after their staminate counterparts have lost the ability to pollinate them. That would be an unprecedented form of dioecism in the Plantæ kingdom, with the only known seeded flowers ever, and where male and female iterations would enter flowering at different periods.
From a different angle, yet fully aligned with modern botanical sciences, considering ‘bud’ not as bractlets, flowers, or inflorescences, but as fruits or infructescences, might be insightful. In the past defined as ‘structures bearing seeds’, fruits are currently described in a more sensible way as ‘matured pistil or ovary of the flower, with or without accessory structures’ (Blumenthal et al., 1998: 59) or as ‘a derivative of the gynoecium or whatever extracarpellary part it may be united with during the fruiting stage’ (Esau, 1977: 430; Scagel et al., 1967: 559). Botanical sciences do contemplate the development of fruits from unfertilised female flowers, a mechanism termed ‘parthenocarpy’ and widely spread among dicots (supplementary Appendix II).
These observations suggest that the basic ‘cannabis product’ might not be composed of flowers, but fruits – more precisely, parthenocarpic fruits. Because C. sativa naturally has both abilities to produce seeded and seedless fruits, the genus would be more accurately classified as a ‘facultative parthenocarpic plant’ (Koltunow et al., 2002), and when some seeds remain in an otherwise seedless Cannabis ‘top’ it would be called a ‘partially parthenocarpic infructescence’.
The phenomenon of parthenocarpy in C. sativa and possible reasons for its oversight in literature are discussed and illustrated in supplementary Appendix II. In this study, the expression ‘parthenocarpic fruits’, or ‘parthenocarpic infructescence of C. sativa’ (which can be abbreviated as ‘pioC’), is therefore preferred to ‘inflorescence’, ‘top’, ‘bract’, or other terms, when referring to the mature seedless reproductive parts of pistillate C. sativa plants.
Defining products derived from the Cannabis plant
In popular and folk lexica of extracts, tinctures, resins, and other derivatives of C. sativa for human consumption (except pioC themselves), a seemingly standard corpus of terms is used globally (Backes, 2014; Cannazza and Citti, 2018a, 2018b; Chambers, 2019; Daniulaityte et al., 2017; Jaffe, 1995: 982–989; Ministry of Health and Family Welfare of India, 1989; Nicoletti Motta, 2018; Oliver-Bever, 1986; UN, 1958; UNODC, 2009; World Health Organisation, 2016; Zavřelová, 2017) although it refers to different concepts, depending on sources, area, and time. No universally accepted terminology is used, but some terms recur. For instance, while the word ‘hashish’ in Morocco refers to the living plant from which resin is extracted (Bellakhdar, 1997: 233), the Encyclopedia of Drugs and Alcohol explains it is ‘the Arabic word for a particular form of Cannabis sativa … the resin derived principally from the flowers, bracts and young leaves’ (Jaffe, 1995: 541). For Oliver-Bever (1986: 78) it is a ‘purified alcoholic extract’ and for the UNODC (2009: 16) hashish consists of ‘resinous secretions of the plant, produced in glandular trichomes’. The Martindale explains that the word ‘is often applied to the resin, although in some countries, hashish is applied to any cannabis preparation’ (Sweetman, 2005: 1666). Many C. sativa products linked to traditional medical practice or entheogenic uses, still partially undocumented (Abbott, 2014; Abdool, 2013; Bellakhdar, 1997: 232–234; Kutesa, 2018) are victims of similar terminological confusions. Another example is the inappropriate use of the word ‘oil’ (Daniulaityte et al., 2017; Szendrei, 1997; WHO, 2016) to describe all sorts of products very distinct from the common understanding of ‘oil’, mostly limited to fatty oils and essential oils. Although a lot of these ‘cannabis’-specific terms seem universally known and used, they have no consistent, nor universal definition.
Generally, folk distinctions between ‘resin’ and ‘extracts’ recur (Nicoletti Motta, 2018; Satchel, s.d). Resin is often described as the product of a simple and often traditional extraction process. The Encyclopedia of Drugs and Alcohol sees hashish as ‘a concentrated resin containing increased amounts of Δ9-THC’, ‘derived principally from the flowers, bracts, and young leaves of the female hemp plant’, and that ‘contains cannabinoids’ (Jaffe, 1995: 429, 541). Some products like rosin, dry sift, or bubble hash are alternatively referred to as either a form of hashish or as extracts (Cannazza and Citti, 2018a, 2018b; Medicaljane, s.d). This confusion is absent from another widely used approach, which distinguishes products in two complementary categories based on the use of solvent during the extraction process, or not (Zavřelová, 2017; see also supplementary Appendix III).
Defining resin
Because ‘cannabis’ is defined in C61 as tops of C. sativa ‘from which the resin has not been extracted’, it is suggested that extraction is the method of obtention of ‘resin’ from ‘cannabis’. Such definition corresponds to ‘manufacture’ in Article 1(n): ‘all processes, other than production, by which drugs may be obtained [including] refining as well as the transformation of drugs into other drugs’. Additionally, Article 1(d) explains that resin is ‘separated’ from C. sativa, which conflicts with Article 1(t) for which this operation is ‘production’ (‘the separation of […] cannabis and cannabis resin from the plants from which they are obtained’). ‘Resin’ can therefore, under the Convention, be obtained by extraction, by manufacture, and by separation, as outlined in Table 2.
An additional detail about ‘resin’ is its state of being either ‘crude or purified’. The Commentary notes: ‘the separated resin is “cannabis resin” not only when it is “purified”, but also in its “crude” state, i.e. when it is still mixed with other parts of the plant’ (UN, 1973: 5 §3). The concept of purification is invoked to mean the elimination of residual botanical elements. It relates to the processes of refining described in Article 1(n) as being ‘manufacture’. Production and manufacturing are the two genuine ‘operations by which “drugs” […] are obtained’ (UN, 1973: 15 §2). However, a more complex set of terminology, summarised in Table 2, is used to refer to the processes of elaboration of C. sativa and pioC derivatives. Boundaries in C61 are unclear and leave a margin for bias and errors of interpretations. Because a different régime of control under the IDCC is currently applied to these products, this may have non-negligible legal implications.
Besides the language of the IDCC, the word resin is widely used to describe complex mixtures of phytochemicals secreted by plants (Evans, 2009: 298). Bone’s Principles of herbal pharmacology (2013) explains the polysemy of the word resin: The term is used in several contexts. When certain plants are damaged, either by incision or naturally due to the action of animals or the environment, they secrete a viscous fluid that soon hardens … Such resins are often associated with essential oils (oleoresins), with gums (gum resins) or with oil and gum (oleo-gum resins). Their resin components, which mainly comprise diterpenes known as resin acids, resin alcohols and resin phenols, are soluble in alcohol and ether but are insoluble in water and hexane. In another context, the term ‘resin’ (or occasionally ‘resinoid’) means the part of the plant that is soluble in ether or alcohol … These resins are chemically diverse and can contain resin acids, pyrones, lignans, esters and glycosides amongst others. (38)
The WHO ECDD (2018: 18) considers that ‘resin’ originates from ‘resinous secretions’ of the plant’s EGT which contain, in addition to numerous phenols and terpenoids, dronabinol and other phytocannabinoids. Analytical explorations of the exudate secreted by EGT show the most present components numerically are terpenoids, phytocannabinoids, hydrocarbons, sugars, nitrogenous compounds, phenols, and flavonoids, with fatty, simple and amino acids, ketones, esters, lactones, aldehydes present in a much less significant yield. These substances ‘have all been identified as a constituent of some preparation of Cannabis: herbal plant material, whole extracts, and chromatographic fractions, or illicit material such as hashish’ (Hazekamp et al., 2010: 1038–1039).
All of these compounds correspond to those described as characteristics of plant resins in literature. They are obtained by the secretion from a plant’s glands and are all known for their ether soluble properties (Clarke, 1981; Evans, 2009: 525–527, 557; Happyana et al., 2013). In other words, all acceptations of ‘resin’ (glandular origin, composition, solubility in alcohol) match with the material contained in C. sativa trichomes’ glandular heads. The systematic and substantial yield of aromatic metabolites in addition to phytocannabinoids suggests oleoresin might be a more accurate term than resin to describe the ether soluble separable content from EGTs.
It is generally assumed that the authors of the C61 agreed to use ‘resin’ in the absence of more advanced knowledge about its chemical composition, as a way to ensure dronabinol, by then unidentified, would fall under control in all circumstances. The assumption that the active compound(s) of C. sativa were included in resin is reflected in the Commentary, which refers, for instance, to the ‘pharmacologically strongly active resin’ (UN, 1973: 2 §1). Pure THC/dronabinol itself was eventually placed in the schedules of another IDCC treaty: the C71. Interestingly, countries’ authorities have been encouraged by the International Narcotics Control Board (INCB) to consider dronabinol of synthetic origin under the régime of C71, while dronabinol derived from the C. sativa plant would be considered under C61 rules (INCB, 2019: 8). A gross inconsistency (see Figures 1 and 2) which has been proposed a solution in the ECDD (2019: 45–47, 49–50) recommendations: placing all dronabinol-related isomers and stereoisomers within the same Schedule.

Conflicting ontologies of dronabinol and THC: impact on international control. C61 I: Schedule I of the 1961 Single Convention; C61 III: Schedule III of the 1961 Single Convention; C61 IV: Schedule IV of the 1961 Single Convention; C71 I: Schedule I of the 1971 Convention; C71 II: Schedule II of the 1971 Convention; THC: tetrahydrocannabinol. aOnly when ‘from synthetic origin’ (INCB, 2019: 8); bOnly when obtained from C. sativa (INCB, 2019: 8); cOnly for some preparations for medical use (ECDD, 2019: 55).
Notably, since 1961, the chemical composition of C. sativa oleoresin has been figured out: it was found to be exceptionally diverse and variable (Baram et al., 2019), with components interacting and interplaying above and beyond their individual pharmacological activity (Ben-Shabat et al., 1998; Rather et al., 2013; Russo, 2011). This dramatically influences the oleoresin’s overall therapeutic effects far beyond that of dronabinol, dependent on ‘complex interaction between molecules and multiple targets’ (Atakan, 2012; Baker et al., 2000; Baram et al., 2019; Russo, 2011) – a phenomenon which is typical of herbal medicines (Rather et al., 2013), called ‘cooperative effect’ or ‘entourage effect’ (Ben-Shabat et al., 1998; Oña and Bouso, 2019). This entourage effect results in C. sativa drugs binding to different neuronal targets, an effect in turn called ‘polypharmacology’ (Gertsch, 2011; Oña and Bouso, 2019).
In other words, ‘cannabis resin’ in IDCC as well as folk language, relates to a single concept: the sum of all ingredients contained in the material separated from the glands of the epidermal capitate trichomes from C. sativa plants. In Water is not H2O, Weisberg (2006) warns that there are not always ‘a straightforward connection between scientific kinds and the natural kinds recognised by ordinary language users’ (337). Similarly it can be stated that resin is not THC, refraining from amalgamating ‘cannabis resin’ with THC/dronabinol (or any other single cannabinoid or simplified composition) as an epistemological precondition.
Defining extracts
A changing nomenclature at the level of the IDCC
‘Extracts and tinctures of cannabis’, although present in the Schedules of C61 since its inception (UN, 1961: 239) are nowhere defined in the Treaty. No additional information is provided in the Commentary. To document the ECDD assessment of that particular category of Cannabis products, the WHO tasked five teams of authors to draft reports on, respectively, the chemistry, pharmacology, toxicology, therapeutic use, and epidemiology of the products (Krawitz et al., 2018: 17–22). The five documents result in different understandings of the products covered (or not) by this category, as presented in Table 3.
Ontological conflicts: ‘extracts and tinctures of cannabis’ according to different authors of the pre-review documentation used at the 40th ECDD meeting.

Adapted from Krawitz et al., 2018.
The word ‘extraction’ derives from Latin extraho/extrahere, formed by the lemmas ex- (‘out of’) and trahō (‘to pull’, ‘raw’, ‘drag’), meaning together: to withdraw, drag-out, remove. Popular meaning covered by the word ‘extraction’ is often that of a process consisting in ‘the separation of a substance from a matrix’ (Wikipedia, s.d). Separation is defined in the Encyclopedia of Separation Sciences as a ‘process of any scale by which the components of a mixture are separated from each other without substantial chemical modification’ (Wilson, 2000: VIII); in this approach, ‘extraction’ is seen as a subset of ‘separation’, and defined as ‘the process of moving one or more compounds from one phase to another’. (Wilson, 2000: 1372). The concept of separation also seems to correspond to all four methods of obtention used in C61 (see Table 2). Products can also involve actual separation without properly extracting: an illustration is the preparation of traditional ‘dry’ hashish where trichome heads are separated from the rest of the plant, but where the glands’ external layers are not broken to liberate (extract) the oleoresin contained in the cavity of the trichome’s head.
Associated with ‘extraction’ in C61, the word tincture seems to denote with common understanding likewise with etymology. A tincture is indeed generally a mixture, a dilution of a herbal matrix in alcohol. This process increases the mass of the original matrix by diluting it in a solvent, in apparent divergence from the extracts produced by separation which inevitably involve a reduction in the mass or volume of the final material compared to the starting matrix.
In order for the ECDD (2018, 2019: 2–3, 34–36) to review C. sativa, the WHO collected data, using the nomenclature of C61 as terms of reference (Riboulet-Zemouli and Krawitz, 2019: 4). This explains why products known for their total absence of similarity with dronabinol or other phytocannabinoids’ effects were de facto included. ‘Hemp oil’ and ‘essential oil’ were indeed considered in the scope of the review (WHO, 2016) aside products such as ‘CO2 oil’, ‘butane hash oil, propane hash oil and solvent extracts’, ‘wax’, ‘budder’, ‘live resin’, ‘shatter’, ‘taffy’, ‘distillate’, ‘pie crust/honeycomb’, ‘caviar’, ‘jelly hash’, ‘rosin’, ‘edibles’, ‘e-liquids’ (Cannazza and Citti, 2018b: 5–7). On the other hand, the ECDD (2019) noted that ‘some preparations with high Δ9-THC concentrations are produced in such a way that they are not captured within the definition of an extract or tincture’ (35).
The results of the ECDD assessments suggested editing the Schedules of the C61 to replace the terminology extracts and tinctures with that of preparations. In the Convention’s Article 1(s), preparations are defined as any ‘mixture, solid or liquid, containing a drug’ and subject to the same dispositions as the drug they contain. Such a move makes the definition more accurate by covering processes other than extraction/separation, such as dilution (mixing, compounding, composition, emulsion) or concentration (further mechanical or chemical processing). It would also provide a consistent division of C. sativa derivatives in three clear categories able to match evidence-based definitions:
‘Cannabis’ as pioC and other botanical material of minor importance, ‘Cannabis resin’ as the raw, unprocessed oleoresin separated from EGTs, ‘Preparations of cannabis’ as those products obtained by further processing (a) or (b).
C. sativa derivatives, defined as drugs in the IDCC (i.e. products whose ‘medical use continues to be indispensable for the relief of pain and suffering’, UN, 1961: Preamble), also correspond to the definition of a ‘medical product’ laid out in BP (Medicine and Healthcare products Regulatory Agency, 2017: I-22): (a) Any substance or combination of substances presented as having properties for treating or preventing disease in human beings and/or animals; or (b) any substance or combination of substances that may be used in or administered to human beings and/or animals with a view either to restoring, correcting or modifying physiological functions by exerting a pharmacological, immunological or metabolic action, or to making a medical diagnosis.
Historical nomenclatures of Cannabis in pharmacopœias
C. sativa was considered a valuable drug (i.e. essential drug) at the moment of the first International conference on the unification of formulæ of potent medicaments held at Brussels in 1906, which resulted in an Agreement seen as an early basis for the harmonisation of pharmacopœias, and later for The International Pharmacopœia (Ph.Int.) (WHO Expert Committee on Specifications for Pharmaceutical Preparations, 2008: 6). Remarkably, C. sativa drugs were not included in the first harmonised list because too little of their phytoconstituents were known (Power, 1903: 9). In contrast, the second conference, and Agreement, of September 1925 (International Agreement revising the Agreement signed at Brussels, 29 November 1906, on the Unification of Pharmacopoeial Formulas for Potent Drugs, see: Seconde conférence internationale pour l’unification de la formule des médicaments héroïques, 1925a, 1926; Indian Med Gaz, 1932) did incorporate three C. sativa-based ‘formulæ’ (Real Academia Nacional de Medicina, 1930: 897–905; Seconde conférence internationale pour l’unification de la formule des médicaments héroïques, 1925b): Cannabis indicæ herba (raw plant), Extractum cannabis indicæ and Tinctura cannabis indicæ. The proceedings of the conference show that prior to 1925, extractum cannabis was found in the pharmacopœias of at least 10 countries (detailed in Table 4), and that Cannabis tincturæ were present in very diverse formulations in all countries reviewed except in the Austrian, Belgian, and Dutch pharmacopœias (General Council of Medical Education and Registration of the United Kingdom, 1914; Seconde conférence internationale pour l’unification de la formule des médicaments héroïques, 1925a, 1926). After the entry into force of the Agreement in 1929, monographs of internationally standardised formulations were progressively included in most national pharmacopœias. While the 1865 edition of the Spanish pharmacopœia only included ‘Cañamo – Cannabis sativa L.’ as a crude drug, pointing at the seeds as the preferred material for formulations (Real Academia de Medicina de Madrid, 1865: 25), the 1930 revision harmonises on the Agreement’s standard by adding extractum (Real Academia Nacional de Medicina, 1930: 335) and tincturæ (Real Academia Nacional de Medicina, 1930: 857). In the UK, the British Pharmacopœia Commission followed in 1932, although with some changes (Cartwright, 2015: 50–51; Indian Med Gaz, 1932). It is safe to assume that C61 partially inherited this nomenclature.
Presence of C. sativa pharmaceuticals in pharmacopœias, 1925-2020; non-exhaustive.
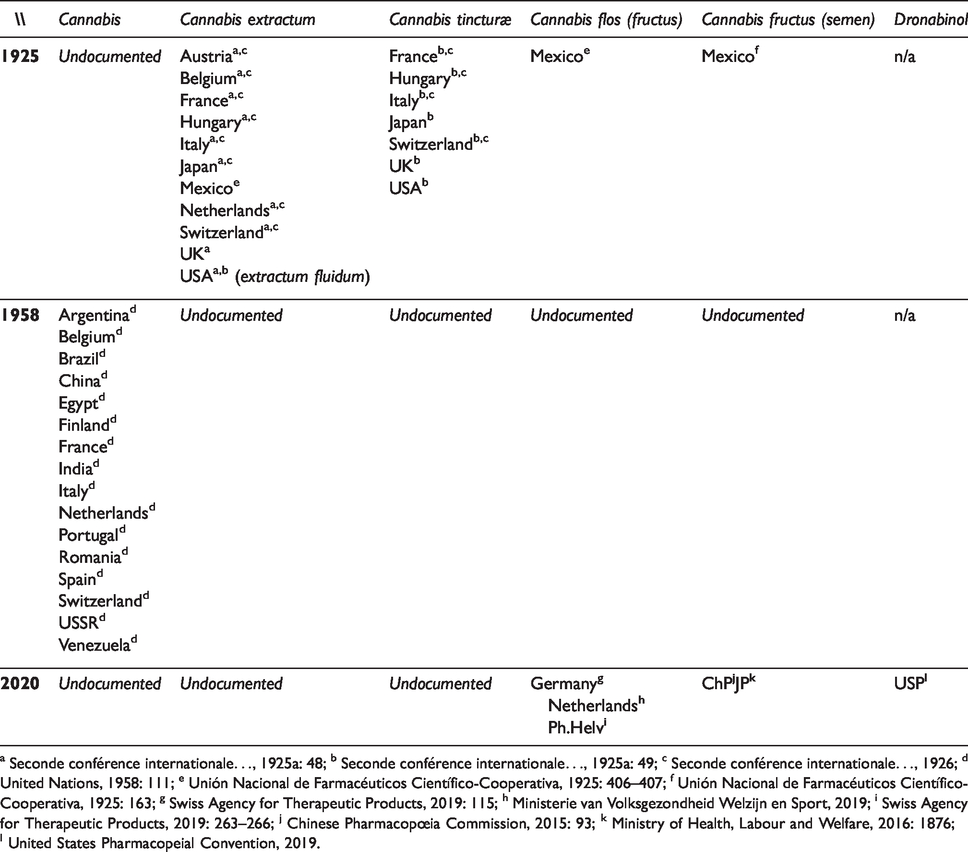
a Seconde conférence internationale…, 1925a: 48; b Seconde conférence internationale…, 1925a: 49; c Seconde conférence internationale…, 1926; d United Nations, 1958: 111; e Unión Nacional de Farmacéuticos Científico-Cooperativa, 1925: 406–407; f Unión Nacional de Farmacéuticos Científico-Cooperativa, 1925: 163; g Swiss Agency for Therapeutic Products, 2019: 115; h Ministerie van Volksgezondheid Welzijn en Sport, 2019; i Swiss Agency for Therapeutic Products, 2019: 263–266; j Chinese Pharmacopœia Commission, 2015: 93; k Ministry of Health, Labour and Welfare, 2016: 1876; l United States Pharmacopeial Convention, 2019.
Conversely, in the 1950s, the body within the recently created WHO with a similar mandate to that of the ECDD today (Danenberg et al., 2013) stated that ‘there is no justification for the medical use of cannabis preparations’ (in 1952 and 1953), adding that ‘there should also be extension of the effort towards the abolition of cannabis from all legitimate medical practice’ (in 1954), and that ‘Cannabis and its preparations are practically obsolete and there is no justification for their medical use’ in 1960 (Riboulet-Zemouli, 2018: 37–39). The records of their meetings on Cannabis feature cherry-picked documentation, and no trace of methodology seems to have been used to reach these conclusions. After receiving the first draft of C61, ‘the Committee was pleased to note the decision … to place cannabis drugs together with diacetylmorphine (heroin) … in the list of prohibited drugs’ although it seems that such decision had not been consulted with them as, in the next paragraph, the Experts ‘expressed the view that [they] would welcome an opportunity to consider, and give advice on, substances that might be placed in that schedule’ (WHO Expert Committee on Drugs Liable to Produce Addiction, 1956: 3–4). Once the Convention was in force, they continued in 1965, and again in 1968, to declare that ‘medical need’ for C. sativa drugs ‘no longer exist’. The placement of C. sativa-related medicines in the Schedules of C61 which involve the most stringent controls, obviously ‘with insufficient scientific support to substantiate those classifications’ (Committee on Economic, Social and Cultural Rights, 2020: 15), but also the subsequent withdrawal of Cannabis drugs from pharmacopœias and pharmacy shelves, was undoubtedly influenced by these statements.
Monographs on Cannabis disappeared from BP in 1932, and 10 years later from USP (Giancaspro et al., 2016). Nonetheless, the UN (1958: 111) still reports, in the 1950s, C. sativa pharmaceuticals in at least 16 legally binding pharmacopœias, on all continents. With the entry into force of the C61 in 1964, these phytopharmaceuticals started to disappear from pharmacopœias as well as from mainstream medical practice, following the gradual decline in the use of phytomedicines in modern healthcare along the latter 20th century (see Table 4, and Chandra et al., 2017).
At the time of writing this article, C. sativa phytopharmaceuticals were almost totally absent from pharmacopœias worldwide, even in jurisdictions where it was allowed for medical prescription. In Asia, Ch.P and JP include monographs of C. sativa fruits (raising questions with regards to pioCs), while in North America, USP has two monographs on dronabinol. In Europe, German Commission E’s monographs (DAB) and Swiss Pharmacopœia reincluded in 2019 monographs of Cannabis parthenocarpic infructescences (although titled ‘Cannabis flos’ [flower], see Swiss Agency for Therapeutic Products, 2019: 115, 263–266). Besides the mere presence of Cannabis or not in pharmacopœias, the way medical products of herbal origin are addressed and classified provides useful guidance on how to approach C. sativa derivatives.
Pharmacopœial nomenclatures of phytopharmaceuticals
All current pharmacopœias reviewed contemplated phytopharmaceuticals, in individual monographs, in general notices/appendices, or both. One pattern of categorification for plant-based medication was found recurrent from the 1865 edition of the Spanish pharmacopœia onwards to the latest edition of European Pharmacopœia (Ph.Eur). It is based on the observation of the physical consistency of phytopharmaceuticals, divided between:
solid/dry (extracta sicca), semisolid/soft (extracta spissa) or fluid/liquid extracts (extracta fluida).
The latest editions of Argentina’s, Brazil’s, Ph.Eur., Russia’s State Pharmacopœia of the Russian Federation (SPRF), and Spain’s pharmacopœia consistently use this model. Others like Herbal Pharmacopœia of Mexico (FHEUM), Indian Pharmacopœia (IP), JP, and French Pharmacopœia (Ph.Fr.) occasionally refer to these terms without systematising them. Most pharmacopœias consider tinctures (tincturæ) distinct from extracta, while others such as Argentinian Pharmacopœia and Ph.Eur. consider tincturæ a subset of extracta fluida. Pharmacopœial monographs however never mention that other alcohols like glycerol, propylene glycol, or polyethylene glycol are increasingly replacing ethanol in the preparations of tincturæ (Schulz et al., 2004: 11). Ph.Eur. separates oleoresina (oleoresins), defined as ‘semi-solid extracts composed of a resin in solution in an essential and/or fatty oil … obtained by evaporation of the solvent(s)’, from extracta spissa that it defines as ‘semi-solid preparations obtained by evaporation or partial evaporation of the solvent’.
In the 2000s, new models of classification were introduced to complement the traditional one (Gaedcke and Steinhoff, 2003: 4–7, 26). One of these newer models divides phytopharmaceuticals between drugs that are genuine extractable matter, termed ‘native extracts’, and drugs that contain added ingredients (whether active compounds or excipients) with regard to the starting botanical material, termed ‘non-native’ (European Directorate for the Quality of Medicines, 2016: 6519, 6521; Gaedcke and Steinhoff, 2003: 6–7). In 1988, Menßen (Gaedcke and Steinhoff, 2003: 26–27) proposed to distinguish between ‘primary extracts’ and ‘refined extracts’ as the refinement process (also referred to as purification or enrichment) produces phytopharmaceuticals with a reduced spectrum of constituents.
Another complementary model pushed forward by Ph.Eur. is that of ordering phytopharmaceuticals according to content standardisation criteria, distinguishing them according to the level of precise knowledge of their composition in active compounds. According to this model rapidly expanding, phytopharmaceuticals are either ‘standardised extracts’, ‘quantified extracts’, or ‘other extracts’ (European Directorate for the Quality of Medicines, 2016: 25–26). Ph.Eur. explains: Standardised extracts are adjusted to a defined content of one or more constituents with known therapeutic activity. This is achieved by adjustment of the extract with inert excipients or by blending batches of the extract. Quantified extracts are adjusted to one or more active markers, the content of which is controlled within a limited, specified range. Adjustments are made by blending batches of the extract. Other extracts are not adjusted to a particular content of constituents. (European Directorate for the Quality of Medicines, 2016: 6515–6516, 6519)
Ch.P., FHEUM, FP, Romanian Pharmacopœia (FR), IP, JP, Egyptian Pharmacopœia, Ph.Fr., Hellenic (Greek) Pharmacopœia, Polish Pharmacopœia, SPRF, USP, and Pharmacopœia of Vietnam (VP) adopted sui generis approaches, often in complement to one of the above patterns. Ch.P references none of the previous models: instead, it separates crude drugs from preparations, which are subdivided into 26 specific formulations (e.g. pills, powders, granules, concentrated decoctions, plasters, ointments, etc.). In the same spirit, JP regards ‘crude drugs’ (including drugs of herbal, animal, or mineral origin), only distinguishing between ‘whole’, ‘cut’, or ‘powdered’ crude drugs (Ministry of Health, Labour and Welfare, 2016: 1, 5–6). It classifies crude drugs simultaneously in eight subcategories: ‘extracts’ (corresponding to both extracta sicca and spissa), ‘fluid extracts’, ‘tinctures’, ‘spirits’, ‘infusions or decoctions’, ‘teabags’, ‘aromatic waters’, and ‘pills’ (Ministry of Health, Labour and Welfare, 2016: 21–23). FHEUM separates ‘vegetal drugs’, ‘triturated herbs’, ‘teas’, ‘tinctures and extracts’, and ‘essential oils’ (Comisión Permanente de la Farmacopea de los Estados Unidos Mexicanos, 2013: 7–8). IP distinguishes ‘crude herbs’ and ‘processed herbs’ (covering either intermediary or traditional preparations), ‘botanical extracts’ which includes fluid, powdered, and semisolid extracts, separates the ‘tinctures’, and presents ‘herbal formulations’ as readily available products for consumers, and potentially for non-medical uses (e.g. food supplement, nutraceutical, cosmetic, see Indian Pharmacopoeia Commission, 2018: 3725–3729). Numerous references such as Ph.Eg, Ph.Indo, Ph.Pl, FR, and VP use long lists of traditional formulations (e.g. elixirs, ointment, potion, granulate, powder, suppository, syrups, etc.), with a rationale mostly reliant on historical addition of marketed formulations types rather than on the intrinsic physical characteristics of the products. USP makes mention of ‘herbals’, ‘crude products of plant origin’, as well as ‘botanical-containing products’ without further definition, nor consistent use throughout the different pharmacopœial monographs. A different detailed scheme of categorification was found in the former USSR pharmacopœia (USSRSP, see Shikov et al., 2014) in force until 2007. Phytopharmaceuticals were distinguished between ‘medicinal plant’, ‘summarised non-refined (or galenic) formulations’ (which includes infusions, decoctions, tinctures, concentrated extractions, and elixirs), ‘novo-galenic formulations’ (herbal material mixed with non-herbal ingredients), ‘combined phyto-preparations’ (mixtures of only herbal ingredients), and ‘active pharmaceutical ingredients’ (APIs).
Finally, among the non- or partially legally binding pharmacopœias reviewed (Table 1), Ph.Int. did not contemplate herbal medicines at all, while others such as British Herbal Pharmacopœia, Ayurvedic Pharmacopœia of India, USHP, and African Herbal Pharmacopœia did not rely on any orderly meta-category for phytopharmaceuticals.
Other kinds of medical products of herbal origin were found in a number of pharmacopœias, particularly those following Ph.Eur. standards. Some of these eventually correspond to C. sativa-derived phytopharmaceuticals, such as:
Essential oils, termed ætherolea (European Directorate for the Quality of Medicines, 2016: 814–815), Teas, plantæ ad ptisanam, and instant herbal teas, præparationes celeres ad ptisanam (European Directorate for the Quality of Medicines, 2016: 820), Products obtained from organisms with recombinant deoxyribonucleic acid (rDNA) biotechnologies, producta ab arte ADN recombinandorum (European Directorate for the Quality of Medicines, 2016: 6526–6528) Products of fermentation, producta ab fermentatione (European Directorate for the Quality of Medicines, 2016: 5909–5910), Vegetable fatty oils, olea herbaria (European Directorate for the Quality of Medicines, 2016: 848–850).
European regulatory nomenclature of phytopharmaceuticals
Alternatively, the European Union (EU) in its Community code relating to medicinal products for human use (European Commission, 2001) considers a simplified approach where all phytopharmaceuticals are termed ‘herbal medicinal products’ and subdivided between substances and preparations: Herbal medicinal products: Any medicinal product, exclusively containing as active ingredients one or more herbal substances or one or more herbal preparations, or one or more such herbal substances in combination with one or more such herbal preparations, Herbal substances: All mainly whole, fragmented or cut plants, plant parts, algae, fungi, lichen in an unprocessed, usually dried, form, but sometimes fresh. Certain exudates that have not been subjected to a specific treatment are also considered to be herbal substances. Herbal substances are precisely defined by the plant part used and the botanical name according to the binomial system (genus, species, variety and author), Herbal preparations: Preparations obtained by subjecting herbal substances to treatments such as extraction, distillation, expression, fractionation, purification, concentration or fermentation. These include comminuted or powdered herbal substances, tinctures, extracts, essential oils, expressed juices and processed exudates.
Ontological conflicts in the pharmacopœial classification of phytopharmaceuticals: comparison between different models and the classification of C. sativa phytopharmaceuticals in international law.
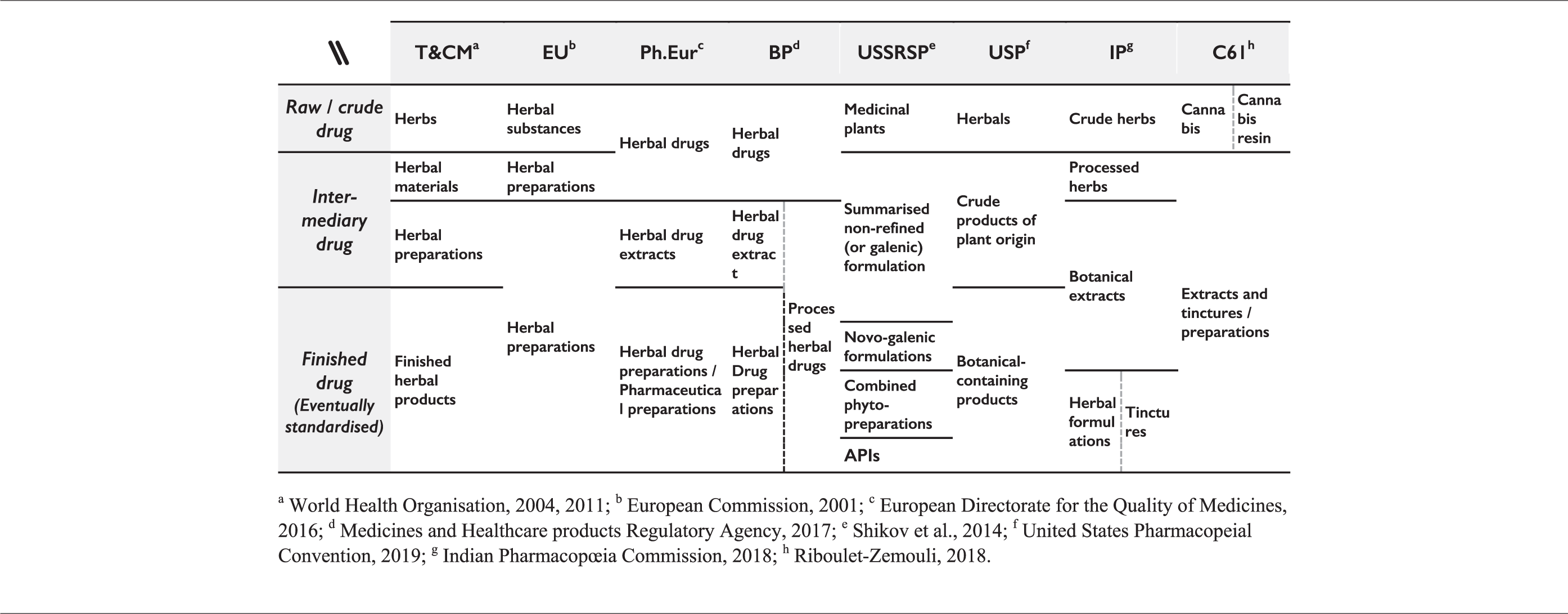
Nomenclature of Traditional & Complementary Medicine (T&CM) by the WHO
Unrelated to C. sativa, to the ECDD and to its mandate under the IDCC, the T&CM unit of WHO classifies phytopharmaceuticals according to their pharmacological properties, while staying consistent with the processes of obtention that are often firmly embedded into traditional processing methods (Abbott, 2014; Bellakhdar, 1997; Kutesa, 2018; World Health Organisation, 2011). Their approach distinguishes phytopharmaceuticals according to processing stages (World Health Organisation, 2011: 129–130): Herbal medicines include herbs, herbal materials, herbal preparations and finished herbal products: • Herbs include crude plant material such as leaves, flowers, fruit, seed, stems, wood, bark, roots, rhizomes or other plant parts, which may be entire, fragmented or powdered. • Herbal materials are either whole plants or parts of medicinal plants in the crude state. They include herbs, fresh juices, gums, fixed oils, essential oils, resins and dry powders of herbs. In some countries, these materials may be processed by various local procedures, such as steaming, roasting, or stir-baking with honey, alcoholic beverages or other materials. • Herbal preparations are the basis for finished herbal products and may include comminuted or powdered herbal materials, or extracts, tinctures and fatty oils of herbal materials. They are produced by extraction, fractionation, purification, concentration, or other physical or biological processes. They also include preparations made by steeping or heating herbal materials in alcoholic beverages and/or honey, or in other materials. • Finished herbal products consist of herbal preparations made from one or more herbs. If more than one herb is used, the term mixture herbal product can also be used. Finished herbal products and mixture herbal products may contain excipients in addition to the active ingredients finished products or mixture products to which chemically defined active substances have been added, including synthetic compounds and/or isolated constituents from herbal materials, are not considered to be herbal.
The definition of ‘herbs’ provided by WHO seems to correspond to EU’s ‘herbal substance’, while EU’s ‘herbal preparations’ include WHO’s ‘herbal materials’, ‘herbal preparations’, and ‘finished herbal products’ (Table 5). The T&CM classification proposed by WHO seems more detailed than EU’s, even though some products could fall under the scope of two of the proposed categories. For instance, a product can match both ‘stir-baking with honey, alcoholic beverages or other materials’ (herbal materials) and ‘steeping or heating herbal materials in alcoholic beverages and/or honey, or in other materials’ (herbal preparations). Regrettably, the WHO T&CM model has not been more followed in pharmacopœias than EU’s.
Often, the same wording is used to encompass different definitions. BP adopted a dichotomous approach to phytopharmaceuticals: on the one hand, it fully follows Ph.Eur. considering ‘herbal drugs’ as non-processed material while it labels processed herbs as ‘herbal drug extracts’ and ‘herbal drug preparation’ for the further homogenised – and eventually standardised – phytopharmaceuticals (Medicines and Healthcare products Regulatory Agency, 2017: I-17, VII-A837). On the other hand, BP maintains in parallel a monograph for what is called ‘processed herbal drugs’, corresponding to those phytopharmaceuticals obtained via traditional processing methods’ (Medicines and Healthcare products Regulatory Agency, 2017: IV-43–49).
Defining an upper limit to phytomedicine
The only consensus that seems to be shared is that of excluding from the category of phytopharmaceutical some finished products that, however, originate from plant material: those herbal derivatives to which non-herbal active compounds have been added. BP (Medicines and Healthcare products Regulatory Agency, 2017: I-22), in a similar fashion to that of DAB and RFE, the EU with the supra category ‘herbal medicinal products’ or the WHO T&CM with ‘herbal medicines’, groups as phytopharmaceuticals only those drugs:
exclusively containing as active ingredients one or more herbal drugs, exclusively containing as active ingredients one or more herbal drug preparations, exclusively containing as active ingredients one or more such herbal drugs in combination with one or more such herbal drug preparations.
WHO (2011) specifies that ‘products to which chemically defined active substances have been added, including synthetic compounds and/or isolated constituents from herbal materials, are not considered to be herbal’ (130). Consequently, herbal-based products containing non-herbal active compounds, or containing non-herbal compounds as excipients, are not regarded as phytotherapy. Gaedcke and Steinhoff (2003: 1) explain that because phytopharmaceuticals ‘are always mixtures of a number of substances’, a medicinal plant (entirely or by parts) is considered as a single active ingredient regardless of the composition (Gaedcke and Steinhoff, 2003; Rather et al., 2013). Besides their multiple constituents, phytopharmaceuticals, working in a synergical ‘entourage’ fashion, are viewed as one single substance (Atakan, 2012).
Also, fully isolated compounds or ‘refined extracts’ (Gaedcke and Steinhoff, 2003: 2–7) obtained from botanical material are not regarded as phytopharmaceuticals. These are rather considered as common APIs for the formulation of conventional pharmaceutical preparations. As an example, in the USSRSP model (Table 5) ‘combined phyto-preparations’ would be considered phytopharmaceuticals, while ‘standardised extracts’ and ‘novo-galenic formulations’ would fall out of this category.
‘Others’: Non-phyto cannabinoid pharmaceuticals, non-Cannabis cannabinoid phytopharmaceuticals, etc
The uppermost limitation of the scope of phytomedicine on which pharmacopœias rely is not complete, as it would not exclude one particular (and diverse) corpus of compounds commonly referred to as synthetic cannabinoids. This broad category encompasses some ‘synthetic’ molecules that can be derived from plant material, without being mixed with non-herbal compounds; hence, pharmacopœial models currently de facto embrace some ‘synthetic cannabinoids’ as phytomedicines, which is problematic.
Alonso (1998) explains that botanical materials can, in addition to their use as a source of active compounds, be utilised as non-active starting materials in laboratory processes in order to obtain compounds that were not genuinely present in the plant. Halfway between natural compounds and molecules designed fully in vitro, these drugs have been referred to as seminaturals (Feher and Schmidt, 2003) or more recently as semisynthetics (Cragg and Newman, 2013; Jones et al., 2006). Mathur and Hoskins (2017) describe them as ‘generally produced by transforming starting materials from natural sources into final products via chemical reactions’, clarifying that these reactions consist in the ‘rearrangement of chemical entities or structural isomers of naturally occurring products in order to generate new molecules’. Chrispeels and Sadava (1994) mention the example of saponins which are structurally ‘so much like human steroids that saponins are used as the starting material for synthetizing steroids used for making birth control pills’ (136).
At the API level, in order to explore the implications for C. sativa medicines, a safe analogy can be established with the extensively studied Papaver somniferum L. from which originate naturally occurring opioids (i.e. opiates). Six primary natural secondary metabolites are biosynthesised in P. somniferum: morphine, codeine, thebaine, papaverine, noscapine, and narceine. Semisynthetic opioids, in comparison, are those obtained by human intervention over the chemical structure of these compounds: diacetylmorphine (heroin) is a well-known example of semisynthetic opioid obtained from morphine (Novak et al., 2000; Solimini et al., 2018); oxycodone is a semisynthetic derivative of thebaine (Cortazzo et al., 2013: 502; Elkader and Sproule, 2005). In what concerns C. sativa, semisynthetics consist of derivatives from naturally obtained phytocannabinoid molecules (Fahrenholtz et al., 1967), having undergone modifications of some of their pharmacophores with significant binding affinity to brain receptors (Bow and Rimoldi, 2016; Razdan and Zitko, 1969; Shevyrin et al., 2016). It is generally accepted that structural modifications corresponding to the natural pathways of cannabinoids biosynthesis and degradation within the C. sativa plant (e.g. decarboxylation, see Baram et al., 2019; Caspi et al., 2017; Hanuš et al., 2016) are not considered products of semisynthesis. However, the product of a semisynthesis can be another, different naturally occurring compound (as in the example of human steroids mentioned). For instance, the process to transform cannabidiol (CBD) into dronabinol, because it does not occur in vivo (not corresponding to the natural biosynthetic pathways of C. sativa, see Caspi et al., 2017) but is possible in vitro (ECDD, 2018: 13; Merrick et al., 2016), is considered a semisynthesis.
Importantly, semisynthetic compounds should not be confused with the second subset of synthetic cannabinoids, the naturally occurring ones obtained only by synthesis in vitro. In the case of opioids, morphine can either be extracted from P. somniferum or created by full chemical synthesis (Gates and Tschudi, 1956; Mechoulam and Hanuš, 2000; Novak et al., 2000). The same goes with molecules structurally identical to naturally occurring phytocannabinoids such as dronabinol or cannabidiol (CBD is the INN of (–)-CBD, whether natural or synthetic) that are designed in vitro without involving initial plant material (Adam Ametovski and Lupton, 2019; Mechoulam and Gaoni, 1965; Petrzilka et al., 1967; Razdan et al., 1974; Trost and Dogra, 2007). Analytically indistinguishable, fully synthesised in vitro phytocannabinoids and naturally obtained in vivo phytocannabinoids were not distinguished by the ECDD which considers them in all points pharmacologically identical (ECDD, 2019: 54–55; Riboulet-Zemouli and Krawitz, 2019).
Yet another class of compounds termed ‘synthetic cannabinoids’ is that of synthetic analogues, also (mostly) obtained via laboratory synthesis without C. sativa botanical ingredients. The resulting substances, however, are not phytocannabinoids found in the environment but novel compounds, mimicking their pharmacological effects without being derived from, nor directly relatable to them. Firman et al. (2019) find that out of the ‘223 compounds identified, a mere ten … bear structural relation to THC’. Examples of synthetic cannabinoid analogues are nabilone (Blanchard and Ryan, 1977a, 1977b; Flemming et al., 2007: 22–23) or the HU- and JWH-type components found in ‘spice’ (National Institute on Drug Abuse, 2018; Seely et al., 2012), while fentanyl and methadone are well-known synthetic opioid analogues.
Synthetic cannabinoid analogues have been extensively studied, resulting in the invention of a wide array of substances. A proposal of a classification system for cannabinoids (Shevyrin et al., 2016) found few compounds structurally related to C. sativa phytocannabinoids – HU-210 and dexanabinol, for instance – and termed them ‘classical cannabinoids’. Shevyrin et al. identified numerous other groups of synthetic compounds with no direct structural relationship to the plant’s phytoconstituents: nonclassical and hybrid synthetic cannabinoid analogues, naphthoylindoles, phenylacetylindoles, benzoylindoles, naphthylmethylindoles, diarylpyrazoles, 3-naphthoylpyrroles, synthetic endocannabinoid analogues, etc. To distinguish these molecules from naturally occurring phytocannabinoids obtained in vitro and from semisynthetics, the expression ‘synthetic analogues’ (Biernat, 2018; European Monitoring Centre for Drugs and Drug Addiction, 2017; Flemming et al., 2007; Luo et al., 2019; Pop, 1999; Shevyrin et al., 2016; Trost and Dogra, 2007) has been proposed. However, the confusing use of the phraseology ‘synthetic cannabinoids’ continues (Bonn-Miller et al., 2018; Carvalho et al., 2017; ECDD, 2019: 19–25; Fattore and Fratta, 2011; National Institute on Drug Abuse, 2018; Pop, 1999; Reekie et al., 2018; Seely et al., 2012). Some authors placed ‘semisynthetics’ as a subset of ‘analogues’ (Bow and Rimoldi, 2016) making the case of the inexistence of both semisynthetics and analogues in a natural environment – the only difference being the use of botanical C. sativa material to obtain the former, not the latter. Nevertheless, some compounds existing in nature can be obtained by the semisynthesis process of chemically altering starting botanical material containing phytocannabinoids, when these alterations are different from those of the natural biosynthetic pathways of the plant. This is illustrated by cases such as the transformation of CBD into dronabinol: both compounds are found in nature, but such transformation has not been reported in natural environments – there is limited evidence that CBD can be processed into some compounds defined thus far as metabolites of dronabinol (Huestis, 2007) in an in vitro environment simulating gastric acids (Bonn-Miller et al., 2017; Grotenhermen et al., 2017; Merrick et al., 2016; White, 2018:10–11), but no confirmation of conversion in animal or human models has been reported (Crippa et al., 2020; ECDD, 2018: 13; Grotenhermen et al., 2017; White, 2018: 10–11; Wray et al., 2017).
The criterion of similarity in the molecular structure of compounds is not always related to the final pharmacological effect, organoleptic properties, the consistency of the product, or to other characteristics, and it is not yet entirely mapped at this time. Hence, if that variable is discarded, a double dichotomous distinction stands out: occurrence in nature versus novelty of the compound; obtention from the transformation of C. sativa material versus that of other material. This allows for cannabinoid APIs to be arranged in four meta-categories:
• In vivo phytocannabinoids: naturally occurring compounds, derived: - from C. sativa plant material (e.g. dronabinol present in EGTs), - from other plant genera (e.g. (−)-cis-perrottetinene present in some plants of the genus Radula; see Gertsch, 2018), • In vitro phytocannabinoids: the same naturally occurring compounds as above, obtained by full ‘chemical synthesis’, • Synthetic cannabinoid analogues (e.g. nabilone, HU-210, dexanabinol): non-naturally occurring compounds, obtained by full chemical synthesis, • Semisynthetic cannabinoids: -Semisynthetic phytocannabinoids, i.e. naturally occurring compounds, derived from C. sativa plant material, obtained by partial chemical synthesis different than those of the plant’s natural phytocannabinoid biosynthetic pathways (e.g. CBD transformed into dronabinol), -Semisynthetic cannabinoid analogues, i.e. non-naturally occurring compounds, derived from C. sativa plant material, obtained by partial chemical synthesis (e.g. the (+)-enantiomer of CBD, not found in natural environments).
An additional layer of complexity is brought by modern biotechnologies and also relates to the method of obtention used (Sirikantaramas et al., 2007). Serious questioning of the traditional Aristotelian breach between nature and artefacts (Bhushan, 2006; Cragg and Newman, 2013; Feher and Schmidt, 2003; Schummer, 2002) arise while innovations such as gene editing, genome mining, and combinatorial biosynthesis (e.g. rDNA) thrive. What were thought to be ‘fundamental differences between combinatorial synthesis and biosynthesis’ (Feher and Schmidt, 2003) are being overcome, also for C. sativa. Already, cannabinoid compounds, just like opioids (Galanie et al., 2015) can be obtained from the design of heterologous expression of biosynthetic pathways, such as in genetically modified (GM) organisms (Carvalho et al., 2017; Luo et al., 2019; Siddiqui et al., 2012; Sirikantaramas et al., 2007). Both naturally occurring and novel cannabinoids were obtained by Luo et al. (2019) from genetically engineered yeasts, a feat achieved by introducing selected C. sativa genes in Saccharomyces cerevisiae using rDNA technologies. Different classes of GM organisms, including GM C. sativa plants (Berahmand et al., 2016; Feeney and Punja, 2003; Sayre et al., 2019) are currently being engineered to produce different yields or ratios of various cannabinoid compounds. All the four meta-categories of cannabinoids listed above can, or will likely soon, be obtainable via genetically engineered organisms, complicating the distinction between the natural and the artificial. This is echoed in pharmacopœial nomenclatures, increasingly tending to distinguish drugs obtained via organisms having been genetically recombined, adding them special requirements such as PhEur (monograph 0784).
Noteworthily, GM in C. sativa can also be unrelated to the biosynthetic mechanisms of the plant. GM can indeed be limited to specific characters, such as resistance to pests, salinity and drought, or tolerance to herbicides or insecticides (Catacora-Vargas, 2011: 10–11, 85–86) which in theory do not alter, or affect only indirectly, the production of phytocannabinoids within EGTs. A distinction is therefore needed between a GM which alters the plant’s biosynthetic pathways and a GM which does not.
Finally, if this was not a complex enough panorama, cannabinoids are naturally produced by other living organisms. Beyond Cannabis, other plant genera naturally biosynthesise cannabinoid compounds (Gertsch, 2018; Hanuš et al., 2016). Beyond the Plantae kingdom, Homo sapiens also produce ‘endocannabinoids’ (Hanuš et al., 2016; Huestis, 2007; Pacioni et al., 2015; Shevyrin et al., 2016) and a series of living organisms (Hanuš et al., 2016) are found to produce their own endogenous cannabinoid substances, similar or not to those known to be biosynthesised in H. sapiens or C. sativa (e.g. anandamide is naturally secreted in humans and in black truffle fungi, see Pacioni et al., 2015).
Discussion
This study is the first to examine the interrelation between different nomenclatures of natural and artificial drugs containing cannabinoids, and between these and the products available nowadays. The study finds mostly confusing terminology, where four salient troubling aspects (which sometimes overlap) recur: terms comprising various distinct ‘relata’ (a relatum is the thing or entity to which a word relates and which it designates); terms whose relata vary according to context, time, or geography; terms inherited from socially constructed, utilitarian, non-science-based language; terms overly specific to a defined field of scientific research.
Terminologies used for synthetic cannabinoids represent useful frameworks for research but are limited in their potential applications outside of the biochemical sectors. Nomenclatures used in international law rely on broad, utilitarian categories that expand, beyond the products targeted for control, to other similar peripheral products, even if they are not relevant to drug control criteria. This is the case for pioC, controlled as ‘fruiting tops’ alongside ‘flowering tops’ from either male, female, hermaphrodite, and monoecious plants, although they might contain insignificant amounts of phytocannabinoids. It is also the case for dronabinol, the ‘main psychoactive substance in the cannabis plant’ (ECDD, 2019: 45): while only dronabinol, the (–)-trans enantiomer of the delta-9 isomer of THC, is a target for public health scrutiny, the three other stereochemical variants, as well as the six other delta isomers of THC are grouped with dronabinol as tetrahydrocannabinols – as a collateral damage – besides the fact that the ECDD (2018) recognised dronabinol ‘is the only [stereochemical variant] that occurs naturally in the cannabis plant’ and that ‘the limited information on the pharmacology of [its] stereochemical variants … suggests that they have little activity’ (33–34). It conflicts, also, with the statement that ‘there are no reports that the THC isomers … induce physical dependence, or that they are being abused or are likely to be abused so as to constitute a public health or social problem’ and ‘there are no reported medical or veterinary uses of these isomers’ (ECDD, 2019: 49).
Way forward: A non-conflicting framework for the nomenclature of ‘cannabis’ products
Besides approximations and scientific inconsistencies, the four-tiered ontology used in C61 is consistent. It can be interpreted as an incremental and mutually exclusive scheme composed of: ‘cannabis plant’ (the living or freshly harvested plant, staminate and pistillate iterations), ‘cannabis’ (pioCs and inflorescences of the former), ‘cannabis resin’ (material separated from EGT found on one of the former), and ‘preparations’ (or ‘extracts and tinctures’ any processed drug derived from either one of the former). Consistency is reached at the expense of clear relata, at least concerning the last three categories, which include everything other than what is in the previous category. Such a categorisation does not carry any practical pharmaceutical information, since it aggregates products of all kinds. It also leaves room for conflicting interpretations: some products are virtually included within this ontological class (because they are prepared from ‘cannabis’ or ‘cannabis plant’) even though they do not comply with the criteria for international drug control (e.g. essential oils, or non-medical cosmetics containing CBD). The IDCC corrects this ontological conflict applying a different, transversal criterion: that of the purpose of use (Riboulet-Zemouli, 2019). As C. sativa products, and their methods of obtention, become increasingly more diverse, the four-tiered categorification of C61 will continue to lose relevance.
While the first two categories, ‘cannabis plant’ and ‘cannabis’ (starting materials, see Table 2) can be easily further defined by modern botanical research, elements are missing to distinguish products within the last two categories, ‘cannabis resin’ and ‘preparations’ (see Table 5). The uniquely complex phytochemical composition of C. sativa, the variability in patterns and routes of administration, and the still uncaptured phytopharmacological mechanisms of action of C. sativa products render insufficient the analytical identification of the composition in phytocannabinoids. It suggests the need for complementary elements of differentiation between products. This study finds that a consideration of the ‘methods of obtention’ (i.e. transformation, processing, and formulation of the products) in addition to the very composition of the final product, might be insightful. Alas, the four terms present in the C61 treaty to describe obtention processes (‘production’, ‘manufacture’, ‘separation’, and ‘extraction’) are used interchangeably (Table 2) and do not convey additional information about the pharmacognosy of the drugs. In popular culture, the most solid distinction criterion – which relates to the methods of obtention – is a dichotomy discriminating the addition of foreign matter (solvent extraction) to the thermomechanical processing of the plant’s EGTs (solventless extraction). Such additional determinant, complementing the four-tiered basis provided by C61, could help tweak a method of differentiation between products.
The criterion relying on the addition or not of eluents (i.e. solvents), although useful, does not provide full coverage of the potential products obtained: within the two subgroups, variations in techniques and resulting products often happen (see supplementary Appendix III). Lu and Luthria (2014: 5, 12) inform that ‘postharvest storage and processing (such as grinding and drying) influence the quantity of phenolic phytochemicals’ and of phytocannabinoids. Concerning the processing methods based on the addition of foreign matter, all basic and widely used techniques to extract and isolate natural biocompounds are reported for obtaining C. sativa derivatives (supplementary Appendix III provides details of these techniques). A series of other secondary methods of extraction exist, likely to be increasingly used in the future. Minor changes in extraction parameters have shown to result in significant chemical and pharmacological differences between final products (for instance cannabinoidless essential oils and high-dronabinol ‘concentrates’ which can both be obtained by distillation). Solventless extraction or processing techniques, although less numerous, also showed substantial variability; additionally, many of these are embedded in traditional folk knowledge and intangible assets, still fully or partially undocumented (Abbott, 2014; Abdool, 2013; Bellakhdar, 1997: 232–234; Clarke, 2007; Kutesa, 2018). Variables such as the size of filter pores, the amount of pressure exerted, the temperature, or the type of movement applied are also determinative in characterising the final product (Devi and Khanam, 2018; Hamayun and Shinwari, 2004; Upton et al., 2014; supplementary Appendix IV). With or without eluent, all extraction or processing techniques report different thresholds of acid or decarboxylated phytocannabinoids depending on trivial variations in parameters along the process.
Laying out in detail the processes of obtention of drugs is a core objective of pharmacopœias. Nevertheless, even going more in-depth than the IDCC, neither the monographs related to Cannabis nor general nomenclature for phytopharmaceuticals provided any thorough pattern for an orderly, non-overlapping and non-arbitrary classification of C. sativa derivatives. Pharmacopœias either focus on precise traditional formulations, like in Ch.P and FHEUM, falling short of including all known products (and in particular the most recent ones, e.g. liquid mixtures for electrical vaporisation devices), or they rely, like Ph.Eur., on wide-ranging categories that would include, in the case of C. sativa, an heterogeneous array of products with different pharmacological effects (varying concentrations, different formulations, different routes of administration).
Ph.Eur’s extracta sicca, extracta spissa, extracta fluida, oleoresina, tincturæ, ætherolea, olea herbaria, plantæ ad ptisanam, præparationes celeres ad ptisanam, producta ab fermentatione, and producta ab arte ADN recombinandorum are relevant to Cannabis, but insufficient. The derivative of C. sativa called ‘rosin’, obtained via a solventless extraction process relying on heat and pressure; ‘butane hash oil’, obtained by percolation of dry pioC using pressurised butane as eluent (Beal, 2019); and ‘supercritical CO2 oil’ (Naz et al., 2017; Omar et al., 2013) have a similar consistency. They could be considered as extracta spissa although their composition and methods of obtention vary greatly; they could also be seen as oleoresina. Another preparation reported already in 1848 (Mechoulam and Hanuš, 2000) and nowadays known as ‘Rick Simpson oil’ or full ethanol-extracted cannabis oil, obtained by maceration of pioC in alcohols followed by partial or total evaporation (mechanically or via distillation), could be viewed either as tincturæ, extracta spissa, or oleoresina. ‘Dry sift’ or ‘dry sieve’ (Beal, 2019, Daniulaityte et al., 2017), a type of ‘hashish’ obtained via repeated microfiltrations of ground dry pioCs, without involving the use of solvent, is another illustration. The process results in a fine dry powder almost entirely made of capitate heads of EGTs. As such, it corresponds to extracta sicca. However, under very light pressure and heat (e.g. from fingers) it immediately takes the consistency of an extracta spissa or oleoresina, since EGTs’ external layers (cuticles) break and liberate the content of the glands, which then stick together (Graham, 1976: 6; Nicoletti Motta, 2018). The UNODC (2009) talks about a resinous secretion that ‘appears as loose or pressed sticky powder, depending on the method of production’ (16). Two pharmacopœial categories, oleoresina and extracta spissa, likely apply to these four very different products that have (or can have under certain thermophysical conditions) a consistency in appearance similar. Among all these products, many are popularly referred to as ‘oils’ although none correspond to the pharmacopoeial category of herbal fatty oils olea herbaria.
Not only the method of obtention seem to be a common blind spot in IDCC and pharmacopoeial nomenclatures (notably, with the exception of WHO T&CM’s), but this criterion is also a determinative element of a drug’s pharmacological effect, since ‘preparations with different therapeutic properties can be made from the same herbal material, depending on the manufacturing process employed’ (Schulz et al., 2004: 5). The fitness of a phytopharmaceutical to one route of administration or another also relates to the processes involved to prepare it (European Directorate for the Quality of Medicines, 2016: 6519; Gaedcke and Steinhoff, 2003).
From harvest to the last stage of product formulation, processes of obtention are of singular relevance in the case of C. sativa phytopharmaceuticals, regarding the large variety of pharmacologically active secondary metabolites present in the plant as well as the complex molecular interactions involved. The presence and yield of flavonoids, phytocannabinoids, and other terpenoids can be subject to variations depending on the method employed. In this regard, C. sativa derivatives appear to be chemically unstable since straightforward factors (temperature, time, humidity, light) target degradation or other types of chemical reactions. Variations dramatically affecting the pharmacological properties of a product can be induced by basic thermomechanical stimuli (Agarwal et al., 2018; Naz et al., 2017), for instance, heating at 200°C for seven minutes (Verhoeckx et al., 2006) or ageing (Fairbairn, 1976: 15; Mechoulam and Hanuš, 2000; Zamengo et al., 2019). This is the case when phytocannabinoids, obtained in acidic form when separated from the plant (Happyana et al., 2013; Kimura and Okamoto, 1970; Perrotin-Brunel et al., 2010, 2011, Pertwee, 2006) decarboxylate into compounds with enhanced psychopharmacological effects (Reekie et al., 2018; Verhoeckx et al., 2006).
From many perspectives, the most compelling evidence suggests the ‘method of obtention’ is a common, and overlooked criterion in classification systems that fail to overarch C. sativa products. Significantly, this study shows that, if the identification of different methods of extraction, separation, fractionation, isolation, purification, refinement and concentration (terms somehow used interchangeably) and of crucial parameters within those processes, might not per se provide all sufficient information on C. sativa products, it seems likely to provide a consistent and useful complement of information on the characteristics and properties of C. sativa products, to achieve a thinner, more precise cartography of them. The variabilities could be used to frame the lowest taxon of ‘products’, the smallest common denominator, in such hypothetical classification.
A metachemistry of cannabinoids (and beyond)
The very same methods which are used to extract or separate phytopharmaceutical material from pioC and C. sativa are also sometimes employed to transform botanical C. sativa material into ‘semisynthetic’ ingredients. Moreover, these same techniques can also be used to process GM C. sativa material, and even biotechnologically crafted EGT containing non-natural cannabinoid analogues. Borrow limitations between herbal drugs and non-herbal drugs could lead to even greater confusion in the future. Such reversibility and porosity between natural and unnatural cannabinoid-containing products was determinative for including ‘synthetic’ products in the scope of this study: limiting the study only to natural herbal drugs would have required preexisting boundaries that neither the IDCC nor the pharmaceutical references provided for.
Historically, the myriad of new cannabinoid molecules discovered in the last decades (Firman et al., 2019; Shevyrin et al., 2016) have been termed ‘synthetic cannabinoids’ by opposition to ‘phytocannabinoids’, the naturally occurring cannabinoids found in Cannabis plants. While the word ‘phytocannabinoid’ links to an unambiguous and delimited relatum, the expression ‘synthetic cannabinoids’ is given by ostentation rather than by description. Eighty years ago, Adams et al. (1940) were the first to synthesise a cannabinoid compound ex vivo: the phytocannabinoid CBN (see Mechoulam and Hanuš, 2000; Pertwee, 2006). The first compounds to be named ‘synthetic cannabinoid’ in the 1960s were all closely related to the molecules found in C. sativa, either phytocannabinoids obtained in vitro or closely related byproducts, obtained during attempts to synthesise and describe naturally occurring phytocannabinoids (Bow and Rimoldi, 2016; Fahrenholtz et al., 1967; Mechoulam and Gaoni, 1965; Mechoulam and Hanuš, 2000; Pertwee, 2006; Petrzilka et al., 1967; Razdan and Zitko, 1969). Hence their definition by the negative insofar they were, by then, the only non-natural, not wholly-phyto cannabinoids. With the years, dozens of new structurally heterogeneous compounds were described, obtained via varying synthesis routes, using different starting materials, and resulting in an increasingly diverse class of molecular structures within what continued to be a single ontological entity: synthetic cannabinoids, encompassing each time more different relata.
A compressive arrangement of those ‘synthetic agonists active at cannabinoid CB1 and CB2 receptors’ (Firman et al., 2019) is still to be found. Shevyrin et al. (2016) made insightful proposals (classifying these substances in six groups: ‘classical’, ‘nonclassical’, ‘hybrid cannabinoids’, ‘aminoalkylindoles’, ‘eicosanoids’, and ‘other cannabinoids’), and Biernat (2018) proposed three categories (‘cannabinoids derived directly from cannabis’, ‘synthetic versions of cannabinoids found in cannabis’, and ‘compounds that are similar to cannabinoids found in cannabis’), among others. It appears however that the six-tiered categories continue to rely on a definition by ostentation, grouping under others a variety of structurally diverse molecules. The suggestion made by Biernat falls in a similar stalemate with the class ‘compounds that are similar to cannabinoids found in cannabis’, probably too heterogeneous, encompassing novel artificial analogues, semisynthetics, endocannabinoids, and any other cannabinoid.
Beyond their ability to bind to the human endocannabinoid receptor system, what brings together cannabinoid compounds is the difficulty in classifying them rightly. Relying solely on their chemical structure might not be the best approach, as, again, the category other will always exist and potentially grow as an increased number of substances may be found to have some interaction with CB1 or CB2 (it is already the case for non-cannabinoid phytoconstituents (Gertsch et al., 2010) and for paracetamol (Klinger-Gratz et al., 2018), suggesting the need for a real metaphysical address of the endocannabinoid system). Heretofore, the lemma cannabinoid itself has not been discussed. Basis of all neologisms in chemistry, the word ‘cannabinoid’ is nonetheless much more a neuro-pharmacological term than a chemical one. From its etymology – appeared in the 1940s (Google Books Ngram Viewer, s.d) built as a derivative of ‘cannabis’ to which was added the suffix -oid derived from classical Greek εἶδος, eîdos: ‘kind’, ‘form’, ‘type’, ‘likeness’ – it designates substances that are of the cannabis kind, that relate, that are similar to ‘cannabis’, from a wholly subjective, empirical point of view. The relatum associated with ‘cannabinoid’ does not necessarily imply a structural resemblance at the molecular level, but rather a similarity in terms of the overall pharmacological effects observed, deemed akin to those produced by botanical Cannabis products. Dewick (2012) makes an enlightening statement: Natural products structures are usually quite complex, some exceedingly so, and fully systematic nomenclature becomes impracticable. Names are thus typically based on so-called trivial nomenclature, in which the discoverer of the natural product exerts their right to name the compound. (3)
Because advancements in chemistry make language continuously drift away from compounds, the need to ‘define and stress the differences between terms that may appear to be synonyms’ (Santaló and Casado, 2016: 39) becomes pressing, particularly for the class(es) of ‘synthetic cannabinoids’, neither stable nor consistent. Such demand is outstanding, not only because of the need for accurate language to match understanding, but also in terms of the strong ethical concerns which arise from the debates around the uses of Cannabis products (Zarhin et al., 2019) presently amplified in the context of moral and epistemological upheaval driven by two ongoing parallel revolution(s): the Cannabis policy reforms one, and the biotechnological one (Perron-Welch, 2019).
Extrapolations from other well-known classes of psychoactive phytoconstituents and their analogues do not seem to provide comprehensive solutions. For instance, cannabinoids differ from opioids on at least three levels: hundreds of different phytocannabinoids have been identified in C. sativa plants (while only a dozen of phyto-opioids are known); the human body naturally produces endogenous cannabinoids; cannabinoids are also naturally secreted by other living organisms (whether plant, animal, or fungus). This latter point is subject, again, to further scrutiny of the metaphysics of animal endocannabinoid systems. Besides come the novel, human-conceived cannabinoid compounds, raising the absolute list of cannabinoids kinds (or taxa), but also virtually the complexity of their systematics, above that of opioids, and most other well-studied classes of molecules.
A bioethical nomenclature for cannabinoid compounds
The exploration of a new classification of cannabinoid substances, in a bioethical rather than purely biochemical fashion, might be a solution. In the same fashion that the methods of obtention of C. sativa derivatives which bring additional analytical information on the composition of a product, a bioethical nomenclature of compounds does not per se conflict with nomenclatures arranged according to chemical structures. It could very well be a complementary tool, in line with Bhushan’s (2006) suggestion to overcome the difficulties in classifying chemical substances by using ‘a kind for every occasion’ on the basis that ‘kinds are real but particular to the occasion of the individuals who choose to work with them, their choices of models and strategies, and what happens to function best in the environment in question’ (327–328). In this direction, kinds, or nomenclatures, used by chemists, might not be the best approach for an environment of law and public health policy, or for clinical contexts.
As exposed, a pattern in four classes of cannabinoids showed two main axes useful to start discriminating compounds: occurrence in nature or not, and obtention from C. sativa material or not.
The first axis, regarding the prevalence of a compound in natural environments, appears to be easily divisible in two clear mutually exclusive groups: one comprises novel and artificial substances, not present in nature, that could be labelled as human inventions. A comparison can be done with the artificial analogues of nicotine: initially defined under a chemical nomenclature as chloronicotinyls, they have been renamed neonicotinoids by Tomizawa and Yamamoto (1993: 97–98) and Sheets (2002) adding the prefix νέος (néos: ‘new’, ‘young’) to ‘nicotine’ followed by the suffix -oid. Likewise, the group of cannabinoid compounds not present in nature could be grouped under the term neocannabinoids. The second group would include cannabinoids occurring in nature (either in raw state, such as dronabinol, or as secondary metabolites processed by a living organism, like THC-COOH; see Huestis, 2007), whether they are derived from living organisms or obtained by processes of human-made synthetic chemistry reproducing the natural biosynthesis pathways of a living organism. These include phytocannabinoids, human endocannabinoids, as well as naturally occurring endogenous cannabinoid substances found in other living, genetically unmodified organisms. Medical and scientific terminologies often articulate the prefix neo- in opposition to paleo- (derived from παλαιός, palaiós: ‘old’, ‘ancient’); as such, this second group, mutually exclusive with the previous one, could be termed paleocannabinoids.
The second axis relies on the methods of obtention. The first level of opposition is found between substances derived from living organisms (the mechanism called biosynthesis), whether GM or not, and substances derived from an inert material, in vitro, via different laboratory processes (traditionally called ‘synthesis’ or ‘chemical synthesis’). The use of the word ‘synthesis’ alone, as a synonym of ‘chemical synthesis’, sometimes in vitro, sometimes ex vivo, is exceptionally confusing: ‘synthesis’ derives from σύνθεσις (súnthesis: ‘arrangement’, ‘putting together’, ‘composition’, ‘combination’) and is the generic, general term to refer to any obtention of a complex chemical compound from simpler precursors, whether in living organisms or not. The Oxford Dictionary of Biology defines synthesis as ‘the formation of chemical compounds from more simple compounds’ (Martin and Hine, 2015: 577).
In order to avoid using ‘synthesis’, ‘laboratory synthesis’, or ‘chemical synthesis’ to oppose ‘biosynthesis’, the neologism poesynthesis is suggested. It is created from ποιέω, poieō: ‘to make’, ‘produce’, ‘create’, ‘compose’, ‘bring into existence’; arranged for phonetic and legibility purposes according to lemmas formed with the same prefix, like poetry. ‘Poesynthesis’ would aggregate all those molecules obtained via in vitro synthesis; it would complement and oppose ‘biosynthesis’ referring only to molecules obtained in laboratory chemical synthesis processes. A cannabinoid obtained in vitro would accordingly be called poesynthetic cannabinoid.
Looking at the holdups, this axis deserves a more complex than a dual approach, in particular in the context of the surge of gene editing technologies targeting and altering biosynthesis pathways. While both paleocannabinoids and neocannabinoids can be poesynthetic, neocannabinoids can, by definition, not be derived from natural biosynthesis . Or can they? Luo et al. (2019), among others, have shown that neocannabinoids can be obtained through a biosynthesis altered by biotechnological means. This suggests the need for a subdivision within biosynthesis, discriminating GM from non-GM biosynthesis. To distinguish between natural biosynthesis and biosynthesis induced artificially (sometimes called ‘synthetic biology’ [sic] in the context of policy, see Perron-Welch, 2019), the use of lemmas inspired in ancient Greek can again bring terminological clarity. Cannabinoids which are obtained by processes of biosynthesis not naturally occurring, having been induced by biotechnological genetic modifications, could be termed dysbiosynthetic cannabinoids as per the prefix δυσ- (dus: ‘disordered’, ‘difficult’, ‘abnormal’) to the existing ‘biosynthesis’ (formed with βίος, bios: ‘life’). By opposition, the prefix εὖ- (eû: ‘true’, ‘real’) could form the terms eubiosynthesis and eubiosynthetic cannabinoids to cover compounds obtained entirely by natural biosynthesis in unmodified environments, and their natural metabolites. Figure 3 details the interrelation between the terms proposed.

Graphic representation of the neologisms proposed for the distinction of different types of synthesis, and hierarchy between them.
Figure 3 shows that the only of these neologisms covering an unnamed ontology is ‘dysbiosynthesis’. It is also worth noting that the proposed set of terms loses the ability to directly designate what corresponds to ‘ex vivo’, which would need to be described as non-eubiosynthetic.
Stretching the model to try it, these five terms can be combined in a double-entry table (Table 6), resulting in virtually six classes of cannabinoids: eubiosynthetic paleocannabinoids, eubiosynthetic neocannabinoids, dysbiosynthetic paleocannabinoids, dysbiosynthetic neocannabinoids, poesynthetic paleocannabinoids, and poesynthetic neocannabinoids (individual definitions can be found in supplementary Appendix IV). As seen earlier, the second of these categories, which combines neocannabinoid with eubiosynthesis, is antithetical and inexpressible. Nonetheless, the five remaining categories offer a coherent nomenclatural basis. At the crossroads of biological and ethical considerations, this five-tiered categorification model is compatible with existing classes of cannabinoids. For instance, phytocannabinoids from C. sativa and endocannabinoids from H. sapiens are two subdivisions of eubiosynthetic paleocannabinoids. In what relates to C. sativa, we can observe that the plant is a source of cannabinoids in all five classes: beyond the ‘classical’ phytocannabinoids from C. sativa (eubiosynthetic paleocannabinoids), both dysbiosynthetic paleocannabinoids and dysbiosynthetic neocannabinoids are obtainable from GM C. sativa. Finally, poesynthetic paleocannabinoids and poesynthetic neocannabinoids can also be obtained, in particular via semisynthesis, with C. sativa infructescences or oleoresin as starting material.
Proposed bioethical nomenclature for cannabinoid compounds.

Unexplored in this study, further subdivisions relying on the same method and scheme could be contemplated: for instance eumycosynthesis to distinguish naturally occurring anandamide in truffles from the dysmycosynthesis of GM yeasts, or euphytosynthetic and dysphytosynthetic cannabinoids.
Admittedly this model presents limitations on at least two levels. First is the classic philosophical problem of induction and extrapolation of classes, or their ability to be projective and normative (see The Kripkenstein Paradox and the Private World, 2011). Despite the split nature versus artefacts represented by the dichotomy ‘phytocannabinoids vs synthetic cannabinoids’ being offered an alternative solution in the model proposed with the use of paleocannabinoids, ‘[broadening] the notion of natural kind so that it now comes to mean naturally occurring things and those identical to them’, Bhushan (2006) illustrates why projective and normative limitations matter: A compound that is synthesized first and later discovered to occur naturally … For example, around 1985 the compound now called buckminsterfullerene was prepared synthetically. This is a 60-carbon compound in which the carbons are at the vertices of five- and six-membered rings positioned like those inscribed on the surface of a soccer ball. Subsequently, buckminsterfullerene was found to be a component of soot, including ancient samples of soot. (332–333) As soon as one lets “time put its mark on substance”, the hypothetical character of persistence drops out, and the old debate of realists versus anti-realists becomes obsolete. It is replaced by the question of how substance is instituted and how its reality becomes physically, socially, consciously realized over the course of time … In other words, substances become increasingly reliable or stable actors in experimental and technological interactions, i.e., as the situations are defined and become defined in which they will assert themselves in certain ways. The trajectory is, therefore, graphed in reference to two variables: The time that passes as the work of science goes on, and a scale that registers the increasing specificity of the characteristics with which the substance becomes identified.
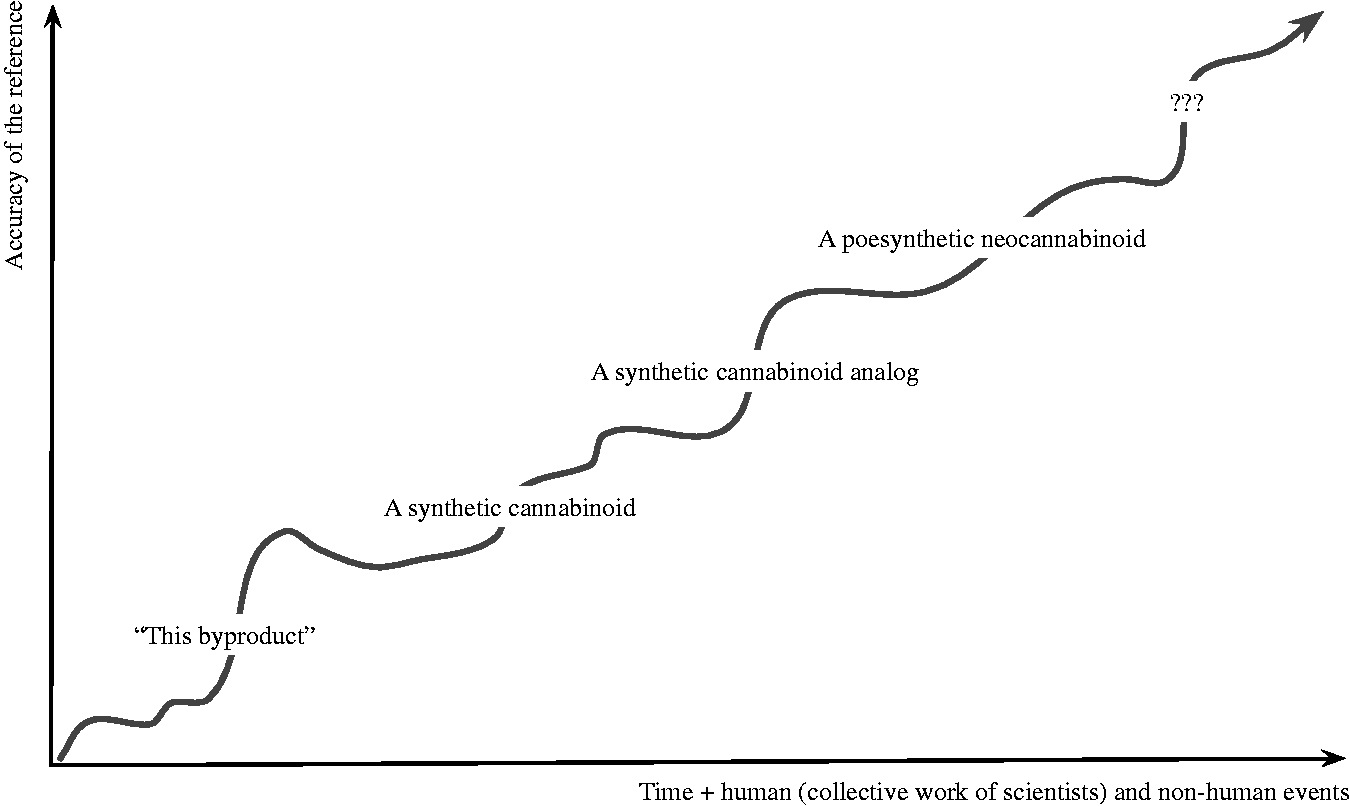
Trajectory of the ontological representation of a ‘poesynthetic neocannabinoid’. Source: Adapted from Latour (1990: 68, 2000: 256) and Nordmann (2006: 355).
Since the phytoconstituents contained in the plant have quite exhaustively been screened, the possibility that, in the future, neocannabinoids could be found to be occurring naturally is unlikely – but still possible anecdotally – a rather common impasse in induction and extrapolation, that the Kripkenstein paradox (Kripke, 1982) illustrates. The division paleo- versus neo- is relevant from the ethical, public health, and regulatory perspectives, undoubtedly useful in the case of Cannabis research. It conveniently adds value in various fields, for this particular moment in time and state of scientific knowledge. Even today, chemists lack a full understanding of most molecular structures, and certain models in use may in fact prove to be wrong. Yet they can meaningfully talk about these molecules and assert that they are speaking of a particular one, when the molecule they have in mind may not be the same after the model becomes revised. (The Kripkenstein Paradox and the Private World, 2011: 6)
Besides these issues, this model of representation provides enhanced clarity. It is just one proposed step ahead in the trajectory of advancement in the collective representation of the realities of our world. A salient feature of this classification is its close relatability with the classes of C. sativa derivatives identified earlier, which are also characterised by their ‘method of obtention’ as a differentiating factor.
Conclusion
Beyond the results of the assessments conducted by WHO, which change some language but mainly suggest the need for a broader, more thought-through assessment of the different nomenclatures currently used, resolving the confusions in naming ‘cannabis products’ is urgently needed since derivatives continue to diversify. Not only is sound nomenclatures missing for C. sativa and cannabinoids, but it is also absent for other phytopharmaceuticals. The lack of clarified, unified, and internationally recognised terminology is salient in herbal medicine and pharmacy generally. Applying to phytomedicine as a whole the exercise of ‘drawing a trajectory’ might result in a flat curve, where time and efforts progress much faster than the accuracy of ontological representations used – at least during the major part of the past Century and the current one. This study locates C. sativa and ‘medicinal plants’ as a whole at a relatively low epistemological standing.
More skilful addresses of the philosophical concerns laid out in this paper than those of the author would considerably help – metaphysicians and epidemiologists could bring enlightenment by addressing the endocannabinoid system. The ongoing developments in science and biotechnologies should be closely monitored to capture the extent of actual and future possible products. In parallel, a new look at the basis is needed, in chemistry but also in botany, with studies of the morphology, histology, and cytology of pioCs and EGT urging.
Over and above this evolving landscape, this study shows that it is possible to stress patterns that can serve to establish evidence-based nomenclatures where they are missing. The method of obtention, best criterion found to distinguish between the myriad of derivatives of C. sativa, is also determinative in establishing a practical, bioethical distinction scheme between isolated cannabinoid APIs. It could be used to refine a nomenclatural classification, in combination with the morphological categorification of extracts and formulations used in pharmacopœias, with the route of administration, and with the absolute or relative content in some identified active compounds. Future attempts to adequately, accurately nomenclature cannabinoid-containing products should consider, in addition to morphology and content analysis, already used, the methods of obtention. These three variables can be articulated in a system allowing a proper identification process for virtually all cannabinoid-containing products. Without the illusion of reaching a one size fits all nor universally acceptable nomenclature of ‘cannabis products’, such a process could at least allow a diversion from current confusions and cross-purposes talk, in a domain where misinterpretations can have important legal, social, or health-related consequences. Cannabis ontologies II, a follow-up to the present study (in press) that favours the contemporary understanding of ontology as an applied information science, pushes forwards a proposal of non-conflicting nomenclature for Cannabis and cannabinoid products, based on a ‘smallest common denominator single product signature’ generated through an identification key.
Beyond the advancement of scientific knowledge and correct understanding of a topic immersed in multiple disciplines and scientific corpora, the development of clearer ontologies for C. sativa products and cannabinoids can have direct applications in numerous domains: bioinformatics, cheminformatics, ethnobotany, ethnopharmacology, phytochemistry or pharmacovigilance and ADR monitoring (see also Abdool, 2013: 15). It can influence the interpretation of ChP and JP Cannabis fructus monographs. It can assist and support the work of documentation, preservation, and transmission of both the genetic and cultural diversities and heritage attached to C. sativa and its derivatives. It would be desirable that such a revised nomenclatural ‘cannabis’ background be considered in the various ongoing standardisation and policymaking workstreams: the potential to clarify ambiguous terms and help translate into regulations the complex reality contemplated is non-negligible. Obviously, it can help in guiding physicians’ practice for C. sativa or cannabinoid therapeutic treatments, but is also a tool for doctors to make better choices in the field of prevention of dependence and use disorders, as well as for harm reduction practitioners; it can assist the design of accurate product labelling, and consumer information (European Directorate for the Quality of Medicines, 2016: 6520; Gaedcke and Steinhoff, 2003: 31–36; World Health Organisation, 2004). The study contains elements that, if popularised, may contribute to enlightening public scrutiny and debate over Cannabis and certain of its controversial products (marketed, or yet to come), thus enhancing the fundamental ‘right to participate in, and to enjoy the benefits of scientific progress and its applications’ (Committee on Economic, Social and Cultural Rights, 2020), for instance in biology, medicine, food sciences, or agriculture.
Accepting that language is ‘not a question about correctness, but rather assertion’ (Kripke, 1982; The Kripkenstein Paradox and the Private World, 2011), it is worth remembering Camus (1970) for whom ‘naming an object inaccurately means adding to the unhappiness of this world’ (238). With this in mind, the most important contribution of this article may not be assessing ‘cannabis’ language, but perhaps asserting the need – and means – for the awakening of evidence-based ‘cannabis’ terminologies and nomenclatures.
Supplemental Material
sj-pdf-1-dsp-10.1177_2050324520945797 - Supplemental material for ‘Cannabis’ ontologies I: Conceptual issues with Cannabis and cannabinoids terminology
Supplemental material, sj-pdf-1-dsp-10.1177_2050324520945797 for ‘Cannabis’ ontologies I: Conceptual issues with Cannabis and cannabinoids terminology by Kenzi Riboulet-Zemouli in Drug Science, Policy and Law
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author has completed the Unified Competing Interest form and declares grants for the later part of the conduct of the study (redaction) from Fundacion CANNA and FAAAT think & do tank, and non-financial support from Hempoint s.r.o; unrelated personal fees during the earlier part of the conduct of the study from CBDepot, s.r.o, EIHA, Fundacion ICEERS, Harmony ltd., Hempoint s.r.o, Satimed, and VoteHemp. The author declares that the manuscript is a novel, honest, accurate, and transparent study undertaken in complete independence.
Acknowledgements
The author wishes to greet Simon Anderfuhren, PhD, and Amy Case King for assistance at all stages, providing crucial inputs, helping with the study design, strengthening the method, and addressing quintessential concerns along the data collection and writing. The author also commends the journal editors and the peer reviewers for their insightful comments, and Angela Bacca for assisting with language. This study would not have been possible without fruitful exchanges with Michael Krawitz; Fabian Steinmetz and Julie P Fry PhDs; Boris Baňas, Ing; Alberto Sainz-Cort PhD; Gilles Forte and Olivier Bertrand MDs; Julian Stobbs, Myrtle Clarke, Òscar Parés Franquero, Farid Ghehiouèche and Susan van Brunschot; Tseng-Chieng Huang and David Nutt PhDs; Stefano Berterame, David Borden, Hanka Gabrielová; Maxime Collin, PhD; Herbert Schmidt, MD; and Yrving Díaz. Importantly, the author also greets the staff of the WHO department of Essential medicines and health products, the ECDD experts, extraordinary librarians (with special thanks to Jacques Oberson and Lee Robertson, League of Nations archives, Reynald Erard and Tomas Allen, WHO library, and the staff of University libraries in Barcelona, Geneva, and Paris), and the UNESCO Chair in Bioethics at the University of Barcelona. Last but not least, this study could not have been possible without a continuous love and support from the ‘cannabis communities’: hundreds of people, users groups, social movements, civil society organisations, labs, chemists and alchemists, growers, breeders, healers, thinkers – incredible biodiversity of skilled, responsible and wise human beings from all corners the globe, most of them still considered dangerous offenders by their own authorities, because of the cultural traditions of their land, because of their craft, of their intimate personal choices, of their knowledge, etc.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


