Abstract
Introduction and objectives:
Cannabis use has been associated with psychosis and with poor outcome in patients with mental illness. Synthetic cannabinoids (SCs) have been suggested to pose an even greater risk to mental health, but the effect on clinical outcome has not been directly measured. In this study, we aimed to investigate the demographics and hospitalisation of psychiatric patients who were SC users.
Methods:
We searched the Biomedical Research Centre Clinical Record Interactive Search register for SC users and age- and sex-matched SC non-users who had been psychiatric patients under the South London and Maudsley NHS Trust. We recorded diagnosis, homelessness, cannabis use and the total number of days admitted as an inpatient to secondary and tertiary mental-health services.
Results:
We identified 635 SC users and 635 age- and sex-matched SC non-users. SC users were significantly more likely to be homeless (χ2=138.0; p<0.0001) and to use cannabis (χ2=257.3; p<0.0001) than SC non-users. SC users had significantly more inpatient days after their first recorded use of SCs than controls (M (SD)=85.5 (199.7) vs. 25.4 (92.32); p<0.0001). Post hoc tests revealed that SC non-users who used cannabis had fewer inpatient days than SC users (p<0.0001), and that non-users of both SC and cannabis had fewer inpatient days than SC non-using cannabis users (p=0.02).
Conclusions:
SC use may lead to an increase in the number of days spent in hospital in patients with psychiatric illness. This highlights the need for clinicians to ask specifically about SC use.
Introduction
There has been considerable interest in the effect of cannabis on mental health, with associations found between cannabis use and the development of psychotic illness (Di Forti et al., 2015; Sideli et al., 2019) and between cannabis use and worse outcome in patients with psychosis (Patel et al., 2016; Stone et al., 2014). There is growing evidence that cannabis use may also be associated with manic symptoms (Gibbs et al., 2015; Sideli et al., 2019; Stone et al., 2014).
Synthetic cannabinoids (SCs) constitute a substantial proportion of the so-called new psychoactive substances (NPSs). Originally designed for pharmacological research purposes, SCs have emerged as new and ever-evolving drugs of abuse since their first appearance as ‘legal highs’ just over a decade ago (UNODC, 2017).
Given their higher affinity for the cannabinoid CB1 receptor and the absence of cannabidiol, there is concern that SCs may have more marked risks to both mental and physical health than cannabis (Castaneto et al., 2014). There is growing evidence to suggest that SC intoxication is associated with symptoms including psychosis and suicidal ideation (Hobbs et al., 2018; Van Amsterdam et al., 2015). Additionally, a withdrawal syndrome exhibiting both physical and psychiatric symptoms is emerging, with reports of patients developing paranoia, anxiety, chest pain and muscle spasm, which requires a novel approach from detoxification services (Macfarlane and Christie, 2015; Van Hout and Hearne, 2017). In a study of attendances at emergency departments, SC use and another naphthyl pyrovalerone, the stimulant methylenedioxypyrovalerone, were associated with acute psychosis in individuals attending emergency departments for treatment, but in contrast, psychosis was less common in users of other stimulant NPSs such as mephedrone and methedrone (Vallersnes et al., 2016).
SC use appears to be more prevalent in vulnerable populations, with SC users being more likely to be homeless (Joseph et al., 2017, 2019; Manseau et al., 2017), and SCs pose a particular problem for inpatient psychiatric services, underscored by previous reports of undetected SC use in forensic facilities (Every-Palmer, 2011). SCs can be smoked, are often odourless and include a multitude of different chemical structures, all of which impede their detection.
Although there have been a number of case reports focusing on the presence of psychotic signs and symptoms associated with SC use (Hurst et al., 2011; Mahgoub and Young, 2017; Van Der Veer and Friday, 2011), there are few case control studies that have analysed the characteristics of SC users and how SC use impacts upon prognostic factors such as length of stay in psychiatric services, and none in a UK-specific sample. A US-based study of patients admitted to a dual diagnosis ward (with drug use and mental-health problems) reported that patients who took SCs were more likely to have a psychotic presentation, and were more likely to have agitation as part of their presentation, were less likely to be Caucasian and had increased length of hospital stay (Bassir Nia et al., 2016). A German study reported that SC users tended to be younger at first use than cannabis users (early to mid-20s), and a study of Turkish patients suggested that SC users were around 14 years younger as a patient group than SC non-users with schizophrenia, although these differences in age may be due to the fact that SCs have not been on the market for a long period of time (Altintas et al., 2016; Welter et al., 2017).
Following the introduction of the Psychoactive Substances Act into UK law on 26 May 2016, the production and supply of all NPSs has been illegal in the UK, as well as the possession of NPSs within a custodian institution. Little is known about the impact of the Psychoactive Substances Act on the number of patients who are also SC users presenting for the first time with a psychiatric illness. Additionally, the psychiatric sequelae of SC use remain uncertain, and larger studies are required to determine the level of risk associated with this relatively new class of drug.
In this study, we compared SC users to age- and sex-matched SC non-users accessing mental-health services in South London to determine whether SC use was associated with a difference in total hospitalisation days. We also investigated whether SC use was associated with differences in home treatment days (the number of days when patients were being seen by the home treatment team – a service to provide more intensive input for patients who are more unwell but who do not require hospitalisation), number of face-to-face contacts (attendances at clinic to see a doctor, nurse or other professional), diagnosis, homelessness and cannabis use.
Methods
The sample population evaluated in this study was collected from the Clinical Record Interactive Search (CRIS) system accessed via the National Institute of Health Research Maudsley Biomedical Research Centre in January 2018. CRIS provides researchers with access to anonymised digital clinical records of patients accessing secondary and tertiary mental-health services across South London and Maudsley NHS Trust (SLaM; Perera et al., 2016; Stewart et al., 2009), and has previously been used to investigate the association of cannabis use on clinical outcomes in first-episode psychosis (Patel et al., 2016). Ethical approval for research using CRIS as a database for secondary analysis has been obtained from the Oxford C Research Ethics Committee. Informed consent was not required for this study, as deemed by the ethics committee (Fernandes et al., 2013).
SLaM is the largest mental-health provider in Europe, serving a population in excess of one million people across the London Boroughs of Croydon, Lambeth, Lewisham and Southwark (Stewart et al., 2009). In addition, SLaM provides a national referral service for individuals with a wide variety of mental-health problems, including an adult addictions complex case service, an adult psychosis service and both an adult and a child and adolescent forensic service.
For this study, CRIS was searched using the search terms ‘synthetic cannabis’ OR ‘synthetic cannabinoids’ OR ‘synthetic marijuana’ OR ‘synthetic weed’ OR ‘K2’ OR ‘Spice’ OR ‘Black Mamba’ OR ‘Kronic’ OR ‘Clockwork Orange’. The search returned a total of 1799 records where one or more of the search terms was mentioned in a patient’s notes. The anonymised clinical records were then hand searched to exclude those where the mention did not relate directly to the use of SCs by the patient. A patient was deemed to have used SCs only where self-reported or, in the absence of self-report, where a positive urine drug screen (UDS) was reported in the medical notes. The date of first recorded use of SCs in the electronic patient record (either self-reported or verified by UDS) was identified. This is referred to as the index date. The number of first recorded uses of SCs per quarter was recorded.
The control group was matched on date of birth (±365 days from the index date), ethnicity and sex. The control group were also matched on being under the care of the same clinical academic group (CAG; ±365 days from the index date). CAGs form the subdivision of psychiatric specialist services in SLaM and include acute care, addictions, behavioural and development, child and adolescent, older and dementia, psychological medicine and integrated care, and psychosis. We used the CAGs for matching in order to ensure that the two groups contained the same number of patients attending for a specific group of conditions (psychosis vs. affective disorders vs. substance misuse). We did not match directly on diagnosis, as we wished to determine whether SC users were more likely to have particular diagnoses, and because diagnoses could change as a result of SC use. We did not specify natural cannabis use as a matching criterion, as we wished to determine the proportion of matched non-SC users who used cannabis (compared to the proportion of SC users who also used cannabis).
After matching, a total of 635 SC users (617 with self-reported use and 18 with a positive UDS) and 635 matched SC non-users were included in subsequent analyses.
Information extracted from CRIS structured fields regarding the study population and control group included: total inpatient hospitalisation days after the index date, total home treatment days after the index date, the number of face-to-face psychiatric contacts after the index date, homelessness (ever), homelessness (before the index date), homelessness (at the index date), homelessness (after the index date) and ICD-10 diagnosis after the index date (or where this was not recorded, the most recent diagnosis prior to the index date). Data on cannabis use (distinct from SC use) were extracted from free text using natural-language processing, as previously reported (Patel et al., 2016). A machine learning approach was employed, allowing correct identification of sentences describing cannabis use with a minimum precision value of 90%. As frequency, strength and amount of cannabis use were not consistently documented in electronic health records, a binary variable defined as any documentation of cannabis use by the patient was used.
Data analysis
Independent t-tests were performed to assess the mean difference in inpatient hospitalisation days, home treatment team days and face-to-face psychiatric contacts between the SC user (SC+) and non-user (SC–) groups. Significant results were tested further using a General Linear Model (GLM), with age, sex, cannabis use (ever), homelessness (ever) and diagnosis (at index date) included as covariates. A post hoc GLM analysis subdividing the SC groups by the use of cannabis was also performed, including age, sex, homelessness and diagnosis as covariates. Chi-square tests or Fisher’s exact tests (where expected values were <5) were performed to assess the relationship between SC use and type of ICD-10 diagnosis, CAG, homelessness and cannabis use.
Results
SC use, sociodemographic characteristics, cannabis use and homelessness
The earliest index date (first SC use recorded in the clinical records) was March 2007. Numbers of index dates per quarter were below 10 up until December 2014, after which there was a sharp increase in recorded SC use. This peaked in April–June 2016 at 91 cases (Figure 1).
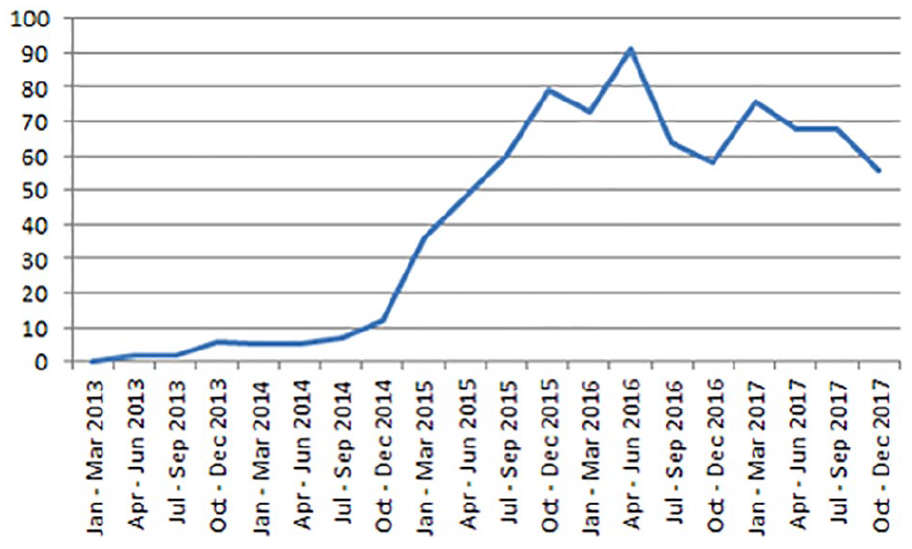
First recorded synthetic cannabis use in South London and Maudsley NHS Trust – number of records per quarter.
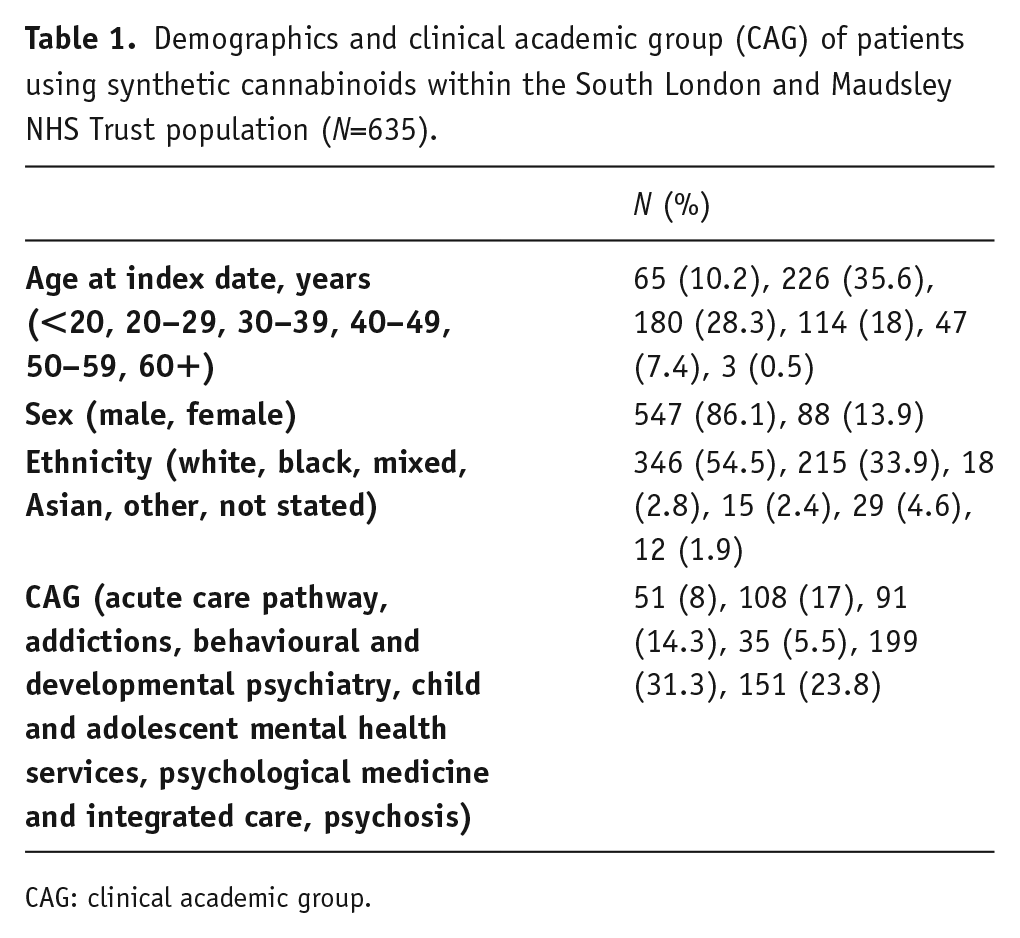
The mean age of SC users was 32.9 years (standard deviation (SD)=10.81), with an age range of 13–64 years, and the highest represented age group of 20–29 years. The majority of SC users were male (86.1% compared to 59.8% across SLaM in 2017), with 54% having their ethnicity recorded as white (compared to 44.2% across SLaM) and 33.8% as black (compared to 38.8% across SLaM; SLaM, 2017).
In terms of CAG allocation, 199 (31.3%) SC users were under the care of psychological medicine and integrated care, 151 (23.8%) were under psychosis, 108 (17%) were under addictions, 91 (14.3%) were under behavioural and developmental psychiatry, 51 (8%) were under acute care pathway and 35 (5.5%) were under child and adolescent psychiatry (Table 1).
Demographics and clinical academic group (CAG) of patients using synthetic cannabinoids within the South London and Maudsley NHS Trust population (N=635).
CAG: clinical academic group.
SC users were significantly more likely to use cannabis compared to SC non-users (χ2=257.3; df=1; p<0.0001), with 629 (99%) cannabis users in the SC user group, and 406 (64%) cannabis users in the SC non-user group. Only six individuals in the SC user group were recorded as non-users of cannabis.
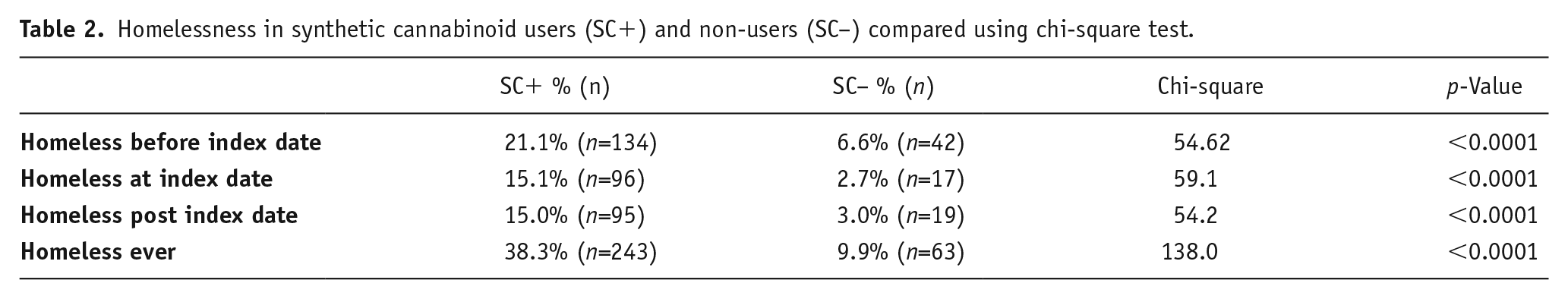
Individuals in the SC user group were significantly more likely to be homeless than those in the SC non-user group at each of the time points measured in this study (p<0.0001; Table 2). A total of 242 (38.3%) SC users were recorded as homeless at any time point in their medical records compared to 63 (9.9%) SC non-users.
Homelessness in synthetic cannabinoid users (SC+) and non-users (SC–) compared using chi-square test.
Diagnosis
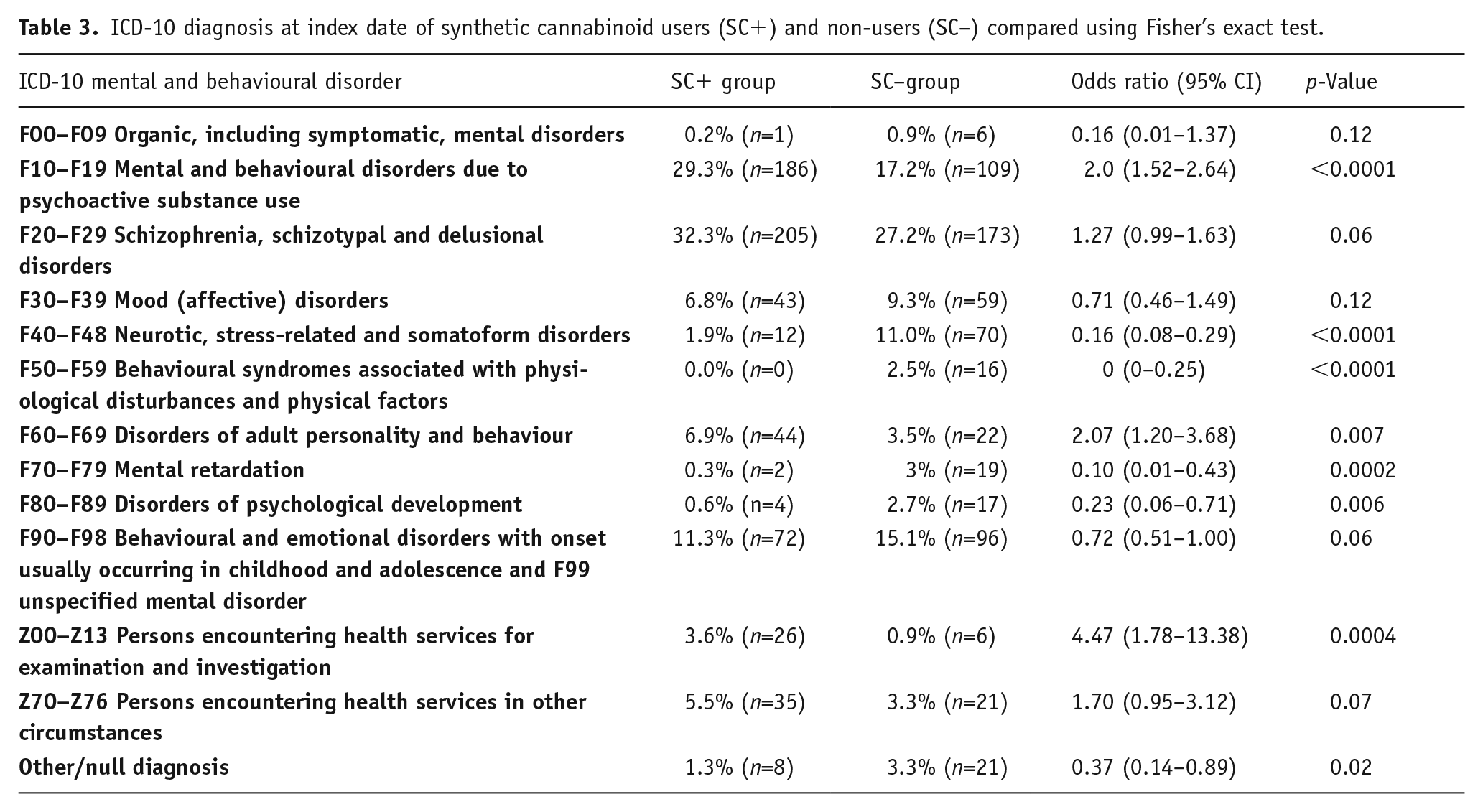
The most common diagnosis for both SC users and SC non-users was F20–F29 ‘schizophrenia, schizotypal and delusional disorders’. There was a non-significant trend for the SC users to have an F20–F29 diagnosis compared to SC non-users (32.3% vs. 27.2%; p=0.09). The two groups differed on other diagnostic categories, with SC users being more likely to have a recorded diagnosis of F10–F19 ‘mental and behavioural disorders due to psychoactive substance use’, and SC non-users being more likely to have a diagnosis of F40–F48 ‘neurotic, stress-related and somatoform disorders’, F50–F59 ‘behavioural syndromes associated with physiological disturbances and physical factors’, F70–F79 ‘mental retardation’ or F80–F89 ‘disorders of psychological development’ (p<0.005; see Table 3).
ICD-10 diagnosis at index date of synthetic cannabinoid users (SC+) and non-users (SC–) compared using Fisher’s exact test.
Admissions and contact with clinical services
Individuals in the SC user group had more hospital inpatient days following the index date than controls (M (SD)=85.54 (199.74) vs. 25.42 (92.32); p<0.0001) but did not significantly differ on number of home treatment team days. They had a trend towards increased face-to-face psychiatric contacts compared to controls (M (SD)=24.12 (40) vs. 19.75 (39.5); p=0.051). The difference in hospital inpatient days between SC users and SC non-users was still present after co-varying for age, sex, cannabis use, homelessness and diagnosis (β=45.7; p<0.0001), with homelessness (β=59.0; p<0.0001) and male sex (β=26.8; p=0.03) having a positive association with hospital inpatient days in the model, but no other variables having a significant main effect.
In a post hoc analysis of the effect of cannabis use on hospital inpatient days including age, sex, homelessness and diagnosis as covariates, SC users who were concurrent cannabis users had significantly more hospital inpatient days following the index date (n= 629; M (SD)=86.34 (200.5)) than controls who used cannabis alone (n=406; M (SD)=37.62 (112.8); β=48.3; p<0.0001), with homelessness (β=61.6; p<0.0001) and male sex (β=31.8; p<0.04) being significantly associated with hospital inpatient days following the index date. In addition, non-users of SCs who were cannabis users had significantly more hospital inpatient days (n=406; M (SD)=37.62 (112.8)) than individuals who were non-users of both cannabis and SC (n=229; M (SD)=3.8 (18.5); β=19.4; p=0.02), with homelessness (β=40.1; p=0.001) being significantly associated with hospital inpatient days following the index date.
Discussion
SC users had significantly higher use of hospital bed days than non-users following their first recorded use of SCs. This could have resulted from an increase in length of stay or more frequent hospitalisations, or both. This is in keeping with a previous study, which reported the length of a single stay in hospital on a dual diagnosis ward being increased if SC use was present at the time of admission (Bassir Nia et al., 2016). In contrast, one study reported a shorter length of stay in individuals experiencing an SC-induced psychotic episode compared to those presenting with schizophrenia (Altintas et al., 2016), and another reported that SC using individuals were likely to be prescribed lower antipsychotic doses and to be discharged more quickly than SC non-users (Deng et al., 2018). These studies examined the association of SC use recorded at hospitalisation on the length of the subsequent admission and may have included individuals presenting due to both acute and subacute effects caused by SCs. It is likely that a psychotic presentation caused by SC use would resolve rapidly on cessation of the drug and that such patients would require a shorter period of treatment than in those whose symptoms were due to schizophrenia. In the present study, we instead examined the total number of days spent in a psychiatric hospital after the first recorded evidence that the individual had taken SCs. This record may have been made in any psychiatric health-care setting, not just during admission to a psychiatric ward.
International studies of SC use, both in psychiatric patients (Every-Palmer, 2011; Manseau et al., 2017), and in other cohorts (Barratt et al., 2013; Loeffler et al., 2016; Vandrey et al., 2012), have found SC users to be predominantly male, non-white and young. Although we found SC users to be predominantly young males, they were also more likely to be white than would be expected from the local demographics. The reason for this is not clear but suggests that SC use in London may occur in different subcultural groups from those in the USA or other European countries.
We found that SC use was first reported in the CRIS system in 2007, but records of first SC use did not increase markedly until 2014. It is unclear whether this change was due to an increase in the number of patients using SCs or an increase in the awareness of clinicians of SCs and their relevance to mental illness, or a combination of the two. It is interesting to note that records of SC use have not continued to increase after the introduction of the Psychoactive Substances Act in May 2016, suggesting that the Act may have had an effect on SC use. This is in keeping with reports from the National Poisons Unit and the TOXBASE database, which both reported reduced enquiries about SC toxicity following the introduction of the Psychoactive Substances Act (Hobbs et al., 2018).
Although a large sample size was identified, this study was limited by reliance on a self-report methodology assessed retrospectively. Confounding effects are possible due to recall bias, reporting bias, clinician documentation bias, as well as potential poly-drug abuse amongst those reporting SC use and the control group. Poly-drug use may be associated with worse outcome and is more common in SC users (Joseph et al., 2019). Furthermore, we were not able to determine the proportion of patients who were taking higher potency cannabis, which has previously been associated with an increased risk of psychosis (Di Forti et al., 2015).
Other factors (including the use of alcohol) may have influenced the number of days of hospitalisation. We found that SC users had very high cannabis use, with almost all SC users taking cannabis. This is in keeping with previous work that has reported that more than 80% of SC users also take cannabis (Mensen et al., 2019). We have attempted to correct for this as far as we can through matching of groups and correcting for potentially confounding variables, but further work is required to establish the role that SC use plays in patients with mental illness and the directionality of this association.
In this study, any confirmed recorded use of SCs was deemed acceptable. This means that individuals who may have only tried SCs on one occasion and reported its use were included alongside those who had a heavier and more regular SC habit. It appears unlikely that single uses of SCs would lead to the type of increased inpatient hospitalisation found in this study, although the SC group included people at all levels of use. It is also possible that patients reporting SC use had mistaken another drug such as cannabis for SCs, although again this is unlikely to explain the increased hospital stay. It is equally possible that SC use was underreported in the studied population, especially after following the change in law that made this drug class illegal. Although relying on self-reported drug use amongst a psychiatric population is far from ideal, in the case of SC, where UDS have thus far failed to keep up with an ever-evolving class of drug, it is currently the best way of establishing use.
It is not possible to clarify the nature of the relationship between SC use and longer hospital admissions. Although one possibility is that SC use worsens psychiatric illness, which leads to longer admissions, it is also possible that it is those with more severe illness who are more likely to take SCs. We were not able to test this hypothesis directly, as we were not able to extract quantitative measures of psychopathology. The fact that SC use is associated with homelessness suggests that SC users may have less favourable social circumstances, which in turn leads to them being admitted for longer.
Although we found SC users were more likely to be young, male, homeless and cannabis users, it is possible that this demographic was subject to more thorough questioning regarding their drug habits compared to female patients, older males and those who were neither homeless nor reported cannabis users.
Lastly, we defined the index date for first recorded SC use by either self-report or positive UDS. Clinical suspicion of SC use often proceeds disclosure by the patient, and it is possible that the true first use of an SC predates the first recorded use in a patient’s electronic notes by months, if not years in some cases. It is also possible that patients who spend longer as inpatients in a psychiatric hospital are more likely to have undergone thorough questioning of their drug-taking habits, including at tribunals, than those who stay for only a number of days, which could in part explain why SC use was associated with more inpatient stays, although we only counted the number of days following the recording in the notes.
Conclusion
This study is in keeping with previous reports of SC users as being more likely to be young, male, homeless and cannabis users. It also suggests that SC use is associated with an increase in time spent admitted to a psychiatric hospital. Further work is needed to ascertain if this is due to increased morbidity associated with SC use, especially when compared to cannabis alone. This study highlights the need for clinicians to perform an adequate drug history, and suggests that targeted drug counselling approaches for SC use may be valuable. Future studies should focus on the efficacy of interventions such as psychoeducation upon reducing the duration of inpatient care.
Footnotes
Acknowledgements
The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the NIHR Biomedical Research Centre for Mental Health at the South London and Maudsley NHS Foundation Trust and Institute of Psychiatry, King’s College London. R.P. has received support from a Medical Research Council (MRC) Health Data Research UK Fellowship (MR/S003118/1) and a Starter Grant for Clinical Lecturers (SGL015/1020) supported by the Academy of Medical Sciences, The Wellcome Trust, MRC, British Heart Foundation, Arthritis Research UK, the Royal College of Physicians and Diabetes UK.
